
Synthesis of aligned carbon nanotube with straight-chained alkanes by nebulization method
LIU Yun-quan(刘云泉), CHEN Xiao-hua(陈小华), YANG Zhi(杨 植), PU Yu-xing(蒲玉兴), YI Bin(易 斌)
College of Materials Science and Engineering, Hunan University, Changsha 410082, China
Received 23 March 2009; accepted 25 August 2009
Abstract: Aligned carbon nanotubes (CNTs) were synthesized by nebulized spray pyrolysis of solutions of organometallics in carbon precursor solvents. Four types of straight-chained alkanes including n-pentane, n-hexane, n-heptane and n-octane were used as precursor solvents for synthesis of aligned CNTs. The results from scanning electron microscopy, transmission electron microscopy and Raman spectroscopy show that the CNTs obtained from them have different diameters and degrees of graphitization. It is found that the n-heptane is the most suitable for the growth of aligned CNTs with high quality and yield. The thermodynamic properties of precursory carbon sources such as boiling point and formation enthalpy are considered to play a decisive role in the synthesis of CNTs. It will be very helpful for the controllable preparation of aligned CNTs at relatively low cost.
Key words: aligned carbon nanotubes; nebulization; straight-chained alkanes; thermodynamic property
1 Introduction
Since carbon nanotube (CNT) was discovered by IIJIMA in 1991[1], considerable efforts have been made to study its preparation, growth mechanism and potential applications[2-4]. Aligned and well separated CNT morphology is important for many potential applications, where a high electric-field intensity is needed in field emission applications and a large surface area is desirable in catalytic reactions, such as fuel cell or battery. To date, a lot of studies have been done on the synthesis of aligned CNTs by catalystic chemical vapor deposition (CCVD) method[5-9]. They can be broadly divided into chemical and physical methods according to the way that carbon atoms are released from carbon-containing precursor molecules. In the physical methods, high-energy input is used to release the carbon atoms. The chemical methods rely on carbon atomization via catalytic decomposition of carbon precursors on the surface of transition metal particles. Conversely, in nebulized spray synthesis, the catalyst particles are suspended in the gas-phase throughout the entire CNT formation process. The formation of the geyser is accompanied by the generation of a spray, resulting from the vibrations at the liquid surface and the cavitation at the gas/liquid interface[10].
Nebulized spray pyrolysis has been employed for the preparation of submicron-sized particles and epitaxial thin films of complex metal oxides[11-12]. The advantage of using nebulized spray is its industrial scale process, as the reactants are fed into the furnace continuously. Furthermore, the nebulized spray method is safer and cheaper than classical CCVD method due to avoiding the use of some relatively expensive, dangerous or toxic gas carbon source such as C2H2, CH4, and CO. In the past a few years, many organic solvents, such as ethanol, xylene, and toluene, have been used as carbon feedstock in this method[13-14]. However, the effects of different precursor carbon sources on the formation of CNTs and quality of aligned CNTs are still uncertain. In this study, four types of straight-chained alkane were selected as carbon sources on the basis of the chemical and structural similarities. The relationship between properties (molecular structure and thermodynamic property) of precursory carbon source and quality of aligned CNTs (diameter, degree of graphitization, and alignment) in a similar reaction system is investigated.
2 Experimental
Silicon substrates were placed in the reactor to collect the product. In a typical synthesis, 2 g of ferrocene was dissolved in 100 mL of the precursor solution nebulized using a 1.54 MHz ultrasonic beam carried in a 40 mm quartz tube and placed in a SiC furnace maintained at the required temperature (900 ℃). Ultra pure argon was used as the carrier gas and the gas flow rate was controlled using mass flow controllers. In a typical procedure, the flow rate of the carrier gas was kept at 1 000 cm3/min. After reaction for 40 min, the supply of the precursor solution was stopped and the furnace was cooled to room temperature in flowing argon. The products were collected after the tube was cooled to room temperature. These samples prepared from four types of straight-chained alkane carbon precursors including n-pentane, n-hexane, n-heptane and n-octane were marked as CNTs-pent, CNTs-hex, CNTs-hept and CNTs-oct, respectively. Morphology of the samples was characterized with scanning electron microscope(SEM, JSM-6700F) and high resolution transmission electron microscope (HRTEM, JEM-3010). Raman spectroscopy was performed using a Jobin Yvon Labran-010 spectrophotometer equipped with a 50 mW diode laser source for 632 nm excitation and a CCD detector.
3 Results and discussion
3.1 Morphologies of aligned carbon nanotubes
Fig.1 shows SEM images of samples prepared from four types of saturated straight-chained alkanes. The images of the CNTs-pent (Figs.1(a) and 1(b)) show that the samples consist of plentiful fiber-bundles with ordered arrangement, where abundant carbon fibers with rough surfaces and a wide diameter ranging from 150 to 400 nm are highly interlaced and enlaced. These suggest that n-pentane is not suitable for the growth of aligned CNTs. Figs.1(c) and (d) show the images for the CNTs- hex sample. It can been found that the sample possesses a relatively good array of carbon nanofibers, and the CNTs are curved and intertwined with each other in the nanoscale and their diameters are approximately 40-50 nm. These observations indicate that the carbon precursor can yield aligned CNTs, but the quality is lower. Fig.1(e) gives a low magnification SEM image of the sample obtained from n-heptane, where several cleaved CNT films with the thickness of about 700 mm can be seen. A high magnification SEM image of the cleaved CNT film shown in Fig.1(f) clearly reveals that the CNTs possess a good alignment with few impurities and have uniform diameter of about 45 nm. Furthermore, it can be observed that all of the CNTs are almost very straight and aligned along a uniform direction, which is an important indication for high-quality aligned CNTs with excellent integration performance. Figs.1(g) and (h) show the SEM images of the CNTs-oct. It is apparent that the CNTs are randomly oriented, and possess the relatively short length. These suggest that n-octane is not favored for the synthesis of aligned CNTs.
The TEM and HRTEM images (Fig.2) of samples prepared from n-heptane show that the aligned CNTs with hollow and multi-walled structure are well crystallized and a little amorphous carbon is present on the external walls. These ordered morphology characteristic and relatively perfect microstructure clearly indicate that n-heptane is a ideal precursor for the preparation of the high-quality aligned CNTs.
3.2 Raman spectroscopy of aligned carbon nanotubes
Raman spectroscopy has been applied to all the samples obtained from four kinds of straight-chained alkane, as shown in Fig.3. All the spectra indicate the presence of G-band (at about 1 590 cm-1) and D-band (at about 1 320 cm-1) with different G/D ratio. The G-band is representative of the amount of graphitization associated with the nanotube growth, while the D-band represents the amount of defects (open ends, disorder, amorphous deposit, etc). It is observed that the G/D ratio of as-grown nanotubes depends on the carbon precursors. For the sample synthesized with n-pentane, n-hexane, n-heptane and n-octane, the G/D ratios are 0.856, 1.106, 1.180 and 0.770, respectively. The narrow D-band and higher G/D ratio observed for n-heptane indicate that the amounts of graphitic impurities and side wall defects are more reduced than those for the other precursors, which further confirms that n-heptane is a good precursor candidate for synthesis of aligned CNTs.
3.3 Effect of carbon precursor on formation of aligned carbon nanotubes
The experimental results show that the synthesis of aligned CNTs is very sensitive to the carbon precursor. Generally speaking, during the chemical vapor deposition process, the carbon precursor is thermally decomposed and dissociated into carbon fragments; then the carbon fragments are deposited on the surfaces of the catalyst particles and diffuse into the metal to precipitate the ACNTs in a region with high catalyst density. In the experiments, the synthesis parameters such as temperature, gas flow rate, ferrocene concentration and ultrasonic frequency were fixed. Thus, the molecular structure and properties of the carbon precursor play a critical role to affect the quality of products[15-16]. Focus on two aspects should be required. One is the decomposition rate of carbon precursor, and the other is the decomposition mechanism of carbon precursor as well as the molecule structures of their in-process products during the process. It is well known that these straight-chained alkanes possess very similar molecule structures and their molecules are made up of methyl (―CH3) and methylene (―CH2―) radicals. This indicates that four kinds of alkanes can yield some in-process products with similar components during the decomposition process. Thus, the decomposition rates of these alkane precursors become a crucial factor for the nanotube formation.

Fig.1 SEM images of samples prepared from different kinds of saturated alkanes: (a), (b) n-pentane; (c), (d) n-hexane; (e), (f) n-heptane; (g), (h) n-octane

Fig.2 TEM (a) and HRTEM (b) images of samples prepared from n-heptane
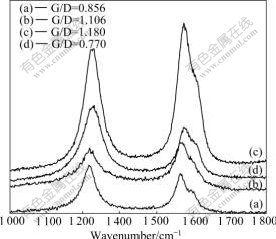
Fig.3 Raman spectra of samples obtained from n-pentane (a), n-hexane (b), n-heptane (c) and n-octane (d)
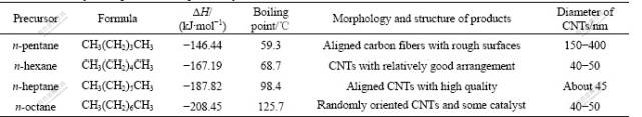
Based on the classical thermodynamics theory, the decomposition rate mainly depends upon physico- chemical properties such as boiling point and formation enthalpy. Table 1 lists the chemical structure, formation enthalpies and boiling points of the precursors. From Table 1, it can be found that the value of the formation enthalpies and the boiling point of these saturated alkanes increase with increasing the relative molecular mass. Higher value of the formation enthalpies and boiling points mean that the decomposition occurs more difficultly. We thus conclude that the decomposition rates of four kinds of alkanes decrease with increasing the relative molecular mass. For n-pentane with low relative molecular mass, the surface of the nanotubes is rough, and the diameter becomes large. For n-octane with high relative molecular mass, the CNTs obtained are relatively short and a lot of non-nanotube materials are present. When n-hexane and n-heptane are used as precursory carbon sources, the aligned CNTs can be synthesized. Especially for n-heptane, the alignment of CNTs is the best, and no amorphous carbon is observed. This phenomenon should be attributed to the variety of the decomposition rate. As carbon precursor is thermally decomposed at a relatively high rate, abundant carbon fragments can be formed in a short time. Some fragments diffuse into the catalyst particles to precipitate in the form of nanotubes, while the superfluous fragments directly deposit around the outer walls of the nanotube and form the amorphous carbon layer. This is consistent with the results in this work. As the decomposition rate is relatively low, carbon fragments cannot be sufficiently supplied to deposit on the surface of the catalyst. Some catalyst clusters will continually congregate with each other and form larger particles until losing their activities. That is to say, this catalyst is not suitable for the synthesis of CNTs. The n-heptane with decomposition rate higher than that of n-octane and lower than that of n-pentane has already been confirmed as excellent precursor for growth of high-quality aligned CNTs, as shown in Figs.1(e) and (f), strongly suggesting that the decomposition rate of n-heptane is very suitable for synthesis of ACNTs under the experimental conditions in this work. We thus conclude that the thermodynamic properties of straight-chained alkanes play a decisive role in the morphology and structure of the CNT samples.
Table 1 Thermodynamic parameters of precursory carbon sources
4 Conclusions
1) Aligned carbon nanotubes with different diameters and degrees of graphitization are prepared with four types of straight-chained alkanes as precursory carbon precursors by nebulized spray pyrolysis method.
2) The thermodynamic properties of precursory carbon sources such as boiling point and formation enthalpy play a decisive role in the morphology and structure of the carbon nanotube samples prepared from four kinds of straight-chained alkanes.
3) The decomposition rate of n-heptane is very suitable for synthesis of aligned carbon nanotubes under this experimental condition.
References
[1] IIJIMA S. Helical microtubules of graphitic carbon [J]. Nature, 1991, 354: 56-59.
[2] KONG J, FRANKLIN N R, ZHOU C W, CHAPLINE M G, PENG S, CHO K J, DAI H J. Nanotube molecular wires as chemical sensors [J]. Science, 2000, 287: 622-625.
[3] CHANG L W, LUE J T. Magnetic properties of multi-walled carbon nanotubes [J]. Journal of Nanoscience and Nanotechnology, 2009, 9: 1956-1963.
[4] KIM D, LIM S H, GUILLEY A J, COJOCARN C S, BOUREE J E. Growth of vertically aligned arrays of carbon nanotubes for high field emission [J]. Thin Solid Films, 2008, 516: 706-709.
[5] EDGAR K, JOHN L. Aerosol-based synthesis of carbon nanotubes [J]. Spencer Current Applied Physics, 2004, 4: 121-124.
[6] IAIA A, MARTY C, NAUD V, LOISEAU A, DI MUOIO E, FOURNIER T, BONNOT A M. Oriented growth of suspended single wall carbon nanotube by hot filament CVD [J]. Thin Solid Films, 2006, 501: 221-223.
[7] ANASTASIOS J H, ALEXANDER H. Rapid growth and flow-mediated nucleation of millimeter-scale aligned carbon nanotube: Structures from a thin-film catalyst [J]. J Phys Chem, 2006, B110: 8250-8257.
[8] LIANG Z, TAN Y Q, DANIEL E. Controlling the growth of vertically oriented single-walled carbon nanotubes by varying the density of CoMo catalyst particles [J]. Chemical Physics Letters, 2006, 422: 198-203.
[9] CHIUA C C, TAIA N H, YEHB M K, CHENB B Y, TSENGA S H, CHANGC Y H. Tip-to-tip growth of aligned single-walled carbon nanotubes under an electric field [J]. Journal of Crystal Growth, 2006, 290: 171-175.
[10] LANGLET M, JOUBERT J C. Chemistry of advanced materials―A chemistry for the 21st century (IUPAC Monograph) [M]. Oxford: Blackwell Scientific Publisher, 1993: 55.
[11] RAJU A R, RAOCN R. Oriented films of LaNiO3 and other members of the Lan+1NinO3n+1 series, LaCuO3- delta and Pb(Zr0.52Ti0.48)O3, obtained by nebulized spray pyrolysis [J]. Chem Mater, 1995, 7: 225-231.
[12] AIYER H N, RAJU A R, SUBBANNA G N. Epitaxial nature of the films of LaNiO3, Pb(Zr0.5Ti0.5)O3, and La0.95Mn0.95O3 obtained by nebulized spray pyrolysis [J]. Chem Mater, 1997, 9: 755-760.
[13] YANG Z, CHEN X H, NIE H G. Direct synthesis of ultralong carbon nanotube bundles by spray pyrolysis and investigation of growth mechanism [J]. Nanotechnology, 2008, 19(25): 5606-5613.
[14] LUO C X, LIU L, JIANG K L. Growth mechanism of Y-junctions and related carbon nanotube junctions synthesized by Au-catalyzed chemical vapor deposition [J]. Carbon, 2008, 46(3): 440-444.
[15] JIANG Z W, SONG R J, BI W G. Polypropylene as a carbon source for the synthesis of multi-walled carbon nanotubes via catalytic combustion [J]. Carbon, 2007, 45(2): 449-458.
[16] MONTORO L A, CORIO P, ROSOLEN J M. A comparative study of alcohols and ketones as carbon precursor for multi-walled carbon nanotube growth [J]. Carbon, 2007, 45(6): 1234-1241.
Foundation item: Projects(50772033, 50972043) supported by the National Natural Science Foundation of China; Project(2007FJ3003) supported by the Natural Science Foundation of Hunan Province, China
Corresponding author: CHEN Xiao-Hua; Tel: +86-731-88821727; Fax: +86-731-88821611; E-mail: hudacxh62@yahoo.com.cn
DOI: 10.1016/S1003-6326(09)60250-8
(Edited by YANG Bing)