Ti-Ni-Oϵ��ͼ�ļ��㼰����TiNi�Ͻ�绯ѧ�����е�Ӧ��
��Դ�ڿ����й���ɫ����ѧ����Ӣ�İ棩2011���8��
�������ߣ����� ��᰽� ���� ³�۸� ���غ�
����ҳ�룺1808 - 1816
�ؼ��ʣ�TiNi�Ͻ𣻼�����ͼ��Ti-Ni-Oϵ���绯ѧ����
Key words��TiNi alloy; Calphad; Ti-Ni-O system; electrochemical deoxidation
ժ Ҫ�����������е�ʵ�����ݣ���CALPHAD������Ti-Ni-O��Ԫϵ���������Ż�������Ti-Ni-O��Ԫϵ��1 173��1 273 Kʱ�ĵ��½��档������������������ʵ�����Ǻϡ�ͨ���Ż����һ����Ǣ������ѧ������ʹ�������½��������ѧ���ļ����Ϊ���ܣ�Ϊ�����Ͻ�������ṩ����ָ����
Abstract:
The Ti-Ni-O ternary system was assessed by means of Calphad method using ternary experimental data in previous study. Isothermal sections at 1 173 and 1 273 K were calculated. The result shows that the present calculated results are in good agreement with most of the experimental results. The consistent model parameter set determined in this work may provide theoretical guidance for the deoxidation of TiNi alloy.

QIU Ai-tao, LIU Lan-jie, PANG Wei, LU Xiong-gang, LI Chong-he
Shanghai Key Laboratory of Modern Metallurgy and Materials Processing,
Shanghai University, Shanghai 200072, China
Received 18 October 2010; accepted 10 December 2010
Abstract: The Ti-Ni-O ternary system was assessed by means of Calphad method using ternary experimental data in previous study. Isothermal sections at 1 173 and 1 273 K were calculated. The result shows that the present calculated results are in good agreement with most of the experimental results. The consistent model parameter set determined in this work may provide theoretical guidance for the deoxidation of TiNi alloy.
Key words: TiNi alloy; Calphad; Ti-Ni-O system; electrochemical deoxidation
1 Introduction
Shape memory materials have attracted considerable attention recently as functional materials in a variety of industrial and medical applications. They have also been considered as important smart materials because of their ability to perform both sensing and actuating functions [1]. Among the practical shape memory materials, TiNi-based alloys are considered to be one of the most important shape memory alloys because of their excellent shape memory effect [2-3], large pseudo elasticity [4-5], high chemical corrosion resistance and good biocompatibility. Oxygen is the interstitial solute element in TiNi alloys, which must be strictly controlled, because it strongly influences the microstructure and mechanical properties.
The Ti-Ni-O system is an interesting system from the view point of the controlling of oxygen content and the deoxidation of TiNi alloy. Therefore, critically evaluated thermodynamic parameters of the Ti-Ni-O system are urgently needed in the deoxidation of TiNi alloy. The first experimental studies of the phase equilibria in the Ti-Ni-O system were performed in 1955 by ROSTOKER [6]. But even now, the Ti-Ni-O phase diagram is not known in detail, no Ti-Ni-O
thermodynamic assessment has been published.
The purpose of the present work is to thermodynamically evaluate the Ti-Ni-O ternary system so that the calculation of isothermal sections and other thermodynamic quantities become possible in the whole composition and temperature range. The present work is based on the thermodynamic descriptions of the binary systems, Ti-Ni by KEYZER et al [7], Ti-O by CANCAREVIC et al [8] and Ni-O by KOWALSKI and SPENCER [9]. The NiO-TiO2 pseudo-binary systems were assessed. Then the thermodynamic parameters of the Ti-Ni-O system were modeled, including the ternary interaction parameters of BCC, HCP structured alloy and liquid, and some interaction parameters involving the third element, and then constructed its phase diagrams at 1 173 and 1 273 K, respectively. The the oxygen potential was calculated using thermodynamic parameters of the Ti-Ni-O ternary system, and the oxygen potential can be transformed into electrical potential using Nernst equation.
2 Review of literature information
2.1 Binary systems
The Ti-Ni system was assessed previously [10-13]. Both BELLEN et al [12] and TANG et al [13] described the B2 phase as an ordered form of the A2 phase. In the assessment by TANG et al [13], the B19' phase was also included. The upadted CALPHAD-type thermodynamic assessment was studied by KEYZER et al [7]. It was considered that the Gibbs energy expression for the pure elements in the Ni3Ti structure should refer to the DHCP structured instead of the HCP structured phase. After changing the Gibbs energy of the pure element, parameters for Ni3Ti were reassessed by KEYZER et al [7]. NiTi, Ni3Ti and Ti2Ni phases were described by the two-sublattice model in thermodynamic assessment [7]. Because the optimization showed better agreement with experiments, thermodynamic parameters [7] for the Ti-Ni system were adopted in this work.
The Ti-O system was recently assessed [8, 14-15]. FISCHER [14] published a thermodynamic calculation of the system neglecting Magneli phases and oxygen solubility of rutile. WALDNER and ERIKSSON [15] presented a complete Ti-O phase diagram from pure titanium to gaseous oxygen including Magneli phases. Most extensive evaluation of Ti-O system was done by CANCAREVIC et al [8]. The results agreed well with the experimental data and were complete thermodynamic parameters of this system, the data was used to model the Ti-Ni-O ternary system.
Ni-O system was assessed by TAYLOR and DINSDALE [16] using the ionic two-sublattice model for the description of the liquid phase. Later, the thermodynamic revaluation of the Ni-O system was made by KOWALSKI and SPENCER [9] using an associated solution model for the liquid phase. Complete thermodynamic parameters of Ni-O system were reported in Ref. [9], and the data was used to model Ti-Ni-O ternary system.
2.2 NiO-TiO2 pseudo-binary system
The first systematic investigation in NiO-TiO2 pseudo binary system at high-temperature was carried out by BIRNBAUM and SCOTT [17], NiTiO3 with ilmenite structure was described as the only binary compound. It was suggested that there also existed a phase with spinel structure, and the composition may be Ni2TiO4 [18]. WERNER and GEBRET [19] considered that the composition should be Ni3TiO5. An improved phase diagram was published in Ref. [20]. The first complete experimental phase diagram of the NiO-TiO2 system was reported [21], as shown in Fig. 1.
The assessment of NiO-TiO2 pseudo-binary systems was scarcely reported. Thus, the NiO-TiO2 system was assessed [22] (Fig. 2).The phase diagram of NiO-TiO2 system was calculated and optimized by means of the CALPHAD method using the experimental data in precious work.
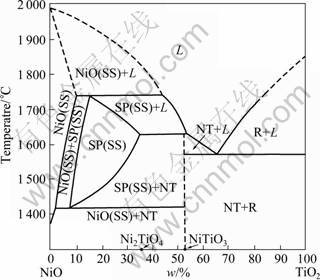
Fig. 1 Phase relations in NiO-TiO2 system according to MUAN [21] (SP-spinel; NT-NiTiO3; R-rutile; L-liquid; SS-solid solution)
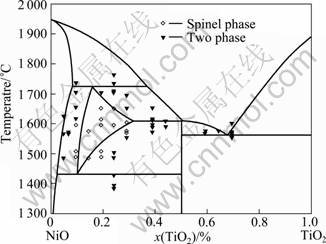
Fig. 2 Calculated NiO-TiO2 phase diagram in this work with experimental points measured by MUAN [21]
2.3 Ti-Ni-O ternary system
Few experimental studies reported Ti-Ni-O system.
ROSTOKER [6] studied the phase relation character- istics of the isothermal section of the Ti-Ni-O system at 1 173 K, the focus was concentrated on the Ti-rich region, twenty-seven alloys were used to delineate phase boundaries in the ternary section. The isothermal section with data points is shown in Fig. 3.
In order to study the influences of oxygen on the properties of TiC-base cermets, HASHIMOTO et al [23] studied the phase relation characteristics of the isothermal section of the Ti-Ni-O system at 1 000 and 2 000 ��C. The isothermal section at 1 000 ��C is shown in Fig. 4. The phase relationships of the Ti-Ni-O system were determined mainly in the region below 50% Ti (molar fraction). At 1 000 ��C, a nickel solid solution (��-phase) was in equilibrium with Ti2O3, Ti3O5, Magneli phases, TiO2, NiTiO3 and NiO.
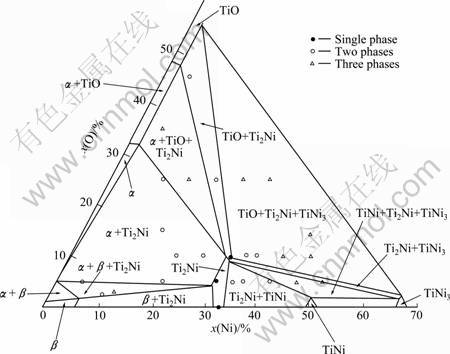
Fig. 3 Experimental phase diagram measured by ROSTOKER at 1 173 K [6]
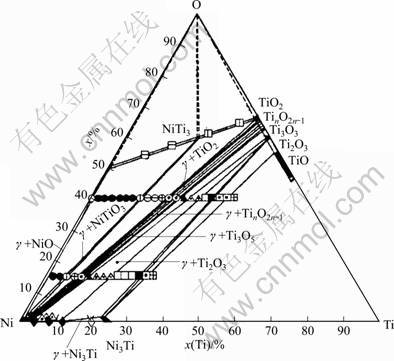
Fig. 4 Experimental phase diagram measured by HASHIMOTO et al at 1 273 K [23]
CHATTOPADHYAY and KLEYKAMP [24] re-examined the isothermal section of Ti-Ni-O system at 1 200 K in the region between Ni(Ti) solid solution and the binary oxides of titanium. The relative partial Gibbs energy of oxygen over the Ti2O3-Ti3O5 region, as well as the Gibbs energy of formation of NiTiO3, Ti3O5 and TiNi3 were determined between 1 100 and 1 300 K by the use of solid electrolyte galvanic cells.
Ti-Ni-O system is an interesting system from the view point of the controlling of oxygen percent and the deoxidation of TiNi alloy. Up to now, no Ti-Ni-O thermodynamic assessment has been published. Therefore, it is of practical interests to model this ternary system based on those experimental results.
3 Thermodynamic modeling
The Gibbs energy functions of all phases are referred to the enthalpy of pure elements in their stable states at 298.15 K under 0.1 MPa. Gibbs energy functions of the pure elements were taken from the SGTE (Scienti?c Group Thermodata Europe) unary database [25].
3.1 Gas phase
The gas phase was described as an ideal mixture containing the species of Ti, TiO, TiO2, O, O2, O3, Ni and NiO. The Gibbs energy of the gas phase is given as
![]() (1)
(1)
where xi is the mole fraction of the species i in the gas phase; ![]() is the standard Gibbs energy of the gaseous species i [26]; R is the gas constant and P0 is the standard pressure of 0.1 MPa.
is the standard Gibbs energy of the gaseous species i [26]; R is the gas constant and P0 is the standard pressure of 0.1 MPa.
3.2 Liquid
The (Ti, O, TiO, TiO1.5, TiO2, Ni and NiO) model was chosen for description of liquid in the present work. The Gibbs energy of the liquid is given as
![]() (2)
(2)
where xi is the mole fraction of the species i in the liquid phase; ![]() is the standard Gibbs energy of the liquid species i; R is the gas constant and GE is the excess Gibbs energy.
is the standard Gibbs energy of the liquid species i; R is the gas constant and GE is the excess Gibbs energy.
The excess Gibbs energy (GE) is given by the general formula using the Redlich�CKister polynomial as
![]()
![]() (3)
(3)
where the first term represents the binary interaction terms, the second one represents the ternary interactions. ![]() is the binary interaction parameter for the i-j binary and Lk is the ternary interaction parameter. Vk is defined as
is the binary interaction parameter for the i-j binary and Lk is the ternary interaction parameter. Vk is defined as
 (4)
(4)
3.3 Interstitial solid solution phases
In Ti-Ni-O ternary system, there existed some
interstitial solid solution phases, such as BCC, HCP and FCC. In the present study, it was assumed that oxygen atoms dissolve interstitially in the BCC ��-Ti and Ni-rich FCC phases as well as in HCP ��-Ti phase. Therefore, these phases were described using a two-sublattice model with formula unit (Ti,Ni)a (O,Va)c. In this model, it was assumed that the metal elements Ti and Ni can substitute for each other on the metal sublattice and oxygen and vacancies on the interstitial sublattice. The subscripts a and c were used to denote the number of sites on each sublattice. In the case of FCC and BCC structred phases, a=c=1; for HCP, a=1 and c=0.5. The Gibbs free energy for one mole of formula unit can be expressed as
![]()
![]() (5)
(5)
![]()
![]() (6)
(6)
where yi denotes the site fraction of component i on the relevant sublattice; 0Gi:Va is the Gibbs energy of pure element i and 0GVa:O is the Gibbs energy of a hypothetical state where all the interstitial sites are filled with oxygen.
3.4 Ti2Ni
In the assessment of Ti-Ni binary system [7], Ti2Ni phase was described using a two sublattice model with formula unit (Ti,Ni) 2(Ti,Ni)1. In the present study, this model was extended into the ternary system by introducing one more sublattice for interstitial components, oxygen and vacancies [27]. In this study, the formula unit becomes (Ti,Ni)2(Ti,Ni)1(O,Va)3. Because Ti2Ni phase possesses the crystal structure that corresponds to FCC, the number of sites on the interstitial sublattice was given a value of 3, which is the same as that in the interstitial solid solution model. The Gibbs free energy for one mole formula unit is expressed as
![]()
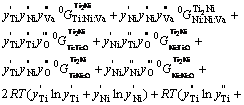
![]() (7)
(7)
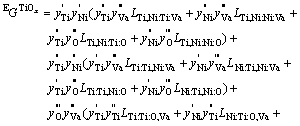
![]() (8)
(8)
3.5 NiTi (BCC_B2)
In the Ti-Ni system, there is a Ti-rich A2 phase which is the disordered form of the B2 phase and the B2 phase can be considered as an ordered state of A2 phase. In KEYZER��s assessment of the Ti-Ni binary system [7], NiTi phase was described using a two sublattice model with formula unit, (Ti,Ni)0.5(Ti,Ni)0.5. This model was extended into the ternary system by introducing one oxygen on each sublattice. In this work, the formula unit of the NiTi phase becomes (Ti,Ni,O)0.5(Ti,Ni,O)0.5. In the case that two or more phases are related by an order-disorder transformation, the Gibbs energy of those phases can be written with one expression [28-29]. The Gibbs free energy of NiTi phase can be described as
![]() (9)
(9)
The first term ![]() (xi) is independent of the ordering state of the B2 phase, it can be described using associated solution model. The second term
(xi) is independent of the ordering state of the B2 phase, it can be described using associated solution model. The second term ![]() (
(![]() ) is the contribution due to long-range ordering, it can be expressed as
) is the contribution due to long-range ordering, it can be expressed as
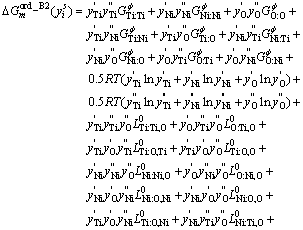
![]() (10)
(10)
3.6 NiTiO3 ternary phases
The ternary phase NiTiO3, which was newly evaluated in this study, was treated as a stoichiometric compound. The (NiO, TiO2) model was chosen for the description of NiTiO3 in the present work. The Gibbs free energy for one mole formula unit can be expressed as
![]() (11)
(11)
where ![]() is the standard Gibbs energy of halite NiO and
is the standard Gibbs energy of halite NiO and ![]() is the standard Gibbs energy of rutile TiO2.
is the standard Gibbs energy of rutile TiO2.
3.7 Other phases
The other phases consisting in the present evaluation of Ti-Ni-O system are Ni3Ti, Ti3O2, ��-TiO, Ti2O3, Ti5O9, Ti7O13, Ti3O5, Ti6O11, Ti8O15, Ti9O17, Ti10O19, Ti20O39, halite, spinel, rutile and TiOx binary phases. The thermodynamic descriptions for these phases were not changed from their original ones. Therefore, Ni3Ti was described using the sublattice model, (Ni,Ti)3(Ni,Ti)1, halite, spinel, rutile and TiOx phases were described using associated solution model and all the other binary phases were treated as stoichiometric compounds.
4 Optimization of thermodynamic parameters
All the thermodynamic parameters are optimized by Pan-optimizer included in the Pandat Phase Equilibrium Calculation software, which is a C/C++ software package for evaluating thermodynamic, kinetic and thermo-physical model parameters from experimental measurements. The optimization is conducted until the sum of the squares of the errors between the calculated and the experimental thermodynamic properties and the phase equilibrium is minimized.
Thermodynamic descriptions of pure elements used in the present work are from the Scientific Group Thermo data Europe databank [25]. The binary descriptions of Ti-Ni [7], Ti-O [8] and Ni-O [9] were accepted as mentioned above. The thermodynamic parameters evaluated in the present work for the Ti-Ni-O ternary description were the G and L parameters of liquid, HCP (��-Ti), BCC (��-Ti), FCC (Ni) and NiTiO3, and the L parameters of TiNi, Ti2Ni.
This procedure can be conducted as follows: firstly, the ternary interaction parameters of liquid, BCC, HCP and FCC were calculated; secondly, the interaction parameters involving the third element in Ti2Ni phase were optimized; finally, the interaction parameters involving the third element in TiNi (B2) phase were optimized.
All the parameters optimized in this work are listed in Table 1.
Table 1 Thermodynamic parameters of Ti-Ni-O system

5 Results and discussion
The calculated phase diagrams of the Ti-Ni [7], Ti-O [8] and Ni-O [9] systems are illustrated in Figs. 5-7, respectively.
Figure 8 shows the calculated isothermal section of the Ti-Ni-O ternary system at 1 173 K. Here, the experimental data by ROSTOKER [6] is also included. The calculated equilibria are in good agreement except for the equilibrium of TiO+Ti2Ni+TiNi3. The calculated solubility of the element O in Ti2Ni phase is much lower than that measured by ROSTOKER [6]. This may be resulted from the solubility of the Ti2Ni phase in the Ti-Ni binary systems assessed by KEYZER et al [7], which is very small.
Figure 9 shows the calculated isothermal section of the Ti-Ni-O ternary system at 1 273 K. Here, the experimental data by HASHIMOTO et al [23] are also included. The calculated equilibria are in good agreement with the majority of experimental results by HASHIMOTO et al [23].
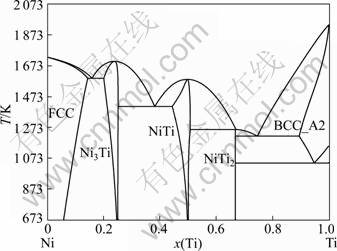
Fig. 5 Calculated Ti-Ni phase diagram using thermodynamic assessment [7]
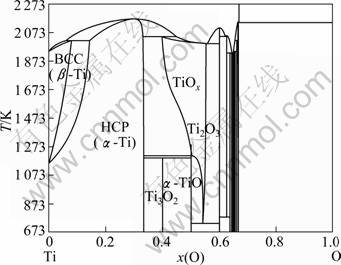
Fig. 6 Calculated Ti-O phase diagram using thermodynamic assessment [8]
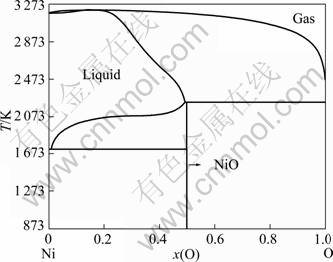
Fig. 7 Calculated Ni-O phase diagram using thermodynamic assessment [9]
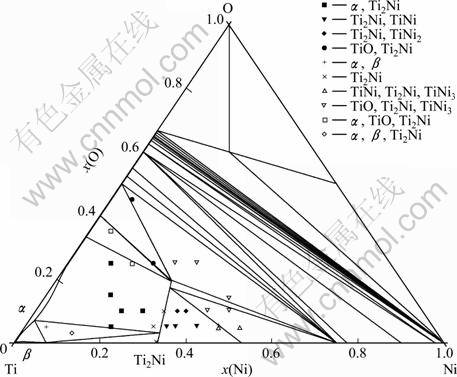
Fig. 8 Calculated 1173 K isothermal section of Ti-Ni-O in this work with experimental points measured by ROSTOKER [6]
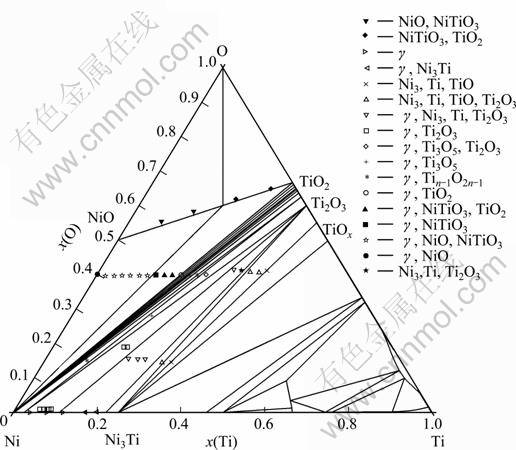
Fig. 9 Calculated 1 273 K isothermal section of Ti-Ni-O in this work with experimental points measured by HASHIMOTO et al [23]
The oxygen potential can be calculated from the relevant calculation of the phase diagram of Ti-Ni-O ternary system. Then the oxygen potential is transformed into electrical potential using the Nernst equation, formulated as
![]() (12)
(12)
where ![]() is the vapor pressure of oxygen in the titanium melt and
is the vapor pressure of oxygen in the titanium melt and ![]() is the partial pressure of oxygen
is the partial pressure of oxygen
in the atmosphere. When the oxygen content of TiNi alloy is 0.08%, the vacuum is 10-2 Pa, the calculated electrical potential is 2.74 V at 1 773 K, through Eq. (12) (Fig. 10).
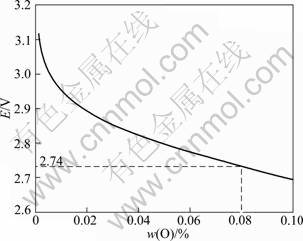
Fig. 10 Relationship between electrical potential obtained by Nernst equation and oxygen content in TiNi alloy at 1 773 K and vacuum of 10-2 Pa
6 Conclusions
1) Based on the previous assessed Ti-Ni, Ti-O and Ni-O systems and our evaluated NiO-TiO2 pseudo binary systems, the thermodynamic description for the Ti-Ni-O ternary system is obtained through the extrapolation by CALPHAD method.
2) The 1 173 and 1 273 K isothermal sections are calculated. It is shown that the present thermodynamic assessment is in good agreement with the majority of experimental results. A consistent model parameter set is determined so that the calculation of isothermal sections and other thermodynamic quantities becomes possible.
3) The oxygen potential was calculated using the thermodynamic parameters of the Ti-Ni-O ternary system. Then the oxygen potential is transformed into electrical potential using Nernst equation. This result may provide theoretical guidance for the electrochemical deoxidation of TiNi alloy.
Acknowledgements
The authors are grateful to Dr. CHEN Shuang-lin from CompuTherm LLC in USA and Prof. ZHANG Jie-yu from Shanghai University in China for the helpful discussion in this work.
References
[1] GRUMMON D S, PENCE T J. Thermotractive titanium-nickel thin films for microelectromechanical systems and active composites [J]. Materials for Smart Systems II, 1997, 459: 331-343.
[2] KAHN H, HUFF M A, HEUER A H.The TiNi shape-memory alloy and its applications for MEMS [J]. Journal of Micromechanics and Microengineering, 1998, 8(3): 213-221.
[3] MIYAZAKI S, OTSUKA K, SUZUKI Y. Transformation pseudoelasticity and deformation behavior in a Ti-50.6 at% Ni alloy [J]. Scripta Metallurgica, 1981, 15(3): 287-292.
[4] MIYAZAKI S, OHMI Y, OTSUKA K, SUZUKI Y. Characteristics of deformation and transformation pseudoelasticity in Ti-Ni alloys [J]. Journal de Physique Colloques, 1982(43): C4-255�CC4-260.
[5] MIYAZAKI S, IMAI T, IGO Y, OTSUKA K. Effect of cyclic deformation on the pseudoelasticity characteristics of Ti-Ni alloys [J]. Metallurgical and Materials Transactions A, 1986, 17(1): 115-120.
[6] ROSTOKER W. Selected isothermal sections in the titanium-rich corners of the systems Ti-Fe-O, Ti-Cr-O and Ti-Ni-O [J]. Transactions of the Metallurgical Society of AIME 203, 1955(7): 113-116.
[7] de KEYZER J, CACCIAMANI G, DUPIN N, WOLLANTS P. Thermodynamic modeling and optimization of the Fe-Ti-Ni system [J]. CALPHAD, 2009, 33(1): 109-123.
[8] CANCAREVIC M, ZINKEVICH M, ALDINGER F. Thermodynamic description of the Ti-O system using the associate model for the liquid phase [J]. CALPHAD, 2007, 31(3): 330-342.
[9] KOWALSKI M, SPENCER P J. Thermodynamic revaluation of the Cr-O, Fe-O and Ni-O systems: Remodeling of the liquid, bcc and fcc phases [J]. CALPHAD, 1995, 19(3): 229-243.
[10] NASH P. Phase diagrams of binary nickel alloys [M]. Materials Park, Ohio: ASM International, 1991: 342-355.
[11] LIANG Hai-yan, JIN Zhan-peng. A reassessment of the Ti-Ni system [J]. CALPHAD, 1993, 17(4): 415-426.
[12] BELLEN P, KUMAR K C H, WOLLANTS P. Thermodynamic assessment of the Ti-Ni phase diagram [J]. Zeitschrift f��r Metallkunde, 1996, 87(12): 972-978.
[13] TANG W, SUNDMAN B, SANDSTROM R, QIU C. New modelling of the B2 phase and its associated martensitic transformation in the Ti-Ni system [J]. Acta Materialia, 1999, 47(12): 3457-3468.
[14] FISCHER E. Thermodynamic calculation of the O-Ti system [J]. Journal of Phase Equilibria, 1997, 18(4): 338-343.
[15] WALDNER P, ERIKSSON G. Thermodynamic modelling of the system titanium-oxygen [J]. CALPHAD, 1999, 23(2): 189�C218.
[16] TAYLOR J R, DINSDALE A T. A thermodynamic assessment of the Ni-O, Cr-O and Cr-Ni-O systems using the ionic liquid and compound energy models [J]. Zeitschrift f��r Metallkunde, 1990, 81(5): 354-366.
[17] BIRNBAUM H, SCOTT R K. X-Ray Diffraction studies of the system: Zn2TiO4-NiTiO3 [J]. Journal of the American Chemical Society, 1950, 72(3): 1398-1399.
[18] BAYER G, FL?RKE O W. A new spinel-type phase in the system NiO-TiO2-SiO2 [J]. Naturwissenschaften, 1973, 60(2): 102.
[19] WERNER H D, GEBRET W. A new compound in the system nickel oxide-titanium dioxide [J]. Neues Jahrbuch f��r Mineralogie- Monatshefte, 1976, 1: 44-48.
[20] LAQUA W, SCHULTZ E W, REUTER B. Phase relations and reactions in the NiO-TiO2 system [J]. Zeitschrift fur anorganische und allgemeine chemie, 1977, 433: 167-180. (in German)
[21] MUAN A. Equilibrium relations in the system NiO-TiO2 in the temperature range 1 300 ��C to 1 750 ��C [J]. Journal of the American Ceramic Society, 1992, 75(6): 1357-1360.
[22] QIU Ai-tao, PANG Wei, GAO Yong-hui. Optimization of the thermodynamic parameters and calculation of phase diagram of NiO-TiO2 binary system [J]. The Chinese Journal of Process Engineering, 2010, 10(Z1): 241-245. (in Chinese)
[23] HASHIMOTO Y, KOYAMA K, OMORI S, ARAMI Y. Isothermal phase relationships in the nickel-rich region of the nickel-titanium-oxygen system [J]. Journal of the Japan Society of Powder and Powder Metallurgy, 1980, 27(5): 155-159. (in Japanese)
[24] CHATTOPADHYAY G, KLEYKAMP H. Phase equilibria and thermodynamic studies in the titanium-nickel and titanium-nickel-oxygen systems [J]. Zeitschrift f��r Metallkunde, 1983, 74(3): 182-187.
[25] DINSDALE A T. SGTE data base for pure elements [J]. CALPHAD, 1991, 15(4): 317-425.
[26] The SGTE substance database [M]. version 1997. Scienti?c Group Thermodata Europe, Grenoble, France, 1997.
[27] LEE B J, SAUNDERS N. Thermodynamic evaluation of the Ti-Al-O ternary system [J]. Zeitschrift f��r Metallkunde, 1997, 88(2): 152-161.
[28] ANSARA I, SUNDAMAN B, WILLEMIN P. Thermodynamic modeling of ordered phases in the Ni-Al system [J]. Acta Metallurgica, 1988, 36(4): 977-982.
[29] KUSOFFSKY A, DUPIN N, SUNDMAN B. On the compound energy formalism applied to fcc ordering [J]. CALPHAD, 2001, 25(4): 549-565.
����, ��᰽�, �� ��, ³�۸�, ���غ�
�Ϻ���ѧ �Ϻ����ִ�ұ�����Ʊ��ص�ʵ���ң��Ϻ� 200072
ժ Ҫ�����������е�ʵ�����ݣ���CALPHAD������Ti-Ni-O��Ԫϵ���������Ż�������Ti-Ni-O��Ԫϵ��1 173��1 273 Kʱ�ĵ��½��档������������������ʵ�����Ǻϡ�ͨ���Ż����һ����Ǣ������ѧ������ʹ�������½��������ѧ���ļ����Ϊ���ܣ�Ϊ�����Ͻ�������ṩ����ָ����
�ؼ��ʣ�TiNi�Ͻ𣻼�����ͼ��Ti-Ni-Oϵ���绯ѧ����
(Edited by FANG Jing-hua)
Foundation item: Project (10520706400) supported by the Science and Technology Commission of Shanghai Municipality, China; Project (2007CB613606) supported by the National Basic Research Program of China; Projects (50774052, 51074105) supported by the National Natural Science Foundation of China
Corresponding author: LI Chong-he; Tel/Fax: +86-21-56332934; E-mail: chli@staff.shu.edu.cn
DOI: 10.1016/S1003-6326(11)60935-7

