Reduction behaviour of Odisha Sands Complex, India ilmenite-coke composite pellets
��Դ�ڿ������ϴ�ѧѧ��(Ӣ�İ�)2020���6��
�������ߣ�D NAYAK N RAY N DASH S S RATH S K BISWAL
����ҳ�룺1678 - 1690
Key words��ilmenite; composite pellet; pre-oxidation; reduction; metallization
Abstract: Presently, ilmenite concentrates from Odisha Sands Complex at Chhatrapur, India are utilized to produce TiO2 slag by direct smelting in an electric arc furnace. However, the process involves the consumption of excess electrical energy and difficulty in handling the arc furnace due to frothing effects. A more efficient process of pre-reducing the ilmenite before smelting has been proposed in the present communication. In particular, studies have been undertaken on the reduction process of ilmenite-coke composite pellets. The difference in the reduction behaviour of raw ilmenite and ilmenite-coke composite pellets has been established and compared with that of the pre-oxidized raw pellets. The effects of various processing parameters like temperature, residence time, and reductant percentage on the metallization of composite pellets in a static bed have been investigated. Metallization of about 90% has been achieved at 1250 ��C for a reduction period of 360 min with a 4% coke composition. Furthermore, the reduced pellets have been characterized through chemical analysis, optical microscopy, field emission scanning electron microscopy and X-ray diffraction analysis. The reduction behaviour of composite pellets has also been found better than that of pre-oxidized pellets indicating the former to be more efficient.
Cite this article as: D NAYAK, N RAY, N DASH, S S RATH, S K BISWAL. Reduction behaviour of Odisha Sands Complex, India ilmenite-coke composite pellets [J]. Journal of Central South University, 2020, 27(6): 1678-1690. DOI: https://doi.org/10.1007/s11771-020-4399-6.

J. Cent. South Univ. (2020) 27: 1678-1690
DOI: https://doi.org/10.1007/s11771-020-4399-6
D NAYAK, N RAY, N DASH, S S RATH, S K BISWAL
CSIR-Institute of Minerals & Materials Technology, Bhubaneswar-751013, Odisha, India
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2020
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2020
Abstract: Presently, ilmenite concentrates from Odisha Sands Complex at Chhatrapur, India are utilized to produce TiO2 slag by direct smelting in an electric arc furnace. However, the process involves the consumption of excess electrical energy and difficulty in handling the arc furnace due to frothing effects. A more efficient process of pre-reducing the ilmenite before smelting has been proposed in the present communication. In particular, studies have been undertaken on the reduction process of ilmenite-coke composite pellets. The difference in the reduction behaviour of raw ilmenite and ilmenite-coke composite pellets has been established and compared with that of the pre-oxidized raw pellets. The effects of various processing parameters like temperature, residence time, and reductant percentage on the metallization of composite pellets in a static bed have been investigated. Metallization of about 90% has been achieved at 1250 ��C for a reduction period of 360 min with a 4% coke composition. Furthermore, the reduced pellets have been characterized through chemical analysis, optical microscopy, field emission scanning electron microscopy and X-ray diffraction analysis. The reduction behaviour of composite pellets has also been found better than that of pre-oxidized pellets indicating the former to be more efficient.
Key words: ilmenite; composite pellet; pre-oxidation; reduction; metallization
Cite this article as: D NAYAK, N RAY, N DASH, S S RATH, S K BISWAL. Reduction behaviour of Odisha Sands Complex, India ilmenite-coke composite pellets [J]. Journal of Central South University, 2020, 27(6): 1678-1690. DOI: https://doi.org/10.1007/s11771-020-4399-6.
1 Introduction
Ilmenite is a mineral resource of great importance in light of the production of TiO2 rich pigment. The demand for TiO2 pigment, a specialty chemical, is increasing rapidly in the paper, plastics, and pigment industries [1]. India��s ilmenite resources are one of the richest grades in the world. However, the ilmenite from Odisha Sands Complex (OSCOM) at Chhatrapur, Odisha, India, is inferior in grade containing about 50% TiO2 content in comparison to Chavra deposit of Kerala, India. These lower TiO2 ilmenite resources are suitable for TiO2 production by direct smelting as the high iron content provides suitable thermodynamic conditions for smelting. However, the process of direct smelting is energy-intensive, which unnecessarily increases the production cost of TiO2. Therefore, a more energy-efficient route of TiO2 production is highly sought-after. An alternative to direct smelting is to pre-reduce the ilmenite before feeding it to an electric arc furnace, thereby reducing the electricity consumption. The reduction of naturally occurring ilmenite has been studied extensively worldwide by various methods, e.g., thermal reduction, selective chlorination, and selective leaching [2-6]. KUCUKKARAGOZ et al [7] investigated the reduction of natural ilmenite by graphite in which the reduction proceeded through Fe3+ to Fe2+, Fe2+ to Fe and Ti4+ to Ti3+. The carbothermic reduction of ilmenite was examined by FRANCIS et al [8] up to 1200 ��C, and the results showed that up to 99% metallization could be achieved at 1200 ��C for a treatment period of 6 h. In another instance, ilmenite was reduced to Fe and TiN by a carbonitrothermic process. The reduction process progressed through the formation of iron by reducing iron oxide components of ilmenite, followed by reduction of TiO2 to produce TiN [9]. In short, high temperature and time are required for sufficient reduction of iron oxides. Hence, extensive research was undertaken on the enhancement of solid-state reduction of ilmenite concentrates on increasing the efficiency of reduction.
It is now well established that the pre-oxidation process enhances the reduction of iron oxides present in the ilmenite. For example, ZHANG et al [10] reported that pre-oxidation increased the attainable extent of the reduction of iron oxides in ilmenite concentrates. GOU et al [11] found that pre-oxidation at 1000 ��C was beneficial for the carbothermic reduction of Panzhihua ilmenite concentrate, China. However, oxidizing the ilmenite before a reduction not only involves an extra capital cost but also increases the power consumption levels. Researchers have also studied other methods of accelerating the reduction rate of carbothermic reduction like milling, the addition of additives and microwave reduction [12-14]. CHEN et al [15] stated that long milling time exerted a positive effect on lowering temperatures of ilmenite reduction. LEI et al [16] found that ilmenite pellets could be reduced at lower temperature and shorter time in a microwave field than by conventional heating. Though these processes have exciting findings, they consume excess energy and will not be realistic for practical use.
So far as iron ores are concerned, composite pellets are regarded as suitable charge materials to improve productivity as they offer higher reduction rates [17-19]. In particular, composite pellets produced by cold-bonding techniques lead to substantial energy savings since they do not require induration. The required strength is attained by curing at very low temperatures [20, 21]. These composite pellets have been utilized as a part of the ferrous burden in blast furnace (BF) or as feed material for a new generation of ironmaking processes to produce direct reduced iron (DRI) [22]. For instance, processes like FASTMET, FASTMELT and Itmk3 have been commercialized, which involve the direct reduction of composite pellets prepared to iron ore fines and pulverized coal in RHF [23, 24]. Besides, the selection of binder in the composite pellet preparation is an essential step since it determines the final composition of the burden. The main drawback of the binders commonly used in iron ore based composite pellets is the presence of impurities like silica and alumina that are undesirable in iron making [19, 25]. In contrast, organic binders have been found suitable for cold-bonding of composite pellets since they do not add impurities into the pellet [26-28].Although the application of composite pellets in the iron ore reduction process has been well established, their use in ilmenite reduction is still under investigation. Nevertheless, some researchers have reported reduction studies of ilmenite based pellets/briquettes [29-32]. However, these studies have been carried out using a single pellet/briquette, and therefore, the scalability of the reduction process is not discussed. Moreover, the in-depth characterization of the ilmenite composite pellets at different levels of operating parameters depicting the reduction mechanism is yet to be established.
In light of the above findings, the current work would focus on the reduction behaviour of cold- bonded composite pellets made from OSCOM ilmenite concentrate, coke fines, and an organic binder. The reduction behaviour of the ilmenite- coke composite pellets has been compared with that of the raw and pre-oxidized pellets because pre- oxidation has been one of the popular methods of enhancing the ilmenite reduction rate. The effect of these parameters on the reduction rate of ilmenite concentrate has been illustrated. Besides, the mechanism underlying the enrichment of reduction has also been discussed vide several characterization studies.
2 Methods
2.1 Materials
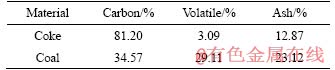
The ilmenite concentrate obtained from OSCOM, Chhatrapur, Odisha, and coke fines and coal were used as raw materials in this study. The chemical composition and mineralogy of the concentrate were characterized by chemical analysis, XRD, and optical microscopy (OM), whereas proximate analysis was carried out for both coke and coal. The results of proximate studies of the coke fines (source of internal carbon) and coal (source of external carbon), which are used as the reductants, are given in Table 1.
Table 1 Proximate analysis of internal coke and external coal (Mass fraction)
2.2 Pelletization
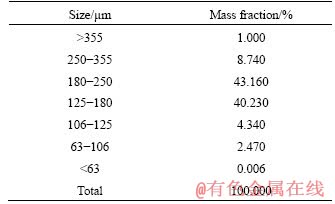
The size analysis of the ilmenite concentrate (Table 2) shows that nearly 84% of the material is in a size range of 125-250 ��m. Hence, the concentrate was milled to below 100 ��m separately in a laboratory-scale ball mill and then mixed with coke fines and sodium lignosulfonate (organic binder) in a high-intensity mixer to produce samples with different coke composition by mass (2%, 4%, 6% and 8%). The binder composition was kept fixed at 3% by mass for all samples. The internal coke composition was limited to 8% by mass as per the inference derived in an earlier report by AGRAWAL et al [33] that the dry strength of pellets decreases with an increase in the amount of carbonaceous materials. Pelletization of the powdered sample was then carried out using a disc pelletizer to make composite pellets of size 5-6 mm. For preparing raw ilmenite pellets, only powdered ilmenite concentrate was used. The raw pellets were oven-dried at 110 ��C for 120 min. The green composite pellets obtained were dried in an ambient atmosphere for 1 d, followed by oven drying at 110 ��C for 120 min.
Table 2 Size analysis of ilmenite concentrate
2.3 Pre-oxidation and reduction
The pre-oxidized ilmenite pellets were prepared by oxidizing the oven-dried raw ilmenite pellets at 1000 ��C in a laboratory-scale muffle furnace under air atmosphere for 1 h to ensure complete oxidation of the samples. The compressive strength of both the pre-oxidized and dried composite pellets was then evaluated using a universal tensile tester. The compressive strength value for each pellet was noted in terms of the maximum load that the pellet could withstand (kg/pellet). For a given pellet type, the average compressive strength of 20 pellets was recorded.
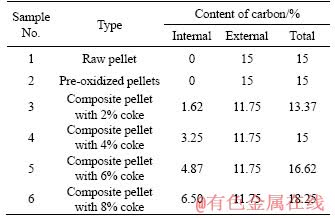
Reduction experiments were carried out in a laboratory-scale muffle furnace. An excess of 50% by mass of carbon to the stoichiometric carbon required for the reduction of iron oxides present in the ilmenite concentrate was partitioned between internal coke and external coal. The carbon distribution in the raw and composite pellets is given in Table 3. A batch of 500 g of raw ilmenite pellets (sample No. 1 in Table 2) was reduced with 217 g of coal (15% by mass of carbon), accounting for an excess of 50% of carbon required stoichiometrically. The same amount of coal was used for the reduction of the pre-oxidized pellets (sample No. 2) as well. In order to compare the reduction results of the raw and pre-oxidized pellets with that of the composite pellets, the same amount of total carbon (15% carbon) was maintained for the reduction of the composite pellets with 4% coke (sample No. 4). Besides, samples No. 3, 5 and 6 (Table 2) were subjected to a reduction to study the effect of the variation in the internal carbon keeping the amount of external carbon (11.75%) the same as that of sample No. 4.
Table 3 Distribution of carbon in different types of ilmenite pellets
The composite pellets were placed as alternative layers with the desired amount of coal in a graphite crucible for the reduction studies. The graphite crucible properly sealed by glass wool and sand was then kept in the furnace. The reduction of the composite pellets of different coke compositions was carried out at varying levels of temperatures of 1250, 1200, 1150 and 1100 ��C at a heating rate of 10 ��C per minute. The residence time at each temperature was varied between 120 and 360 min. On the other hand, the reduction of the raw and pre-oxidized ilmenite pellets was performed at 1200 ��C for the same residence period for the comparison of the results with that of the composite pellets (sample No. 4). The results of metallization under different operating conditions were reported through chemical analysis, as shown in Eq. (1). Metallization (M) is a measure of the conversion of iron oxides into metallic iron during reduction.
M=C(FeM)/C(FeT)��100 (1)
where C(FeM) is the content of metallic Fe, and C(FeT) is the content of total Fe.
The reduced composite pellets were separated from coal by a dry magnetic separator and subjected to XRD, OM and FESEM analyses. All the instruments used for the characterization studies have been calibrated from time to time. The energy dispersive spectroscopy (EDS) attached to the FESEM was calibrated using a universal block of 42 standards.
3 Results and discussion
3.1 Characterization of ilmenite and coke fines
The chemical composition of the as-received OSCOM ilmenite given in Table 4 shows that the sample contains around 50% of TiO2 with 34% FeO suggesting it to be a low-grade resource of ferruginous ilmenite. The XRD analysis of the ilmenite concentrate (Figure 1) indicates that the major components present in the sample are FeTiO3 and Fe2O3. Furthermore, the ilmenite grains are found to be sub-rounded to sub-angular varying in size with complex internal structures, as can be seen from the optical micrographs displayed in Figure 2.
Table 4 Chemical composition of OSCOM ilmenite (mass fraction, %)
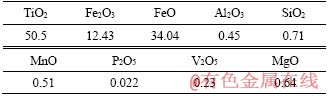
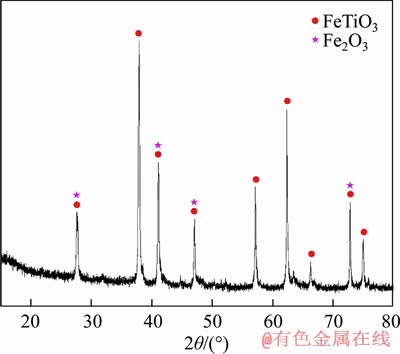
Figure 1 XRD pattern of OSCOM ilmenite concentrates

Figure 2 Optical microscopic images of OSCOM ilmenite concentrate:(I: Ilmenite; H: Hematite)
Also, they have intergrowths and inclusions of other silicate and oxide minerals. The ilmenite grains occur as inclusions within hematite grains, and sometimes vice versa. It is known that the intergrowth or exsolution structures of ilmenite and hematite are very typical as they are isostructural in the rhombohedral series [34].
The particle size distribution of the coke fines used in the pelletization process is shown in Figure 3. It is found that 80% by volume of the particles are below the size of 80 ��m, which meets the requirements for a better pelletization performance. The morphology of the coke, as depicted in Figure 4, shows the distribution of different macerals and pores. During the formation of coke from coal, especially during the softening, the formation of pores occurs in vitrinite, which is shown in Figure 4(a). The size of the pores varies, and in some cases, they are found to be interconnected. The differences in size can be attributed to the various compositions of the parent coals, where the significant difference lies in the scattering of vitrinite and the percentage of inertinite. This coke also produces incipient anisotropic carbon as a minor component as shown in Figure 4(a). Figure 4(b) represents the polished surface of the anisotropic coke showing medium size mosaic texture. The presence of semifusinite infers the spontaneous decomposition of the exinites. It is also revealed from the figure that the shrinkage of the coke due to the emission of volatile matter has not caused any changes in the pores or cell walls of the coke.
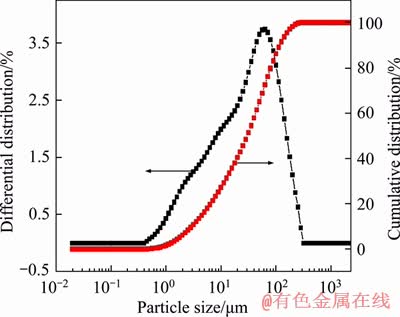
Figure 3 Particle size distribution of coke fines used in pelletization process
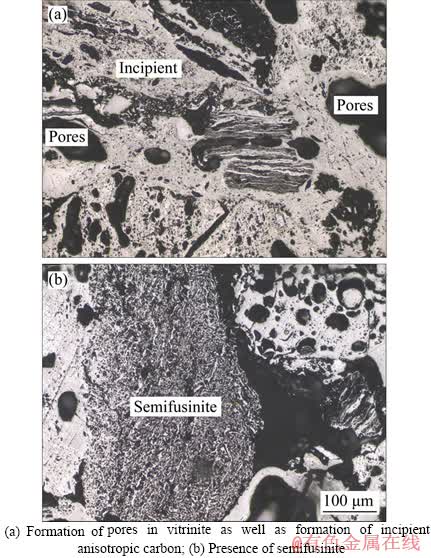
Figure 4 Morphology of coke fines:
3.2 Characterization of pellets
The results from the compression test of different types of pellets are presented in Figure 5. While the pre-oxidized pellets show better strength, the cold compressive strength (CCS) of the composite pellets decreases with an increase in the carbonaceous content. A similar fall of the dry strength of the iron ore-coal composite pellets with the amount of internal coal was also noticed by AGRAWAL et al [33]. In spite of the drop in the CCS values, the developed ilmenite composite pellets exhibit sufficient strength to withstand handling. Except for pellets with 8% internal coke composition, the strength remains within acceptable limits (~10 kg/pellet). However, it is necessary to understand the reduction behaviour of these composite pellets as carbon would play a more vital role in reduction, which has been discussed in the subsequent section.
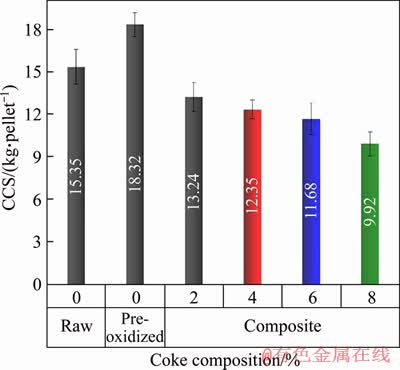
Figure 5 Cold compression strength of different types of ilmenite pellets
3.3 Reduction behaviour of composite pellets
3.3.1 Effect of temperature and time
The reduction experiments of ilmenite-coke composite pellets were performed in the temperature range of 1100-1250 ��C, and the effect of reduction temperature on the degree of metallization of the composite pellets at different coke compositions is presented in Figure 6. It is observed that the reduction temperature has a significant influence on the degree of reduction. At the same reduction time, the degree of reduction increases with an increasing reduction temperature for all coke compositions. The metallization rate increases rapidly in the temperature range of 1150-1250 ��C. For example, in the case of the reduction of the composite pellets having 4% coke composition at 1250 ��C, the reduction degree reaches ~70% in 120 min, and the final degree of reduction after 360 min was ~90%. In contrast, there was only a ~61% reduction at 1100 ��C even after 360 min. It indicates that there is a significant drop in reduction at 1100 ��C. This might be because of an inadequate energy level at this temperature to bring about sufficient reduction. The reduction behaviour is more or less similar to the earlier research report [16].
3.3.2 Effect of coke composition
Attempts were made to understand the effect of coke composition on the degree of reduction at different temperatures while the reduction period was kept fixed at 360 min, and the relevant results are depicted in Figure 7. It is observed that the influence of coke content inside the pellet on the reduction of ilmenite ore is significant. At the same reduction time, the degree of reduction increases with coke composition increasing up to 4% at all reduction temperatures. It is generally agreed that carbon gasification plays a critical role in the carbon-based reduction of iron oxide because it provides the reducing gas (CO) for reduction. Generally, the increase in coke content leads to the increase of contact surface area (between ilmenite ore and coke), which provides more chemically active sites for the reaction of iron oxide with carbon particles increasing the concentration of CO in the muffle furnace. Therefore, the degree of reduction increases with an increase in coke content. The results also indicate that at higher coke content (6%-8%), the degree of reduction has decreased. This may be imputed to the fact that the presence of more carbon could enhance the highly endothermic gasification or the Boudouard reaction that may reduce the overall reaction rate. With the excess presence of internal solid carbon in the pellet, the endothermic nature of the gasification increases resulting in a poor thermal conductivity thereby adversely affecting the reduction process [35].
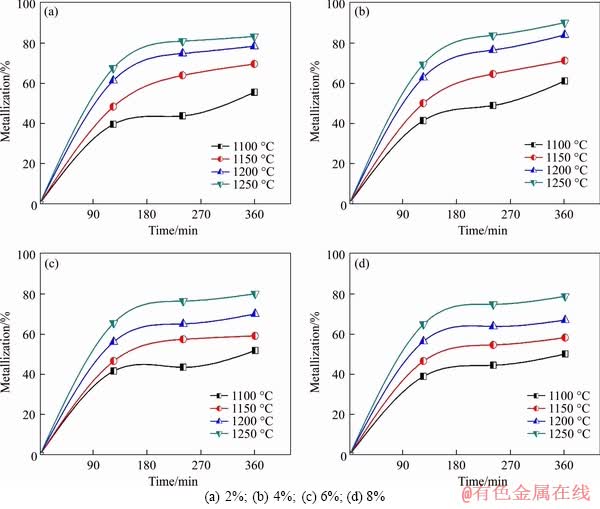
Figure 6 Effect of temperature on metallization of composite pellets at different coke compositions:
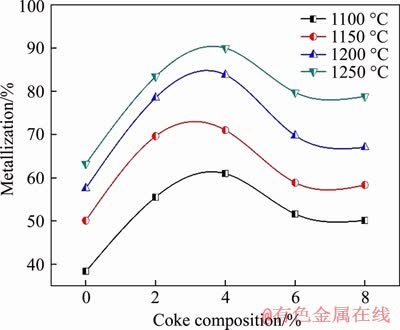
Figure 7 Effect of internal coke addition on metallization of composite pellets reduced at different temperatures for 360 min
3.4 XRD analysis
As per the experimental results discussed so far, the reduction temperature affects the metallization degree significantly. Therefore, XRD studies were carried out for the reduced products at different temperatures and a fixed reduction time of 360 min, and the patterns are shown in Figure 8. The XRD pattern at 1100 ��C, where the metallization degree is 61%, indicates the presence of Fe and FeTi2O5. It is observed that the intensities of both the peaks are getting stronger with an increase in temperature from 1100 to 1250 ��C.This is because FeTiO3 gets converted to FeTi2O5 and Fe at higher temperatures according to the chemical reaction described in Eq. (3), thereby increasing the amounts of Fe and FeTi2O5. Besides, certain amounts of TiO2 are also found at 1100 ��C owing to the conversion of FeTi2O5 to Fe and TiO2 (Eq. (4)). However, as the temperature rises to 1150 ��C, both the peaks of TiO2 and FeTiO3 become weaker, with a higher metallization degree of 71%. This may be due to the formation of suboxides of TiO2, as shown in Eq. (5). However, these were not detected by XRD owing to their minimal presence below the detection limit. At 1200 ��C and 1250 ��C, where the metallization degrees are 84% and 90%, respectively, no new phase appeared, indicating the presence of Fe, TiO2 and FeTi2O5 in the final products.
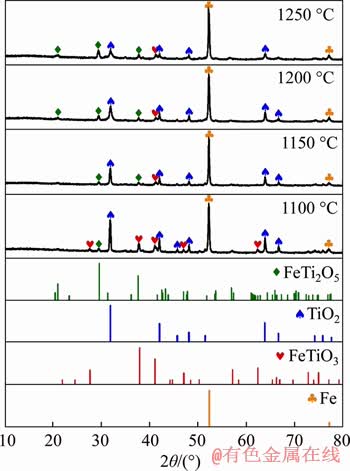
Figure 8 XRD patterns of composite pellets with 4% coke content reduced at various temperatures for 360 min
The XRD patterns of the samples reduced at 1250 ��C as a function of the reduction time are shown in Figure 9. With an increase in time from 120 to 240 min, the peaks of TiO2 and FeTi2O5 become weaker, and those of Fe get stronger. However, the complete reduction of FeTi2O5 couldn��t be easily achieved even after 360 min. This shows that the transformation from FeTi2O5 to Fe is the slowest step in the reduction of the OSCOM ilmenite. Moreover, it can be seen that the intensity of TiO2 increased from 240 to 360 min. At high temperature and time, both the chemical reactions (Eqs. (4) and (5)) co-occur. While TiO2 is being produced from FeTi2O5, it is also being reduced to suboxides like Ti3O5 on the other hand. Given more time for reduction, the transformation of FeTi2O5 to Fe and TiO2 occurs more than the reduction of TiO2. Therefore, a slight increase in the peaks of TiO2 can be found in the XRD pattern at 360 min.
Based on the above XRD analysis and the previous studies, the reduction behaviour of OSCOM ilmenite composite pellets can be suggested by the following reactions [29, 36]:
Fe2O3+3CO��2Fe+3CO2 (1)
2FeTiO3+3CO��FeTi2O5+Fe+CO2 (2)
FeTi2O5+CO��2TiO2+Fe+CO2 (3)
3TiO2+CO��Ti3O5+CO2 (4)
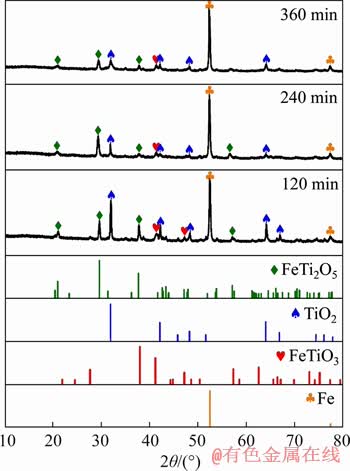
Figure 9 XRD patterns of composite pellets with 4% coke content reduced at 1250 ��C during progress of reduction
3.5 Microstructural analysis
Further studies involving optical microscopy and FESEM were undertaken to substantiate the reduction pathway. The microstructures of the reduced ilmenite samples as analyzed under an optical microscope, shown in Figure 10, display the sequential reduction of ilmenite as a function of temperature. At 1100 ��C, the reductant reacts with the outer layers of ilmenite grains to produce Fe and FeTi2O5. The first-stage formation of the metallic Fe phases can be observed alongside the ilmenite peripheries (Figure 10(a)). At a later stage, when the temperature is increased to 1150 ��C, FeTi2O5 acts as a reactant for the reduction process. As a result, a large amount of metallic iron generated from FeTi2O5 gets dispersed in the sample (Figure 10(b)). With a subsequent rise in temperature to 1200 ��C, the fine grains of metallic iron are found to be uniformly distributed throughout the matrix of the titanium-rich phase (Figure 10(c)). Since the coarsening of the iron particles does not occur even at 1250 ��C, large numbers of fine iron particles are seen within the reduced ilmenite particles (Figure 10(d)). The hindrance in the growth of the iron particles might be due to the presence of titanium oxides and the pores within the structure. GUPTA et al [38], who dealt with the reduction of ilmenite powder, also had a similar observation of iron particle growth at higher temperatures.
In the previous section, the presence of metallic Fe, along with slags comprising of different phases like TiO2, unreacted FeTiO3, and FeTi2O5, was noted in the XRD pattern of the reduced ilmenite. The same phases were also confirmed in the reduced pellet (conditions: 1200 ��C, 240 min, 4% internal coke) by X-ray mapping and EDS analysis. The back-scattered image as given in Figure 11 explains the distribution of iron, titanium and oxygen with the supporting EDS analysis in Table 5. The maximum concentration of iron (~97%) is seen in the white region (point No. 1 in Table 5). The light grey region (points Nos. 3 and 5) in the reduced ilmenite composite pellet has average iron, titanium and oxygen contents of about 58.56%, 14.30% and 27.15%, respectively, and therefore, correspond to FeTi2O5 and unreacted FeTiO3 phases. These are the sites for preferential reduction of oxides to iron, as explained earlier in the optical micrographs. On the other side, the dark grey regions (points No. 2 and 4) consist of 59.66% Ti, 39.56% O and 0.78% Fe, which indicates that they primarily involve titanium oxides.
3.6 Discussion
In the present study, it was found that the reduction rates of composite ilmenite pellets were much higher than that of raw ilmenite, as seen from Figure 12. This is attributed to the fact that in the case of composite pellets, the CO gas that generates inside the composite layers undergoes more interaction with the iron oxides yielding better metallization. The process of reduction of composite ilmenite pellets is also energy efficient as it gives better metallization than the pre-oxidized ilmenite pellets at the same temperature and time. Additionally, the extent of reduction of raw ilmenite is lower than the pre-oxidized ilmenite pellets.
It is well-known that the reduction in pellets with any carbonaceous material inside it follows topo-chemical reactions that proceed from the top surface to the center [39]. However, the composite pellets are better because of the availability of a large number of reaction sites owing to the presence of reductants in situ. Some researchers have also suggested that the topo-chemical reactions start in parallel at several locations within the composite pellet [20, 40]. Besides, many researchers have worked on the reduction kinetics of composite pellets and stated that these are potential materials for enriching the reduction of iron oxides [41-43]. A simple illustration of the microscopic level changes that might have occurred during the reduction of all three types of ilmenite pellets explaining the reduction mechanism of the composite pellets vis-��-vis that of the raw and pre-oxidized pellets is presented in Figure 13.
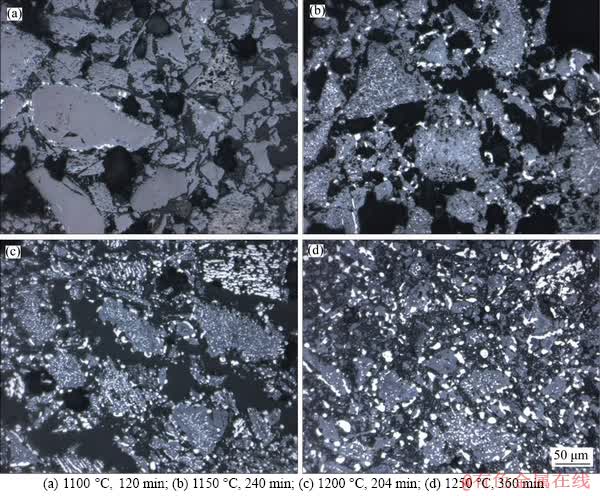
Figure 10 Microstructural changes during reduction of ilmenite composite pellets with 4% internal coke:
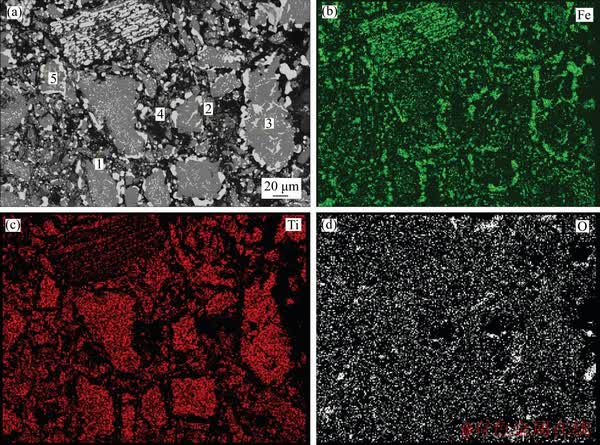
Figure 11 Microstructure (a) and elemental distribution (b, c, d) of reduced composite pellet (1200 ��C, 240 min, 4% internal coke)
Table 5 Elemental composition of reduced composite pellet (EDS analysis, points in Figure 11, at. %)
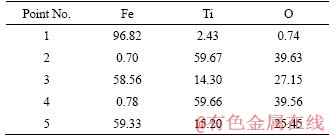
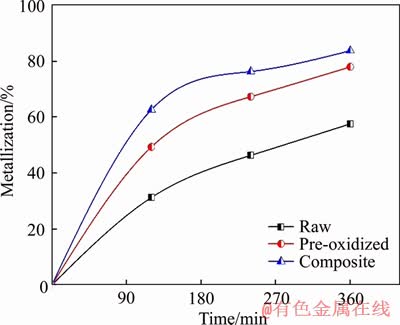
Figure 12 Reduction curves of raw, pre-oxidized and composite ilmenite pellets with 4% internal coke reduced at 1200 ��C
As compared to the reduction of raw and pre-oxidized pellets, the reduction of composite pellets is believed to occur through a series of reactions. It involves the direct reduction of iron oxides (Eq. (6)) as long as the internal carbon, generating the CO gas, which is in close contact with the ilmenite grains. This CO gas reduces the adjacent grains through indirect reduction(Eqs. (2)-(5)) and releases CO2 gas, which is utilized in the gasification reaction (Eq. (7)) to produce CO gas. On the other hand, pre-oxidation treatment induces cracks in the ilmenite grains (Figure 13), which in turn increases the specific surface area and thereby, provides higher metallization than the raw pellets [44].
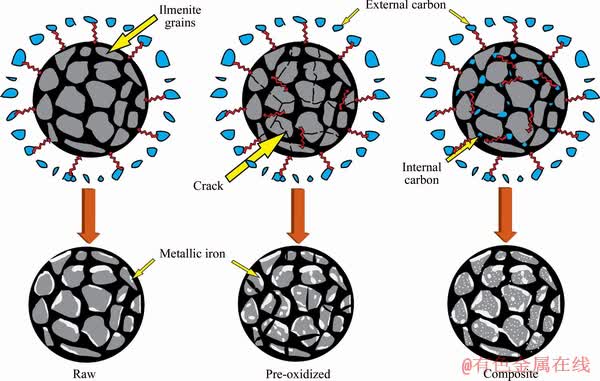
Figure 13 Reduction mechanism of different types of ilmenite pellets
Fe2O3+3C=2Fe+3CO (5)
CO2+C=2CO (6)
4 Conclusions
In India, with issues such as high power tariffs, there is a need for the development of less power-intensive technology for the production of TiO2 slag. The technology can be a potential business model for better utilization of the majority of OSCOM ilmenite produced today. In the context of making the process energy-efficient, the reduction behaviour of ilmenite-coke composite pellets is studied. Experiments for different reduction temperatures, time and coke compositions were carried out to study their effects on metallization. The experimental results led to the following conclusions.
1) All the variables have a pronounced impact on the degree of metallization, while temperature plays the most significant role. The maximum metallization (89.94%) could be obtained at a temperature of 1250 ��C for 360 min with 4% coke composition.
2) XRD and EDS results show that ilmenite is first reduced to Fe, FeTi2O5 and TiO2. FeTi2O5 is then converted to Fe.
3) The morphology of metallic iron particles changes with the reduction temperature. Significant growth of iron particles occurs at higher temperatures (>1200 ��C), resulting in a uniform distribution of the same within the titanium-rich matrix.
4) The extent of reduction for the composite ilmenite pellets is always more significant than that of raw and pre-oxidized ilmenite pellets at the same temperature and time.
Acknowledgements
The authors are thankful to the OSCOM for providing the ilmenite concentrate sample and to Professor Suddhasatwa BASU, the Director of CSIR-Institute of Minerals and Materials Technology for his consent to publish this work.
References
[1] LI W B, YUAN Z F, XU C, PAN Y F, WANG X Q. Effect of temperature on carbothermic reduction of ilmenite [J]. Journal of Iron and Steel Research International, 2005, 12: 1-5.
[2] WANG Yu-ming, YUAN Zhang-fu. Reductive kinetics of the reaction between a natural ilmenite and carbon [J]. International Journal of Mineral Processing, 2006, 81(3): 133-140.
[3] HUANG Run, LIU Peng-sheng, YUE Yue-hui, ZHANG Jin-zhu. Vacuum carbothermic reduction of panzhihua ilmenite concentrate: A thermodynamic study [J]. Mineral Processing and Extractive Metallurgy Review, 2017, 38(3): 193-198.
[4] MAHMOUD M H H, AFIFI A A I, IBRAHIM I A. Reductive leaching of ilmenite ore in hydrochloric acid for preparation of synthetic rutile [J]. Hydrometallurgy, 2004, 73(1): 99-109.
[5] DANG Jie, ZHANG Guo-hua, CHOU Kuo-chih. Kinetics and mechanism of hydrogen reduction of ilmenite powders [J]. Journal of Alloys and Compounds, 2015, 619: 443-451.
[6] LV Wei, LV Xue-ming, LV Xue-wei, XIANG Jun-yi, BAI Chen-guang, SONG Bing. Non-isothermal kinetic studies on the carbothermic reduction of Panzhihua ilmenite concentrate [J]. Mineral Processing and Extractive Metallurgy, 2018, 128(4): 1-9.
[7] KUCUKKARAGOZ C S, ERIC R H. Solid state reduction of a natural ilmenite [J]. Minerals Engineering, 2006, 19(3): 334-337.
[8] FRANCIS A A, EL-MIDANY A A. An assessment of the carbothermic reduction of ilmenite ore by statistical design [J]. Journal of Materials Processing Technology, 2008, 199(1): 279-286.
[9] MUKHERJEE A, RAJE N, KRISHNAMURTHY N. Effect of carbon on the beneficiation of OSCOM ilmenite by carbonitrothermy [J]. Thermochimica Acta, 2014, 575: 206-211.
[10] ZHANG Guang-qing, OSTROVSKI O. Effect of preoxidation and sintering on properties of ilmenite concentrates [J]. International Journal of Mineral Processing, 2002, 64(4): 201-218.
[11] GOU Hai-peng, ZHANG Guo-hua, CHOU Kuo-chih. Influence of pre-oxidation on carbothermic reduction process of ilmenite concentrate [J]. ISIJ International, 2015, 55(5): 928-933.
[12] KELLY R M, ROWSON N A. Microwave reduction of oxidised ilmenite concentrates [J]. Minerals Engineering, 1995, 8(11): 1427-1438.
[13] LIAO Xue-feng, PENG Jin-hui, ZHANG Li-bo, HU Tu, LI Jie. Enhanced carbothermic reduction of ilmenite placer by additional ferrosilicon [J]. Journal of Alloys and Compounds, 2017, 708: 1110-1116.
[14] GUO Sheng-hui, CHEN Guo, PENG Jin-hui, CHEN Jin, LI Dong-bo, LIU Li-jun. Microwave assisted grinding of ilmenite ore [J]. Transactions of Nonferrous Metals Society of China, 2011, 21(9): 2122-2126.
[15] CHEN Min, TANG Ai-tao, XIAO Xuan. Effect of milling time on carbothermic reduction of ilmenite [J]. Transactions of Nonferrous Metals Society of China, 2015, 25(12): 4201-4206.
[16] LEI Ying, LI Yu, PENG Jin-hui, GUO Sheng-hui, LI Wei, ZHANG Li-bo, WAN Run-dong. Carbothermic reduction of panzhihua oxidized ilmenite in a microwave field [J]. ISIJ International, 2011, 51(3): 337-343.
[17] YUUKI M, MEGUMI N, NAOTO T, TOMOHIRO A. Reduction behavior and crushing strength of carbon-containing pellet prepared from COG tar [J]. Fuel Processing Technology, 2016, 142: 287-295.
[18] HUANG Zhu-cheng, ZHONG Rong-hai, YI Ling-yun, JIANG Tao, WEN Liang-ming, LIANG Zhi-kai. Reduction enhancement mechanisms of a low-grade iron ore�Ccoal composite by NaCl [J]. Metallurgical and Materials Transactions B, 2018, 49(1): 411-422.
[19] NIKAI I, GARBERS-CRAIG A M. Use of iron ore fines in cold-bonded self-reducing composite pellets [J]. Mineral Processing and Extractive Metallurgy Review, 2016, 37(1): 42-48.
[20] SAH R, DUTTA S K. Kinetic studies of iron ore-coal composite pellet reduction by TG-DTA [J]. Transactions of the Indian Institute of Metals, 2011, 64(6): 583-591.
[21] MANTOVANI M C, TAKANO C, BUCHLER P M. Electric arc furnace dust-coal composite pellet: Effects of pellet size, dust composition, and additives on swelling and zinc removal [J]. Ironmaking and Steelmaking, 2002, 29(4): 257-265.
[22] YOSHITAKA S, TETSUYA Y, KANJI T, HIROSHI I. New coal-based process to produce high quality DRI for the EAF [J]. ISIJ International, 2001, 41(Suppl): S17-S21.
[23] MATHEISON J G, SOMERVILLE M A, DEEV A, JAHANSHAHI S. 19-Utilization of biomass as an alternative fuel in ironmaking [M]// Iron Ore. UK: Woodhead Publishing, 2015: 581-613.
[24] LU W K. The search for an economical and environmentally friendly ironmaking process [J]. Metallurgical and Materials Transactions B, 2001, 32(5): 757-762.
[25] EISELE T C, KAWATRA S K. A review of binders in iron ore pelletization [J]. Mineral Processing and Extractive Metallurgy Review, 2003, 24(1): 1-90.
[26] CHIZHIKOVA V M, VAINSHTEIN R M, ZORIN S N, ZAINETDINOV T I, ZINYAGIN G A, SHEVCHENKO A A. Production of iron-ore pellets with an organic binder [J]. Metallurgist, 2003, 47(3): 141-146.
[27] de MORAES S L, KAWATRA S K. Laboratory study of an organic binder for pelletization of a magnetite concentrate [J]. Mining, Metallurgy & Exploration, 2010, 27(3): 148-153.
[28] AMMASI A, PAL J. Replacement of bentonite in hematite ore pelletisation using a combination of sodium lignosulphonate and copper smelting slag [J]. Ironmaking & Steelmaking, 2016, 43(3): 203-213.
[29] EL-HUSSINY N A, SHALABI M E H. Studying the pelletization of rosseta ilmenite concentrate with coke breeze using molasses and reduction kinetics of produced pellets at 800-1150 ��C [J]. Science of Sintering, 2012, 44(1): 113-126.
[30] DEWAN M A R, ZHANG Guang-qing, OSTROVSKI O. Phase development in carbothermal reduction of ilmenite concentrates and synthetic rutile [J]. ISIJ International, 2010, 50(5): 647-653.
[31] ZHANG Jian-liang, ZHENG Chang-le, TANG Yun-teng, CHAI Yi-fan. Reduction of Panzhihua titanium-bearing oxidized pellets by CO-N2-H2 gas [J]. Journal of Central South University, 2016, 23(5): 1015-1022.
[32] LV Wei, BAI Chen-guang, LV Xue-wei, HU Kai, LV Xue-ming, XIANG Jun-yi, SONG Bing. Carbothermic reduction of ilmenite concentrate in semi-molten state by adding sodium sulfate [J]. Powder Technology, 2018, 340: 354-361.
[33] AGRAWAL B B, PRASAD K K, SARKAR S B, RAY H S. Cold bonded ore-coal composite pellets for sponge ironmaking. Part 1. Laboratory scale development [J]. Ironmaking & Steelmaking, 2000, 27(6): 421-425.
[34] RAMDOHR P. The ore minerals and their intergrowths [M]. Germany: Pergamon Press, 1969.
[35] AHMED H M, VISWANATHAN N, BJORKMAN B. Composite pellets�CA potential raw material for iron-making [J]. Steel Research International, 2014, 85(3): 293-306.
[36] SHE X F, SUN H Y, DONG X J, XUE Q G, WANG J S. Reduction mechanism of titanomagnetite concentrate by carbon monoxide [J]. Journal of Mining and Metallurgy B: Metallurgy, 2013, 49(3): 263-270.
[37] HU Tu, LV Xue-wei, BAI Chen-guang, LUN Zhi-gang, QIU Gui-bao. Reduction behavior of panzhihua titanomagnetite concentrates with coal [J]. Metallurgical and Materials Transactions B, 2013, 44(2): 252-260.
[38] GUPTA S K, RAJAKUMAR V, GRIEVESON P. The influence of weathering on the reduction of llmenite with carbon [J]. Metallurgical Transactions B, 1989, 20(5): 735-745.
[39] KHATTOI S C, ROY G G. Reduction efficiency of iron ore�Ccoal composite pellets in tunnel kiln for sponge iron production [J]. Transactions of the Indian Institute of Metals, 2015, 68(5): 683-692.
[40] CHOWDHURY G M, MURMU C S, ROY S K, ROY G G. Some studies to establish the reaction mechanism for the reduction of iron ore-graphite composite pellets in a packed bed reactor [J]. Steel Research International, 2010, 81(11): 925-931.
[41] YANG Jian, MORI T, KUWABARA M. Mechanism of carbothermic reduction of hematite in hematite�Ccarbon composite pellets [J]. ISIJ International, 2007, 47(10): 1394-1400.
[42] SUN K, LU W K. Mathematical modeling of the kinetics of carbothermic reduction of iron oxides in ore-coal composite pellets [J]. Metallurgical and Materials Transactions B, 2009, 40(1): 91-103.
[43] COETSEE T, PISTORIUS P C, de VILLIERS E E. Rate-determining steps for reduction in magnetite-coal pellets [J]. Minerals Engineering, 2002, 15(11): 919-929.
[44] WANG Zhen-yang, ZHANG Jian-liang, JIAO Ke-xin, LIU Zheng-jian, BARATI M. Effect of pre-oxidation on the kinetics of reduction of ironsand [J]. Journal of Alloys and Compounds, 2017, 729: 874-883.
(Edited by FANG Jing-hua)
���ĵ���
Odisha Sands Complex��˾����������-��̿�������Ż�ԭ��Ϊ
ժҪ��Ŀǰ��ӡ��Chhatrapur��Odisha Sands Complex��˾ͨ���ڵ绡¯��ֱ����������������ԭTiO2������Ȼ������һ�����漰���������ܵ����ĺ�������ĭЧӦ�����Դ����绡¯�����⡣���������һ�ָ���Ч��������ǰԤ��ԭ������Ĺ��գ��ر��������-��̿�������ŵĻ�ԭ���̽������о���������ԭ�������������-��̿�������Ż�ԭ��Ϊ�IJ��죬����Ԥ������ԭ���ŵĻ�ԭ��Ϊ�����˱Ƚϡ��о����¶ȡ���ԭʱ�䡢��ԭ�������ȹ��ղ����Ծ�̬���ϸ������ŵĽ�������Ӱ�졣��1250 ��C����ԭʱ��Ϊ360 min����̿����Ϊ4%�������£�ʵ����Լ90%�Ľ����������⣬ͨ����ѧ��������ѧ������������ɨ�����������X������������Ի�ԭ���Ž����˱�������������������ŵĻ�ԭ��Ϊ����Ԥ�������ţ�˵��ǰ�߸���Ч��
�ؼ��ʣ������������ţ�Ԥ��������ԭ��������
Foundation item: Project(MLP-52) supported by the Council of Scientific and Industrial Research (CSIR), India
Received date: 2019-10-10; Accepted date: 2020-01-30
Corresponding author: D NAYAK, Master; Tel: +91-98610-78942; E-mail: deepak@immt.res.in; ORCID: 0000-0002-2345-3841

