
Effect of minor Sc and Zr on microstructure and mechanical properties of
Al-Zn-Mg-Cu alloy
HE Yong-dong(贺永东), ZHANG Xin-ming(张新明), YOU Jiang-hai(尤江海)
School of Materials Science and Engineering, Central South University, Changsha 410083, China
Received 26 September 2005; accepted 24 January 2006
Abstract:A series of Al-8.2Zn-2.1Mg-2.3Cu based as-cast alloys and some plates with thickness of 4 mm containing minor Sc and Zr were prepared. The effect of joint addition of minor Sc and Zr on microstructure and mechanical properties of Al-Zn-Mg-Cu alloys were investigated by using OM, SEM with EDS and TEM. The results show that by adding 0.18% Zr (mass fraction) in the cast alloy, the grains can be refined to a certain degree, and by adding 0.18% Sc a little as well. Adding Sc and Zr can generate strong grain refinement effect and obtain a fine equiaxed grain structure, because primary Al3(ScxZr1-x) precipitation forms in front of the α-Al grains. The microstructure and tensile test results show that 0.18% addition of Zr does not bring higher tensile strength and elongation to the alloy of adding Sc, but a better inhibition to recrystallization. Recrystallization inhibiting effect is the strongest in the alloys with joint addition of Sc and Zr. When the content of Zr is unchanged, the strength and elongation of the alloys increase with increasing Sc addition. The increase of strength and elongation in the alloys is related to the refine grain strengthening, precipitation particles strengthening and substructure strengthening principles.
Key words:Sc; Zr; Al-Zn-Mg-Cu alloy; microstructure; strengthening mechani
1 Introduction
The microstructure and properties of aluminium alloys are strongly affected by adding small quantities of scandium. Minor Sc may improve the temperature of recrystallization and fracture toughness, decrease the sensitivity of stress corrosion cracking[1-3]. The alloy containing minor Sc precipitates primary Al3Sc particles from the melt, numerous potent heterogeneous nucleis are dispersed in the melt, and becomes active and nucleate α-Al during solidification[4, 5]. Al3Sc particle with the structure of LI2 precipitated from the matrix during homogenization and solution, pins dislocation, restricts the nucleation of recrystallization, increases the mechanical properties and improves weldability in the aluminum alloys[6,7]. The particle, as a stable heterogeneous nucleus, intensively refines grains of aluminium, but whether forming ScAl3 primary phase or not determines the effect of grain refinement with addition of Sc to Al-Zn-Mg-Cu alloys, and the concentration of Sc in the melt must exceed the critical limit in order to refine the grain size. These would increase the cost of fabricating the alloy and affect the investigation and application of the alloy containing trace Sc[8-10]. The effect of Sc is amplified by the simultaneous addition of Zr as precipitating the complex phase of Al3(Sc1-xZrx). It is apparent that, the addition of Sc and Zr play a critical role in Al-Zn-Mg-Cu based alloys in providing the precipitating phase which impedes grains growth[11]. Due to the presence of Al3(Sc1-xZrx) precipitates, the alloys retain their deformed microstructure even when annealed at very high temperatures[12]. Deformation restoration is proceeded by recovery and it is very difficult to obtain a fully recrystallized microstructure. By combining Al3(Sc1-xZrx) precipitation hardening and the stable deformed substructure, different microstructures can be obtained. As a result, the alloy containing minor Sc and Zr exhibits unusual physical and mechanical properties [13]. At present, joint addition of minor Sc and Zr to Al-Mg or Al-Zn-Mg based alloys is among the most frequent studies, but investigates on the microstructure and mechanical properties of Al-8.2%Zn-2.1%Mg-2.3%Cu based alloys are scarce, and its refinement mechanism is still not clear yet[14]. In this paper, the relationship between the mechanical behavior and microstructure characteristics of Al-Zn-Mg-Cu based alloys with and without Sc and Zr is investigated to understand the mechanism of grain refining and dispersive precipitation.
2 Experimental
The nominal compositions of the studied alloys are listed in Table 1. The starting materials were 99.8%Al, 99.99%Cu, 99.92%Zn, 99.5%Mg, Cu-10%Zr and Al- 3.6%Sc master alloys. The alloys were melted in clay-bonded-graphite crucible by a closed electric heat- treating furnace and then cast into chill molds. After homogenization at 400 ℃ for 5 h, 450 ℃ for 20 h and 470 ℃ for 12 h, the ingots were quenched by cold water, and held at 420 ℃ for 2 h, then hot rolled to 4 mm plates. Tensile samples were taken from the hot-rolled plates. Before testing, the samples were solution treated at 470 ℃ for 2 h and then held at 480 ℃ for 1 h, finally quenched by cold water. The ageing process is shown in Fig.1. The specimens of metallograph were divided into three groups. One was anode coating after being electropolished and then observed the grain size of the cast alloy by OM. Grain size analysis was carried out using linear intercept method. The second was etched with modified keller’s reagent (2 mL HF, 3 mL HCl, 20 mL HNO3) and then observed the pattern and distribution of the second-phase particles by OM and SEM. The composition of the second-phase particles were quantitatively analyzed by spectrometer. Thin specimens for TEM were prepared by twin-jet polishing with an electrolyte solution consisting of 30% HNO3 and 70% methanol below -20℃. The foils were examined on TecnaiG220 electron microscope.
Table 1 Chemical composition of alloys(mass fraction, %)


Fig.1 Treatment process for Al-Zn-Mg-Cu-Xi alloys
3 Results and discussion
3.1 Grain refining and its microstructure
Fig.2 shows the grain structure of as-cast alloys. It can be seen that the grain size of Al-Zn-Mg-Cu without trace element(Fig.2(a)) is about 200-300 μm, that with adding 0.18% Zr(Fig.2(b)) is about 80-130 μm, and that with adding 0.18% Sc gets no refining efficiency Cu (Fig.2(c)). But by joint adding 0.10%Sc and 0.20% Zr, the average grain size decreases to 25-30 μm; when joint adding 0.30%Sc and 0.10%Zr, the average grain size is as fine as 20-25 μm. And in special when joint adding 0.30%Sc and 0.18%Zr the finest equiaxed grains of 10-15 μm is gotten (Fig.1(d)). So joint addition of Sc and Zr has better grain refining effect compared to addition of single Sc or single Zr.

Fig.2 Polarized light optical micrographs of as-cast alloys: (a) Al-Zn-Mg-Cu; (b) Al-Zn-Mg-Cu-0.18Zr; (c) Al-Zn-Mg-Cu-0.18Sc; (d) Al-Zn-Mg-Cu-0.30Sc-0.18Zr
From Fig.3, it is evident that the dendritic structure is coarse and a large number of second-phase particles exist within the grains and on the grain boundaries for the alloy with adding 0.18%Zr. For the alloy of adding 0.18%Sc, the dendritic structure is still coarse but the second-phase within the grains disappears. Adding minor Sc and Zr to the alloys obtains equiaxed structure, the dendrite structure and the second-phase within the grains disappears completely.
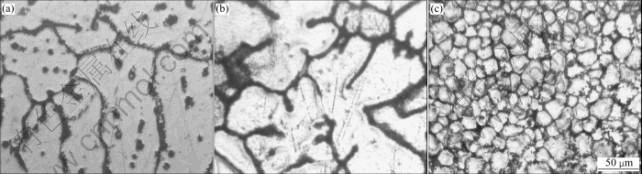
Fig.3 Micrographs of as-cast alloys: (a) Al-Zn-Mg-Cu- 0.18Zr; (b) Al-Zn-Mg-Cu-0.18Sc; (c) Al-Zn-Mg-Cu-0.15Sc- 0.18Zr
Fig.4(a) presents a lot of the second phases within the central grains of the alloy. Fig.4(b) shows that this kind of particles is rectangular and actually composite particle. EDS analysis indicates that Zr is rich in the center of the particle and Sc enriches in the outside shell relatively. The chemical formula of the compound can be written as Al3(Scx, Zr(1-x)). It is suggested that Al3Zr precipitates from the melt at first, and is followed by Al3Sc precipitation on the Al3Zr surface, and then Al3Zr precipitation on the Al3Sc surface and so on, finally forms much layers containing Al3Zr or Al3Sc complex particle. The second phase is the nucleus of the α-Al during solidification.
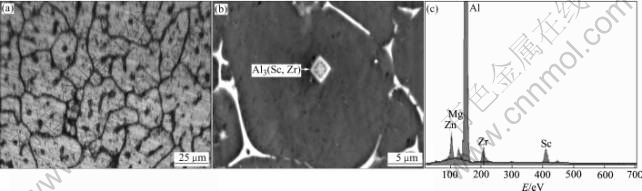
Fig.4 Grain refining mechanism analysis of Al-Zn-Mg-Cu- 0.30Sc-0.10Zr alloy: (a) Particles within grains; (b) Second phase nucleates α-Al; (c) EDS of second phase
The binary Al-Zr equilibrium phase diagram is dominated at the Al-rich end by a peritectic reaction between liquid and Al3Zr intermetallic phase, and Zr solid solubility is about 0.28% at the equilibrium temperature [11]. Solid solubility of Zr maybe reaches up 2.0%-2.5% under the condition of non-equilibrium solidification. In practical experience, when the content of Zr excesses 0.16%, the primary Al3Zr intermetallic phase will directly precipitate from the melt. The primary phase belongs to tetragonal system, its melting point is about 1 850 K, lattice constant is a=4.05×10-10 m, disregistry to the matrix is about 2.9% and that of LI2 Al3Zr is only 0.70%, lattice of Al3Zr is similar to α(Al), which are all effectively heterogeneous. Adding singly zirconium to aluminium melt, the refinement mechanism varies with the content of Zr. When the concentration of Zr is lower, it mainly interacts with atom cluster, forms steady atom clusters, and finally grows up and becomes nuclei; when the concentration of Zr is higher, mechanism for the formation of α(Al) is based on the peritectic reaction: L+Al3Zr=α(Al) (solid solution). The binary Al-Sc equilibrium phase diagram is dominated at the Al-rich end by a eutectic reaction between α(Al) and Al3Sc phase, there is a narrow freezing rang for hypoeutectic alloys, and the eutectic composition is at the low Sc level 0f 0.55%[13]. Under the condition of non-equilibrium solidification, Sc mainly trends to form Al-Sc solid solutions, only a little form Al3Sc primary phase, which belongs to face central cubic system of type AuCu3. The melting point is about 1 593 K, lattice constant is a=4.106×10-10 m, disregistry to α(Al) is about 1.5%[15]. Al3Sc phase is a stable heterogeneous particle and intensively refines grain of aluminium, but whether forming ScAl3 primary phase determines the effect of grain refinement with adding Sc to Al-Zn-Mg- Cu alloys. The concentration of Sc in the melt must exceed the critical limit in order to refine the grain size, otherwise, it is unefficient. The solvent/solute atomic size ratio and chemical affinity have an influence on concentration fluctuation and microstructure wave in the melt. With joint addition of trace Sc and Zr, concentra- tion fluctuation and microstructure wave in the melt enhance interpolymerization of Sc and Zr. Addition Zr to alloys containing Sc generates Al-Sc-Zr crystal cell in the melt, and the nearest Sc―Zr bond has the highest bond energy of covalent bond in Al-Zn-Mg-Cu-Sc-Zr alloy, and has a great influence on combined copolymeri- zation of Sc and Zr. Concentration of Sc and Zr in the solid solution alloy is higher, which leads to more intensive interpolymerization of Sc and Zr, Sc and Zr much easier to generate rich-region in the melt. With increasing the concentration of Sc and Zr, the degree of rich-region becomes highly intensive, and the rich-region becomes unstable. At first, atom Zr replaces atom Al of crystal cell of face centre cubic to form Al-Zr crystal cell, which promotes atom Sc substitute for atom Al of the unit cell, and generates Al-Sc-Zr unit cell, eventually generates insoluble particle of Al3(ZrxSc1-x), dispersed into melt. The particle belongs to face central cubic system, lattice constant is similar to α(Al), and dis- registry to the α(Al) is only about 1%-2%. This com- pound is of small size, high density and homogeneous distribution, and its size is 1-15 nm. When it solidified normally, it is perfect heterogeneous. For Al-8.2Zn- 2.1Mg-2.3Cu-0.30Sc-0.18Zr alloy, the grain refining mechanism can be understood from the experimental.
3.2 Dispersive precipitation in as-cast alloys
The TEM microstructures of as-cast alloys containing Zr is shown in Fig.5(a). Many dispersive precipitates in the matrix can be seen, pea-like strain contrast of the precipitates in Fig.5(a). The TEM microstructures of as-cast alloys containing minor Sc is shown in Fig.5(b). Many dispersive precipitates in the matrix can be seen, and these particles look like U-shaped. Under the condition of simultaneously adding of Sc and Zr, not only the precipitate content increases but also the precipitate size decreases. Coffee bean-like strain contrast of the precipitates and EDP in Fig.5(d) indicate that those precipitates are coherent with the matrix. According to Al-Sc, Al-Zr, Al-Sc-Zr phase diagram, the precipitates should be Al3Zr in Fig.5(a), Al3Sc in Fig.5(b), Al3(Scx, Zr(1-x)) in Fig.5(c). Fig.5(d) shows the super-lattice structure of LI2 Al3Sc. In general, It is hard to see the dispersive precipitates in as-cast Al alloys, but so many precipitates can be seen in the investigated alloys. Blake suggested that the incubation period of coherent Al3Zr, Al3Sc, Al3(Scx, Zr(1-x)) precipitates are much shorter than that in traditional Al alloy. In order to verify this suggestion, following experiment was performed. When taking the samples of Al-Zn-Mg-Cu-0.18%Zr, Al-Zn-Mg-Cu- 0.18%Sc, Al-Zn -Mg-Cu-0.20%Sc-0.18%Zr alloys from cast mold in 10-2 s just after solidification into cold water, no above precipitates can be found on TEM. The results show that dispersive particles mentioned above precipitate from Al solid solution during cooling of solidification. Copper is the main alloying element in the studied alloy, which can interact with element Sc and form W phase (ScCu6.6-4Al5.4-8), which is coarsen, insoluble and primary phase, and harms the properties of the matrix. Rokhlin and Dobatkina suggested that Al-Zn-Mg-Cu-Sc- Zr system consists of six components. At present, the investigation of the phase diagram is limited to a few regions from the point of view of industry for improving the properties of the alloys. From the phase diagram of Al-Zn-Mg-Cu-Sc-Zr, we know that there would be no W phase existing when the concentration of Mg is above 0.8% and Cu keeping in 2.0% fixation and temperature below 200 ℃. The studied alloy is suitable for the conditions.

Fig.5 Dispersive precipitions of Al3Zr and Al3Sr and Al3(Sc, Zr) in studied alloys: (a) Al-Zn-Mg-Cu-0.18%Zr; (b) Al-Zn-Mg-Cu- 0.18%Sc; (c) Al-Zn-Mg-Cu-0.20%Sc-0.18%Zr; (d) EDP corresponding to Fig.5(c)
3.3 Recrystallization behaviour
The microstructure of as-hot rolled alloys after solution treatment is shown in Fig.6. Grain structure of Al-Zn-Mg-Cu-0.18Zr is the mixture with mainly fiber structure and a small amount of equiaxial grains, showing that the partial recrystallization has been occurred (Fig.6(a)). Grain structure of Al-Zn-Mg-Cu- 0.18%Sc alloys is equiaxial grains showing complete recrystallization of the alloy occurred during hot rolling. Rystads suggested that the alloy containing minor Zr would occur recrystallization where the density of Al3Zr dispersive precipitates is relatively lower during plastic deformation and following solution treatment, and this phenomenon is also found in our experiments(shown in Fig.6(b)) and is thought to be related to the atom zirconium segregation within the grains or on the grain boundary. This segregation induces a strong solute depletion of Zr on the matrix. The area which is adjacent the rich region of Zr would come forth depletion of solid solution, resulting in a decrease of the solute available for precipitation, the dispersive precipitates of Al3Zr would be scare in the poor region, and the ability of restricting nucleation of recrystallization and growth is weak and, therefore, inhibiting subsequent fine-scale uniform precipitation during hot-rolling and ageing, a reduction in the restricting potential. The recry- stallization would first occur in the poor region of Al3Zr precipitates. Grain structure with adding 0.30%Sc and 0.18%Zr of the alloys presents a complete fiber characteristics, which shows that no recrystallization occurs during hot-rolling. Recrystallization behavior of the alloys is related to the dispersive precipitates existed in the alloys. On one hand, Al3Zr and Al3Sc and Al3(ScxZr1-x) particles are intense obstacle to the movement of dislocation didn’t free themselves from powerful pinning dislocation(Fig.7). On the other hand, the interface energy between Al3Zr, Al3Sc or Al3(ScxZr(1-x)) and matrix is very low and very stable because those precipitates are coherent with the matrix. Under this condition, it is very difficult for grain boundaries to pass through the low energy interface. Dislocations and the substructure are strongly pinned, so recrystallization of the Al-Zn-Mg-Cu-030%Sc-0.18%Zr alloy is completely restricted during solution treatment. In particular, the initial high concentration of the solute elements including Sc and Zr at the dendrite edges is predicted to promote dispersoid formation in these regions, and leads to a substantial reduction in the width of the dispersoid free zone. This is expected to have an influence on the fraction of recrystallization observed in solution treated plate material. However, this effect may be hidden by the joint addition of minor transition elements which will affect the distribution of the solute elements during solidification and homogenization and solution treatment.

Fig.6 Microstructures of as-hot rolled studied alloys: (a) Al-Zn- Mg-Cu-0.18Sc; (b) Al-Zn-Mg-Cu-0.18Zr; (c) Al-Zn-Mg-Cu- 0.10Sc-0.18Zr; (d) Al-Zn-Mg-Cu- 0.20Sc-0.18Zr; (e) Al-Zn- Mg-Cu-0.30Sc-0.18Zr
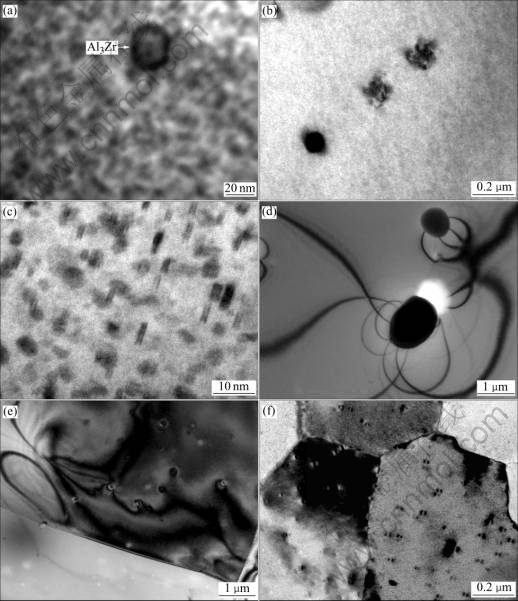
Fig.7 Dispersive precipites of Al3Zr and Al3Sc and Al3(Sc, Zr) on studied alloys after ageing: (a) Al-Zn-Mg-Cu-0.18%Zr; (b)-(e) Al- Zn-Mg-Cu-0.18%Sc; (c) Al-Zn-Mg-Cu-0.20%Sc-0.18%Zr
Tensile properties of as-hot rolled alloys after solution and ageing are listed in Table 2. It shows that by adding of 0.18%Zr to the alloy, the strength increment caused by simultaneous adding Zr (mass fraction) is up to 99 MPa and the elongation is up to 3.6%.
Table 2 Tensile properties of studied alloys after ageing at 120℃ for 24 h
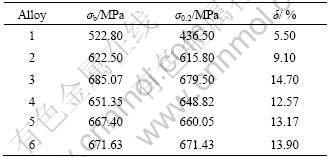
By adding 0.18%Sc to the alloy, the strength increases to 162.27 MPa and the elongation is up to 9.2%. Joint addition of 0.10%Sc and 0.18%Zr increase the strength of the alloy strongly and the strength increment caused by adding of Sc and Zr is larger than that of adding Zr singly. The strength increment caused by simultaneous adding Sc and Zr is up to 128.55 MPa and the elongation is up to 7.07%. Joint addition of 0.20%Sc and 0.18%Zr gets the higher strength than that of alloy 4. The strength increment is up to 144.60 MPa and the elongation is up to 7.67%. Joint addition of 0.30%Sc and 0.18%Zr gets the higher strength than that of alloy 5. The strength increment is up to 148.83 MPa and the elongation is up to 8.4%. In case of keeping the content of Zr unchangeable and increasing the amount of Sc, the strength of the alloys increases from 622.50 MPa to 679.63 MPa. Mechanical behavior is closely related to the microstructures of the alloys. Adding Zr to Al-Zn-Mg-Cu alloy can refine grain size to a certain degree, produces dispersive Al3Zr precipitates and obtains mainly the fiber structure after hot-rolling, which leads to strengthening of the alloy. Adding Sc to Al-Zn-Mg-Cu alloy can produce dispersive Al3Sc precipitates (shown in Fig.7(b) which act as the basis of GP zone and η′ phase(Fig.7(c)). Al3Sc particles with the structure of LI2 precipitating from the matrix during homogenization and solution treatment, pin dislocation restricts the nucleation of recrystallization, which leads to strengthening of the alloy. The dislocation structure of the studied alloy is shown in Figs.7(d) and (e). Joint addition of Sc and Zr to Al-Zn-Mg-Cu alloy not only generates strong grain refinement and obtains a fine equiaxed grain, but also produces dispersive Al3(ScxZr(1-x)) precipitates and leads to a complete fiber structure after hot-rolling. Obviously, the strength increment produced by simultaneously adding Sc and Zr is mainly attributed to refine grain strengthening, Al3(ScxZr(1-x)) dispersive strengthening and substructure strengthening mechanisms (shown in Fig.7(f)).
4 Conclusions
1) Adding 0.18%Zr to Al-Zn-Mg-Cu cast alloy refines its grain size to a certain degree, and the average grain is about 80-130 μm. And the grain structure of the as-hot alloy is mixture of fiber and small amount of equiaxial grains. Partial recrystallization occurs during hot-rolling and solution. The strength increases by 99 MPa and the elongation increases by 3.6%.
2) Adding 0.18%Sc to Al-Zn-Mg-Cu alloy has a little effect on its refining effect of grain size. And the grain structure is equiaxial grains, showing complete recrystallization of the alloy occurred during hot rolling. The tensile strength increases by 166 MPa and the elongation increases by 9.2%. The strengthening mechanism is mainly Al3Sc precipitating, the particle pinning dislocation and the dislocation cutting dispersive precipitates strengthening.
3) Joint addition of Sc and Zr to Al-Zn-Mg-Cu alloy generates strong grain refinement and obtains a fine equiaxed grain. The grain structure presents a complete fiber structure, the strength is up to 148.83 MPa and the elongation is up to 7.67%. When the content of Zr is unchanged, the strength and elongation of the alloy improve with the increase of the Sc addition. The strengthening mechanism is mainly refine grain strengthening, substructure strengthening and dislocation cutting dispersive dispersoids strengthening.
References
[1] NAKAI M, ETO T. New aspects of development of high strength aluminum alloys for aerospace applications [J]. Materials Science and Engineering, 2000, A285: 62-68.
[2] WU Y L, FROES F H, ALVAREZ A, LI C G, LIU J. Microstructure and properties of a new super-high-strength Al-Zn-Mg-Cu alloy C912 [J]. Materials and Design, 1997, 18(4): 211-215.
[3] PENG Kai-ying, CHEN Wen-zhe, ZHANG Hao-gao, QIAN Kuang-wu. Features of dynamic strain ageing in high strength Al-Zn Mg-Cu alloy [J]. Materials Science and Engineering, 1997, A234-236: 138-141.
[4] GAO Ying-jun, ZHONG Xia-ping, LIU Hui, WU Wei-ming. Valence electronic structures of Al-Mg alloy with minor Sc and Zr and its effect on grain refinement [J]. Guangxi Sciences, 2003, 10(1): 32- 35.
[5] KAIBYSHEV O A, FAIZOVA S N, HAIRULLINA A F. Diffusional mass transfer and superplastic deformation [J]. Acta Mater, 2000, 48: 2093-2100.
[6] DESCHAMPS A, BR?CHET Y. Nature and distribution of quench-induced precipitation in an Al-Zn-Mg-Cu alloy [J]. Scripta Materialia, 1998, 39(11): 1517-1522.
[7] NAKAI M, ETO T. New aspects of development of high strength aluminum alloys for aerospace applications [J]. Materials Science and Engineering A, 2000, A285: 62-68.
[8] ROKHLIN L L, DOBATKINA T V. Investigation of phase equilibria in alloys of Al-Zn-Mg-CuZr-Sc system [J]. Journal of Alloys and Compounds, 2004, 367: 10-16.
[9] HANSEE LIV, GJONNES J, et al. Hrem study and structure modelling of the η′ phase, the harding precipitates in commercial Al-Zn-Mg alloy [J]. Acta Mater, 1999, 47: 2651-2659.
[10] IWAMURA S, MIURA Y. Loss in coherency and coarsening behavior of Al3Sc precipitates [J]. Acta Materialia, 2004, 52: 591-600.
[11] WARD D, BEERNSEN D J. Grain structure and quench-rate effects on strength and toughness of AA7050 Al-Zn-Mg-Cu-Zr alloy [J]. Matallurgical Transaction A, 1995, 26A(9): 2481-2484.
[12] XIE You-hua. Effect of Minor Zr on the Microstructure and Mechanical Properties of ultra Aluminium Alloys [D]. Beijing: Bejing Voyage Academe, 1999: 6-20.
[13] HE Yong-dong, ZHANG Xin-ming. Refinement mechanism of trace Cr, Mn, Ti and Zr as-cast 7A55 alloys [J]. The Chinese Journal of Nonferrous Metals, 2005, 15(10): 1594-1601.
[14] HE Yong-dong, ZHANG Xin-ming. Effect of minor Cr, Mn, Ti and Zr on microstructure and mechanical properties of Al-Zn-Mg-Cu based alloy [J]. The Chinese Journal of Nonferrous Metals, 2005, 15(12): 1917-1924. (in Chinese)
[15] HE Yong-dong, ZHANG Xin-ming, CHEN Jian-mei, TANG Jian-guo. Refinement mechanism of trace Sc and Zr on as-cast 7A55 alloys [J]. Journal of Central South University(Science and Technology), 2005, 36(6): 919-923. (in Chinese)
Foundation item: Project(2005CB623706) supported by the National Advanced Technology Research and Development Committee of China
Corresponding author: HE Yong-dong; Tel: +86-935-8827599; E-mail: hydongq@126.com
(Edited by LI Xiang-qun)