Fe-Mn/ETS-10�����Ʊ�����CO-SCR����������
��Դ�ڿ����й���ɫ����ѧ��2020���11��
�������ߣ������� �κ��� ���� ��Ӣ
����ҳ�룺2616 - 2627
�ؼ��ʣ�Fe-Mn��������������CO-SCR�����ܣ�ETS-10����ɸ
Key words��Fe-Mn catalyst; catalytic denitrification; CO-SCR catalyst property; ETS-10 Molecular sieve
ժ Ҫ��ͨ�����¹��෨�Ʊ���ͬ��������Fe-Mn/ETS-10����������X����������(XRD)������Ҷ������(IR)��ɨ���������(SEM)��X���߹����������(XPS)�ȶԴ������б����������˲�ͬ��������Mn-Fe/ETS-10��ò���ṹ��ɡ��Լ�ѡ���Դ���ԭ(SCR)���ܵ�Ӱ�졣���������Fe-Mn/ETS-10�����������õľ�һ�Ժͷ�ɢ�ԣ�������ETS-10ԭ����ò�����ͣ�5%Fe-1%Mn/ETS-10���и���Ļ���λ���ߵ��������ܡ�CO-SCR�����ܲ��Խ����ʾ��5%Fe-1%Mn/ETS-10�������¶�300 ���400 ��ʱ�����ʷֱ�ﵽ80%��85%����ѵ�Fe-Mn/ETS-10�������ر�(5%Fe-1%Mn/ETS-10)�������SCR��Ӧ�Ĵ����ԡ�
Abstract: Fe-Mn/ETS-10 catalysts with various compositions were synthesized by high temperature solid state reaction. These catalysts were characterized by X-ray diffractometry(XRD), Fourier transform infrared spectroscopy(IR), scanning electron microscopy(SEM) and X-ray photoelectron spectroscopy(XPS). The effect of different amounts of Fe-Mn/ETS-10 on the morphology, composition and the catalytic reduction performance was studied. The results show that Fe-Mn/ETS-10 has good uniformity and dispersion, the morphology and crystal structure of ETS-10 are kept. 5%Fe-1%Mn/ETS-10 has more active sites and higher deNOx performance. The catalysts activity of CO-SCR results show that denitrification rates of 5%Fe-1%Mn/ETS-10 are 80% and 85% at 300 �� and 400 ��, respectively. So, it is concluded that an optimal Fe-Mn/ETS-10 catalyst support ratio (5%Fe-1%Mn/ETS-10) enhances the catalytic activity in the selective catalytic reduction reaction.
DOI: 10.11817/j.ysxb.1004.0609.2020-35899
������1���κ���1, 2, 3���� ��1���� Ӣ4
(1. ���ϴ�ѧ ұ���뻷��ѧԺ����ɳ 410083��
2. �人�Ƽ���ѧ ���һ���������ұ��Դ��������Ⱦ�����ص�ʵ���ң��人 430081��
3. ���չ�ҵ��ѧ ұ���������Դ�ۺ����ý������ص�ʵ���ң�����ɽ 243002��
4. ���廷���ɷ�����˾����ɳ 410001)
ժ Ҫ��ͨ�����¹��෨�Ʊ���ͬ��������Fe-Mn/ETS-10����������X����������(XRD)������Ҷ������(IR)��ɨ���������(SEM)��X���߹����������(XPS)�ȶԴ������б����������˲�ͬ��������Mn-Fe/ETS-10��ò���ṹ��ɡ��Լ�ѡ���Դ���ԭ(SCR)���ܵ�Ӱ�졣���������Fe-Mn/ETS-10�����������õľ�һ�Ժͷ�ɢ�ԣ�������ETS-10ԭ����ò�����ͣ�5%Fe-1%Mn/ETS-10���и���Ļ���λ���ߵ��������ܡ�CO-SCR�����ܲ��Խ����ʾ��5%Fe-1%Mn/ETS-10�������¶�300 ���400 ��ʱ�����ʷֱ�ﵽ80%��85%����ѵ�Fe-Mn/ETS-10�������ر�(5%Fe-1%Mn/ETS-10)�������SCR��Ӧ�Ĵ����ԡ�
�ؼ��ʣ�Fe-Mn��������������CO-SCR�����ܣ�ETS-10����ɸ
���±�ţ�1004-0609(2020)-11-2616-11���� ��ͼ����ţ�TB333���� ���ױ�־�룺A
��������(NOx)�Ǵ�����Ⱦ����̬�ƻ�����Ҫ��ȾԴ֮һ����������ꡢ������ն����������⡣ѡ���Դ���ԭ����(SCR)������������;������������ԭ����ѡ���Ǻ��ġ�CO��һ�ֵ��͵Ļ�ԭ ��[1-2]�����㷺����ѡ���Դ���ԭNO(CO-SCR)��ͬʱ���㷺����������β������ҵ�����У���ˣ��ɴﵽ�Է��ηϵ�Ŀ��[3]������CO��NOx��SO2[4]֮����Ժܺõ�����ã��Ӷ�������ͬʱ��������[5]��Ŀǰ���о��������������ǽ����������������Ҫ����Pt��Pd��Ir�ȹ�����������Լ�Fe��Ni[6]��Co[7]��Mn��Cu[8-9]��Ce[10]�ȹ��ɽ����������������Դ���ޣ��ɱ����ߣ���չ�ܵ����ơ���ˣ����������ͼۻ����ʹ����ܵ�Խ��Խ��Ĺ�ע��
1989�꣬Engelhard��˾�״α����ϳ��ѹ�����ETS-10(���Ϊ(Na,K)2Si5TiO13��4H2O)�����е��͵��ṹ[11]��ETS-10�����[SiO4]�������[TiO6]��������ɣ�����ͨ������������ETS-10��ά�ṹ�а���5Ԫ����7Ԫ����12Ԫ������ͬ��֮����Ի�ͨ��������Խϴ�(0.76 nm��0.49 nm)���ȱ�����Ϳ�����ϸߣ����ص��ѹǼ���λ��ʹ��������õ����ȶ��Ժ�ѡ����[12]��
ETS-10�Ⱦ����������η�ʯ�����������־��нᾧ�����η���ɸ�ķǼ�������[13]����ˣ��ڴ��� ��[14]�����ӽ���[15]�����[16]�ͷ�����������[17]�������й�����Ӧ��ǰ��������ETS-10������������о����١�BORDIGA��[18]���ú������֤��Cu-ETS-10��NO�ֽ��о��д����ԡ�SULTANA��[19]���ý��շ��Ʊ���Mn/ETS-10����NH3-SCR��SONG��[20]�Ʊ����Ե�Cu-ETS-10�ѹ����δ�������NOx����ѡ���Դ���ԭ��JIN��[21]ͨ����ѹ���ͷ���CeO2������һϵ��Mn-Mo-W-Ox/TiO2-SiO2����������Эͬ��ȥ��CO��NO��C3H8�������о�������ETS-10�����������Ӧ�ü���Ӧ�����д��о���Ȼ������ETS-10��������CO-SCR��ϵ���о����б�����
�о����������̽��������ڷ���ɸ��Al2O3��TiO2��̼���ܵȣ��Ʊ��ɸ����ʹ������������SCR���ԡ�WANG��[22]ͨ�����շ��Ʊ�һϵ������������ͬ��Mn-Fe/ZSM-5���������������Դ�����NH3-SCR���ԣ�����ͨ��������������ǿ�˿���SCR��Ӧ��NO���������еĴ����ԡ�ZHOU��[23]ѡ��Beta����ɸ��Ϊ���壬�Ʊ�Fe-Mn/Beta�������о����ڸ���������C3H6-SCR���ܣ�350 ����߿ɴ�99.4%�����ʡ�WANG��[24]�Ʊ���һϵ��Fe-Mn/Al2O3�����������˵���ѡ���Դ��о�(NH3-SCR)���о���Fe-Mn������NOת���ʺʹ���ʧ���Ӱ�죬����Թ�������������������ߵķ�ɢ�ȡ����õ�������ԭ���ܺͱ�����ȡ�����ı������������͵Ľ���ܡ�HUANG��[25]�Ʊ��˷���״Ho���Ե�Fe-Mn/TiO2�������о��˽ṹ�����ȶԳ��ʹ���NH3-SCR���ܵ�Ӱ�졣ZHANG��[26]����������ԭ���������Ʊ�����NH3-SCR��ϵ��Mn-FeOx/CNTs�����������߱��Ǿ�̬�ṹ������̬�ȶ��ԣ���ѧ�������������ߣ���ԭ��ǿ����32000 h-1�ĸ߿�������140~180 ����ֳ�80%~10%�������ʡ�Fe-Mn�������ֳ��ߵĴ�����[27]����ˮ������[28]��
��ˣ�ѡ��Fe��Mn������ETS-10���Ƴɴ��������о������ѹ����ɸ���ع��ɽ�����Ϊ�����������о����������ԣ����۸����ʹ����й��ɽ��������̴�����̬��CO-SCR��Ӧ������֮��Ĺ�ϵ��
1 ʵ��
1.1 �����Ʊ�
���ø��¹��෨�Ʊ�������Fe-Mn/ETS-10������
1.1.1 ETS-10�����Ʊ�
ͨ��ˮ�ȷ��Ʊ�ETS-10��ʼ������ɣ�n(SiO2): n(TiO2):n(Na2O):n(K2O:n(H2O):n(H2SO4)=5.5:1.0:3.7: 0.95:171:2.82���������Ϊ��1.56 g H2SO4��4.32g Ti(SO4)2������18 mLȥ����ˮ�У�����1 h����ΪA��Һ��5.34 g NaOH��3.21 g KF��2H2O������23 mL ȥ����ˮ�У�����1 h����ΪB��Һ����A��B��Һ���Ȼ�ϣ����Ƚ���4 h������50 mL���ķ���ϩ��ѹ���У�200 ����·�Ӧ48 h���������ġ�ϴ�ӡ�����õ�ETS-10��ɫ��ĩ��
1.1.2 Fe-Mn/ETS-10�����Ʊ�
ʵ��ǰ�������Fe/ETS-10������̽��ʵ�飬�õ�Fe��ETS-10������Ϊ5%��ѡ����5%Fe/ETS-10�����Ϸֱ��ز�ͬ������Mn���������趨Ϊ5%Fe-xMn/ETS-10(x=0%��0.5%��1%��2.5%��5%������xΪMn��ETS-10��������)��
���ռ���õı�����ȷ����ETS-10��Ʒ����������50%(��������)������������ˮ�Ҵ���2) ��ˮԡ80 �������½������ɣ����12 h��3) ��ĥ0.5 h����������¯���� 5 ��/min�����ٶ�����550 ��(������¶���ǰ���ս��¶�����ʵ����ȷ��)������4 h������Ȼ���������£����õ�Ŀ����
1.2 �����ﻯ���ܱ���
�����ձ�Rigaku D��˾TTR ����X����������(XRD)�����ϳɵIJ��Ͻ�������ṹ�ļ�⣬Cu��(��=1.5406 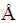 )��ɨ�跶Χ2��Ϊ10��~80�㣬ɨ���ٶ�Ϊ10 (��)/min��JSM-6360LV�͵���ɨ�������Բ��Ͻ��б�������ò�۲��������ɨ��Ԫ�سɷַ�����Nico-let6700����Ҷ���������(FT-IR)�Է��ӽṹ���ж��Զ���������Thermo Fisher��˾ESCALABXi+�ⶨ�������ĵ��ӽṹ�ͱ�����ɵĻ�ѧ�ɷ֡�
)��ɨ�跶Χ2��Ϊ10��~80�㣬ɨ���ٶ�Ϊ10 (��)/min��JSM-6360LV�͵���ɨ�������Բ��Ͻ��б�������ò�۲��������ɨ��Ԫ�سɷַ�����Nico-let6700����Ҷ���������(FT-IR)�Է��ӽṹ���ж��Զ���������Thermo Fisher��˾ESCALABXi+�ⶨ�������ĵ��ӽṹ�ͱ�����ɵĻ�ѧ�ɷ֡�
1.3 �����������ܱ���
ͨ��ʵ���ҶԺϳɵĴ��������������ܼ��ʵ�顣ʵ����ϵ������ϵͳ����ϵͳ�����ϵͳ3��������ɣ�����ϵͳ��װ��CO��NO�����ĸ�ƿ����������ɣ���ϵͳ�ɹ̶���Ӧ����ʽ����¯��ɣ����ϵͳΪ�¹�MRU��˾VARIO PLUS����ǿ�����������ǡ�
���ÿ�����ʯӢ��0.5 g �����̶���ʯӢ���в��������ڳ�����µĹ�ʽ����¯�н��з�Ӧ����500 ���£�1% CO+99% N2 (Ħ������)��ԭ�����У�����Ԥ����1 h��Ȼ��ͨ�뷴Ӧ���壺w(CO)= 600��10-6��w(NO)=500��10-6��ƽ������Ϊ�������پ������������¿�����5 ��/min���ٶȴ�50 ������600 ����������ʲ��ԡ��������������Ǽ�Ⲣʵʱ��¼���ڴ�NOŨ��������( )���㹫ʽ���£�
)���㹫ʽ���£�
 (1)
(1)
ʽ�У� Ϊ���ڴ�NOŨ�ȣ�
Ϊ���ڴ�NOŨ�ȣ� Ϊ���ڴ�NOxŨ��, ����NO��NO2��
Ϊ���ڴ�NOxŨ��, ����NO��NO2��
2 ���������
2.1 XRD����������
ͼ1(a)��ʾΪETS-10��XRD�ף���12.3�㡢12.9�㡢20.1�㡢23.8�㡢24.7�㡢25.8�㡢27.1�㡢31.8�㡢35.3�㡢35.6�㡢36.5�㡢37.0�㴦��������壬�ֱ��ӦETS-10��(101)��(004)��(105)��(200)��(202)��(116)��(204)��(109)��(217)��(208)��(224)��(110)�;���[29-30]������ETS-10����ɸ�ṹ����������ģ��������λ�����Ӧ[31-32]��ͼ1(b)��ʾΪFe-Mn/ETS-10��Ʒ��XRD���������Fe��Mn���ص�ETS-10�Ϻ���Ҫ�����λ�þ�δ�����仯��˵��Fe��Mn�ĸ��ز���Ӱ��ETS-10�ľ���ṹ��������ǿ�����н��ͣ�������2��Ϊ24.7���35.6��λ�ã��ֱ��ӦԭETS-10��(202)��(208)���档
��Fe-Mn/ETS-10��XRD����δ�۲쵽��Ϊ���Ե���Fe��Mn������صķ塣����������ױ������ڱ�ʵ�������£�Fe��Mn���ʵĴ�����ʽһ����������(Fe2O3��MnO2��)[33-34]����ͼ����δ������������������塣�����ƶϣ�����������������״̬���ڣ���ߴ�dz�С�Ľ��������������ɢ����Ʒ�У�С��XRD�ļ����ֵ[35]��
2.2 SEM����������
ͼ2��ʾΪETS-10��Fe-Mn/ETS-10��Ʒɨ���������ͼƬ��ͼ2(a)�ɿ�����ETS-10Ϊ��ȡ���˵�˫�壬��ò����������ߴ�Ϊ3 ��m���ң�������[30]�����������֤�ɹ��ϳ�ETS-10��ͼ2(b)~(f)ΪFe-Mn/ETS-10��Ʒ��ò���Ա�ͼ2(a)�ɿ�����ETS-10������ò�������֣���С����������ETS-10����ı��棬�Ʋ�С����Ϊ�̼��������������
5%Fe-1%Mn/ETS-10��Ʒ��X��������Ԫ����ֲ�����(SEM-EDAX)�����ͼ3��ʾ���ֱ�Ϊɨ������Na��K��Ti��Si��O��Mn��Fe�ķֲ�ͼ��֤ʵ��Fe��Mn���ʵĴ��ڣ�����ETS-10��������ɢ���ȡ�
ͼ1 ETS-10��Fe-Mn/ETS-10��XRD��
Fig. 1 XRD patterns of ETS-10(a) and Fe-Mn/ETS-10(b)

ͼ2 ETS-10��Fe-Mn/ETS-10��SEM��
Fig. 2 SEM images of ETS-10 and Fe-Mn/ETS-10

ͼ3 5%Fe-1%Mn/ETS-10��SEM����Ӧ��Ԫ����ɨ��ֲ�
Fig. 3 SEM image and EDAX element mappings distribution of 5%Fe-1%Mn/ETS-10
2.3 FTIR����������
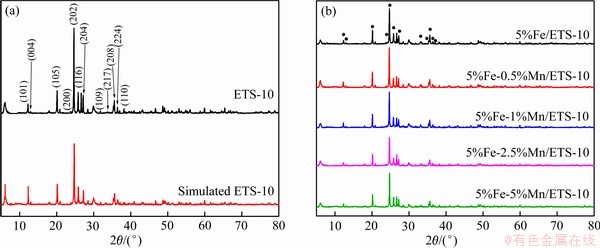
ͼ4��ʾΪFTIR�ס���ƷETS-10��500~4000 cm-1��Χ�ڳ��ֵ����շ��ǿ�Ⱥ�λ�������� [36-37]�е����������547.69 cm-1��Ӧ��Si��O���������Լ�O��Ti��O�����������壬744.40 cm-1���ķ��ӦTi��O�����������壬��973.89 cm-1���ķ�������ETS-4�е�Si��O��Ti��������ETS-10������ģ�1029.81 cm-1���ķ��ӦSi��O�����������壬1637.29 cm-1�����շ��Լ���3200 cm-1��3600 cm-1�Ŀ�����ˮ����������������������������塣ͨ�������������ͼ�����Ը�����֤ETS-10����ɸ�ijɹ��ϳɣ�ͬʱ����ETS-10��ʯ���ȶ��Ĵ������塣Fe-Mn/ETS-10�����е�Mn��Fe���ʼ�������������ͨ��FTIR��������������[38-39]���Ʋ������ںϳɵ�Fe-Mn/ETS-10��Ʒ�е�Mn��Fe�����͡�
2.4 XPS����������
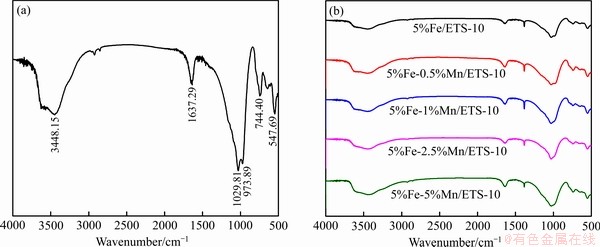
��Fe-Mn/ETS-10����XPS��⣬ͨ�����㲻ͬ���ر�����Ʒ��Fe��Mn��̬���������Աȷ�������ܵ����������ߵ�˳��ͼ5��ʾΪFe-Mn/ETS-10��Fe 2p���ף�����Fe 2p3/2�塢Fe 2p1/2�塢Fe3+���Ƿ�[40]��ͼ��ʾ�����ز����н���Fe��Ҫ�Զ��ۺ�����̬��ʽ���ڡ�Fe 2p3/2��Fe 2p1/2�ϵͽ����λ�ã��ֱ��Ӧ710.3 eV��723.8 eV��������Fe2+��Fe 2p3/2��Fe 2p1/2�ϸ߽����λ�ã���Ӧ712.7 eV��726.510 eV��������Fe3+ [41]���������Ƿ�Ҳ������Fe3+��
ͼ4 ETS-10��Fe-Mn/ETS-10�ĺ������ͼ
Fig. 4 Infrared spectra of ETS-10(a) and Fe-Mn/ETS-10(b)
ͼ5 Fe-Mn/ETS-10��Fe 2p����
Fig. 5 Fe 2p XPS spectra of Fe-Mn/ETS-10
ͨ��ͼ5����ͨ�����������Fe-Mn/ETS-10�еIJ�ͬFe���ֵ���Ժ�����������1��ʾ��ͨ��NOx�������ͻ�ԭ����ȡ���ڴ��������ϵ�Fe������Fe����������أ���ԭ��Ӧ�ɿ��������ؼ����裺1) CO�ڴ�����������������¾����������ӵ���ʧ���Ӷ������뻹ԭ�Ľ���������ϵ�����λ��2) NO�ڻ�ԭ�ı����ϵ�������Ȼ��������(�Ǽ���)CO��Ӧ[42]����������������������Ӻ�С����������������Գ䵱NOx�����Ļ�������[43]��Fe2O3ͨ����������λʹ����Fe3+��Fe2+״̬֮�䴩�����������������䷴Ӧ���ƣ���˷���ĺ͵;۵�Fe3+���ּ�С��Fe2O3�Ŵ���CO-SCR��Ӧ�Ļ�������[44-45]�������������������Fe���ֲַ�����Ҫ�ԣ���Fe3+��������NOx���������������õ�Fe���ֲַ�����˴����ԡ�5%Fe-1%Mn/ETS-10�в�ͬFe���ֵ���Ժ����Ͼ��ȡ�ͬʱ��5%Fe-1%Mn/ETS-10��Fe3+���ּ�Fe2O3������ߣ����������ĵĺ�����࣬��ˣ�������������Ӧ�Ľ��С�
��1 Fe-Mn/ETS-10��FeԪ�غ�������
Table 1 Analysis of Fe element content in Fe-Mn/ETS-10
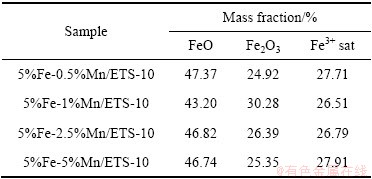
ͼ6��ʾΪFe-Mn/ETS-10��Mn 2p���ף�����Mn2O3�ߡ�MnO2����ͼ��ʾ������Mn����Ʒ����Ҫ�����ۺ��ļ�̬��ʽ���ڣ�������ڸ߽���ܴ�û�г����壬˵���ڴ������治����Mn2+���֡�
ͼ6 Fe-Mn/ETS-10��Mn 2p����
Fig. 6 Mn 2p XPS spectra of Fe-Mn/ETS-10
Mn 2p3/2������Լ641.4 eV��Mn 2p1/2������Լ653.2 eV[46]�������о�����[47]��Mn 2p3/2�Ľϸ߽���ܺ�Mn 2p1/2�Ľϵͽ����λ�÷ֱ��Ӧ641.2 eV��652.6 eV��ΪMn3+��Mn 2p3/2�Ľϸ߽���ܺ�Mn 2p1/2�ϵ͵Ľ����λ�ö�Ӧ643.4��655.0 eV��ΪMn4+��ʵ�����ݵ���Ͻ����ͼ6��ʾ��
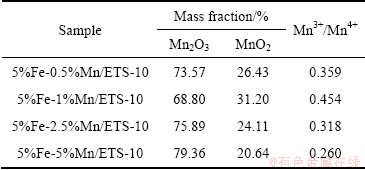
ͨ��ͼ6����ͨ�����������Fe-Mn/ETS-10�еIJ�ͬMn���ֵ���Ժ�����������2������ʾ�����У�����������ֳ�Mn3+��Mn4+��̬�����̬ʹ����������ֳ������������ԭ���ܣ�������ڶ������͵IJ��ȶ�������[48]����������ߴ����ԡ������й��ɽ����������У�Mn2O3���ֳ��ȶ���CO�������ܡ�5%Fe-1%Mn/ETS-10��Mn3+/Mn4+������ߣ����������ۼ��ļ�����֮���ת���������������������ֱ����ء�
��2 Fe-Mn/ETS-10��MnԪ�غ�������
Table 2 Analysis of Mn in Fe-Mn/ETS-10
2.5 ����CO-SCR���Բ��Լ�����
ͼ7��ʾΪ������50~600 ��������������ߣ���ͼ��֪����ͬ���ر�����Fe-Mn/ETS-10��Ʒ���������ܾ������¶ȵ����߳���ǿ���ơ��Աȷ��֣���50~600 �淶Χ�ڣ�5%Fe-1%Mn/ETS-10�������Խ�������Ʒ���ߣ�300 ��ʱ��5%Fe-1%Mn/ETS-10�������Դﵽ80.00%����400 ��ʱ��5%Fe-1%Mn/ETS-10�������Դ�85.33%��֮�������ʳ�������������������Ʒ��400 �������ʴﵽ80%���ϡ����������ܶԱȿ��Կ���Mn-Fe˫�������ضԴ����ĸ�����Ҫ�����ڵ����������ܵ���ߡ���ͬ���ر�����5%Fe-1%Mn/ETS-10Ϊ������ߵĴ���������Ĵ����������ԭ�ԣ�����Ľṹȱ�ݺ������̵�Эͬ�����й�[49]��
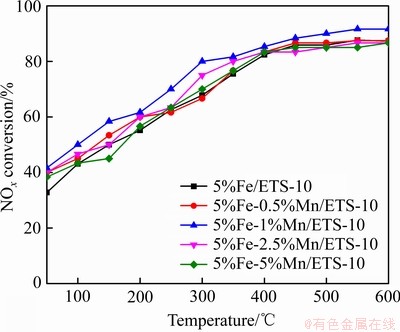
ͼ7 Fe-Mn/ETS-10������������
Fig. 7 Denitrification rate curves of Fe-Mn/ETS-10
3 ����������
����ǰ��XRD��XPS�ȱ����ɷ����õ���˫�������ص�Fe-Mn/ETS-10�����������������������Ӱ�죺1) ������������Fe3+��Fe2O3����λ���������2) ��������������IJ�ͬ���(MnO2��Mn2O3)�����ڲ�ͬ�¶ȷ�Χ�ڵ��������ܣ�����ؿ��˻��Դ��ڡ�
����CO-SCR��Ӧ���������뷴Ӧ����NO��CO�Ӵ�ʱ��NO�������������ڻ��Բ�λ�ϣ��γɲ�ͬ������̬NO���֣�Ȼ���յ�����ΪN��O��֮������Nԭ����NO���ӽ���γ�N2O�����CO���Ӻ��м����N2Oת��Ϊ��̬CO2�����ղ�ƷN2���ձ���Ϊ��Ӧ��ѭLangmuir-Hinshelwood(L-H)����[50]�����У�NO�Ľ�����ͨ��COȥ��NO�Ĺؼ����裬����λ���Լ���N��O���Դٽ�NO�Ľ���[51]��
��Fe-Mn/ETS-10�����У�Fe2O3��������λʹ����Fe3+��Fe2+��̬ת����ͬʱ��������֮�������ÿ�����Ч�ز�������Эͬ����λ�������ģ�����Эͬ����λֻ����˫�������������ɣ��������ڵ����������Ļ�е�����������[52]�����ڱ�������λ������Эͬ����λ�Ĺ��棬Fe-Mn˫��������������ߡ�
ͬʱ������NO+CO��Ӧ��Fe3++Mn3+ Fe2++Mn4+������ԭƽ�⣬���Դٽ������ڸ��ֻ������֮���ת�ƺͽ�������ˣ�ƽ�������������(Fe3+��Fe2+)��������(Mn3+��Mn4+)�����̬�������ӱ���Ӱ�����ת��Ч�ʣ�����N��O��ǿ�ȣ������ڱ�������NO���ֵĽ��룬�Ӷ�������ǿCO��ԭNO�Ĵ����ԡ������5%Fe-1%Mn/ETS-10����������ߵ�ԭ��
Fe2++Mn4+������ԭƽ�⣬���Դٽ������ڸ��ֻ������֮���ת�ƺͽ�������ˣ�ƽ�������������(Fe3+��Fe2+)��������(Mn3+��Mn4+)�����̬�������ӱ���Ӱ�����ת��Ч�ʣ�����N��O��ǿ�ȣ������ڱ�������NO���ֵĽ��룬�Ӷ�������ǿCO��ԭNO�Ĵ����ԡ������5%Fe-1%Mn/ETS-10����������ߵ�ԭ��
4 ����
1) ͨ�����¹��෨�ɹ��Ʊ���Fe-Mn/ETS-10�������Դ������ϵĽṹ��������ò���ṹ��������ɱ������֣��ս����Ĵ���������ETS-10����ԭ�еľ���ṹ������ԭ�н�ȡ���˵İ�������ò�������̽����Ը߶ȷ�ɢ����������ʽ������ETS-10������棬����������Ӧ��
2) ��5%Fe-1%Mn/ETS-10�����´�������������ߣ��¶�Ϊ300 �漰450 ��ʱ���������ʷֱ�ﵽ80%��90%��
3) Fe-Mn/ETS-10�����У������̵�Эͬ���������ڻ������ʵIJ�����ʹFe��Mn���ʲ�ͬ��̬�ķֲ���ƽ�����ٽ�Fe3++Mn3+ Fe2++Mn4+������ԭƽ�ⷴӦ����ߵ�������Ʒ�������ԡ�
Fe2++Mn4+������ԭƽ�ⷴӦ����ߵ�������Ʒ�������ԡ�
REFERENCES
[1] PATEL A, SHUKLA P, PAN Guan-ting, CHONG S, RUDOLPH V, ZHUA Zhong-hua. Influence of copper loading on mesoporous alumina for catalytic NO reduction in the presence of CO[J]. Journal of Environmental Chemical Engineering, 2017, 5(3): 2350-2361.
[2] LIU Tang-kang, QIAN Jun-ning, YAO Yan-yan, SHI Zhang-fu, HAN Li-ying, LIANG Cai-yuan, LI Bin, DONG Li-hui, FAN Min-guang, ZHANG Ling-ling. Research on SCR of NO with CO over the Cu0.1La0.1Ce0.8O mixed-oxide catalysts: Effect of the grinding[J]. Molecular Catalysis, 2017, 430: 43-53.
[3] OTON L F, OLIVEIRA A C, DE ARAUJO J C S, ARAUJO R S, DE SOUSA F F, SARAIVA G D, LANG R, OTUBO L, SILVA DUARTE G, CAMPOS A. Selective catalytic reduction of NOx by CO (CO-SCR) over metal-supported nanoparticles dispersed on porous alumina[J]. Advanced Powder Technology, 2020, 31(1): 464-476.
[4] ZHANG Li, QIN Yi-hong, CHEN Bai-zhen, PENG Ya-guang, HE Han-bing, YUAN Yi. Catalytic reduction of SO2 by CO over CeO2-TiO2 mixed oxides[J]. Transactions of Nonferrous Metals Society of China, 2016, 26(11): 2960-2965.
[5] �� ��, �����, �°���. CO NOx-SO2��ϵ��ѧƽ�����[J]. ���ϴ�ѧѧ��(��Ȼ��ѧ��), 2015, 46(1): 27-33.
ZHANG Li, QIN Yi-hong, CHEN Bai-zhen. Chemical equilibrium analysis of CO NOx SO2 system[J]. Journal of Central South University (Science and Technology), 2015, 46(1): 27-33.
[6] ���Ѿ�, �κ���, �����, �� ��. Ni������OMS-2���¿������������о�[J]. ���ܲ���, 2019, 50(8): 8084-8090.
LONG Jia-ju, HE Han-bing, LI Zhong-pu, ZHANG Hui. Low-temperature and anti-sulfur catalytic performance of Nix-OMS-2 for NO-CO[J]. Journal of Functional Materials, 2019, 50(8): 8084-8090.
[7] �κ���, ��С��, �����, �� ��, �� ٻ. ����˫�����л��Ǽܼ���̿������ĵ������������о�[J]. ���ܲ���, 2018, 49(12): 12001-12006.
HE Han-bing, MA Xiao-peng, LI Zhong-pu, ZHANG Hui, REN Qian. Low-temperature catalytic performance of cobalt-iron bimetallic organic framework and its carbonized products for NO-CO[J]. Journal of Functional Materials, 2018, 49(12): 12001-12006.
[8] ZHANG Li, HUANG Lei, QIN Yi-hong, CHEN Bai-zhen. Structure and denitration performance of carbon-based catalysts prepared from Cu-BTC precursor[J]. Transactions of Nonferrous Metals Society of China, 2018, 28(5): 980-988.
[9] WANG Jian-cheng, CHEN Ying, TANG Lei, BAO Wei-ren, CHANG Li-ping, HAN Li-na. One-step hydrothermal synthesis of Cu-SAPO-34/cordierite and its catalytic performance on NOx removal from diesel vehicles[J]. Transactions of Nonferrous Metals Society of China, 2013, 23(11): 3330-3336.
[10] ������, ���ܿ�, ���, ������, �ƽ, ������, �ž�Ӫ. CeO2-TiO2��������Эͬ����ȼúNOx�빯[J]. ���ϴ�ѧѧ��(��Ȼ��ѧ��), 2016, 47(3): 1049-1057.
LI Li-qing, WU Shao-kang, LI Hai-long, MA Wei-wu, YANG Jian-ping, ZHAO Yong-chun, ZHANG Jun-ying. Simultaneous removal of NOx and mercury over CeO2-TiO2 catalyst at low flue gas temperature[J]. Journal of Central South University (Science and Technology), 2016, 47(3): 1049-1057.
[11] KUZNICKI S M. Large-pored crystalline titanium molecular sieve zeolites, US Patent 4853202[P]. 1989-08-01.
[12] SHIRAISHI Y, TSUKAMOTO D, HIRAI T. Selective photocatalytic transformations on microporous titanosilicate ETS-10 driven by size and polarity of molecules[J]. Langmuir, 2008, 24(21): 12658-12663.
[13] MANI F, SAWADA J A, KUZNICKI S M. A comparison of the adsorptive behaviour of ETS-10, 13X and highly siliceous ZSM-5[J]. Microporous and Mesoporous Materials, 2015, 214: 32-40.
[14] DATTA S J, YOON K B. Co-ETS-10 and Co-AM-6 as active catalysts for the oxidation of styrene to styrene oxide and benzaldehyde using molecular oxygen[J]. Chinese Journal of Catalysis, 2015, 36(6): 897-905.
[15] SHARIATY P, LASHAKI M J, HASHISHO Z, SAWADA J, KUZNICKI S, HUTCHEON R. Effect of ETS-10 ion exchange on its dielectric properties and adsorption/ microwave regeneration[J]. Separation and Purification Technology, 2017, 179: 420-427.
[16] REN Yuan-hang, GU Min, HU Yi-chen, YUE Bin, JIANG Lei, KONG Zu-ping, HE He-yong. Preparation and photocatalytic activity of lanthanide loaded microporous titanosilicate ETS-10 catalysts[J]. Chinese Journal of Catalysis, 2012, 33(1): 123-128.
[17] SORRIBAS S, COMESANA-GANDARA B, LOZANO A E, ZORNOZA B, TELLEZ C, CORONAS J. Insight into ETS-10 synthesis for the preparation of mixed matrix membranes for CO2/CH4 gas separation[J]. RSC Advances, 2015, 5(124): 102392-102398.
[18] BORDIGA S, PAZE C, BERLIER G, SCARANO D, SPOTO G, ZECCHIANA A, LAMBERTI C. Interaction of N2, CO and NO with Cu-exchanged ETS-10: A compared FTIR study with other Cu-zeolites and with dispersed Cu2O[J]. Catalysis Today, 2001, 70: 91-105.
[19] SULTANA A, SASAKI M, HAMADA H. Influence of support on the activity of Mn supported catalysts for SCR of NO with ammonia[J]. Catalysis Today, 2012, 185(1): 284-289.
[20] SONG Li-yun, ZHAN Zong-cheng, LIU Xiao-jun, HE Hong, QIU Wen-ge, ZI Xue-hong. NOx selective catalytic reduction by ammonia over Cu-ETS-10 catalysts[J]. Chinese Journal of Catalysis, 2014, 35(7): 1030-1035.
[21] JIN Qi-jie, SHEN Yue-song, SUI Guo-rong, TAO Xing-jun, PAN You-chun, ZHU She-min. Synergistic catalytic removals of NO, CO and HC over CeO2 modified Mn-Mo-W-Ox/TiO2-SiO2 catalyst[J]. Journal of Rare Earths, 2018, 36(2): 148-155.
[22] WANG Tao, WAN Zhen-tian, YANG Xiao-chu, ZHANG Xin-yu, NIU Xiao-xuan, SUN Bao-min. Promotional effect of iron modification on the catalytic properties of Mn-Fe/ZSM-5 catalysts in the Fast SCR reaction[J]. Fuel Processing Technology, 2018, 169: 112-121.
[23] �� ��, ������, �Ի�ˬ, ��ʿ��, Ҷ�س�, ������. ����������Fe-Mn/Betaѡ���Դ���ϩ��ԭ��������[J]. ȼ�ϻ�ѧѧ��, 2019, 47(6): 751-761.
ZHOU Hao, LI Meng-yu, ZHAO Hui-shuang, WU Shi-guo, YE Bi-chao, SU Ya-xin. Selective catalytic reduction of nitric oxide with propylene in excess oxygen over Fe-Mn/Beta catalysts[J]. Journal of Fuel Chemistry and Technology, 2019, 47(6): 751-761.
[24] WANG Xiao-bo, WU Shi-guo, ZOU Wei-xin, YU Shuo-han, GUI Ke-ting, DONG Lin. Fe-Mn/Al2O3 catalysts for low temperature selective catalytic reduction of NO with NH3[J]. Chinese Journal of Catalysis, 2016, 37: 1314-1323.
[25] ���콿, ����ƽ, ׯ ��, ½ ��, ��һ��, ��. ����״Ho����Fe-Mn/TiO2�������Ʊ��������ѡ�����ԭ(SCR)��������[J]. ȼ�ϻ�ѧѧ��, 2018, 46(3): 319-327.
HUANG Tian-jiao, ZHANG Ya-ping, ZHUANG Ke, LU Bin, ZHU Yi-wen, SHEN Kai. Preparation of honeycombed holmium-modified Fe-Mn/TiO2 catalyst and its performance in the low temperature selective catalytic reduction of NOx[J]. Journal of Fuel Chemistry and Technology, 2018, 46(3): 319-327.
[26] ZHANG Yan-bing, ZHENG Yu-ying, WANG Xie, LU Xiu-lian. Preparation of Mn-FeOx/CNTs catalysts by redox co-precipitation and application in low-temperature NO reduction with NH3[J]. Catalysis Communications, 2015, 62: 57-61.
[27] �� ��, �� ��, ��ԪԪ, ����ǫ, �� ��, �ſ���, ������. ˮ�ȷ��Ʊ����̴����������ܼ���ˮ���������о�[J]. ���Ӵ�, 2018, 32(3): 240-248.
TANG Nan, HUANG Yan, LI Yuan-yuan, ZHAO Yong-qian, ZHOU Ting, ZHANG Jun-feng, YANG Liu-chun. Low temperature selective catalytic reduction of NO with NH3 over Fe-Mn catalysts prepared by hydrothermal method[J]. Journal of Molecular Catalysis, 2018, 32(3): 240-248.
[28] �¶, ������, ���, �߷���, ���ӱ, �� ��, �����. Mn������SCR�����Ŀ�H2O����SO2�о���չ[J]. ������չ, 2017, 36(3): 934-943.
LI Chen-lu, TANG Xiao-long, YI Hong-hong, GAO Feng-yu, LI Jing-ying, CHU Chao, ZHANG Run-cao. Review on manganese based catalysts resistant to H2O and SO2 for SCR reduction at low temperature[J]. Chemical Industry and Engineering Progress, 2017, 36(3): 934-943.
[29] GALIOGLU S, ISMAIL M N, WARZYWODA J, SACCO A, AKATA B. Preparation and microstructural characterization of oriented titanosilicate ETS-10 thin films on indium tin oxide surfaces[J]. Microporous and Mesoporous Materials, 2010, 131(1/3): 401-406.
[30] JI Zhao-xia, ISMAIL M N, CALLAHAN J, DENNIS M, PANDOWO E, CAI Zhu-hua, GOODRICH T L, ZIEMER K S, WARZYWODA J, SACCO J A. The role of silver nanoparticles on silver modified titanosilicate ETS-10 in visible light photocatalysis[J]. Applied Catalysis B (Environmental), 2011, 102(1/2): 323-333.
[31] ZHU Chao-jie, SHEN Run-sheng, FANG Zhong-xue, ZHANG Lei, WU Dong-fang, WU Meng-yu, TANG Tian-di , FU Wen-qian, CHEN Qun. Basic mesoporous zeolite ETS-10 supported Ni catalyst with bi-functional properties for efficiently catalyzing arenes fluorination[J]. Catalysis Communications, 2018, 117: 63-68.
[32] ANDERSON M W, TERASAKI O, OHSUNA T, PHILIPPOU A. Structure of the microporous titanosilicate ETS-10[J]. Nature, 1994, 367(6461): 347-351.
[33] XU Wen-jin, ZHANG Guang-xu, CHEN Han-wei, ZHANG Guo-meng, HAN Yang, CHANG Yi-chuan, GONG Peng. Mn/beta and Mn/ZSM-5 for the low-temperature selective catalytic reduction of NO with ammonia: Effect of manganese precursors[J]. Chinese Journal of Catalysis, 2018, 39(1): 118-127.
[34] XIN Ying, LI Qian, ZHANG Zhao-liang. Zeolitic materials for deNOx selective catalytic reduction[J]. Chem Cat Chem, 2018, 10(1): 29-41.
[35] CANEPA A L, ELIAS V R, VASCHETTI V M, SABRE E V, EIMER G A, CASUSCELLI S G. Selective oxidation of benzyl alcohol through eco-friendly processes using mesoporous V-MCM-41, Fe-MCM-41 and Co-MCM-41 materials[J]. Applied Catalysis A (General), 2017, 545: 72-78.
[36] SU L L F, ZHAO X. S. Synthesis and characterization of microporous titanosilicate ETS-10 with different titanium precursors[J]. Journal of Porous Materials, 2006, 13(3/4): 263-267.
[37] NGUANG S Y, WONG S R, LAW J S, KHOO L C, LAM W H, CHAN E S, MOH P Y. Enhancing adsorption property of Engelhard Titanosilicate-10 through incorporation of graphene oxide[J]. Microporous and Mesoporous Materials, 2017, 252: 125-139.
[38] DEYU S, DHAL G C, MOHAN D, PRASAD R. Low-temperature complete oxidation of CO over various manganese oxide catalysts[J]. Atmospheric Pollution Research, 2018, 9(4): 755-763.
[39] GOTIC M, MUSIC S. Mossbauer, FT-IR and FE SEM investigation of iron oxides precipitated from FeSO4 solutions[J]. Journal of Molecular Structure, 2007, 834/836: 445-453.
[40] YAMASHITA T, HAYES P. Analysis of XPS spectra of Fe2+ and Fe3+ ions in oxide materials[J]. Applied Surface Science, 2008, 254(8): 2441-2449.
[41] ARONNIEMI M, SAINIO J, LAHTINEN J. Chemical state quantification of iron and chromium oxides using XPS: The effect of the background subtraction method[J]. Surface Science, 2005, 578(1/3): 108-123.
[42] ZHANG Xing-yu, MA Chun-yuan, CHENG Xing-xing, WANG Zhi-qiang. Performance of Fe-Ba/ZSM-5 catalysts in NO+O2 adsorption and NO+CO reduction[J]. International Journal of Hydrogen Energy, 2017, 42(10): 7077-7088.
[43] RIVALLAN M, RICCHIARDI G, BORDIGA S, ZECCHINA A. Adsorption and reactivity of nitrogen oxides (NO2, NO, N2O) on Fe-zeolites[J]. Journal of Catalysis, 2009, 264(2): 104-116.
[44] CHENG Dang-guo, ZHAO Xiao-xu, CHEN Feng-qiu, ZHAN Xiao-li. Effect of preparation method on iron-containing AlPO-5 for selective catalytic reduction of N2O with methane in the presence of steam[J]. Catalysis Communications, 2009, 10(10): 1450-1453.
[45] REDDY B V, KHANNA S N. Self-stimulated NO reduction and CO oxidation by iron oxide clusters[J]. Phys Rev Lett, 2004, 93(6): 068301.
[46] L�� Gang, BIN Feng, SONG Chong-lin, WANG Kun-peng, SONG Jin-ou. Promoting effect of zirconium doping on Mn/ZSM-5 for the selective catalytic reduction of NO with NH3[J]. Fuel, 2013, 107: 217-224.
[47] LIU Yang, LI Xiao-Song, SHI Chuan, LIU Jing-Lin, ZHU Ai-Min, JANG Ben-WL. Ozone catalytic oxidation of adsorbed benzene over AgMn/HZSM-5 catalysts at room temperature[J]. Catalysis Science & Technology, 2014, 4(8): 2589-2598.
[48] LIU Tang-kang, YAO Yan-yan, WEI Long-qing, SHI Zhang-fu, HAN Li-ying, YUAN Hao-xuan, LI Bin, DONG Li-hui, WANG Fan, SUN Chuan-zhi. Preparation and evaluation of copper-manganese oxide as a high-efficiency catalyst for CO oxidation and NO reduction by CO[J]. The Journal of Physical Chemistry C, 2017, 121(23): 12757-12770.
[49] TARJOMANNEJAD A, FARZI A, NIAEI A, SALARI D. NO reduction by CO over LaB0.5B��0.5O3 (B=Fe, Mn, B��=Fe, Mn, Co, Cu) perovskite catalysts, an experimental and kinetic study[J]. Journal of the Taiwan Institute of Chemical Engineers, 2017, 78: 200-211.
[50] BAXTER R J, HU P. Insight into why the Langmuir- Hinshelwood mechanism is generally preferred[J]. The Journal of Chemical Physics, 2002, 116(11): 4379-4381.
[51] ZHANG Yu, ZHAO Ling, DUAN Jun, BI Si-ning. Insights into deNOx processing over Ce-modified Cu-BTC catalysts for the CO-SCR reaction at low temperature by in situ DRIFTS[J]. Separation and Purification Technology, 2020, 116081.
[52] XIONG Yan, YAO Xiao-jiang, TANG Chang-jin, ZHANG Lei, CAO Yuan, DENG Yu, GAO Fei, DONG Lin. Effect of CO-pretreatment on the CuO-V2O5/��-Al2O3 catalyst for NO reduction by CO[J]. Catalysis Science & Technology, 2014, 4(12): 4416-4425.
FU Guo-you1, HE Han-bing1, 2, 3, ZHANG Li1, MA Ying4
(1. School of Metallurgy and Environment, Central South University, Changsha 410083, China;
2. National Key Laboratory of Environmental Protection Mining and Metallurgy Resources Utilization and Pollution Control, Wuhan University of Science and Technology, Wuhan 430081, China;
3. Key Laboratory of Metallurgical Emission Reduction and Resources Recycling, Ministry of Education, Anhui University of Technology, Ma��anshan 243002, China;
4. Yongqing Environmental Protection Co., Ltd., Changsha 410001, China)
Abstract: Fe-Mn/ETS-10 catalysts with various compositions were synthesized by high temperature solid state reaction. These catalysts were characterized by X-ray diffractometry(XRD), Fourier transform infrared spectroscopy(IR), scanning electron microscopy(SEM) and X-ray photoelectron spectroscopy(XPS). The effect of different amounts of Fe-Mn/ETS-10 on the morphology, composition and the catalytic reduction performance was studied. The results show that Fe-Mn/ETS-10 has good uniformity and dispersion, the morphology and crystal structure of ETS-10 are kept. 5%Fe-1%Mn/ETS-10 has more active sites and higher deNOx performance. The catalysts activity of CO-SCR results show that denitrification rates of 5%Fe-1%Mn/ETS-10 are 80% and 85% at 300 �� and 400 ��, respectively. So, it is concluded that an optimal Fe-Mn/ETS-10 catalyst support ratio (5%Fe-1%Mn/ETS-10) enhances the catalytic activity in the selective catalytic reduction reaction.
Key words: Fe-Mn catalyst; catalytic denitrification; CO-SCR catalyst property; ETS-10 Molecular sieve
Foundation item: Project(2017GK4010) supported by the Scientific Technology of Strategic Emerging Industries and Major Achievement Transformation of Hunan Province, China; Project(2019JJ40378) supported by the Natural Science Foundation of Hunan Province, China; Project(HB201908) supported by Open Foundation of State Environmental Protection Key Laboratory of Mineral Metallurgical Resources Utilization and Pollution Control; Project(JKF20-02) supported by Open Foundation of Key Laboratory of Metallurgical Emission Reduction & Resources Recycling of Anhui University of Technology, Ministry of Education, China
Received date: 2019-12-19; Accepted date: 2020-05-13
Corresponding author: HE Han-bing; Tel: +86-13875985605; E-mail: hehanbinghhb@163.com
(�༭ ����)
������Ŀ������ʡս�������˲�ҵ�Ƽ��������ش�Ƽ��ɹ�ת��������Ŀ(2017GK4010)������ʡ��Ȼ��ѧ����������Ŀ(2019JJ40378)�����һ���������ұ��Դ��������Ⱦ�����ص�ʵ���ҿ��Ż������������Ŀ(HB201908)�����չ�ҵ��ѧұ���������Դ�ۺ����ý������ص�ʵ���ҿ��Ż���������Ŀ(JKF20-02)
�ո����ڣ�2019-12-19�������ڣ�2020-05-13
ͨ�����ߣ��κ����������ڣ���ʿ���绰��13875985605��E-mail��hehanbinghhb@163.com

