Electrochemical characteristics of electroplating and impregnation Ni-P/SiC/PTFE composite coating on 316L stainless steel
��Դ�ڿ������ϴ�ѧѧ��(Ӣ�İ�)2020���12��
�������ߣ��ⰲ�� ��ƽƽ ������ ��С�� ������ ���� �ܺ��� ��С�� л־��
����ҳ�룺3615 - 3624
Key words��316L stainless steel; electroplate; Ni-P; PTFE; SiC; corrosion resistance
Abstract: Ni-P/SiC/PTFE coating was obtained on the surface of 316L stainless steel by electrodeposition of Ni-P/SiC coating and immersion of PTFE (polytetrafluoroethylene). The surface morphology and composition were analyzed by scanning electron microscope and energy dispersive spectrometer. The corrosion resistance of the coating in 0.5 mol/L H2SO4+2��10-6 HF solution was studied by electrochemical method. Surface contact angle was used to test the hydrophobic properties of the coating. The results indicated that the Ni-P/SiC/PTFE coating prepared on the surface of stainless steel was uniform and compact, which significantly improved the self-corrosion potential of stainless steel. The self-corrosion current density decreased from 7.62 to 0.008 ��A/cm2. The durability performance of coating was tested under 0.6 V voltage and the stable corrosion current density value was 0.19 ��A/cm2, then wetting angle was tested after durability experiment and the value is 134.5 ��.
Cite this article as: GAO Ping-ping, GAO Mei-lian, WU An-ru, WU Xiao-bo, LIU Chun-xun, ZHANG Yang, ZHOU Hai-kun, PENG Xiao-min, XIE Zhi-yong. Electrochemical characteristics of electroplating and impregnation Ni-P/SiC/PTFE composite coating on 316L stainless steel [J]. Journal of Central South University, 2020, 27(12): 3615-3624. DOI: https://doi.org/10.1007/s11771-020-4508-6.

J. Cent. South Univ. (2020) 27: 3615-3624
DOI: https://doi.org/10.1007/s11771-020-4508-6
GAO Ping-ping(��ƽƽ)1, 2, 3, GAO Mei-lian(������)1, 2, WU An-ru(�ⰲ��)1, WU Xiao-bo(��С��)2,
LIU Chun-xun(������)3, ZHANG Yang(����)3, ZHOU Hai-kun(�ܺ���)1,
PENG Xiao-min(��С��)1, XIE Zhi-yong(л־��)2
1. Hunan Provincial Key Laboratory of Vehicle and Transmission System,Hunan Institute of Engineering, Xiangtan 411104, China;
2. State Key Laboratory of Powder Metallurgy, Central South University, Changsha 410083, China;
3. Hunan Gold Sky Aluminum Industry High-tech Co., Ltd., Changsha 410205, China
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2020
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2020
Abstract: Ni-P/SiC/PTFE coating was obtained on the surface of 316L stainless steel by electrodeposition of Ni-P/SiC coating and immersion of PTFE (polytetrafluoroethylene). The surface morphology and composition were analyzed by scanning electron microscope and energy dispersive spectrometer. The corrosion resistance of the coating in 0.5 mol/L H2SO4+2��10-6 HF solution was studied by electrochemical method. Surface contact angle was used to test the hydrophobic properties of the coating. The results indicated that the Ni-P/SiC/PTFE coating prepared on the surface of stainless steel was uniform and compact, which significantly improved the self-corrosion potential of stainless steel. The self-corrosion current density decreased from 7.62 to 0.008 ��A/cm2. The durability performance of coating was tested under 0.6 V voltage and the stable corrosion current density value was 0.19 ��A/cm2, then wetting angle was tested after durability experiment and the value is 134.5 ��.
Key words: 316L stainless steel; electroplate; Ni-P; PTFE; SiC; corrosion resistance
Cite this article as: GAO Ping-ping, GAO Mei-lian, WU An-ru, WU Xiao-bo, LIU Chun-xun, ZHANG Yang, ZHOU Hai-kun, PENG Xiao-min, XIE Zhi-yong. Electrochemical characteristics of electroplating and impregnation Ni-P/SiC/PTFE composite coating on 316L stainless steel [J]. Journal of Central South University, 2020, 27(12): 3615-3624. DOI: https://doi.org/10.1007/s11771-020-4508-6.
1 Introduction
Due to the excellent corrosion resistance of 316L stainless steel (316LSS), this ultra-low carbon austenitic stainless steel has been extensively applied in petroleum engineering, chemical engineering, bioengineering, etc [1]. The growing development in the contemporary industries puts forward higher requests on materials (e.g., filter screen, ship products, fuel cell bipolar plates, extreme low-altitude aircraft) [2, 3]. These applications need materials with better corrosion protection, abrasive resistance and self-lubrication, hence 316LSS has been widely employed, and traditional surface modifications such as physical vapor deposition (PVD) [4], surface cladding, chemical vapor deposition (CVD) [5], chemical coating, electrochemical coating [6] are normally required to further improve the corrosion resistance of the corresponding components. PVD is one of the most frequently used techniques to obtain dense anti-corrosion coating, where TiN [7], TiAlN [8] and ZrC [9] are the typical coating materials. However, this technique has problems such as high equipment cost, complicate parameter control and product shape limitation, which largely restrict its popularization and the applications in the coating of complex-shaped components. Surface cladding is a technique using high temperature to spray melted metal powders onto the components. Its high heat input will yield cracking of coating and oxidation of substrate material. CVD tends to exhibit similar issues, where the high reaction temperature, low fabrication efficiency and recrystallization could embrittle the substrate material.
For fighter missile suspension position wear-resistant components, all special-shaped regions are normally surface-treated by thermal spraying and PVD (magnetron sputtering). In practical production, insufficient coverage in these techniques demonstrates that there is still a room for improving the coating techniques. And the coating material should be capable of withstanding the corrosion from seawater, acid rain and other corrosive environment, while maintaining a good hardness, abrasive resistance and self-lubrication. Electrodeposition is a cheap and highly-efficient coating technique that could patch up the issue of insufficient coverage during preparing the coatings on complex-shaped components. The simplicity and low production input make this technique suitable for the large-scale industrial manufacturing and coating repairing at normal temperature. LI et al [10] have applied Ni-P/SiC coating on the oil-well pump. Good comprehensive coating performances were obtained due to the high hardness and good abrasive resistance of this composite coating material. ZHANG et al [11] have successfully synthesized the Ni-P/SiC coating on tin alloys and characterized its hydrophobicity, where the optimal hydrophobicity (contact angle) was measured to be 113��. Using composite co-deposition, YING et al [12] have prepared Ni-P/SiC/WS2 on stainless steel, whose micro-hardness, corrosion resistance, abrasive resistance and self-lubrication performances were improved to a large extent. Based on the previous works, this work utilizes the electrodeposition to synthesize Ni-P/SiC coating on 316LSS. The as-received samples were further impregnated with PTFE emulsion to make it more suitable for coating applications in harsh environment requiring better corrosion resistance, abrasive resistance, self-lubrication and service life.
2 Experimental
The flow chart of sample preparation was schematically illustrated in Figure 1. In this work, 316L plates (substrates) with dimensions of 2 mm��2 mm��1 mm were surface-treated as following: as-polished 316L plate and graphite electrode were installed as cathode and anode respectively in electrolyte containing 6% HClO4 and 95% (CH2OH)2. The working voltage was set to 20 V, and then anodizing was implemented at -10 ��C for 10 min to prepare porous surface. After this, Ni-P plating solution (20 g/L NiSO4��5H2O, 30 g/L NaCl, 20 g/L sodium hypophosphite, 30 g/L sodium citrate, pH ~8) was ultrasonically mixed with SiC nanoparticles with size of 20-50 nm (Xiangtan Jinbo New Material Co. Ltd.). The as-anodized samples were electroplated in this plating solution to form Ni-P/SiC composite coating (current density: 3 A/dm2). Eventually, the electroplated samples were impregnated with 20 wt% PTFE, which was followed by a heat treatment at 350 ��C for 2 h to obtain final samples.
SEM, EDS and FTIR (Fourier transform infrared spectroscopy) were employed to characterize the surface morphology, elemental distribution and surface functional groups, respectively. DSC (differential scanning calorimetry) and TG (thermogravimetry) were used to determine the heat treatment temperature of PTFE coating. 3-electrode systems (reference electrode-calomel electrode, counter electrode-Pt plate, working electrode-samples) were connected to an electrochemical workstation (Chenhua Shanghai, CHI660e) to test electrochemical performances, during which the exposed sample surface was fixed at 1 cm��1 cm by sealing with epoxy. The electrolyte with composition of 0.5 mol/L H2SO4+2��10-6 HF was used to simulate strong acidic environment at high temperature. All the samples were soaked and wetted with this solution for 30 min before testing the electrochemical performances. After the open-circuit voltage becomes constant, the plates with coatings are used as working electrode at a sweep rate of 1 mV/s and voltage ranging from -0.8 V to 0.8 V. The temperature of the whole test was 70 ��C. Potentiostatic polarization curve was measured in the same apparatus at 0.6 V for 5 h.
3 Results and analysis
3.1 Coating morphology
Figure 2(a) presents the SEM surface morphology of pretreated 316L samples. Uniform-distributed nanosized pores with diameter of 20 nm was observed on the sample surface. Those nanopores can provide mechanical interlocking sites for the bonding between coating and substrate. Meanwhile, those nanopores can serve as nucleation sites of Ni-P/SiC composite coating, which can facilitate the nucleation of Ni-P particles as compared to its nucleation at smooth sample surface. The surface morphology of electroplated samples is shown in Figure 2(b), which displays a non-uniform coating surface containing interstices/bumps. These defects would decrease the corrosion resistance and hydrophobicity of samples. Figures 2(c) and (d) compare the influence of heat treatment on surface morphology of Ni-P/SiC/PTFE. It can be noted that PTFE particles tend to agglomerate after impregnation, which will leave several uncoated regions and hence poor corrosion protection of substrate. After heat treatment, the coating smoothness was improved substantially, where the PTFE flow into those interstices/bumps formed after the electroplating of Ni-P/SiC, becoming a uniform PTFE coating on top surface of the composite coating. The sideview SEM observation (Figure 3) shows that the thickness of the composite coating is 23.2 ��m.
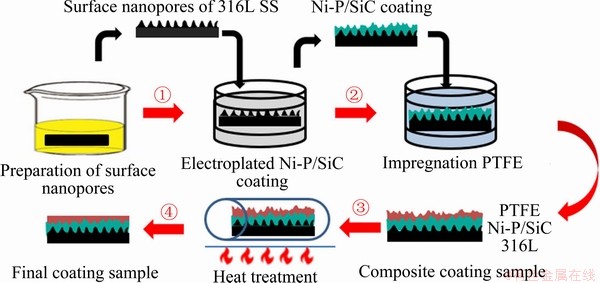
Figure 1 Experimental flow chart of sample preparation

Figure 2 SEM images of 316L stainless steel after surface treatment:

Figure 3 Cross section of 316L stainless steel treated with Ni-P/SiC/PTFE coating
3.2 Coating composition
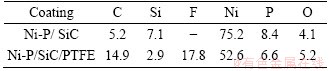
Table 1 compares the composition (EDS mapping) of samples coated with Ni-P/SiC and Ni-P/SiC/PTFE. The composition of Ni-P/SiC sample is C 5.2 wt%, Si 7.1 wt%, Ni 75.2 wt%, P 8.4 wt%, O 4.1 wt%. After impregnated with PTFE emulsion, the ratio of carbon and fluorine was enhanced a lot to 14.9% and 17.8%, respectively. This signifies that PTFE was coated onto the Ni-P/SiC coating, which hampered the detection of signal from Si, Ni and P of the lower surface.
Table 1 EDS analysis results of two coatings (mass fraction, %)
DSC and TG were applied to determine the heat treatment of PTFE coating. It can be seen from Figure 4 that the softening of PTFE initializes at 320 ��C, after which a decomposition at 500 ��C can be observed.The thermal treatment of PTFE coating was designed to be 350 ��C, at which not only the PTFE coating got densified and flattened, but also the stress of electroplate substances in the Ni-P composite coating would experience an alleviation. Additionally, there are C��O, C��H and other functional groups in coating material, which can guarantee the fluidity of PTFE, hence making PTFE fills the Ni-P/SiC coating easily. FTIR result (Figure 4(b)) displays that defects in PTFE molecular chains tends to bond with the oxygen- containing functional groups of Ni-P composite coating. The defective parts of the polymer chain are prone to form new bonds with oxygen- containing the coating surface. The combination of Ni-P/SiC with PTFE macromolecular chain is helpful to improve the binding force of Ni-P/SiC with PTFE [13].
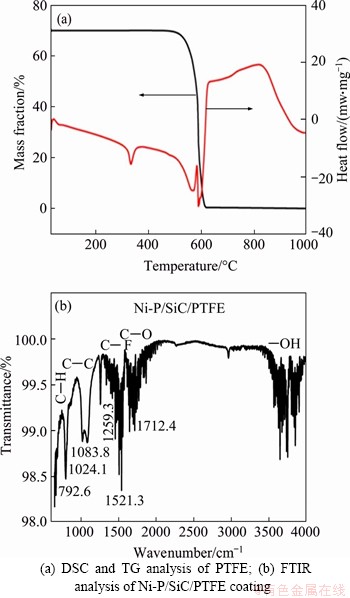
Figure 4 DSC and FTIR analysis of Ni-P/SiC/PTFE coating:
3.3 Coating electrochemical performances
Figure 5 presents the effect of surface modification on Tafel plot of 316L sample. The extrapolation results show that the self-corrosion potential of 316L stainless steel is -0.40 V, and the self-corrosion current density is 39.6 ��A/cm2. After the Ni-P/SiC coating was plated, the self-corrosion potential of the sample increased to -0.38 V, while the corrosion current density decreased to 1.6 ��A/cm2. Further modification by PTFE results in a positive shift of the self-corrosion voltage to 0.13 V, whereas the corrosion current density decreases exponentially to 0.09 ��A/cm2. The kinetic potential polarization curves of the three samples all exhibit four regions of metal passivation, namely cathode region, activation region, passivation region and overpassivation region. Solely using Ni-P/SiC coating to modify 316LSS yields minor improvements on the self-corrosion potential and current density, as well as the stability of the passivation region. After treated with PTFE, the potential of the cathodic protection zone witnessed an increase, and the current density in passivation region is also small. Both the two observations validate that the corrosion resistance of 316LSS can be significantly improved by applying Ni-P/SiC/ PTFE composite coating.
The electroplated Ni-P/SiC coatings are mainly amorphous, and the amorphous structure has good corrosion resistance. Stainless steel corrosion resistance mainly depends on the alloy elements of Ni and Cr, and Ni-base coating also has those performance, so in the test of Ni-P, composite coating showed better performance, but the surface of electroplating coating has holes and other defects, following with the increase of voltage, based on the above reasons, the corrosion rate of the coating significantly accelerated. When hole is populated by modification of PTFE coating and the increased difficulty of electron transfer coating, surface corrosion current decreased, the coating shows excellent corrosion resistance [14, 15].
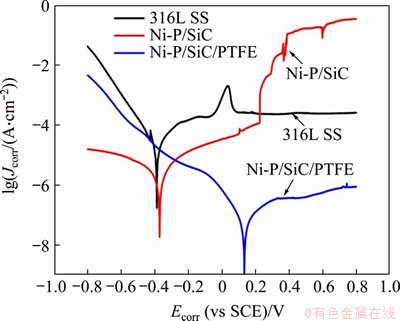
Figure 5 Tafel curves of 316L stainless steel and coating modified 316L stainless steel
Table 2 Self-corrosion current and voltage corresponding to electrodynamic polarization curve
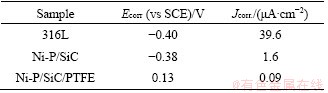
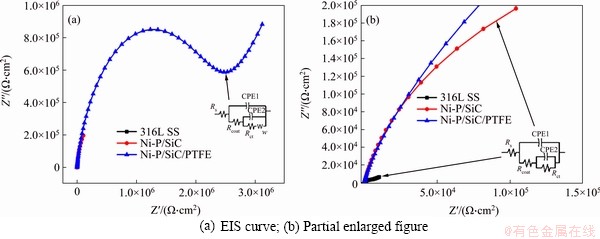
Figure 6 Electrodynamic impedance spectroscopy of 316L stainless steel and coating modified 316L stainless steel:
Table 3 EIS parameter fitted values of 316L stainless steel and coated 316L stainless steel
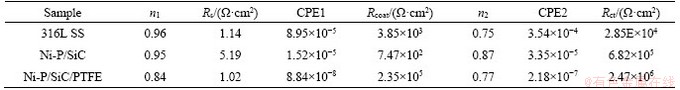
According to the structure of the coating, the appropriate equivalent circuit can be selected to represent coating surface corrosion. The surface of 316LSS is smooth and the thickness of the surface oxide film is about 20 nm. Therefore, its AC impedance spectrum is mainly composed of a single capacitive reactance, whose impedance spectrum generally shows a simple semicircle. After the surface treatment, a new capacitance and resistance are introduced due to the presence of composite coating, thereby the spectrum exhibits a double- capacitance arc ended with Warburg impedance. Among them, Rct is attributed to the charge transfer resistance on the surface of the sample, and the larger value indicates better corrosion resistance of the coating; Rcoat represents the impedance at the intersection of the high frequency curve and the real axis, which is attributed to the internal resistance and proton conduction of the sample. CPE stands for constant phase element. It is generally believed that the larger Rct value indicates higher difficulty in the surface electron transfer and the better corrosion resistance of the coating [16]. The electrochemical parameters obtained from the Nyquist diagram are shown in Table 3. The n values of the Ni-P and Ni-P/TiN coatings are between 0.7 and 0.9, showing that the data fit was reasonable. As shown in Table 3, the comparison of Rct values shows that the interface charge transfer of the Ni-P/SiC/ PTFE coating in the simulated strong acid working environment is the most difficult, whose Rct value is 2.47��106 ����cm2, manifesting again that Ni-P/SiC/ PTFE composite coating can obviously enhance the corrosion resistance of sample.
Potentiostatic polarization curves of 316LSS and surface-modified 316LSS were measured (0.6 V, 70 ��C, 0.5 mol/L H2SO4+2��10-6 HF) to verify the corrosion resistance and durability of the surface- modified sample. Figures 7(a) and (b) show the corresponding potentiostatic polarization curves and enlarged diagram. During the test, all the samples have an initial increase in the interface resistance due to the formation of corrosion products, leading to a rapid decrease in current. This is then followed by a dynamic equilibrium, where the current density remains constant. Specifically, the corrosion current density of 316LSS, Ni-P/SiC, and Ni-P/SiC/PTFE samples approaches 26.2, 23.3 and 0.19 ��A/cm2 after stabilization. The Ni-P/SiC/PTFE- coated sample has the lowest corrosion current density and provides best protection on the substrate (316LSS). Ni-P/SiC electroplating coating generally presents amorphous state, in which the difficulty of electron transfer increases in the corrosion process. Therefore, compared with 316SS at the same voltage, the corrosion current density is smaller and the corrosion resistance is better.
SEM observation of the sample after the potentiostatic test is shown in Figure 8, where the untreated 316LSS (Figure 8(a)) exhibits peeling marks on its surface after potentiostatic experimental test at a voltage of 0.6 V. The surface has been severely corroded, which can also be corroborated by the aforementioned corrosion current density of the coating (large corrosion current density, fast electron transfer speed of the coating, which means metal elements of coating and substrate can be ionized off quickly). On the surface of 316LSS modified by Ni-P/SiC coating, several corrosion cracks can be observed. This is because during the potentstatic test, the defect sites and the composition segregation at the grain boundary are preferentially corroded by the electrolyte. A large number of bubbles were generated on the surface during the corrosion process, and the corrosion products were peeled off leaving these cracks on the surface. The formation of cracks in the coating indicates that the corrosion resistance of the coating can be further improved.
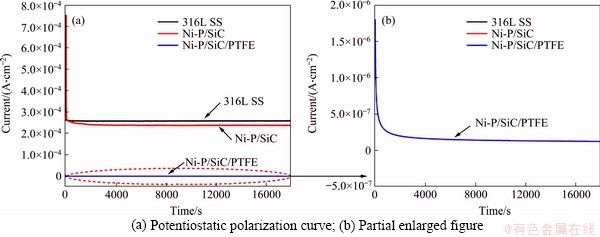
Figure 7 Potentiostatic polarization patterns of composite coatings in 0.5 mol/L H2SO4+2��10-6 HF solution at 70 ��C:

Figure 8 SEM of potentiostatic polarization patterns in 0.5 mol/L H2SO4+2��10-6 HF solution at 70 ��C, 0.6 V:
It can be seen from Figure 8(c) that after subjected to the potentiostatic test, the coating surface of Ni-P/SiC/PTFE coating was flat, where no cracks and peeling corrosion were observed. After adding the SiC nanoparticles into the electroplated coating, the amorphous phase in the coating structure can be enhanced to make ionization of the coating atoms become difficult. The corrosion resistance hence improves because the electron transfer difficulty increases. By filling the pores with PTFE and covering the entire surface of the electroplated coating, the coating can effectively obtain the protection from PTFE. And since the C-F bond is highly stable, it can resist the occurrence of electrochemical corrosion and further avoid pitting and intergranular corrosion of the coating. Hence, it outperforms other samples in corrosion resistance.
3.4 Hydrophobic properties of coatings
Figure 9 shows the coating contact angle after the potentiostatic test. It can be observed that with the addition of PTFE, the hydrophobicity of the coating is improved. Fluorine has a strong polarity and a small atomic radius, while electrons are mainly biased toward fluorine, forming a stable chemical bond performance with carbon atoms [17, 18]. Water is a polar substance that can form hydrogen bonds inside, giving it many unique properties [19, 20]. The surface free energy of PTFE molecules is low, and PTFE molecules show very weak polarity, so it is difficult to form hydrogen bonds with water. The insulating properties of PTFE prevent the conversion of electrons on the coating surface and provide the best hydrophobicity [16]. When PTFE is added to the coating, the coating has the best hydrophobicity, whose wetting angle is 138.1��. After the heat treatment, the Ni-P/SiC/PTFE exhibited the largest wetting angle because the surface morphology and structure were not damaged in the corrosive environment (shown in Figure 9(c)). After completing the potentiostatic test, the surface of 316LSS is severely corroded. There is a certain amount of corrosion peeling, which leads to a decrease in surface flatness and a decrease in hydrophobicity [21, 22]. Its contact angle decreases from 84.3�� to 73.1��, demonstrating the presence of hydrophilic corrosion products. Similarly, for sample treated with Ni-P/SiC, its corrosion occurs preferentially in the weak position of corrosion resistance, which causes the occurrence of corrosion cracks, leading a decrease in contact angle from 115.8�� to 109.4��. The corrosion rate of Ni-P/SiC/PTFE coating after electrochemical test is slow, and the sample still maintains a high wetting angle (134.5��) after the test. Compared with traditional metal coatings by magnetron sputtering coating (such as Ag and Cr-N) [23, 24], this composite has great performance advantages. Hydrophobic surfaces can repel corrosive acids and alkaline solutions and the absence of aqueous electrolyte on the coating surface can reduce the probability of galvanic reactions.

Figure 9 Surface contact angle:
4 Conclusions
Ni-P/SiC/PTFE impregnated 316L stainless steel was obtained by two steps of electroplating Ni-P/SiC and impregnated PTFE. The comprehensive performance of the coating was verified in a strong acid solution of 0.5 mol/L H2SO4+2��10-6 HF with air at 70 ��C. The main conclusions are as follows:
1) The thickness of the Ni-P/SiC/PTFE coating prepared by the two-stage process of electroplating Ni-P/SiC and impregnated composite PTFE reaches 23.2 ��m. The surface morphology of the coating is smooth after heat treatment (350 ��C, 2 h) because the defects such as Ni-P/SiC pores are filled by PTFE particles.
2) Compared with Ni-P/SiC modified layer, the corrosion resistance of Ni-P/SiC/PTFE modified 316L stainless steel has been significantly improved. The self-corrosion current density (in simulated strong acid and high temperature working environment) has been changed from 39.6 ��A/cm2 to 0.09 ��A/cm2, and potentiostatic test for corrosion durability showed that the corrosion current density of Ni-P/SiC/PTFE coating approached 0.19 ��A/cm2 after working for 5 h.
3) The Ni-P/SiC/PTFE modified layer has a uniform and dense multilayer structure, whose contact angle after potentiostatic test is 134.5 ��.
Contributors
GAO Ping-ping provided the concept and edited the draft of manuscript.GAO Mei-lian completed the experiment of the paper; WU An-ru provided directional guidance and financial support for the whole study; WU Xiao-bo, LIU Chun-xun and ZHANG Yang edited the pictures; PENG Xiao-min examined the manuscript several times, and XIE Zhi-yong provided directional guidance.
Conflict of interest
GAO Ping-ping, GAO Mei-lian, WU An-ru, WU Xiao-bo, LIU Chun-xun, ZHANG Yang, ZHOU Hai-kun, PENG Xiao-min and XIE Zhi-yong declare that they have no conflict of interest.
References
[1] WANG Yan, DEREK O. An investigation into polypyrrole- coated 316L stainless steel as a bipolar plate material for PEM fuel cells [J]. Journal of Power Sources, 2006, 163(1): 500-508. DOI: 10.1016/j.jpowsour.2006. 09.048.
[2] YU Lin-ping, JIANG Yao, HE Yue-hui, LIU C T. The corrosion behavior of sinteringmicro-porous Ni-Cu alloy in hydrofluoric acid solution [J]. Journal of Alloys and Compounds, 2015, 638: 7-13. DOI: 10.1016/j.jallcom. 2015. 01.281.
[3] WANG Sheng-li, HOU Ming, ZHAO Qing, JIANG Yong-yi, WANG Zhen, LI Hui-zhe, FU Yu, SHAO Zhi-gang. Ti/(Ti,Cr)N/CrN multilayer coated 316L stainless steel by arc ion plating as bipolar plates for proton exchange membrane fuel cells [J]. Journal of Energy Chemistry, 2017, 26(1): 168-174. DOI:10.1016/j.jechem.2016.09.004. (in Chinese)
[4] MEHRAN Q M, FAZAL M A, BUSHROA A R, RUBAIEE S. A critical review on physical vapor deposition coatings applied on different engine components [J]. Critical Reviews in Solid State and Materials Sciences, 2017, 43(2): 158-175. DOI: 10.1080/10408436.2017.1320648.
[5] CHAI Bin-bin, XIONG Ji, GUO Zhi-xing, LIU Jun-bo, Ni Lei, XIAO Ya, CHEN Cheng. Structure and high temperature wear characteristics of CVD coating on HEA-bonded cermet [J]. Ceramics International, 2019, 45(15): 19077-19085. DOI: 10.1016/j.ceramint.2019.06.152.
[6] WANG H C, HOU K H, LU C E, GER M D. In situ electrochemical coating mechanism of NASICON-structured AgTi2(PO4)3 for Sodium-Ion Batteries [J]. ACS Appl Mater Interfaces, 2020. DOI: 10.1021/acsami.9b20539.
[7] ZHANG Jie, HE Hao, XIE Yon-neng. Comparative investigation on the tribological performances of TiN, TiCN, and Ti-DLC film-coated stainless steel[J]. JOM, 2019, 71: 4872-4879. DOI: 10.1007/s11837-019-03718-y.
[8] WANG L N D, NIE X, HOUSDEN J, SPAIN E, LEYLAND A, MATTHEWS A. Corrosion properties and contact resistance of TiN, TiAlN and CrN coatings in simulated proton exchange membrane fuel cell environments [J]. Journal of Power Sources, 2010, 195(12): 3814-3821. DOI: 10.1016/j.jpowsour.2009.12.127.
[9] BI Fei-fei, PENG Lin-fa, YI Pei-yun, LAI Xin-min. Multilayered Zr-C/a-C film on stainless steel 316L as bipolar plates for proton exchange membrane fuel cells [J]. Journal of Power Sources, 2016, 314: 58-65. DOI: 10.1016/ j.jpowsour.2016.02.078.
[10] LI Meng-long, ZHOU Rui-fen, MA Chun-yang. Electroless plating Ni-P-SiC coatings on surface of barrel in oil pump [J]. Ordnance Material Science and Engineering, 2017, 40(5): 110-112. DOI: 10.14024/j.cnki.1004-244x.20170909.002. (in Chinese)
[11] ZHANG X Z, WU X L, LIU G W, LUO W Q, GUO Y J, SHAO H C, QIAO G J. Wetting of molten Sn-3.5Ag-0.5Cu on Ni-P(-SiC) coatings deposited on high volume faction SiC/Al composite [J]. Transactions of Nonferrous Metals Society of China, 2018, 28(9): 1784-1792. DOI:10.1016/ S1003-6326(18)64822-8.
[12] YING Li-xia, LIU Ying, LIU Guan-nan, LI Zheng-hui, WANG Gui-xiang. Preparation and properties of electroless plating wear-resistant and antifriction composite coatings Ni-P-SiC-WS2 [J]. Rare Metal Materials and Engineering, 2015, 44(1): 28-31. DOI: 10.1016/S1875- 5372(15)30006-0. (in Chinese)
[13] YANG Piao-piao, WU Xiao-bo, XIE Zhi-yong, WANG Pan, LIU Chun-bo, HUANG Qi-zhong. Durability improving and corrosion-resistance mechanism of graphene oxide modified ultra-thin carbon paper used in PEM fuel cell [J]. Corrosion Science, 2018, 130: 95-102. DOI: 10.1016/j.corsci.2017. 10.025.
[14] HAO Feng, JIANG Zhou-hua, LI Hua-bing, LU Peng-chong, ZHANG Shu-cai, ZHU Hong-chun, ZHANG Bin-bin, ZHANG Tao, XU Da-ke, CHEN Zhi-gang. Influence of nitrogen on corrosion behaviour of high nitrogen martensitic stainless steels manufactured by pressurized metallurgy [J]. Corrosion Science, 2018, 144: 288-300. DOI: 10.1016/ j.corsci.2018.09.002.
[15] HAO Feng, Li Hua-bing, JIANG Zhou-hua, ZHANG Tao, DONG Nan, ZHANG Shu-cai, HAN Pei-de, ZHAO Si, CHEN Zhi-gang. Designing for high corrosion-resistant high nitrogen martensitic stainless steel based on DFT calculation and pressurized metallurgy method [J]. Corrosion Science, 2019, 158: 108081. DOI: 10.1016/j.corsci.2019.07.007.
[16] SANGEETHA S K G, ANTHUVAN J T. Pulse electrodeposition of self-lubricating Ni-W/PTFE nanocomposite coatings on mild steel surface [J]. Applied Surface Science, 2015, 359: 412-419. DOI: 10.1016/ j.apsusc.2015.10.127.
[17] GAO Ping-ping, XIE Zhi-yong, OUYANG Chun, TAO Tao, WU Xiao-bo, HUANG Qi-zhong. Electrochemical characteristics and interfacial contact resistance of Ni-P/TiN/PTFE coatings on Ti bipolar plates [J]. Journal of Solid State Electrochemistry, 2018, 22(7): 1971-1981. DOI: 10.1007/s10008-018-3896-0.
[18] VEERAMASUNENI S D J M J, YAMAUCHI G. Hydrophobicity of ion-plated PTFE coatings [J]. Progress in Organic Coatings, 1997, 31: 265-270. DOI: 10.1016/ S0300-9440(97)00085-4.
[19] CARBONE E A D, BOUCHER N, SFERRAZZA M, RENIERS F. How to increase the hydrophobicity of PTFE surfaces using an r.f. atmospheric-pressure plasma torch [J]. Surface and Interface Analysis, 2010, 42(6, 7): 1014-1018. DOI: 10.1002/sia.3384.
[20] ZHOU Yi, LI Meng-yao, ZHONG Xian, ZHU Zhi-ping, DENG Pan, LIU Hai. Hydrophobic composite coatings with photocatalytic self-cleaning properties by micro/ nanoparticles mixed with fluorocarbon resin [J]. Ceramics International, 2015, 41(4): 5341-5347. DOI: 10.1016/ j.ceramint.2014.12.090.
[21] HASHEMZADEH M R K, ASHRAFIZADEH F, KHORSAND S. Effect of ammonium chloride on microstructure, super-hydrophobicity and corrosion resistance of nickel coatings [J]. Surface & Coatings Technology, 2015, 283: 318-328. DOI: 10.1016/j.surfcoat. 2015.11.008.
[22] FORREST E W E, BUONGIORNO J, HU L W, RUBNER M, COHEN R. Augmentation of nucleate boiling heat transfer and critical heat flux using nanoparticle thin-film coatings [J]. International Journal of Heat and Mass Transfer, 2010, 53(1-3): 58-67. DOI: 10.1016/j.ijheatmasstransfer. 2009. 10.008.
[23] FU Yu, HOU Ming, XU Hong-feng, HOU Zhong-jun, MING Ping-wen, SHAO Zhi-gang, YI Bao-lian. Ag-polytetrafluoroethylene composite coating on stainless steel as bipolar plate of proton exchange membrane fuel cell [J]. Journal of Power Sources, 2008, 182(2): 580-584. DOI: 10.1016/j.jpowsour.2008.04.051.
[24] FU Yu, LIN Guo-qiang, HOU Ming, WU Bo, LI Hong-kai, HAO Li-xing, SHAO Zhi-gang, YI Bao-lian. Optimized Cr-nitride film on 316L stainless steel as proton exchange membrane fuel cell bipolar plate [J]. International Journal of Hydrogen Energy, 2009, 34(1): 453-458. DOI: 10.1016/ j.ijhydene.2008.09.104.
(Edited by HE Yun-bin)
���ĵ���
316L����ֱ���Ni-P/SiC/PTFE����Ϳ��������о�
ժҪ�����õ����Ni-P/SiCͿ����ӽ���PTFE���ι�����316L����ֱ�����Ni-P/SiC/PTFEͿ�㣬Ӧ��ɨ���������(SEM)������(EDS)�ȷ���������ò�ͳɷݣ����õ绯ѧ����������0.5 mol/L H2SO4+2��10-6 HF��Һ�е���ʴ���ܽ������о���ͨ��OCOA��ʪ��ϵͳ����Ϳ��绯ѧ��ʴ��������ˮ���ܡ�����������ڲ���ֱ����Ʊ���Ni-P/SiC/PTFEͿ��������ܣ����������˲���ֵ��Ը�ʴ��λ, ���Ը�ʴ�����ܶ���7.62 ��A/cm2��С��0.008 ��A/cm2��Ϳ����0.6 V��ѹ���;��������죬��ʴ�����ܶ��ȶ�ֵΪ0.19 ��A/cm2�����Ժ�Ϳ�����ʪ��Ϊ134.5�㡣
�ؼ��ʣ�316L����֣��������Ni-P����ʴ�ԣ���ˮ��
Foundation item: Project(2018YFB1502500) supported by the National Key Research and Development Program of China; Project supported by State Key Laboratory of Powder Metallurgy, Central South University, China; Projects(2020JJ5100��2018JJ3101) supported by Natural Science Foundation of Hunan Province, China; Project(51671085) supported by the National Natural Science Foundation of China
Received date: 2020-03-07; Accepted date: 2020-09-01
Corresponding author: WU An-ru, PhD, Professor; Tel: +86-731-58688521, E-mail: war891230@163.com, ORCID: https://orcid.org/ 0000-0003-2180- 8554

