
HA coating on titanium with nanotubular anodized TiO2 intermediate layer via electrochemical deposition
WANG Yue-qin(������), TAO Jie(�� ��), WANG Ling(�� ��),
HE Ping-ting(�����), WANG Tao(�� ��)
College of Material Science and Technology, Nanjing University of Aeronautics and Astronautics,Nanjing 210016, China
Received 27 April 2007; accepted 10 December 2007
Abstract: Hydroxyapatite(HA) coating has been prepared on titanium substrate through an electrochemical deposition approach. In order to improve the bonding strength between HA coating and Ti substrate, a well oriented and uniform titanium oxide nanotube array on the surface of titanium substrate was applied by means of anodic oxidation pre-treatment. Then the calcium hydrogen phosphate(CaHPO4?2H2O, DCPD) coating, as the precursor of hydroxyapatite coating, was electrodeposited on the anodized Ti. At the initial stage of electro-deposition, the DCPD crystals, in nanometer precipitates, are anchored in and between the tubes. With increasing the deposition time, the nanometer DCPD crystals are connected together to form a continuous coating on titanium oxide nanotube array. Finally, the DCPD coating is converted into hydroxyapatite one simply by being immersed in alkaline solution.
Key words: anodic oxidation; nanotubes; hydroxyapatite; electrochemical deposition
1 Introduction
Hydroxyapatite(HA) is a calcium phosphate biomaterial with good biocompatibility and bioactivity. However, the poor mechanical properties of HA result in the limit of its application in load bearing. Titanium and its alloys have been extensively used as biomedical implant materials owing to the excellent mechanical properties[1]. But, when titanium or titanium alloy is implanted in bone, the bonding of the implant with living bone often does not develop[2] or a long time of several months is required to achieve the integration of the implants with bone tissue. Therefore, the preparation of HA coating on Ti or its alloys is a promising way to obtain a bioactive implant with better mechanical properties[3-8].
Electrochemical deposition of hydroxyapatite(HA) coating on titanium surface has unique advantages [9-12], which is an attractive process because highly irregular objects can be coated relatively quickly at low temperatures. Additionally, the thickness and chemical composition of coating can be well controlled through adequate conditions of the electro-deposition process. Furthermore, HA can be electrochemically deposited on the internal surfaces of porous metal substrates[11]. The coating morphology can be controlled in the process of electrochemical deposition easily through the domination of the deposition rate by varying the electrochemical potential, current density, electrolyte concentration and temperature[12-13].
The presence of a potentially weak HA coating/Ti alloy interface may compromise the function of the implant devices. It was reported by SPIVAK et al[14] that the failure of HA coating/Ti/bone specimen under tensile testing occurred consistently at HA coating/Ti interface, indicating clearly that the interface bonding was not strong enough to reach a reliable requirement. Therefore, concerns should be paid on the promotion of bonding at HA coating/Ti alloy interface.
The surface roughness of the substrate is particularly important not only because a rough surface can provide increased wettability of the HA precursor solution on the substrate, but also because mechanical interlocking between the HA-coated layer and substrate may be enhanced to avoid the failure of the coated HA layer under shear stress[15].
The vertically aligned TiO2 nanotubes on Ti substrate can be fabricated by anodization process[16-17], which can act as an intermediate layer for improving the bonding condition between HA coating and Ti substrate. However, the investigations on the preparation of hydroxyapatite coating on the TiO2 nanotubes were seldom reported. The purpose of present work is to strengthen the HA coating/Ti interface by anodization of titanium, which might provide a mechanical interlocking between the HA coating and nanotubes in titania intermediate layer on the outer surface of Ti substrate.
2 Experimental
The coating process involves two steps: 1) lower potential anodization of Ti to form nanotubes with controlled nanotube diameter; and 2) cathodic deposition of CaHPO4?2H2O coating, as the precursor of HA coating.
2.1 Formation of nanotubular oxide layer on titanium
The anodization process was conducted as follows. Titanium substrates were ultrasonically cleaned in acetone, ethanol and distilled water, respectively. Then they were chemically cleaned/etched in the solution of HNO3 and HF(V(HNO3)?V(HF)=1?1), rinsed by distilled water and dried at room temperature. The electrolyte solution for anodization process consisted of 0.5 mol/L H3PO4 and 0.138 mol/L NaF in water. The Ti substrate to be anodized was connected to the positive terminal of the power source, and a titanium net connected to the negative terminal was used as the cathode. The distance between anode and cathode was maintained at about 4 cm. The TiO2 nanotubes were prepared by using a 20 V anodization voltage for 28 min at room temperature. The Ti samples with nanotube layer were then heat treated at 500 �� for 2 h in order to crystallize the amorphous TiO2 into the anatase.
2.2 Cathodic deposition of calcium phosphate onto nanoporous titanium
Electro-deposition of calcium phosphate was conducted at room temperature in an electrolyte containing 0.04 mol/L Ca(NO3)2, 0.027 mol/L (NH4)2- HPO4 and 0.1 mol/L NaNO3. This solution was of a molar ratio of Ca to P as 1.67 and a pH value adjusted to be 4.5. The deposition was carried out by applying a constant potential of 3 V. The solution was stirred during electro-deposition to obtain a uniform electrolyte concentration. Titanium substrates after anodic oxidation were used as cathodes for electro-deposition. The obtained coatings were further treated in 0.1 mol/L NaOH for 2 h. Then the coated titanium cathodes were rinsed with distilled water and dried at room temperature in air.
2.3 Electrochemical polarization tests and immersion tests in simulated body fluid (SBF)
The SBF with ion concentrations close to those of human blood plasma was prepared by dissolving reagent grade chemicals of NaCl, NaHCO3, KCl, K2HPO4��3H2O, MgCl2��6H2O, CaCl2 and Mg2SO4 into distilled water and buffering at pH 7.4 with tris-hydroxyl methyl aminomethane ((CH2OH)3CNH3) and hydrochloric acid at 36.5 ��. The titanium, anodized titanium without annealing, and anodized titanium annealed at 500 ��, were electrochemically polarized in SBF solution by the CHI660A electrochemical workstation at a scanning rate of 2 mV/s.
2.4 Evaluation of coating
The crystallinity and the structure of the coatings were examined using X-ray diffractometer (Bruker D-8). The morphology of titanium oxide nanotubes, the surface morphology of coated specimens, and the tensile tested surface where the coating was removed were observed by means of scanning electron microscopy (FE-SEM, LEO��1530VP). An energy dispersive X-ray spectrum was also used to analyze the composition of coating. The corrosion resistance in SBF was evaluated according to polarization curves measured by the CHI660A electrochemical workstation.
3 Results and discussion
3.1 Anodization of titanium substrate
On the surface of Ti substrate, TiO2 nanotubes were produced after anodization for 28 min. The nanotubes, about 100 nm in average diameter, were grown in a vertically aligned, parallel configuration, as shown in Fig.1, which had a hollow core and were slightly separated from each other. The nanotubes fabricated by anodization were found to be amorphous according to the XRD diffraction pattern plotted in Fig.2(a). With subsequent heat treatment at 500 ��, the amorphous nanotubes transformed into the anatase structure (Fig.2(b)).

Fig.1 Top view(a) and side view(b) of TiO2 nanotubes formed on anodized Ti substrate
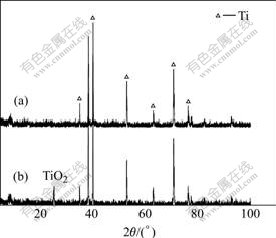
Fig.2 XRD patterns of anodized titanium before(a) and after(b) heat treatment at 500 ��
It was well established that the anatase phase TiO2 was much more efficient in nucleation and growth of HA than the rutile phase TiO2 presumably because of the better lattice match with HA phase[1]. Thus the anatase thin layer with vertically aligned nanotube array could be effective in improving both deposition and bonding of HA bioactive coating on Ti substrate.
3.2 Electro-deposition of hydroxyapatite
Fig.3 shows the XRD patterns of the directly electrodeposited coatings before and after alkaline solution treatment. On the anodized surface of Ti, the directly electrodeposited coating mostly consisted of calcium hydrogen phosphate(CaHPO4��2H2O, DCPD), as the precursor of HA, which transformed to HA coating in alkaline solution.
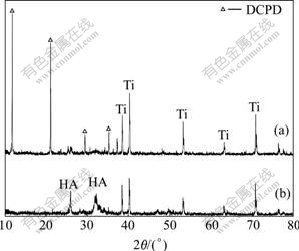
Fig.3 XRD patterns of coatings before(a) and after (b) alkali treatment
However, the previous researches[1, 9] in this field have concentrated on the deposition of the hydroxyapatite on nanotubes. It would be better if the authors had given the pictures of the accurate location of crystals nucleation. Therefore, it would thus be of interest to learn how the hydroxyapatite deposited on the nanotubes.
Fig.4 shows SEM micrograph of the surface and the section of the deposited coating after electrodeposition at the initial stage. With increasing the deposition time, the nanometer DCPD crystals were connected together to form a continuous coating on titanium oxide nanotube array(Fig.5). Fig.6 shows the typical morphology of deposited coating after alkali treatment. When the coating was removed, some crystals were still inserted in the tubes and the space of the tubes(Fig.7). This formed an anchor dropping effect originated from the rough surface treatment of titanium alloy substrate. And the bonding strength of the interface between the HA coatings and the substrate was 7.41 MPa when the substrate was treated by anodic oxidation, compared with 3.29 MPa without anodic oxidation.

Fig.4 SEM micrograph of surface of deposited coating at initial stage

Fig.5 SEM micrograph of surface of deposited coating after electrodepositon for 30 min

Fig.6 Morphology of coating by electrochemical deposition for 2 h after alkali treatment

Fig.7 SEM micrograph of tensile tested surface after removing coating
The nanotubes as shown in Fig.1 have contributed to the increase of surface roughness and surface area provided for the subsequent deposition of coating. In addition, since the nanotube structure of TiO2 should serve as anchorage of HA top coating, the toughness of TiO2 was likely to play a role in the bonding strength. Rougher surface provided by TiO2 promoted mechanical interlocking between HA and TiO2. Although it still needed extra work to gain further evidences, the toughness of TiO2 has presumably been considered to aid in the whole bonding strength.
3.3 Corrosion behavior in SBF
Results of potentiodynamic polarization are given in Fig.8. Anatase titania exhibited a better electrochemical behavior than pure titanium and amorphous titania. It could prevent the deterioration of the metallic substrate.
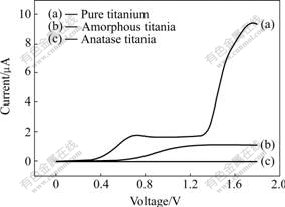
Fig.8 Phenomenon of polarization in SBF
4 Conclusions
A vertically aligned nanotube arrays of titanium oxide on the surface of titanium substrate have been prepared by anodization. HA coating is deposited on anodized titanium by electrodeposition in an electrolyte containing calcium and phosphate ions. The surface morphology of TiO2 nanotube promotes mechanical interlocking between HA and TiO2. The deposited crystals are anchored in and between the tubes. The insert mode makes a contribution to the improvement of bonding strength.
References
[1] ZHANG Y Y, TAO J, PANG Y C, WANG W, WANG T. Electrochemical deposition of hydroxyapatite coatings on titanium[J]. Trans Nonferrous Met Soc China, 2006, 16(3): 633-637.
[2] AFSHAR A, YOUSEFPOUR M, YANG X D, LI X D, YANG B C, WU Y. CHEN J Y, ZHANG X D. Investigation of morphology and bioactive properties of composite coating of HA/vinyl acetate on pure titanium[J]. Materials Science and Engineering B, 2006, 128: 243-249.
[3] YOUSEFPOUR M, AFAHAR A, YANG X D, LI X D, YANG B C, WU Y. Nano-crystalline growth of electrochemically deposited apatite coating on pure titanium[J]. Journal of Electroanalytical Chemistry, 2006, 589: 96-105
[4] PARK J H, LEE D Y, OH K T, LEE Y K, KIM K M, KIM N. Bioactivity of calcium phosphate coatings prepared by electrodeposition in a modified simulated body fluid[J]. Materials Letters, 2006, 60: 2573-77.
[5] OKIDO M, KURODA K, ISHIKAWA M, ICHINO R, TAKAI O. Hydroxyapatite coating on titanium by means of thermal substrate method in aqueous solutions[J]. Solid State Ionics, 2002, 151(1/4): 47�C 52.
[6] PARK J H, LEE Y K, KIM K M, KIM K N. Bioactive calcium phosphate coating prepared on H2O2-treated titanium substrate by electrodeposition[J]. Surface & Coatings Technology, 2005, 195: 252- 257
[7] XIAO X F, LIU R F, ZHENG Y Z. Characterization of hydroxyapatite/titania composite coatings codeposited by a hydrothermal-electrochemical method on titanium[J]. Surface & Coatings Technology, 2006, 200(14/15): 4406-13.
[8] KIM H W, KOH Y H, LI L H, LEE S, KIM H E. Hydroxyapatite coating on titanium substrate with titania bufferlayer processed by sol-gel method[J]. Biomaterials, 2004, 25: 2533-2538.
[9] RAJA K S, MISRA M, PARAMGURU K. Deposition of calcium phosphate coating on nanotubular anodized titanium[J]. Materials Letters, 2005, 59: 2137-2141.
[10] YEN S K, LIN C M. Cathodic reactions of electrolytic hydroxyapatite coating on pure titanium[J]. Materials Chemistry and Physics, 2002, 77: 70-76.
[11] ZHANG Q Y, LENG Y, XIN R L. A comparative study of electrochemical deposition and biomimetic deposition of calcium phosphate on porous titanium[J]. Biomaterials, 2005, 26: 2857-2865.
[12] HU H B, LIN C J, HU R, LENG Y. A study on hybrid bioceramic coatings of HA/poly(vinyl acetate) co-deposited electrochemically on Ti-6Al-4V alloy surface[J]. Materials Science and Engineering C, 2002, 20(1/2): 209-14.
[13] BAN S, HASEGAWA J. Morphological regulation and crystal growth of hydrothermal electrochemically deposited apatite[J]. Biomaterials, 2002, 23: 2965-72.
[14] SPIVAK J M, RICCI J L, BLUMENTHAL N C, ALEXANDER H. A new canine model to evaluate the biological response of intramedullary bone to implant materials and surfaces[J]. J Biomed Mater Res, 1990, 24(9): 1121-49.
[15] YOU C, OH S, KIM S. Influences of heating condition and substrate-surface roughness on the characteristics of sol-gel-derived hydroxyapatite coatings[J]. Journal of Sol-Gel Science and Technology, 2001, 21(1-2): 49-54.
[16] OH S, JIN S. Titanium oxide nanotubes with controlled morphology for enhanced bone growth[J]. Materials Science and Engineering C, 2006, 26: 1301-1306.
[17] CAI Q Y, YANG L X, YU Y. Investigations on the self-organized growth of TiO2 nanotube arrays by anodic oxidization[J]. Thin Solid Films, 2006, 515: 1802-1806.
![]() Foundation item: Project(BK2004129) supported by the Natural Science Foundation of Jiangsu Province, China
Foundation item: Project(BK2004129) supported by the Natural Science Foundation of Jiangsu Province, China
Corresponding author: TAO Jie; Tel: +86-25-52112900; E-mail: taojie@nuaa.edu.cn
(Edited by YANG Bing)