J. Cent. South Univ. Technol. (2007)01-0078-06
DOI: 10.1007/s11771-007-0016-1
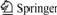
Ship-in-bottle synthesis of NaY zeolite encapsulated Cosalen complex and its catalytic performance for cyclohexane oxidation
YUAN Xia(袁 霞), LI Fang(李 芳), LUO He’an(罗和安)
(College of Chemical Engineering, Xiangtan University, Xiangtan 411105, China)
Abstract: N, N’-bis (salicylidene) ethylenediiminocobalt (Cosalen) was encapsulated into microporous NaY zeolite via the technique of “ship-in-bottle”. The encapsulated complex (Cosalen-NaY) was characterized by Fourier-transform infrared spectrum, ultraviolet-visible spectrum, Brunauer-Emmett-Teller surface areas, X-ray diffraction, thermogravimetry-differential thermal analysis and scanning electron microscope. The reaction of cyclohexane oxidation using oxygen was chosen to investigate the catalytic performance of Cosalen-NaY, and the effects of oxygen pressure, temperature and reaction time were also studied. The results show that Cosalen complex is encapsulated into the supercage of the zeolite and the structure of NaY zeolite remains integrity and the thermal stability of Cosalen is greatly enhanced after encapsulation. Cosalen-NaY shows the better activity in the oxidation of cyclohexane without reductant and solvent. The conversion of cyclohexane is up to 13.4% at 150 ℃ for 3 h under oxygen pressure of 0.85 MPa, with the higher total selectivity to cyclohexanol, cyclohexanone, cyclohexyl hydroperoxide (CHHP) and acid (79.2%) than the neat complex (55.5%). NaY zeolite carrier maybe contributes to the results. There is no obvious induction period to initiate the reaction; furthermore, the amount of CHHP among the products is small, which indicates that the Cosalen-NaY has the strong ability to accelerate the decomposition of CHHP. Recycling tests show that the hybrid material can be used repeatedly with a negligible loss of active sites.
Key words: Cosalen; zeolite; encapsulation; oxygen; cyclohexane; oxidation
1 Introduction
Cytochrome P-450 is an example of biologically important enzyme with iron porphyrin as active center. It is an efficient catalyst for the selective oxidation of hydrocarbon compounds. In the case of enzymes, their high activity and selectivity are connected with the fact that a protein mantle assisting in spatial organization of reactants surrounds the active complex. Therefore, transition metal complexes encapsulated within the pores of zeolites receive special attention as inorganic mimics of enzymes[1-2], the metal complex imitates the role of the active site, and the zeolite host replaces the surrounding protein. Up to now, catalytic oxidation of C―H bonds in saturated hydrocarbons under mild conditions is a key step in the functionalization of many organic compounds and continues to be an important challenge. Molecular oxygen, which is most environmentally benign and economic, would be an ideal ultimate oxidant for oxygenation of hydrocarbons. Cobalt complex of bis (salicylidene) ethylenediamine(Cosalen) is analogue of metalloporphyrin with ability to activate oxygen by the formation of monomeric metaloxygen adducts (Co-O2), which is expected to play an important role in the said field. Cosalen is chosen as catalyst in aerobic oxidation of alcohols, phenolic substrates and benzylic type substrates with the performance of mimic cytochrome P-450[3-7]. While the cobalt schiff base is easily deactivated due to the formation of μ-oxo-dimers and simple dimmers[8], which could be effectively prevented by employing the site isolation effect of polymers or inorganic materials[9-12].
In this study, the three-dimensional micropore NaY zeolite, with the large supercage of 1.2 nm and relatively small open window of 0.7 nm, was chosen as a host framework for encapsulating Cosalen. Heterogenization of Cosalen complexes inside of NaY pores can be achieved via ship-in-bottle technique[13]. It could be prepared from the complex between the Co exchanged NaY zeolite and bis (salicylidene) ethylenediimine (salen) ligand. Salen is an example of a tetradentate ligand, which contains two oxygen and two nitrogen atoms. Its minimum kinetic diameter reaches 0.65 nm, but after the complexation of a metal cation it increases to about 0.9 nm[14]. The size of NaY zeolite supercage enables the synthesis and the accommodation of Cosalen inside the supercage. The final coordination complex is too large and rigid to go out once assembled inside. When confined in the zeolite voids, the Cosalen may lose some of its degrees of freedom and adopt a unique geometry, which is differ from that existing in the solution. The surrounding zeolite framework may also affect the selectivity of catalytic reactions.
In order to prove the encapsulation, the hybrid catalyst Cosalen-NaY was characterized by Fourier- transform infrared (FT-IR) spectrum, ultraviolet-visible (UV-vis) spectrum, Brunauer-Emmett-Teller (BET) surface areas, X-ray diffraction (XRD), thermo- gravimetry-differential thermal analysis (TG-DTA) and scanning electron microscope (SEM). The application of the zeolite encapsulated Cosalen for moderate pressure oxidation of cyclohexane with dioxygen in the absence of reducing agent and solvent for the first time was described.
2 Experimental
2.1 Reagents
The NaY zeolite (molar ratio of SiO2 to Al2O3 is 5.2) was provided by SINOPEC Changling Company, which was dehydrate at 200 ℃ in vacuum for 2 h before use. The salen ligand and cobalt complex were synthesized according to Ref.[15]. The other chemicals used were analytically pure.
2.2 Synthesis of Cosalen-NaY
1.75 g Co(OAc)2?4H2O was dissolved in 350 mL warm distilled water, and then 3.75 g NaY zeolite was added and the contents were refluxed for 24 h under the protection of N2. A pink colored solid obtained was collected by filtration and washed several times with hot water. The cobalt-exchanged zeolite CoNaY was dried in vacuum overnight at 110 ℃ and used for the preparation of Cosalen-NaY.
3 g Salen was dissolved in 114 mL t-butyl alcohol, and then 1 g CoNaY was added and this mixture was stirred under reflux at 80 ℃ for 16 h in nitrogen atmosphere. The resulting slurry was filtered and the solid was Soxhlet extracted for 48 h with t-butyl alcohol, methanol and acetonitrile, respectively. This solid was further refluxed with 50 mL of 1 mol/L NaCl solution for 16 h to replace the uncomplexed Co ions adhering to the outer surface of zeolite by Na+. After this, the solid product was filtered and washed with hot water to remove the adsorbed chloride ions (silver nitrate test). The above product was dried at 110 ℃ for 6 h and used in oxidation experiment.
2.3 Characterization
FT-IR spectra of the solid samples were recorded on a model Spectrom One instrument from PE Co. USA in KBr pellets over the range of 400-4 000 cm-1 under the atmospheric conditions. The electronic spectra of the neat and encapsulated complexes were taken on a PE lambda 25 UV-Vis scanning spectrophotometer. XRD of the samples were carried out using a Rigaku (Model D/Max-2550, Japan), set-up with Cu Kα radiation (40 kV, 300 mA) and a graphite monochromatic with scan speed 8 (?) /min and scanning in the 2θ range from 3?-60?. Silicon was used to calibrate the instrument. The cobalt content of the samples was measured by atomic absorption spectrometer (AAS-Hitachi Model Z-8000, Japan). BET surface area and pore volume of the samples were determined by Tristar 3000 (Micromeritics, USA) at -196 ℃ from nitrogen adsorption. TG-DTA analyses were recorded on Japan Shimadzu instrument (Model BT-40). The SEM of sample was observed on Japan JSM-5600 SEM.
2.4 Catalytic performance
The cyclohexane oxidation experiments were carried out in a 250 mL stainless steel autoclave. In a typical experiment, reaction mixture containing known amounts of cyclohexane and catalyst were placed into the autoclave. After sealing, the reactor was purged by oxygen for several minutes at lowest flow rate, then charged with O2 to the needed pressure and heated to the desired temperature with constant magnetic stirring. When the reaction was stopped, the catalyst was separated by filtration after the reaction mixture was diluted with ethanol to dissolve the by-products. The filtrated catalyst was washed with ethanol three times, dried at 120 ℃ for 6 h and then used for recycling. The products were analyzed by PE Autosystem gas chromatography (GC) and titration. The quantitative analyses of cyclohexanol and cyclohexanone were carried out by GC, which was equipped with AC-10 capillaries of 30 m in length, 0.25 mm in interior diameter and 0.3 μm in film thickness, and the internal standard was chlorobenzene. The concentration of cyclohexyl hydroperoxide (CHHP) was determined by iodometric titration, and the other products acid and ester by acid-base titration.
3 Results and discussion
3.1 Catalyst characterization
3.1.1 Chemical analysis
The cobalt content of the encapsulated complex catalyst, Coslaen-NaY, was estimated by dissolving known amount of the catalyst in concentrated HCl, from these solutions the cobalt contents were estimated using atomic absorption spectrometer (AAS). The Co content of the catalyst is equal to 1.86%(mass fraction).
3.1.2 FT-IR spectroscopy
The FT-IR spectra of Cosalen, NaY and Cosalen- NaY are shown in Fig.1. The IR bands of all encapsulated complexes are weak in comparison with the neat complexes due to their low concentrations in zeolite cages. It can be seen that no absorption band is observed between 1 630 cm-1 and 1 300 cm-1 in the spectrum of NaY, while some bands appear in the spectrum of the extracted Cosalen-NaY. These bands are attributed to the vibrations of C==C and C==N, suggesting the presence of Cosalen. Since the Cosalen-NaY sample underwent a thorough extraction, it is not possible for free salen to be embedded in CoNaY, because uncomplexed salen with a much smaller linear diameter than the pore opening of zeolite Y can be easily removed during the process of extraction. The comparison of the spectra indicates the presence of Cosalen inside the zeolite cages.

Fig.1 FT-IR spectra of samples
1―Cosalen; 2―NaY; 3―Cosalen-NaY
3.1.3 UV-Vis spectroscopy
The UV-Vis spectra of Fig.2 confirm the inclusion of Cosalen complex within the zeolite cages. The free Cosalen complex in HCl solution has the typical absorptions at 260 and 327 nm. After encapsulation, the absorption peak suggested the existence of Cosalen in the cage of zeolite, though the peaks have some shift and are broadened which may be attributable to some distort of Cosalen after encapsulated into NaY zeolite.
3.1.4 Surface area and pore volume analysis
The surface area and micropore volume of NaY zeolite are 756 m2/g and 0.358 mL/g, respectively. The encapsulation of Cosalen complexes reduces the surface area and micropore volume of zeolite to 506 m2/g and 0.288 mL/g. The lowering of the pore volume and surface area supports the fact that Cosalen complexe is present within the zeolite cages and not on the external surface.
3.1.5 XRD analysis
The XRD patterns of NaY and Cosalen-NaY are shown in Fig.3. There is no significant loss in zeolite crystallinity for encapsulated Cosalen-NaY samples as compared with the crystallinity of NaY zeolite. This indicates that complexing and extraction processes have little influence on the structure of the parent NaY materials. Nevertheless, slight modification occurs, as confirmed by the alteration of the relative intensity of the peaks at 220 and 311 reflections (2θ at 10?-12?). Qualitative information about site occupancies can be
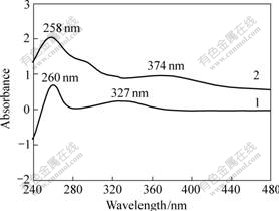
Fig.2 UV-Vis spectra of samples
1―Cosalen; 2―Cosalen-NaY
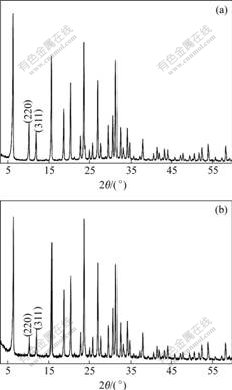
Fig.3 XRD patterns of samples
(a) NaY; (b) Cosalen-NaY
obtained from comparison of the relative intensities of selected XRD lines. It is suggested that a relationship exists between the relative intensities of 311(I311) and 220(I220) XRD peaks and cation location in faujasite-type zeolites[16]. If I220>I311, sodium cations are randomly distributed within the lattice, upon ion exchange with Co2+, and being coordinated to salen, I220 becomes lower than I311. It can be presumed, therefore, that the large molecules of the Cosalen complex displace the sodium cations and locate in the supercages, accompanied by significant cation redistribution.
3.1.6 TG-DTA analysis
The TG-DTA curves of Cosalen and Cosalen-NaY are shown in Fig.4. The endothermic peak observed at below 200 ℃ in the DTA curve of Cosalen-NaY is due to the desorption of physically adsorbed and occluded water, whereas the exothermic peaks at 465.6-602.4 ℃ are ascribed to the combustion of Cosalen complex encaged in the host. As compared to the neat complex, which decomposes beginning at about 283.4 ℃, the thermal stability is greatly enhanced. This gives another evidence for the inclusion of Cosalen in NaY.
Fig.4 TG-DTA curves of samples
(a) Cosalen; (b) Cosalen-NaY
3.1.7 SEM analysis
The SEM image of Cosalen-NaY is show in Fig.5. It can be seen that the crystal shape of NaY zeolites is clear and keeps integrity. It is confirmed that the processes of cooperation and Soxhlet washing during the preparation have no influence on the structure of NaY zeolite.

Fig.5 SEM image of Cosalen-NaY
3.2 Catalytic oxidation of cyclohexane
3.2.1 Catalytic activity of neat and encapsulated complex
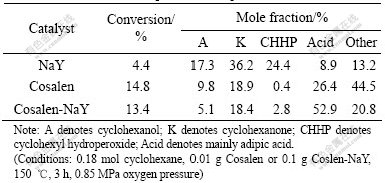
The results of liquid phase oxidation of cyclohexane using molecular oxygen as oxidant, Cosalen-NaY and unsupported Cosalen as catalyst are listed in Table 1. It can be seen that the conversion of cyclohexane is only 4.4% in the blank experiment. The Cosalen-NaY and unsupported Cosalen show nearly equal activity for oxidation reaction. It seems that there is no obvious induction time to start the reaction by activation of dioxygen, which is different from the chain reaction started with the activation of the hydrocarbon molecule by the axial ligand, in the case of other cobalt complexes. And the distributions of CHHP over Cosalen and Cosalen-NaY are much lower than those over NaY and this implies that neat and encapsulated complex have stronger ability to decompose CHHP. However, as regards the selectivity of the reaction products, the molar ratio of cyclohexane to cyclohexanol increases from 1.9 (Cosalen as catalyst) to 3.6(Cosalen-NaY as catalyst). To some extent, Cosalen-NaY favored the formation of acid especially of adipic acid. The total selectivity to cyclohexanol, cyclohexanone, CHHP and acid is 79.2%, much higher than that over Cosalen(55.5%). The carrier may play an important role. Since aluminium rich faujasites have a hydrophilic nature, it is anticipated that polar products cyclohexanol and cyclohexanone will be preferentially adsorbed by the catalyst compared with apolar molecule cyclohexane, therefore, the relative bigger amount of acid is formed among the products.
Table 1 Results of oxidation of cyclohexane over neat and encapsulated complex
3.2.2 Effect of oxygen pressure
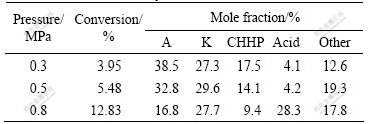
The effect of oxygen pressure on oxidation of cyclohexane with 0.1 g catalyst at 150 ℃ for 3 h is listed in Table 2. It can be seen that the conversion of cyclohexane increases with increasing oxygen pressure as for the solubility of oxygen is improved. At low oxygen pressure, the major products are cyclohexanone, cyclohexanol and CHHP. Selectivities to cyclohexanone and acid increase obviously at relative higher pressure. It indicates that the higher oxygen pressure is in favor of forming cyclohexanone.
Table 2 Effect of oxygen pressure on oxidation of cyclohexane
3.2.3 Effect of temperature
The effect of temperature on the conversion of cyclohexane under 0.85 MPa oxygen pressure is shown in Fig.6. The conversion of cyclohexane increases markedly at above 130 ℃, when the temperature is below 130 ℃, only less CHHP and little cyclohexanol and cyclohexanone are formed and the conversion of cyclohexane remains lower level. So the reaction temperature must be high enough to initiate the reaction; otherwise, the reaction can’t proceed effectively.
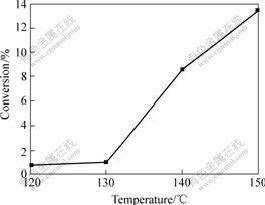
Fig.6 Effect of temperature on conversion of cyclohexane
3.2.4 Effect of time
The cyclohexane conversion and yield of products with increasing time are shown in Fig.7. The products of oxidation have less acid and by-products and give cyclohexanone and cyclohexanol as the major products at the initial reaction time. The conversion of cyclo- hexane increases with increasing time obviously. On the other hand, the amount of cyclohexanol and cyclohexan- one remains little change while the amount of acid increases greatly after the reaction time is over 2 h. The catalyst shows the potential to form the acid in one step from cyclohexane.
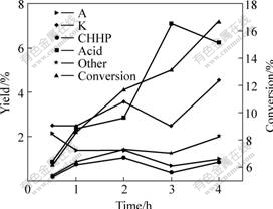
Fig.7 Cyclohexane conversion and yield of products at different reaction time
3.2.5 Reuse of catalyst
The encapsulated catalyst Cosalen-NaY is recovered after oxidation of cyclohexane and reused for three times at the same reaction conditions as shown in. Fig.8. It can be seen that the conversion of cyclohexane decreases about 2.4% after two times recycle and then no obvious loss of activity is observed.
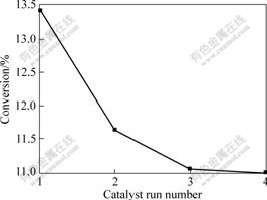
Fig.8 Recycle test of Cosalen-NaY catalyst
4 Conclusions
1) NaY zeolite encapsulated Cosalen complex (Cosalen-NaY) is prepared by the ship-in-bottle technique. Characterizations show that Cosalen is encaged in the supercage of NaY zeolite and the synthesis process has no influence on the structure of the NaY zeolite.
2) Cosalen-NaY exhibits better activity for the oxidation of cyclohexane that utilized dioxygen as the oxidant under moderate pressure and temperature without reductant and solvent. The conversion of cyclohexane is up to 13.4% at 150 ℃ for 3 h at oxygen pressure of 0.85 MPa. Among the products, the amount of CHHP is very small, at the same time, Cosalen-NaY shows the higher total selectivity to cyclohexanol, cyclohexanone, CHHP and acid than the neat complex. NaY zeolite carrier maybe contribute to the results.
3) The encapsulated complexes could be recycled and reused with small changes in catalytic activity.
References
[1] RUDY F P, GUNTER J P, PATRICIA E N, et al. Cyclohexane oxidation with tertiary-butylhydroperoxide catalyzed by iron-phthalocyanines homogeneously and occluded in Y zeolite[J]. Journal of Molecular Catalysis A: Chemical, 1996, 113: 445-454.
[2] HABER J, PAMIN K, POLTOWICA J. Cationic metalloporphyrins and other macrocyclic compounds in zeolite matrix as catalysts for oxidation with dioxygen[J]. Journal of Molecular Catalysis A: Chemical, 2004, 224: 153-159.
[3] EZIO B, SIMONE M, MARCO O, et al. Substituent effects in the cobalt-catalyzed oxidative carbonylation of aromatic amines[J]. Journal of Molecular Catalysis A: Chemical, 1996, 111: 281-287.
[4] EZIO B, LUISELLA B C, CARMEN J, et al. Oxidation of propenoidic phenols catalysed by N,N-ethylenebis (salicylideneiminato) cobalt(II)[Cosalen]: reactivity and spectroscopic studies[J]. Journal of Molecular Catalysis A: Chemical, 1996, 112: 347-351.
[5] KAISA K, HWEIKKI K, MARKKU L, et al. Oxidation of veratyl alcohol by molecular oxygen in aqueous solution catalyzed by cobalt salen-type complexes:the effect of reaction conditions[J]. Journal of Molecular Catalysis A: Chemical, 2003, 203: 9-19.
[6] VISHAL B S, SUMAN L J, KBIR S. Cobalt(Ⅱ)-schiff base catalyzed aerobic oxidation of secondary alcohols to ketones[J]. Journal of Molecular Catalysis A: Chemical, 2004, 212: 55-59.
[7] SUBHABRATA D, PUNNIYAMURTHY T. Cobalt(Ⅱ)-catalyzed oxidation of alcohols into carboxylic acids and ketones with hydrogen peroxide[J]. Tetrahedron Letters, 2003, 44: 6033-6035.
[8] VARKEY S P, RATNASAMY C, RATNASAMY P. Zeolite-encapsulated manganese(III) salen complexes[J]. Journal of Molecular Catalysis A: Chemical, 1998, 135: 295-306.
[9] HU D D, CUI Y L, DONG X L, et al. Studies on Cosalen immobilized onto N-(4-pyridylmethylicene)-chitosan[J]. Reactive & Functional Polymers, 2001, 48: 201-207.
[10] MA Hui-xuan, HU Dao-dao, FANG Yu. Chitosan coordinated Cosalen and its catalytic activity in oxidation of ethanethiol[J]. Journal of Applied Chemistry, 2001, 18 (4) : 290-294. (in Chinese)
[11] TRISSA J, HALLIGUDI S B, SATYANARAYAN C. Oxidation by molecular oxygen using zeolite encapsulated Co(Ⅱ)saloph complexes[J]. Journal of Molecular Catalysis A: Chemical, 2001, 168: 87-97.
[12] TRISSA J, DHANASHREE P S, GOPINATH C S, et al. Zeolite encapsulated ruthenium and cobalt schiff base complexes catalyzed allylic oxidation of a-pinene[J]. Journal of Molecular Catalysis A: Chemical, 2002, 184: 289-299.
[13] KENNETH J, BALKUS J, KHANMAMEDOVA A K, et al. Oxidation catalyzed by zeolite ship-in-a-bottle complexes[J]. Applied Catalysis A: General, 1996, 143: 159-173.
[14] HERRON N. A cobalt oxygen carrier in zeolite Y. A molecular “ship in a bottle”[J]. Inorganic Chemistry, 1986, 25: 4714-4717.
[15] TRISSA J, SAJANIKUMARI C S, DESHPANDE S S, et al. Oxidation of p-cresol catalyzed by neat and zeolite encapsulated cobalt salen complexes[J]. Indian Journal of Chemistry, 1999, 38A: 792-796.
[16] UMEMURA Y, MINAI Y, TOMINAGA T. Structural distortion of 6-coordinated Fe(Ⅱ) complexes in zeolite Y[J]. Journal of Physics Chemistry B, 1999, 103: 647-652.
(Edited by YANG You-ping)
Foundation item: Project ( 04JJ3042 ) supported by the Hunan Provincial Natural Science Foundation of China
Received date: 2006-05-06; Accepted date: 2006-07-13
Corresponding author: LUO He’an, Professor, PhD; Tel: +86-732-8293545; E-mail: hluo@xtu.edu.cn