
Growth restriction effects during solidification of aluminium alloys
CHEN Zhong-wei(陈忠伟), HE Zhi(何 志), JIE Wan-qi(介万奇)
State Key Laboratory of Solidification Processing, Northwestern Polytechnical University, Xi’an 710072, China
Received 13 August 2008; accepted 28 November 2008
Abstract: The effects of solute elements during solidification on the grain size are very important and can be quantified by the growth-restriction parameter Q, and Q possesses the better correlation with the grain size. Based on the constitutional undercooling generated by the growth of an adjacent grain during the initial solidification, the growth-restriction parameter Q is deduced and a comprehensive physical basis of Q is obtained by using an initial solute distributing equation. For the alloys with more potent nucleants, Q is a suitable predictor of the grain size. For less potent nucleants, the relative grain size(RGS) is a more accurate prediction of the grain size. This prediction coincides with the experimental behaviors for Al-Ti and Al-Cu alloys with lower solute content.
Key words: growth restriction; constitutional undercooling; solidification; aluminium alloys
1 Introduction
The grain refinement of alloys is very important in materials processing. Whether aluminium alloys or other alloys, the desirable microstructure consists of fine equiaxed grains. The grain refinement of aluminium alloys is achieved by the addition of inoculant particles (e.g. TiB2, TiC) in the form of master alloys[1-2]. The nucleation of α(Al) is initiated on the surfaces of these particles at small undercoolings[3-4]. The presence of solute elements causes the melt undercooling, which leads a greater proportion of the particles to initiate nucleation[5-6]. The generation of this constitutional undercooling can be quantified by the growth-restriction parameter Q[7-9],
Q=m1c0(k0-1) (1)
where c0 is the average bulk solute content, ml is the gradient of the liquidus line, and k0 is the partition coefficient. It is the build-up of solute at the S/L interface that restricts the growth of α(Al) grains, and the growth-restriction parameter Q is inversely proportional to the growth rate. As reviewed in Refs.[10-11], Q possesses the better correlation with grain size.
But, recently, XU et al[12] indicated that the relationship between the grain size and Q value is not monotonic in Al-(1.3%-30.1%)Cu alloys. EASTON and
StJOHN[8], and QUESTED et al[13] illustrated that the derivative of the fraction solid fs with respect to undercooling, which is calculated by either the Lever rule or Scheil analysis, is inversely proportional to the growth-restriction parameter Q, and Q is not a more accurate prediction of grain size for the melt of less potent nucleants.
In this work, based on solute distributing during the initial solidification and the assumption that the rate of development of constitutional undercooling significantly affects the final grain size, a comprehensive physical basis of Q is further described. The predictors of the grain size are identified for different alloys with lower solute content.
2 Growth restriction effect during initial solidification
2.1 Definition of growth-restriction parameter Q
Partitioning during solidification leads to an enrichment or depletion of solute in the liquid adjacent to the solid. Solute partitioning during solidification can be described by the partition coefficient k0,
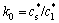 (2)
(2)
where  and
and 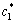 are the equilibrium solute contents of the solid and liquid at the interface (Fig.1). As QUESTED et al[13] described, the degree of partitioning,
are the equilibrium solute contents of the solid and liquid at the interface (Fig.1). As QUESTED et al[13] described, the degree of partitioning,  , is often approximated to c0(1-k0), when ΔTc<< (Tm-Tle) (where ΔTc is the solutal undercooling, Tm is the melting temperature of pure aluminium and Tle is the equilibrium liquidus temperature at the average bulk solute content c0). The partition degree is approximately proportional to solid growth restriction.
, is often approximated to c0(1-k0), when ΔTc<< (Tm-Tle) (where ΔTc is the solutal undercooling, Tm is the melting temperature of pure aluminium and Tle is the equilibrium liquidus temperature at the average bulk solute content c0). The partition degree is approximately proportional to solid growth restriction.
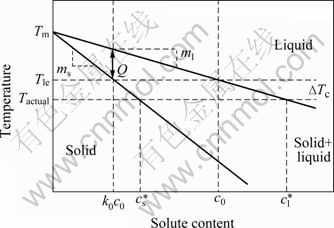
Fig.1 Aluminium-rich portion of idealized binary system[13]
The liquidus gradient ml denotes the change in liquidus temperature with solute content. The gradient in solute content ahead of the solid-liquid interface is proportional to 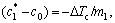 that is, ml is inversely proportional to the driving force for diffusion and is proportional to solid growth restriction. Integrating the above two aspects, the effects of solid growth restriction are parameterized in the factor Q, as shown in Eq.(1) and Fig.1.
that is, ml is inversely proportional to the driving force for diffusion and is proportional to solid growth restriction. Integrating the above two aspects, the effects of solid growth restriction are parameterized in the factor Q, as shown in Eq.(1) and Fig.1.
The growth restriction parameter Q is convenient for the effects of solute partitioning on solid growth-restriction, but it can be valid with two assumptions: 1) the thermal gradient in front of S/L interface is negligible; 2) ΔTc<< (Tm-Tle).
2.2 Growth restriction effect during initial solidification
Assuming that the thermal gradient in front of S/L interface is zero and there is no thermal undercooling and latent heat, the maximum constitutional undercooling ?Tc can be calculated by:
?Tc=Tle-Tactual=Tm+m1c0-(Tm+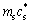 )=
)=
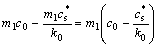 (3)
(3)
where Tactual is the actual temperature of the melt.
In order to meet the assumption (1) in section 2.1, an initial solidification of a single grain is considered and the initial solute distributing equation is[14]
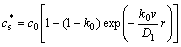 (4)
(4)
where v is the solidification rate of the solid-liquid interface, Dl is the solute diffusion coefficient in the liquid phase and r is the solid growth distance.
Assuming that there are effective nucleants in the solid-liquid interface, then the amount of constitutional undercooling development as initial solidification proceeds can be related to the solid growth distance r by substituting Eq.(4) into Eq.(3):
 (5)
(5)
By using Eq.(5), a physical basis of the growth- restriction parameter Q can be obtained. The initial rate of development of constitutional undercooling is defined by d(?Tc)/dr at r→0, that is,
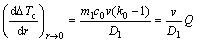 (6)
(6)
Eq.(6) indicates that the initial rate of development of ?Tc is not only proportional to Q but also proportional to v/D1. Using Eq.(6), the growth restriction parameter Q can be obtained:
 (7)
(7)
Eq.(7), which is different from the results of previous literatures [8,13], describes further a comprehensive physical basis of Q.
For estimating the grain size, the relative grain size (RGS) can be defined as being the solid growth distance rn at which the constitutional undercooling ?Tc reaches the undercooling required for effective nucleation ?Tn. Then, using Eq.(5),
RGS=rn=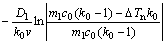 =
=
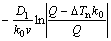 (8)
(8)
For the more potent nucleants, their effective nucleation undercooling ?Tn is smaller. When ΔTn<< Q/k0, which is consistent with assumption (2) in section 2.1, the relative grain size (RGS) in Eq.(8) is substituted for RGS≈(D1?Tn)/(Qv). It shows that, for the grain refinement with the more potent nucleants, Q is a suitable predictor of the grain size. This is the same with the grain refinement in aluminium melts containing TiC or TiB2. However, for the less potent nucleants, in the condition that is not consistent with assumption (2) in section 2.1, the use of RGS is more appropriate than Q in the prediction of the effect of solute on the grain refinement. This can explain that the relationship between the grain size and the Q value is not monotonic in aluminium alloys[12].
This model can not be used when ?Tn>Q/k0, for the logarithm in Eq.(8) can not be negative. Columnar grains will also form if substrates present in the melts require more undercooling than the undercooling parameter Q/k0, as heterogeneous nucleation sites. Therefore, to prevent columnar growth in practice, nucleants with ?Tn less than Q/k0 need to be added to the melts.
3 Results and discussion
3.1 Al-Ti alloys
Fig.2(a) shows the predicted variation of RGS with titanium content of Al-Ti alloy system at various nucleant potencies using Eq.(8). Fig.2(b) is the experimental data showing the effect of solute titanium additions on the grain size for more potent nucleants, e.g. TiB2, and for less potent nucleants where TiB2 is not present[15].
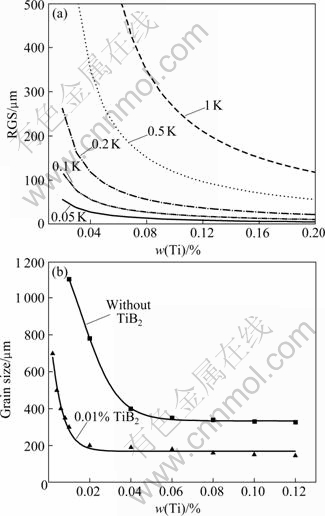
Fig.2 Calculated variation of grain size with titanium content for range of ?Tn in Al-Ti system (a); trendlines of grain size data for titanium additions to pure aluminium with and without TiB2 additions[15] (b)
Fig.2(b) shows that the trend in the grain size with titanium additions is similar to the trend in RGS in Fig.2(a). It was found in the experiments that, after an initial decrease in grain size with the addition of solute titanium, the grain size remained reasonably constant with the further additions of solute. The RGS versus w(Ti) curves in Fig.2(a) for the more potent nucleants (smaller values of ?Tn), also initially decrease rapidly. However, as found experimentally, the addition of more solute does not decrease the grain size much further and the grain size almost appears to be constant for these further additions. The curves for the less potent nucleants (larger values of ?Tn) have a similar behavior but the decrease in grain size occurs more gradually and requires more solute to obtain a small grain size. This is similar to the case where no TiB2 particles are present in Fig.2(b). Therefore, Q is more appropriate to predict the effect of lower solute content on the grain refinement for the more potent nucleants.
3.2 Al-Cu alloys
Fig.3(a) shows the predicted variation of RGS with copper content of the Al-Cu alloy system, at various nucleant potencies using Eq.(8). Fig.3(b) is the experi- mental data showing the effect of solute copper additions on the grain size[12].
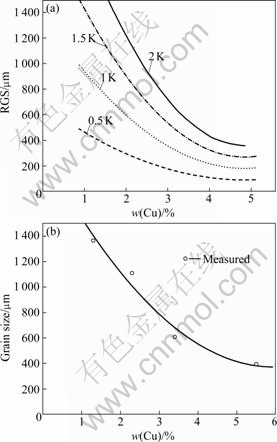
Fig.3 Calculated variation of grain size with copper content for range of ?Tn in Al-Cu system (a); experimental trendlines of grain size data for copper additions to pure aluminium[12] (b)
Fig.3(b) shows that the trend in the grain size with copper additions is similar to the trend in RGS in Fig.3(a). As found experimentally, the RGS versus w(Cu) curves for the less potent nucleants (larger values of ?Tn) have a further similar behavior of the grain size with the additions of solute copper. In fact, the less potent nucleants would be in the melts in Al-Cu alloys (w(Cu) <5.65%)[16]. Therefore, the RGS is more appropriate than Q to predict the effect of lower content solute on the grain refinement, where w(Cu)<5.65% in Al-Cu alloys for no other phase transition. The Q of melts with high solute content or phase transition is beyond the scope of this paper.
4 Conclusions
1) Based on the constitutional undercooling ahead of the solid-liquid interface and by using an initial solute distributing equation, the growth-restriction parameter Q is obtained.
2) For more potent nucleants, Q is a suitable predictor of the grain size. For less potent nucleants,
RGS=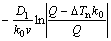 is a more accurate prediction
is a more accurate prediction
of the grain size.
3) This prediction coincides with the experimental behaviors for Al-Ti and Al-Cu alloys with lower solute content.
Acknowledgements
The authors gratefully acknowledge the financial support from the National Basic Research Program of China (No.G2000067202-1). Authors thank Dr. R. P. THACKRAY additional assistance.
References
[1] EASTON M A, StJOHN D H. Improved prediction of the grain size of aluminum alloys that includes the effect of cooling rate [J]. Mater Sci Eng A, 2008, 486: 8-13.
[2] Y?CEL B. Grain refining efficiency of Al-Ti-C alloys [J]. Journal of Alloys and Compounds, 2006, 422: 128-131.
[3] IQBAL N, DIJK N H, HANSEN T, KATGERMAN L, KEARLEY G J. The role of solute titanium and TiB2 particles in the liquid-solid phase transformation of aluminum alloys [J]. Mater Sci Eng A, 2004, 386: 20-26.
[4] JOHNSSON M, B?CKERUD A L, WORTH G K. Study of the mechanism of grain refinement of aluminum after additions of Ti- and B-containing master alloys [J]. Metall Mater Trans A, 1993, 24A: 481-491.
[5] QUESTED T E, GREER A L. Grain refinement of Al alloys: Mechanisms determining as-cast grain size in directional solidification [J]. Acta Mater, 2005, 53: 4643-4653.
[6] MAXWELL I, HELLAWELL A. A simple model for grain refinement during solidification [J]. Acta Metall, 1975, 23: 229-237.
[7] LI Jian-guo, HUANG Min, MA Mo, YE Wei, LIU Dong-yu, SONG Dong-ming, BAI Bing-zhe, FANG Hong-sheng. Performance comparison of AlTiC and AlTiB master alloys in grain refinement of commercial and high purity aluminum [J]. Trans Nonferrous Met Soc China, 2006, 16: 242-253.
[8] EASTON M A, StJOHN D H. A model of grain refinement incorporating alloy constitution and potency of heterogeneous nucleant particles [J]. Acta Mater, 2001, 49: 1867-1878.
[9] GREER A L, BUNN A M, TRONCHE A, EVANS P V, BRISTOW D J. Modelling of inoculation of metallic melts: Application to grain refinement of aluminium by Al-Ti-B [J]. Acta Mater, 2000, 48: 2823-2835.
[10] SIGWORTH G, GUZOWSKI M. Grain reining of hypo-eutectic Al-Si Alloys [J]. AFS Trans, 1985, 93: 907-912.
[11] EASTON M A, StJOHN D H, SWEET L. Light metals [M]. TABEREAUX A T, ed. Warrendale, PA: TMS, 2004: 827-831.
[12] XU H, XU L D, ZHANG S J, HAN Q. Effect of the alloy composition on the grain refinement of aluminum alloys [J]. Scripta Mater, 2006, 54: 2191-2196.
[13] QUESTED T E, DINSDALE A T, GREER A L. Thermodynamic modelling of growth-restriction effects in aluminium alloys [J]. Acta Mater, 2005, 53: 1323-1334.
[14] KURZ W, FISHER D J. Fundamentals of solidification [M]. Switzerland: Trans Tech Publications, 1984: 125.
[15] EASTON M A, StJOHN D H. Grain refinement of aluminum alloys (Part II): Confirmation of, and a mechanism for, the solute paradigm [J]. Metall Mater Trans A, 1999, 30A: 1625-1633.
[16] JOHNSSON M, B?CKERUD L. The influence of composition on equiaxed crystal growth mechanisms and grain size in Al alloys [J]. Z Metallk, 1996, 87: 216-226.
Foundation item: Project(G2000067202-1) supported by the National Basic Research Program of China
Corresponding author: CHEN Zhong-wei; Tel: +86-29-88460445; E-mail: chzw@nwpu.edu.cn
DOI: 10.1016/S1003-6326(08)60287-3
(Edited by YUAN Sai-qian)