
Effect of metallic phase content on mechanical properties of (85Cu-15Ni)/(10NiO-NiFe2O4) cermet inert anode for aluminum electrolysis
ZHANG Gang(�� ��), LI Jie(�� ��), LAI Yan-qing(������), TIAN Zhong-liang(������)
School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China
Received 6 September 2006; accepted 16 April 2007
Abstract: (85Cu-15Ni)/(10NiO-NiFe2O4) cermets were prepared with Cu-Ni mixed powders as toughening metallic phase and 10NiO-NiFe2O4 as ceramic matrix. The phase composition, microstructure of composite and the effect of metallic phase content on bending strength, hardness, fracture toughness and thermal shock resistance were studied. X-ray diffraction analysis indicates the coexistence of (Cu-Ni), NiO and NiFe2O4 phases in the cermets. Within the content range of metallic phase from 0% to 20% (mass fraction), the maximal bending strength (176.4 MPa) and the minimal porosity (3.9%) of composite appear at the metallic phase content of 5%. The fracture toughness increases and Vickers�� hardness decreases with increasing metal content. When the thermal shock temperature difference (?t) is below 200 ��, the loss rate of residual strength for 10NiO-NiFe2O4 ceramic is only 8%, but about 40% for (85Cu-15Ni)/(10NiO-NiFe2O4) cermets. As ?t is above 200 ��, the residual strength sharply decreases for sample CN0 and falls slowly for samples CN5-CN20.
Key words: NiFe2O4; cermet inert anode; aluminum electrolysis; metallic phase; fracture toughness; thermal shock resistance
1 Introduction
The use of inert or non-consumable anodes and wettable cathodes for replacement of consumable carbon anodes and cathodes in Hall-Heroult electrolysis cells for the production of aluminum has been a technical and commercial goal for many decades[1-3]. During electrowinning in the novel cells with inert anodes, the specific criteria that an inert anode material used in aluminum electrolysis are[4]: 1) acceptable electric conductivity comparable to carbon anodes at operating temperature; 2) low reactivity with bath and oxygen when being anodic-polarized; 3) adequate mechanical strength; 4) ability to be fabricated into large shapes; 5) stable electrical connection with metal rod; 6) relatively low cast and ready availability, and 7) ability to produce commercial purity aluminum.
The current research on cermet for inert anode mainly focuses on the properties of electrolysis corrosion and conductivity, but only few studies are about its mechanical characteristics. Since 1980, Aluminum Company of America (Alcoa) has conducted[5], supported by US Department of Energy, a considerable work about cermets inert anodes. One of the major successes was to determine 17Cu-18NiO-NiFe2O4 cermet to be the promising material because of its high electric conductivity and good corrosion resistance during electrolysis. Thereafter, an intensive and extensive work was concentrated on the electric conductivity and corrosion behavior of this material. However, the work of the mechanical characteristics was almost neglected. Only a few investigations had studied the bending strength of 17Cu-18NiO-NiFe2O4[5]. The pilot scale experiments[2,5-6] exhibited that the inert anode samples chapped in the electrolysis process due to their poor mechanical properties, though the samples showed favorable corrosion resistance and conductivity. So the poor mechanical properties have become a main technical obstacle to development of cermet inert anodes. The previous work[7] showed that the cermets with 10NiO-NiFe2O4 as ceramic matrix appeared high relative density and favorable corrosion resistance. For metal composition, Cu is one of favorable choices because of its combination of the advantages of perfect conductivity and corrosion resistance. However, during sintering process, its low melting point and poor wettability with NiFe2O4 affected the mechanical properties of materials because of its sweating out and inhomogeneous dispersion in cermets[8]. The previous research[9] indicated that Ni could improve the wettability between Cu and NiFe2O4 and promote the sintering process.
In this study, Cu-Ni/(10NiO-NiFe2O4) cermets, using Ni and Cu as metallic phase and 10NiO-NiFe2O4 as ceramic phase, were fabricated by cold pressing and sintering. The effects of content of metallic phase on the mechanical properties of (85Cu-15Ni)/(10NiO-NiFe2O4) cermet inert anode were investigated.
2 Experimental
(85Cu-15Ni)/(10NiO-NiFe2O4) cermet composites were fabricated with conventional cold pressing and sintering process with reagent grade raw materials of Ni, Cu, NiO and Fe2O3. A proper amount of NiO and Fe2O3 (the excess content of NiO, compared with that of the stoichiometric NiFe2O4, in the ceramic phase is 10%, mass fraction) were mixed by ball milling and then calcined in a muffle furnace at 1 200 �� for 6 h at static air atmosphere to obtain 10NiO-NiFe2O4 ceramic powders. The X-ray diffraction pattern of the synthesized ceramic powder is illustrated in Fig.1. The calcined powders were mixed with metal powders (85Cu-15Ni) by ball milling again and then compressed to form cylindrical blocks (20 mm in diameter and 45 mm in thickness) at a pressure of 200 MPa. The electrodes were sintered at 1 250 �� for 4 h in nitrogen atmosphere of efficaciously controlled oxygen partial pressure[9-10]. The compositions and theoretical densities of samples are listed in Table 1. The mass fractions of metallic phases with Cu and Ni were 85% and 15% (mass fraction), respectively. With respect to CN0, CN5, CN10, CN15 and CN20, the mass fractions of metal phase were 0%, 5%, 10%, 15%, 20% (mass fraction), respectively.
Table 1 Compounds and theoretical densities of samples
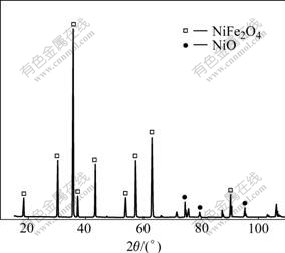
Fig.1 XRD patterns of 10NiO-NiFe2O4 ceramic
Bulk density and apparent porosity of samples were tested according to the Archimedes�� method. Phase composition of the composite was identified by X-ray diffraction analysis using Philips PW1390 X-ray diffractometer with Cu K�� radiation. Microstructure was analyzed with JSM-5600LV scanning electron microscope and XJP-6A metallurgical microscope. Three-point-bending strength of the sintered specimens (3 mm��4 mm��36 mm) was evaluated with a CSS-44100 electrical universal testing machine using span of 30 mm and cross-head speed of 0.5 mm/min. Bending strength data were achieved by average value with testing for six bars. Fracture toughness and hardness were measured with indentation strength(IS) method based on the equation given by GUAN et al[11] using a AKASHI(AVK-A) Vickers�� hardness tester at 98 N with a load time of 10 s.
3 Results and discussion
3.1 Phase composition of cermet samples
The X-ray diffraction patterns of the prepared cermets samples are shown in Fig.2. The major peaks detected include those of (Cu, Ni), NiO and NiFe2O4. Compared with the diffraction peak of Cu[12], the peak position of Cu corresponds to that of (Cu-Ni), and the difference is just a decrease of crystal face spacing of (Cu-Ni) due to solid solution from Ni to Cu.

Fig.2 XRD patterns of (85Cu-15Ni)/10NiO-NiFe2O4 cermets
3.2 Effect of metallic phase content on bending strength
The mechanical property is of great importance to desirable inert anode material. Generally speaking, proper metal will improve the mechanical property of cermet. Given low metallic content, it is hard for cermet to exhibit the advantage of metallic strengthening. The predominance of corrosion resistance of cermet decreases with the increase of metal content because the metal dissolves in cryolite melt prior to the ceramic matrix. The bending strength and porosity of composites are displayed in Fig.3. As the content of Cu-Ni increases from 0 to 5%, the bending strength increases and the porosity decreases. However, as the metallic content ranges from 5% to 20%, the changing trends of bending strength and porosity are contrary to the former. The composite with 5% metallic content achieves a maximum value of bending strength of 176.4 MPa and a minimum value of porosity of 3.9%, respectively. Besides, the changes of bending strength and porosity of cermets are not apparent when the metallic content ranges from 10% to 20%.

Fig.3 Relationship between room temperature bending strength, porosity and content of metal phase of (85Cu-15Ni)/ 10NiO-NiFe2O4 cermets
In general, the bending strength of cermets is related to several factors such as ceramic grain size[11], porosity [13], metallic phase dispersion, binding strength between ceramic phases and metallic phases[14]. Eqns.(1) and (2) illustrate that decreasing grain size and porosity can increase material strength:
��=��0+Kd -1/2 (1)
��=��0exp(-bp) (2)
where �� is material strength; ��0 is yield stress; K is a constant and d is grain size; p is porosity; �� is strength for sample with porosity of p; ��0 is strength for sample without pores; and b is a constant.
As showed in Fig.4, there is no obvious change on ceramic grain size (10-20 ��m) for the composites (CN5-CN20) with various metallic contents. Though the composite CN0 has smaller grain size (5-10 ��m) than others, the strength decreases reversely due to large porosity. Fig.3 shows that the porosity obviously increases when the metallic content is above 5%. In this study, the sintering temperature (1 250 ��) is higher than the melt point of 85Cu-15Ni alloy (1 175 ��), so these cermet composites are treated in liquid sintering process. From Fig.4, the wettability between metal phase (Cu-Ni) and ceramic phase (NiO-NiFe2O4) is not improved and particles are contacted with each other to form continuous framework, which are isolated by the liquid outside. Increasing sintering temperature or holding time can produce strong conglutination and capillarity of particles, which may cause the metal to be squeezed out from ceramic matrix. This trend will be enhanced with increase of metallic content. In this research, it can be found that the metal is overflowed on the surface of sample when metallic content is over 10%. The effect of metallic toughing weakened is related with the decrease of metallic content and the strength declined is related to the increase of porosity and the decrease of densification. These results are coincident with conclusions from Fig.4. In addition, Eqn.(3) suggests that high interfacial energy and small wetting angle are propitious to increased interfacial binding strength:

Fig.4 SEM photographs of (85Cu-15Ni)/ 10NiO-NiFe2O4 cermets with various metallic contents: (a) 0; (b) 5%; (c) 10%; (d) 15%; (e) 20%
W=��LV(1+cos��) (3)
where W is interfacial binding strength; ��LV is interfacial energy; �� is wetting angle.
Generally, the homogeneous dispersion of metal phase can improve interfacial contact between ceramic and metal and promote function of metallic toughening. However, as seen from Fig.4, metallic particles most locate in the isolated pores with unexpected aggregation,
thus the strength is not determined by metal but by ceramic. So increasing metallic content may result in an increase of metallic aggregation and decrease of metallic toughening.
3.3 Effect of metallic phase content on fracture tough- ness
From Fig.5, as metallic content increases, the hardness is decreased and the fracture toughness is increased. The hardness decreases from 4 972.3 MPa to 3 809.2 MPa and the fracture toughness increases from 1.22 MPa��m1/2 to 2.80 MPa��m1/2 with increasing metallic content from 0 to 20%.
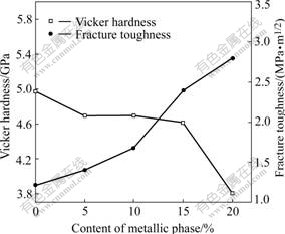
Fig.5 Vicker hardness and fracture toughness of (85Cu-15Ni)/ 10NiO-NiFe2O4 cermets with various metallic contents
The toughening of cermet mainly depends on metallic particle[15]. The crack always extends through the way with low energy. It is difficult for crack to extend through metallic phase but easy to extend across the interface between ceramic and metal due to the relatively low fracture energy. Hence, This increases the extending distance and resistance of fracture growth and strengthens the function of the bridging toughening and crack deflection toughening simultaneously.
3.4 Effect of metallic phase content on thermal shock resistance
The cermets exhibit various abilities of thermal shock resistance in process of thermal shock experiments. Fig.6 shows the residual strength of specimens quenched at different thermal shock temperature differences. The results suggest that the decline of residual strength is apparent for samples CN5-CN0 but unobvious for samples CN0 quenched at thermal shock temperature difference(?t) below 200 ��. As ?t is above 200 ��, the residual strength sharply decreases for sample CN0, but falls slowly for samples CN5-CN20.

Fig.6 Plot of retained flexural strength vs temperature difference for (85Cu-15Ni)/10NiO-NiFe2O4 cermets
According to the theory of thermal shock resistance by HASSELMAN[16], on condition of thermal shock, the presence and spread of crack are related with elastic strain energy and fracture surface energy deposited in composite. Small elastic strain energy and high fracture surface energy can decrease the possibility of spread of crack and increase the ability of thermal shock resistance of composite. In current work, when ?t is between 0 and 200 ��, the ceramic (CN0) shows the promising thermal shock resistance property compared with that of cermets (CN5-CN20), which is related with much pore and other defects in the ceramic (CN0). These defects are not propitious to increasing strength, but improving the capability to resist the spread of catastrophic crack. In the other word, the existence of these defects provides a certain space to relax the stress and cause a decrease of the elastic strain energy and an increase of fracture surface energy. Besides, fine grain particle also benefits to improving the thermal shock resistance of composite. It is generally thought that metallic addition will improve the thermal shock resistance of cermets. But at high temperature, a sharp decline of strength is attributed to the oxidation of metallic phase. Furthermore, the aggregation of metal also weakens the effect of metallic toughening. When the thermal shock temperature change is between 200 �� and 400 ��, the cermet composites CN5-CN20 show better thermal shock properties than ceramic composite CN0. Although the full densification of cermets composites are not achieved, the relative densities are all exceeded 92%, so metal on the surface of composite is oxidized and compact film is formed to postpone or prevent the further oxidation during heating process[17]. Consequently, the effect of metallic toughening can be exhibited with the increase of thermal shock temperature difference and the crack is either passivated or deflected by the metal inclusions.
4 Conclusions
1) As metallic content is increased from 0 to 20%, there is a sharp change in bending strength and porosity of (85Cu-15Ni)/(10NiO-NiFe2O4) composites. A maximum strength of 176.4 MPa and a minimum porosity of 3.9% are presented when the metallic phase content is 5%. Increasing the metallic content from 5% to 20%, the strength decreases and the porosity increases, but the change is not obvious when the metallic content is over 10%.
2) The hardness decreases and the fracture toughness increases with an increase of metallic content. The fracture toughness increases from 1.22 MPa?m1/2 to 2.80 MPa?m1/2 and hardness decreases from 4 972.3 MPa to 3 809.2 MPa with the metallic content increasing from 0 to 20%.
3) As the thermal shock temperature difference (?t) is below 200 ��, a change of strength of 10NiO-NiFe2O4 ceramic is not apparent, but (85Cu-15Ni)/10NiO- NiFe2O4 cermet shows obvious change with a decrease ratio of about 40%. As ?t is above 200 ��, the strength of ceramic distinctly falls and decrease ratio of strength is more than 70%. While the residual strengths of cermets remain at the same level and decrease ratios of residual strengths fall with increasing metallic content.
References
[1] LIU Ye-xiang. Advance on the research and development of inert anode and wettable cathode in the aluminum electrolysis [J]. Light Metals, 2001(5): 26-29. (in Chinese)
[2] PAWLEK R P. Inert anodes: An update [C]// SCHNEIDER W. Light Metals. Warrendale, PA: TMS, 2002: 449-456.
[3] GRAY P T. Corrosion and passivation of cermet inert anodes in cryolite-type electrolytes [C]// MILLER R E. Light Metals. Warrendale, PA: TMS, 1986: 309-320.
[4] BENEDYK C J. Status report on inert anode technology for primary aluminum [J]. Light Metal Age, 2001, 59(1/2): 36-37.
[5] WEYAND J D, DEYOUNG D H, RAY S P, TARCY G P, BAKER F W. Inert anodes for aluminum smelting [R]. DOE/CS/40158-20, Washington D. C: Aluminum Company of America, 1986.
[6] ALCOM T R, TABEREAUX A T, RICHARDS N E, WINDISCH C F, STRACHAN D M. Operational results of pilot cell test with cermet inert anodes [C]// SUBODH K D. Light Metals. Warrendale, PA: TMS, 1993. 433-443.
[7] LI Jie, DUAN Hua-nan, LAI Yan-qing, TIAN Zhong-liang, LIU Ye-xiang. Effect of NiO content on the corrosion behavior of Ni-xNiO-NiFe2O4 cermets in Na3AlF6-Al2O3 melts [J]. Trans Nonferrous Met Soc China, 2004, 14(6): 1180-1186.
[8] ZHANG Gang, LAI Yan-qing, TIAN Zhong-liang, QIN Qing-wei, LI Jie, LI Wen-xian. Preparation of nickel ferrite based cermets for aluminum electrolysis [J]. Journal of Material Science and Engineering, 2003, 21(4): 510-513. (in Chinese)
[9] LAI Yan-qing, ZHANG Gang, LI Jie, LIU Ye-xiang. Effect of adding Cu-Ni on mechanical capacity and electrical conductivity of NiFe2O4-based cermets [J]. Journal of Central South University of Technology, 2004, 35(6): 880-884. (in Chinese)
[10] ZHANG Lei, ZHOU Ke-cao, LI Zhi-you, ZHANG Xiao-yong. Effect of atmosphere on densification in sintering nickel ferrite ceramic for aluminum electrolysis [J]. Trans Nonferrous Met Soc China, 2004, 14(6): 1002-1006. (in Chinese)
[11] GUAN Zhen-duo, ZHANG Zhong-tai, JIAO Jin-sheng. Physical capability of inorganic materials [M]. Beijing: Tsinghua University Press, 2002: 103-106. (in Chinese)
[12] SHAO Wen-zhu, SHA De-sheng, YANG De-zhuang, ZVANNOR V V. Experimental research on conductivity of Cu porous oxide based cermet [J]. J Material Science and Technology, 1999, 7(2): 109-112. (in Chinese)
[13] SCHICKER S, ERNY T, GARCIA D E, JANSSEN R, CLAUSSEN N. Microstructure and mechanical properties of Al-assisted Sintered Fe/Al2O3 Cermets [J]. Journal of the European Ceramic Society, 1999, 19(13/14): 2455-2463. (in Chinese)
[14] SHI Jin. Cermet [M]. Shanghai: Shanghai Science and Technology Press, 1964. (in Chinese)
[15] QUENARD O, LAURENT C, PEIGNEY A, ROUSSET A. Zirconia-spinel composites (Part II): Mechanical properties [J]. Materials Research Bulletin, 2000, 35(12): 1979-1987.
[16] HASSELMAN D P H. Unified theory of thermal shock fracture initiation and crack propagation in brittle ceramics [J]. J Am Ceram Soc, 1969, 52(11): 600-604.
[17] TIAN Zhong-liang, LAI Yan-qing, LI Jie, LIU Ye-xiang. Study on anti-oxygen performance of nickel ferrite based cermets at high temperature [J]. Journal of Central South University of Technology, 2004, 35(6): 902-906. (in Chinese)
Foundation item: Project(2005CB623703) supported by the National Key Basic Research Program of China; Project(50474051) supported by the National Natural Science Foundation of China; Project(03JJY3080) supported by the Natural Science Foundation of Hunan Province, China
Corresponding author: ZHANG Gang; Tel: +86-731-8830474; E-mail: zg0805@163.com
(Edited by YANG Bing)