水硅铀矿及其他常见铀矿的铀浸出动力学比较
来源期刊:中国有色金属学报(英文版)2018年第10期
论文作者:H. S. REYNOLDS R. RAM M. I. POWNCEBY Y. YANG M. CHEN J. TARDIO L. JONES S. K. BHARGAVA
文章页码:2135 - 2142
关键词:水硅铀矿;铀;钛铀矿;浸出动力学
Key words:coffinite; uraninite; brannerite; acid leaching; extraction kinetics
摘 要:水热合成并表征水硅铀矿,在此基础上对水硅铀矿在硫酸体系中的浸出过程进行研究,并将结果与其他相关的合成U4+矿物如氧化铀(UO2)和钛铀矿(UTi2O6)在类似条件下的浸出速率和浸出率进行比较。浸出时间对铀溶出影响的研究表明,水硅铀矿在36~48 h被完全浸出,该反应的活化能为38.4 kJ/mol。与水硅铀矿相比,氧化铀矿的溶出速率明显较快,可以在3 h内达到完全溶出(Ea=42~84 kJ/mol)。比较而言,合成钛铀矿的浸出速率较水硅铀矿的明显更低,其144 h铀的最大浸出率只有18%。上述3种矿物溶出速率和提取率的显著区别与文献报道相符合,即矿物的可溶出性为氧化铀>水硅铀矿>钛铀矿。在天然水硅铀矿中,由于杂质的存在,其可溶出性可能会被进一步抑制。
Abstract: A synthetic coffinite was hydrothermally prepared and characterized before conducting a series of acid sulfate leach tests under conditions of relevance to uranium extraction. The results were then compared with similar studies on synthetic versions of the related U4+ minerals uraninite (UO2) and brannerite (UTi2O6) to identify and differentiate the rate and U extraction among these important uranium minerals. Tests examining the influence of residence time on uranium dissolution from synthetic coffinite, uraninite and brannerite showed that under similar experimental conditions, complete dissolution of uranium from coffinite was obtained between 36 and 48 h. The activation energy for this reaction was calculated to be 38.4 kJ/mol. This represented a significantly slower rate of dissolution than that indicated for uraninite which dissolved in 3 h (Ea=15.2 kJ/mol). The synthetic brannerite was leached at a much slower rate than the coffinite and reached a maximum dissolution of ~18% U in 144 h (Ea=42-84 kJ/mol). The clear differentiation in rates and U extraction among the three minerals is consistent with previous literatures which suggest that in terms of leachability, uraninite>coffinite>brannerite. It is expected that the presence of impurities in natural coffinites would further inhibit leachability.
Trans. Nonferrous Met. Soc. China 28(2018) 2135-2142
H. S. REYNOLDS1, R. RAM2, M. I. POWNCEBY3, Y. YANG3, M. CHEN1,3, J. TARDIO1, L. JONES1, S. K. BHARGAVA1
1. Centre for Advanced Materials and Industrial Chemistry (CAMIC), School of Applied Sciences, RMIT University, GPO Box 2476, Melbourne VIC 3001, Australia;
2. School of Earth Atmosphere and Environment, Monash University, 9 Rainforest Walk, Clayton, Victoria 3800, Australia;
3. CSIRO Mineral Resources, Private Bag 10 Clayton South, Victoria 3169, Australia
Received 19 December 2017; accepted 7 May 2018
Abstract: A synthetic coffinite was hydrothermally prepared and characterized before conducting a series of acid sulfate leach tests under conditions of relevance to uranium extraction. The results were then compared with similar studies on synthetic versions of the related U4+ minerals uraninite (UO2) and brannerite (UTi2O6) to identify and differentiate the rate and U extraction among these important uranium minerals. Tests examining the influence of residence time on uranium dissolution from synthetic coffinite, uraninite and brannerite showed that under similar experimental conditions, complete dissolution of uranium from coffinite was obtained between 36 and 48 h. The activation energy for this reaction was calculated to be 38.4 kJ/mol. This represented a significantly slower rate of dissolution than that indicated for uraninite which dissolved in 3 h (Ea=15.2 kJ/mol). The synthetic brannerite was leached at a much slower rate than the coffinite and reached a maximum dissolution of ~18% U in 144 h (Ea=42-84 kJ/mol). The clear differentiation in rates and U extraction among the three minerals is consistent with previous literatures which suggest that in terms of leachability, uraninite>coffinite>brannerite. It is expected that the presence of impurities in natural coffinites would further inhibit leachability.
Key words: coffinite; uraninite; brannerite; acid leaching; extraction kinetics
1 Introduction
Coffinite is one of the major tetravalent (U4+) minerals in economically exploitable reduced U-ores and is the second most abundant source of uranium in the world behind uraninite [1]. Coffinite is an orthosilicate with the general formula of ABO4 [2-4]. The most commonly used formula for coffinite is U(SiO4)1-x(OH)4x, first proposed by STIEFF et al [5,6] based on chemical data coupled with infrared (IR) analysis. More recent studies [7-10] were unable to confirm the presence of hydroxyl substitution, instead suggesting that water bound to coffinite is of a molecular nature. This led to the proposal of the chemical formula USiO4・nH2O, with n ≈ 2 [11].
Uranium is typically extracted from its ores via acid leaching procedures, usually using sulphuric acid solutions [12]. Simple oxide minerals like uraninite and pitchblende are relatively easily leachable, while more complex uranium ores are not so leachable. Depending upon the ease of dissolution/leachability, the uranium minerals are categorised as (1) slightly refractory (oxidation is required prior to leaching, e.g. coffinite and uranothorite), (2) moderately refractory (requires higher leach temperatures, greater free acid and oxidant concentration and longer leach periods, e.g. brannerite and davidite), and (3) highly refractory (requires extreme leach conditions, e.g. betafite and pyrochlore). Past observations regarding the degree of refractoriness of various uranium minerals, however, have largely been made using natural ores, often containing a complex matrix of uranium minerals [13]. In leach circuits using natural uranium ores, competing effects such as grain size, liberation, gangue mineralogy (potential acid consumers), and the composition of the uranium-bearing ore will all play a significant role in comparative dissolution kinetics.
The synthesis of coffinite has been the subject of many studies based on dry processes [10,14], sol-gel chemistry or hydrothermal [7,15-19] protocols. Although many attempts have been made, there have been persistent difficulties encountered in the preparation of pure, single-phase coffinite. POINTEAU et al [18] adapted the hydrothermal method of FUCHS and HOEKSTRA [7] and determined that the difficulties were because coffinite was sensitive to several experimental parameters including pressure (<50×106 Pa) and Eh/pH. They reported that coffinite synthesis required a narrow pH range of 8-9.5 for thermodynamic stability and the reaction conditions must remain oxygen-free (low Eh to prevent oxidation to U6+). Reaction kinetics was also assumed by POINTEAU et al [18] to be a key factor in effective coffinite synthesis. This was recently confirmed by COSTIN et al [20] who showed that the mechanism of formation of Th1-xUxSiO4 solid solutions through a dissolution/ reprecipitation process under hydrothermal conditions slows down with increasing U content.
To provide a definitive comparison of leach rates of coffinite versus other commonly exploited uranium minerals, synthetic coffinite was hydrothermally prepared and characterised before being used in a series of acid sulfate leach tests under conditions of relevance to uranium extraction. The results were then contrasted with similar studies on synthetic versions of the related U4+ minerals uraninite (UO2) and brannerite (UTi2O6) to differentiate the rate and U extraction between these three common U-bearing minerals in uranium ores.
2 Materials and methods
2.1 Coffinite synthesis
The synthetic coffinite was produced by a hydrothermal technique using a method similar to that described by POINTEAU et al [18] and detailed in Ref. [21]. Briefly, previously prepared UCl4 [22] was dissolved in water and added drop-wise to a dissolved sodium metasilicate solution in which the silicate ratio was slightly above stoichiometric one. Sodium hydroxide was added drop-wise until it formed a gel (approximately pH 8-9) and the solution was then buffered with sodium bicarbonate to a pH of 9. The buffered solution was placed in an autoclave and heated to 250 °C for 24 h, cooled slowly, washed, centrifuged with water, and allowed to air dry. Any uraninite remaining in the product due to slow reaction kinetics was washed away with sulphuric acid. The resultant coffinite was dry sieved to a particle size of P100 ≤75 μm. Similarly, the uraninite and brannerite used for comparative leach tests were also dry sieved to P100 ≤75 μm.
2.2 Characterisation methods
The hydrothermally prepared coffinite was characterized by X-ray diffraction (XRD), X-ray photoelectron spectroscopy (XPS), and electron probe microanalysis (EPMA).
2.2.1 X-ray diffraction (XRD)
Standard X-ray diffraction patterns were obtained on a Bruker D8 Advance fitted with a copper tube (Cu Kα radiation), monochromator and scintillation detector. Diffraction patterns were run at an accelerating voltage of 40 kV and current of 35 mA using a 1° fixed divergence slit. Data were collected over a 2θ range of 10°-90° with a step size of 2θ=0.02° and 2.5 s per step. The instrument was calibrated with a Si calibration standard prior to use.
2.2.2 X-ray photoelectron spectroscopy (XPS)
X-ray photoelectron spectroscopy (XPS) analysis was conducted using a thermo Kα XPS instrument at a pressure of <1.333×10-7 Pa. Samples were prepared by pressing powdered samples in a die press at 7.84×104 N of pressure and affixing the pressed sample to carbon tape on the sample plate. The position of the C 1s peak (285 eV) was used as the internal standard and U 4f core level spectra were analyzed with monochromatic aluminium Kα radiation (photon energy of 1486.6 eV) at pass energy of 20 eV and an electron take-off angle at 90°. The overall resolution was 0.1 eV.
2.2.3 Electron probe microanalysis (EPMA)
Approximately 0.7-0.8 g of the synthetic coffinite was set in epoxy resin and polished flat for EPMA. Two types of EPMA information were obtained. Initially, the sample was mapped using a high resolution field emission gun (FEG) equipped EPMA (JEOL 8500F Hyperprobe) in order to examine the homogeneity of the coffinite grains. Following mapping, the chemistry was measured by quantitative EPMA techniques. The operating conditions for each of the techniques are described separately below.
1) EPMA mapping
An area was mapped using a combination of wavelength dispersive (WD) and energy dispersive (ED) spectroscopic techniques. The distribution of Si and U was mapped using the WD spectroscopy with wollastonite (CaSiO3) and uranium oxide (UO2) as standards. Elements not measured by WD spectroscopy were measured using two energy-dispersive (ED) spectrometers operating in parallel. Operating conditions for the microprobe were: an accelerating voltage of 12 kV, a current of 50 nA, a step size of 0.2-1.0 mm and counting time of 25 ms per step.
2) Quantitative EPMA
The following suite of elements was analyzed: Si, U, S and O. For each element, the counting time on the peak was 20 s and half of that time measured on both sides of the peak (to measure the background). The standards used for calibration, the X-ray peak used, and the calculated detection limits (2σ) are as follows: natural wollastonite (CaSiO3) for Si (Kα), 270×106, synthetic pyrite (FeS2) for S (Kα), 400×106, and natural UO2 for U (Mα), 1700×106. Oxygen was measured directly using the Kα X-ray line that was calibrated using the natural uraninite standard. The accelerating voltage and beam current were 20 kV and 30 nA, respectively, and the electron beam diameter was defocused to 5 μm for all analyses.
3 Results and discussion
3.1 Characterisation of synthetic coffinite
3.1.1 X-ray diffraction (XRD)
Coffinite is an orthosilicate, isostructural with zircon ZrSiO4, and thorite ThSiO4. The synthesis of pure coffinite was confirmed by XRD analysis with the expected tetragonal zircon-type pattern for space group I41/amd [23]. Qualitative investigation of the pattern indicated the synthetic coffinite to be pure, with possible impurities such as uraninite and quartz not apparent. The unit cell parameters of the synthetic coffinite were: a=b= (6.9980±0.0002)  and c=(6.2720±0.0002)
and c=(6.2720±0.0002)  . These are in good agreement with previously reported values (Table 1).
. These are in good agreement with previously reported values (Table 1).
Table 1 Crystallographic unit cell data of synthetic coffinite prepared in this work compared with other published synthetic coffinite data

3.1.2 X-ray photoelectron spectroscopy
XPS analysis results from the synthetic coffinite are shown in Fig. 1. Deconvolution of the main components associated with the uranium 4f core levels gave peaks at (381.1±0.2) eV and (392.0±0.2) eV for the U 4f7/2 and U 4f5/2 levels, respectively. A shake-up satellite U 4f7/2 peak was observed at (386.8±0.2) eV with the difference in binding energies between the main photoelectric peak and the satellite peak being ~6 eV, consistent with uranium being in the U(IV) valence state [18,24]. A second contribution associated with uranium levels was observed at (382.5±0.2) eV, present as a shoulder on the U 4f7/2 peak. This was attributed to the U 4f7/2 level peak characteristic of U(VI) and most likely caused by surface oxidation (after being stored in air).
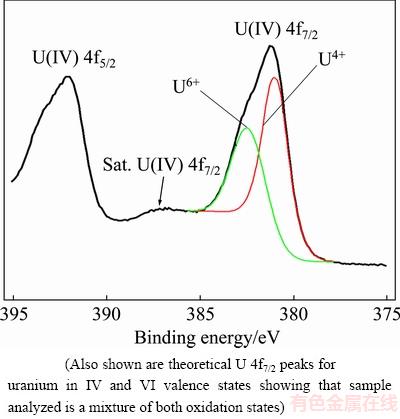
Fig. 1 Uranium U 4f7/2 and U 4f5/2 core level XPS data for synthetic coffinite
3.1.3 Scanning electron microscopy (SEM) and electron probe micro-analysis (EPMA)
Scanning electron microscopy (SEM) imaging of individual synthetic coffinite pieces indicated that the final product consisted of polycrystalline material exhibiting heterogeneous contrast caused by variability in average atomic number between individual crystals (Fig. 2(a)). Textural inhomogeneity was also noted with the central parts of the grains being coarsely crystalline and the edges of the particles exhibiting a finer-grained texture. The internal texture of the synthetic coffinite was consistent with that previously observed by POINTEAU et al [18], where they described their synthetic coffinite as also comprising heterogeneous, polycrystalline aggregates. A comparison of the synthetic material with the internal texture of naturally occurring coffinite particles from Roxby Downs, South Australia showed almost identical textures (Fig. 2(b)).
An EPMA map was obtained over large ~300 μm sized polycrystalline particle shown in Fig. 2(a) which showed a patchy, heterogeneous BSE contrast. Individual element maps for uranium (Fig. 3(b)) and silicon (Fig. 3(c)) showed that the BSE contrast was correlated strongly with variations in both elements. The brighter areas in the BSE image corresponded to regions with greater amounts of uranium (higher Z) while the slightly darker regions indicated areas of higher silicon. The edges of the coffinite grain had a greater amount of silicon present with some internal crystallites also exhibiting variations in the uranium to silicon ratio, e.g. yellow and green patches evident in Fig. 3(c). The oxygen content was also highly correlated with the distribution of silicon (Fig. 3(d)).

Fig. 2 Scanning electron microscopy (BSE) image of synthetic coffinite showing polycrystalline character of particles and significant variation in Z-contrast between individual crystals (a), and image of natural coffinite grain (Roxby Downs, South Australia) showing characteristic heterogeneous and polycrystalline texture (b)

Fig. 3 EPMA mapping results from hydrothermally prepared synthetic coffinite sample shown in Fig. 2(a)
Quantitative analysis of the synthetic coffinite indicated that the mass fraction of uranium and silicon varied over wide ranges of 62.72%-65.44% U and 8.93%-11.33% Si, respectively. The sample also contained 0.124%-0.494% sulphur. The overall U to Si ratio was low, 0.73, compared with the expected U to Si ratio of coffinite (i.e. 1.00) indicating excess silicon and depletion in U compared to ‘stoichiometric’ USiO4 (72.1% U and 8.5% Si, mass fraction). While this result may suggest that coffinite has not been formed during the synthesis experiments, it may be an erroneous assumption to make. XRD measurements confirmed that coffinite was present in substantial quantities within the sample. The EPMA results suggest, however, that the coffinite is likely to be extremely fine-grained, below the analysis volume of the electron beam, and intimately mixed with a nanocrystalline silica-rich phase (as no significant excess quartz was identified during XRD analysis, it is postulated that the excess silica is amorphous in character). The presence of amorphous silica leads to higher than expected Si levels and lower U contents (a dilution effect) when being probed by the electron beam. The presence of fine-grained coffinite and amorphous silica can only be detected using higher resolution microscopic techniques such as transmission electron microscopy.
Attempts to synthesize coffinite by hydrothermal methods carried out by POINTEAU et al [18] also led to the formation of patchy polycrystalline grains that exhibited U contents of ~63% and Si contents of ~13% (mass fraction), similar to those measured in the current study. These gave a corresponding low U to Si ratio of ~0.6. They also attributed the low U to Si ratio to result from the presence of a Si-rich phase in the sample, which was most prominent around the edges of grains but was assumed to be present within the interior as amorphous, nanocrystalline material. In contrast to the current results, however, POINTEAU et al [18] managed to synthesize coffinite that appeared to be stoichiometric (and water-free). They proposed that long synthesis times caused decomposition of coffinite over time to produce UO2 and Si-rich amorphous material.
3.2 Synthetic coffinite leach tests
Tests were conducted to examine the effect of leach temperature on the extraction of uranium from synthetic coffinite under conditions of relevance to industrial processing of [U] ores. Besides temperature, operating conditions were kept constant at values used in typical industrial leach circuits (i.e. 100 mg/L U; [Fe3+]=1 g/L; [Fe2+]=3 g/L; [Fe]Total=4 g/L; [H2SO4]=15 g/L (0.15 mol/L); Oxidation reduction potential (ORP)=460 mV). Temperature was varied among 50, 60, 80 and 95°C. The results are presented in Fig. 4 and can be summarised as:
1) There was minimal change in rate and extent of uranium dissolution when the temperature was below 60 °C. Uranium extraction levels were about 10% U at 50 °C and 20% U at 60 °C.
2) Increasing the temperature to 80 °C substantially increased both the rate and extent of uranium dissolution reaching a maximum of 75% U after 90 min.
3) A further increase in temperature to 95 °C caused the rate of uranium dissolution to increase substantially in the first 30 min before a plateau was reached. A maximum dissolution of 80% U was achieved after 90 min.
Based on these data, the activation energy for uranium dissolution from synthetic coffinite was calculated to be 38.4 kJ/mol.
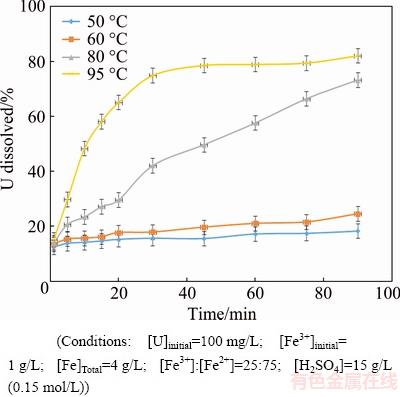
Fig. 4 Uranium dissolution of synthetic coffinite at various temperatures
The coffinite leach test results were compared with similar studies conducted using synthetic uraninite and synthetic brannerite to examine differences in the rate and U extraction among these commercially exploited uranium minerals. Experimental conditions are provided in Table 2 and results are shown in Fig. 5. Further, it is noted that all three minerals were synthesised to be primarily in the U4+ oxidation state for effective determination of kinetics and mechanism.
Table 2 Conditions for synthetic coffinite, uraninite and brannerite leach tests

Results in Fig. 5 showed that under the dissolution experimental conditions (Table 2) complete dissolution of uranium from coffinite was obtained between 36 and 48 h. This was significantly higher than uraninite which dissolved in 3 h [25]. The synthetic brannerite was leached at a much slower rate than the coffinite and uraninite reaching a maximum dissolution of only ~18% after 144 h. The slower rate was attributed to the formation of a TiO2-rich passivation layer [26].
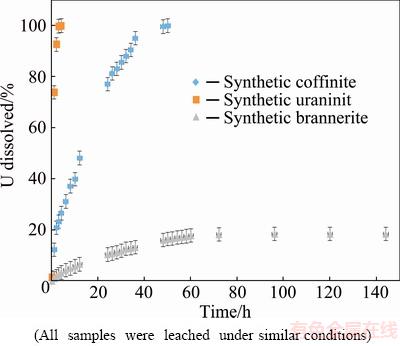
Fig. 5 Uranium dissolution results for synthetic coffinite, uraninite and brannerite
As previously indicated, the activation energy for uranium dissolution from synthetic coffinite was calculated to be 38.4 kJ/mol. This value is larger compared with uranium dissolution from synthetic uraninite under similar conditions where the activation energy was calculated to be 15.2 kJ/mol [25,27]. The differences are consistent with statements in the literature comparing the dissolution of uraninite and coffinite, where uraninite is quoted to be more readily dissolved in dilute acid compared to coffinite [28]. The coffinite activation energy was also lower when being compared with uranium dissolution from brannerite which, although variable, has previously been reported to be 42-84 kJ/mol [29,30].
The clear differentiation in U extraction rates and calculated activation energies among the three most commonly processed uranium minerals is consistent with previous literatures which indicate that uraninite is the easiest to leach (<1 d), followed by coffinite (2-3 d) and brannerite (>7 d) [13,31,32].
3.3 Implications for processing
It is widely accepted that a number of factors influence the dissolution of uranium from uranium bearing minerals [12]. The factors that have been reported to date to have the most influence on the extent and/or rate of dissolution include lixiviant type, temperature, oxidant and redox potential. The influence of chemistry on leachability of uranium minerals has been demonstrated on both simple oxides such as uraninite [25,33] and complex multiple-oxide type minerals such as brannerite [29]. RAM et al [33] conducted studies on Pb-doped and Th-doped uraninite and found variations in rate and U extraction compared to pure UO2. They attributed these changes directly to the compositional and structural variations in the prepared samples. In the case of brannerite, CHARALAMBOUS et al [29] showed significant increases in U extraction from natural brannerite samples compared to synthetic versions. They attributed the increase in leaching to the natural samples being highly metamict and undergoing further degree of alteration due to alpha decay. Upon heat treatment of the natural samples to restore the crystal lattice, a significant reduction in rate and U extraction was observed. They postulated that the composition and structure of brannerites within the ores would further influence their leachability. Based on evidence for broad variation in chemistry and degree of metamictisation exhibited by natural coffinites (e.g. Refs. [34-41]), the chemical and microstructural aspects of coffinite are likely to play an important role in any process to extract uranium from coffinite-containing ores.
The majority of works on uranium dissolution are mostly focused on ‘bulk’ uranium dissolution from uranium-bearing ores and do not discuss the results with any specificity regarding minerals such as coffinite. Although coffinite may be common uranium mineral in many deposits, it often only comprises a small percentage of the total uranium in the ore (e.g. 1.7%-2.7% [31]), making assumptions based on the general findings on coffinite dissolution from these studies difficult to interpret. Very few studies have been reported in the open literature on the dissolution of uranium from coffinite. The lack of studies on uranium dissolution from coffinite is most likely due to a combination of (1) difficulty in isolating the pure mineral/obtaining high purity samples of the mineral, and (2) coffinite not present in significant quantities in ore bodies that have been selected for mining to date. Although there have been very few studies on the dissolution of uranium from coffinite reported in the open literature, multiple researchers have stated that coffinite dissolution is slower in sulphuric acid when being compared to uraninite dissolution [13,31,32,42].
The most recent and comprehensive study on the dissolution of uranium from coffinite was published by MALEY et al [43]. This study was conducted using a sample mixture containing natural coffinite. The exact amount of coffinite in the sample was not reported; however, it was reported that the sample contained ~1500×106 of uranium (measured as U3O8), and almost 100% of the uranium present in the sample was identified as coffinite. The dissolution studies conducted by MALEY et al [43] on the sample mixture containing coffinite were conducted at 30 °C, with sulphuric acid lixiviant at a pH of 1.7; ferric sulphate was added at varying amounts to study the effect of iron. The authors found a strong relationship between ferric concentration and uranium dissolution. At a solution ORP of 475 mV (versus a Ag/AgCl reference electrode) and [Fe]Total between 15 and 1500 mg/L, uranium dissolution above 80% U was achieved after 6 h. Additional studies have been conducted involving low grade uranium ores containing trace levels of coffinite by MACNAUGHTON et al [13] and LOTTERING et al [32]. The general consensus of studies conducted where coffinite is specifically mentioned in the sample is that acid, temperature and oxidant influence the reaction rate and extraction of uranium from coffinite.
4 Conclusions
1) A hydrothermally synthesized coffinite was used in series of acid sulfate leach tests under conditions of relevance to uranium extraction, and complete dissolution of uranium from coffinite was obtained between 36 and 48 h.
2) Tests comparing uranium dissolution from synthetic coffinite, uraninite and brannerite showed that the dissolution of uraninite was significantly faster which dissolved completely in 3 h, while brannerite was leached at a much slower rate reaching a maximum dissolution of only ~18% after 144 h.
3) The U extraction rates of the coffinite, uraninite and brannerite are consistent with previous literatures which suggest that U extraction rate is in the sequence of uraninite>coffinite>brannerite.
4) It is expected that the presence of impurities in natural coffinites would further inhibit leachability.
Acknowledgements
The authors would like to thank Mr. Frank ANTOLASIC (RMIT University) and Assoc. Prof. Johan Du PLESSIS (RMMF, RMIT University) for their assistance in the collection and interpretation of X-ray diffraction and X-ray photoelectron spectroscopy data. The authors would also like to thank Dr. Nick Wilson for assisting in the collection of quantitative EPMA data and for setting up and running the EPMA maps, Aaron TORPY for processing the map data and Cameron DAVIDSON for preparing the polished samples for analysis.
References
[1] PLANT J A, SIMPSON P R, SMITH B, WINDLEY B F. Uranium ore deposits: Products of the radioactive earth [J]. Reviews in Mineralogy and Geochemistry, 1999, 38: 254-319.
[2] WOODY R J, GEORGE D R. Acid leaching of uranium ores [C]//CLEGG J W, FOLEY D D. Uranium Ore Processing 1958. New York: Addison-Wesley Publishing Company Inc., 1958: 115-152.
[3] BURNS P C. The crystal chemistry of uranium [J]. Reviews in Mineralogy and Geochemistry, 1999, 38: 23-90.
[4] ABDELOUAS A. Uranium mill tailings: Geochemistry, mineralogy and environmental impact [J]. Elements, 2006, 2: 335-341.
[5] STIEFF L R, STERN T W, SHERWOOD A M. Preliminary description of coffinite―A new uranium mineral [J]. Science, 1955, 121: 608-609.
[6] STIEFF L R, STERN T W, SHERWOOD A M. Coffinite, a uranous silicate with hydroxyl substitutions: A new mineral [J]. American Mineralogist, 1956, 41: 675-689.
[7] FUCHS L H, HOEKSTRA H R. The preparation and properties of uranium(IV) silicate [J]. American Mineralogist, 1959, 44: 1057-1063.
[8] ABDEL-GAWRAD A M, KERR P F. Urano-organic mineral association [J]. American Mineralogist, 1961, 46: 402-419.
[9] BELOVA L N, TANANAEVA G A, FROLOVA K E. The coffinite problem [J]. Soviet Atomic Energy, 1969, 27: 751-752.
[10] ZIMMER P. Experimental study of the UO2-SiO2-ThO2-H2O system at high temperature and high pressure: Synthesis tests of coffinite (USiO4) [J]. Rapport CREGU, 1981, 81(4): 4-18. (in French)
[11] JANECZEK J, EWING R C. Coffinitization―A mechanism for the alteration of UO2 under reducing conditions [J]. Mater Res Soc Symp Proc, 1992, 257: 497-504.
[12] MERRITT R C. The extractive metallurgy of uranium [M]. Boulder, Colorado: Colorado School of Mines Research Institute, Johnson Publishing Company, 1971.
[13] MACNAUGHTON S J, RING R J, DAY A, COLLIER D E, TAN L K P. Optimisation of the leach conditions for a copper/uranium ore. [C]//Proceeding of Sessions and Symposia Minerals, Metals and Materials Society. San Diego, 1999: 509-522.
[14] NGUYEN C. Geochemistry of uranium deposites: Analogues to the Natural Environment [R]. 2003: 259. (in French)
[15] MULAK J. Crystal field parameters in USiO4 from temperature dependence of paramagnetic susceptibility [J]. Journal of Solid State Chemistry, 1977, 21: 117-126.
[16] UZIEMBLO NH, THOMAS L E, SCHOENLEIN L H, MASTEL B, JENSON E D. Solids characterization from hydrothermal tests with spent fuel [C]//Scientific Basis for Nuclear Waste Management X. Boston, Massachusetts, 1986: 161-171.
[17] AMME M, RENKER B, SCHMID B, FETH M P, BERTAGNOLLI H, DOBELIN W J. Raman microspectrometric identification of corrosion products formed on UO2 nuclear fuel during leaching experiments [J]. Journal of Nuclear Materials, 2002, 306: 202-212.
[18] POINTEAU V, DEDITIUS A P, MISERQUE F, RENOCK D, BECKER U, ZHANG J, CLAVIER N, DACHEUX N, POINSSOT C, EWING R C. Synthesis and characterization of coffinite [J]. Journal of Nuclear Materials, 2009, 393: 449-458.
[19] LABS S, HENNIG C, WEISS S, CURTIUS H, ZANKER H, BOSBACH D. Synthesis of coffinite, USiO4, and structural investigations of UxTh(1-x)SiO4 solid solutions [J]. Environmental Science and Technology, 2014, 48: 854-860.
[20] COSTIN D T, MESBAH A, CLAVIER N, DACHEUX N, POINSSOT C, SZENCKNECT S, RAVAUX J. How to explain the difficulties in the coffinite synthesis from the study of uranothorite [J]. Inorganic Chemistry, 2011, 50: 11117-11126.
[21] REYNOLDS H S. Synthesis, characterisation and dissolution studies of the uranium mineral coffinite [D]. Melbourne, Australia: RMIT University, 2013: 206.
[22] KHAN I A, AHUJA H S, BAGNALL K W, SINF L. Uranium (IV) chloride for organometallic synthesis [J]. Inorganic Syntheses, 2007, 21: 187-190.
[23] FUCHS L H, GEBERT E. X-ray studies of synthetic coffinite, thorite and uranothorites [J]. American Mineralogist, 1958, 43: 243-248.
[24] BERA S, SALI S K, SAMPATH S, NARASIMHAN S V, VENUGOPAL V. Oxidation state of uranium: An XPS study of alkali and alkaline earth urinates [J]. Journal of Nuclear Materials, 998, 255: 26-33.
[25] RAM R. An investigation on the dissolution of synthetic and natural uraninite [D]. Melbourne, Australia: RMIT University, 2013: 282.
[26] CHARALAMBOUS F A. Synthesis, characterisation and dissolution of brannerite [D]. Melbourne, Australia: RMIT University, 2013: 196.
[27] RAM R, CHARALAMBOUS F A, M I, TARDIO J, BHARGAVA S K. An investigation on the effects of Fe (Fe3+, Fe2+) and oxidation reduction potential on the dissolution of synthetic uraninite (UO2) [J]. Hydrometallurgy, 2009, 109: 125-130.
[28] BHARGAVA S K, RAM R, POWNCEBY M I, GROCOTT S, RING B, TARDIO J, JONES L. A review of acid leaching of uraninite [J]. Hydrometallurgy, 2015, 151: 10-24.
[29] CHARALAMBOUS F A, RAM R, MCMASTER S, POWNCEBY M I, TARDIO J, BHARGAVA S. Leaching behaviour of natural and heat-treated brannerite-containing uranium ores in sulfate solutions with iron(III) [J]. Minerals Engineering, 2014, 57: 25-35.
[30] GILLIGAN R, NIKOLOSKI A N. The extraction of brannerite―A literature review [J]. Minerals Engineering, 2015, 70: 34-48.
[31] LOTTERING M J, LORENZEN L, PHALA N S, SMIT J T, SCHALWYK G A. Mineralogy and uranium leaching response of low grade South African ores [J]. Minerals Engineering, 2008, 21: 16-22.
[32] LOTTERING M J, LORENZEN L. The development of a diagnostic leaching method for South African low grade uranium ores [C]// Proceedings of the 24th International Mineral Processing Congress. Beijing, China: Science Press, 2008: 2899-2904.
[33] RAM R, CHARALAMBOUS F A, MCMASTER S, POWNCEBY M I, TARDIO J, BHARGAVA S K. The effect of [Fe]TOT on the dissolution of synthetic Pd-doped UO2 and Th-doped UO2 [J]. Minerals Engineering, 2014, 58: 26-38.
[34] HANSLEY P L, FITZPATRICK J J. Compositional and crystallographic data on REE-bearing coffinite from the Grants uranium region, northwestern New Mexico [J]. American Mineralogist, 1989, 74: 263-270.
[35] SMITS G. (U,Th)-bearing silicates in reefs of the Witwatersrand, South Africa [J]. Canadian Mineralogist, 1989, 27: 643-655.
[36] JANECZEK J, EWING R C. Phosphatian coffinite with rare earth elements and Cerich francoisite (Nd) from sandstone beneath a natural fission reactor at Bangombé, Gabon [J]. Mineralogical Magazine, 1996, 60: 665-669.
[37] FAYEK M, JANECZEK J, EWING R C. Mineral chemistry and oxygen isotopic analysis of uraninite, pitchblende, and uranium alteration minerals from the Cigar Lake deposit, Saskatchewan, Canada [J]. Applied Geochemistry, 1997, 12: 549-565.
[38] FINCH R J, MURAKAMI T. Systematics and paragenesis of uranium minerals [J]. Reviews in Mineralogy and Geochemistry, 1999, 38: 91-179.
[39] JENSEN K A, EWING R C. Microtexture and chemistry of "unaltered" uraninite in the Oklo, Okélobondo, and Bangombé Natural Fission Reactors [C]//LOUVAT D, MICHAUD V, von AREVIC H. Proceedings of the 2nd Oklo Phase II Workshop, Nuclear Science and Technology. Vienna: IAEA, 1998: 61-91.
[40] JANECZEK J. Mineralogy and geochemistry of natural fission reactors in Gabon [J]. Reviews in Mineralogy and Geochemistry, 1999, 38: 321-392.
[41] DEDITUS A P, UTSUNOMIYA S, EWING R C. The chemical stability of coffinite, USiO4・nH2O: 0 [42] SMITS G. Uranium-bearing minerals in the Witwatersrand rocks and their behaviour during leaching [C]//Mintek 50 (Proc. Conf. Sandton, 1984). The Council for Mineral Technology. Randburg, South Africa, 1985: 527-538. [43] MALEY M, BURLING S, RING R. The effect of oxidation-reduction potential and ferric concentration on leaching of uranium ores [C]//Proceedings of the Third International Conference on Uranium, 40th Annual Hydrometallurgy Meeting. Saskatoon, Saskatchewan, Canada, 2010: 563-573. H. S. REYNOLDS1, R. RAM2, M. I. POWNCEBY3, Y. YANG3, M. CHEN1,3, J. TARDIO1, L. JONES1, S. K. BHARGAVA1 1. Centre for Advanced Materials and Industrial Chemistry (CAMIC), School of Applied Sciences, RMIT University, GPO Box 2476, Melbourne VIC 3001, Australia; 2. School of Earth Atmosphere and Environment, Monash University, 9 Rainforest Walk, Clayton, Victoria 3800, Australia; 3. CSIRO Mineral Resources, Private Bag 10 Clayton South, Victoria 3169, Australia 摘 要:水热合成并表征水硅铀矿,在此基础上对水硅铀矿在硫酸体系中的浸出过程进行研究,并将结果与其他相关的合成U4+矿物如氧化铀(UO2)和钛铀矿(UTi2O6)在类似条件下的浸出速率和浸出率进行比较。浸出时间对铀溶出影响的研究表明,水硅铀矿在36~48 h被完全浸出,该反应的活化能为38.4 kJ/mol。与水硅铀矿相比,氧化铀矿的溶出速率明显较快,可以在3 h内达到完全溶出(Ea=42~84 kJ/mol)。比较而言,合成钛铀矿的浸出速率较水硅铀矿的明显更低,其144 h铀的最大浸出率只有18%。上述3种矿物溶出速率和提取率的显著区别与文献报道相符合,即矿物的可溶出性为氧化铀>水硅铀矿>钛铀矿。在天然水硅铀矿中,由于杂质的存在,其可溶出性可能会被进一步抑制。 关键词:水硅铀矿;铀;钛铀矿;浸出动力学 (Edited by Bing YANG) H. S. REYNOLDS and R. RAM are co-first authors. Corresponding author: M. CHEN; E-mail: Miao.Chen@rmit.edu.au DOI: 10.1016/S1003-6326(18)64858-7水硅铀矿及其他常见铀矿的铀浸出动力学比较

