ƽ���������Ʒ��ϳ�����״����п�������ò�ͷ�������
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2011���9��
�������ߣ���ϣ÷ ������ ��� ���
����ҳ�룺2056 - 2060
�ؼ��ʣ�����п�����룻��������������
Key words��ZnO; whisker; oxidation; crystal growth
ժ Ҫ���Խ���пΪԭ�ϲ������κδ�������700 ��ʱͨ��ƽ���������Ʒ��ϳ�����״����п���롣�о��˳ɳ�ʱ�����Ʒ�ṹ�ͷ������ܵ�Ӱ�졣����������Ʊ�����Ʒ��������״����п��ɵģ�����״����п��ij��Ⱥ�ֱ����ɳ�ʱ����ӳ������ӣ�����Ʒ�����·������У���385~391 nm������ǿ������ⷢ�䣬��û���κ���ˮƽ���䣬�����ⷢ���ǿ������Ʒ�ɳ�ʱ����ӳ�����ǿ��
Abstract: The zinc oxide whiskers were synthesized by the equilibrium gas expanding method at the temperature of 700 �� with metallic zinc as the main raw material without any catalysts. The effects of the growth time on microstructure and photoluminescence properties were investigated. The results show that the as-grown samples are composed of uniform tetrapod-like ZnO whiskers. The length and diameter of the arms of the tetrapod-like ZnO whiskers increase obviously with the increase of the growth time. The strong single ultraviolet (UV) emission centering 385-391 nm without any accompanying deep-level emission is observed in the room temperature photoluminescence (PL) spectra of the whiskers. The intensity of UV emission increases markedly with the increase of growth time.

FAN Xi-mei, ZHOU Zuo-wan, WANG Jie, TIAN Ke
Key Laboratory of Advanced Technologies of Materials of Ministry of Education,
School of Materials Science and Engineering, Southwest Jiaotong University, Chengdu 610031, China
Received 2 November 2010; accepted 6 April 2011
Abstract: The zinc oxide whiskers were synthesized by the equilibrium gas expanding method at the temperature of 700 ��C with metallic zinc as the main raw material without any catalysts. The effects of the growth time on microstructure and photoluminescence properties were investigated. The results show that the as-grown samples are composed of uniform tetrapod-like ZnO whiskers. The length and diameter of the arms of the tetrapod-like ZnO whiskers increase obviously with the increase of the growth time. The strong single ultraviolet (UV) emission centering 385-391 nm without any accompanying deep-level emission is observed in the room temperature photoluminescence (PL) spectra of the whiskers. The intensity of UV emission increases markedly with the increase of growth time.
Key words: ZnO; whisker; oxidation; crystal growth
1 Introduction
Zinc oxide (ZnO), an exceptionally important semiconductor with wide gap energy (3.37 eV), large exciton banding energy (60 meV) at room temperature, high mechanical and thermal stabilities, exhibits excellent properties in piezoelectricity [1] and optics [2]. ZnO nanostructure has aroused much interest due to its promising applications in laser diodes and light emitting diodes [3], sensors [4] and microwave absorbing materials [5]. Recently, various ZnO low-dimensional nanostructure, such as nanorods, nanotubes, nanotetrapods and nanoscale whiskers [6-9] have been fabricated by different approaches, including vapor phase transportation [10], metal organic vapor phase epitaxy [11], electrochemical deposition [12] and template-based growth [13]. In these reports the main investigation concentrated on the ultraviolet (UV) emission, yellow-green emission and violet emission from ZnO nanostructure.
Among these fabrication methods, the equilibrium gas expanding method has several advantages, such as low cost, simple equipment and processes suitable for scale-up production [14]. Although the electronic and optical properties of the various ZnO nanostructures have been widely studied [15-16], the successful development of advanced optoelectronic devices likely requires a better understanding of the photoluminescence mechanism on ZnO nanostructure. In this work, zinc oxide whiskers were synthesized by the equilibrium gas expanding method using metallic zinc as the main raw material in a separator. The microstructure and photoluminescence property of the as-grown whiskers were investigated and the relationship between them was also discussed.
2 Experimental
The tetrapod-like zinc oxide whiskers were prepared using the metallic zinc pellets or pieces (chemical-grade purity) as the main raw material by the simple oxidization approach without any catalysts, additives, carrier gas or any protective gas at high temperature. A boat-like stainless steel container carrying pellets or pieces of metallic zinc was placed in a muffled furnace pre-heated to 700 ��C and maintained at this temperature for 8, 12, 16 and 20 min, respectively.
The inner surface of the furnace was made from SiC. The gate of the furnace was closed without special sealing during the whisker growth process. When the stainless steel boat was dragged out, a white fluffy product was obtained.
The structure of as-grown samples was investigated by X-ray diffraction (XRD, Rigaku Dymax) with a Cu target and a monochronmator at 50 kV and 300 mA. Scanning electron microscope (SEM, Camscan-4) with a working voltage of 20 kV was used to characterize the surface morphology of the samples. The optical properties of the zinc oxide whiskers were characterized by photoluminescence with an Ar ion laser as a light source using an excitation wavelength of 325 nm. All spectra were measured at room temperature.
3 Results and discussion
3.1 Structure of ZnO whiskers
The X-ray diffraction patterns of the samples fabricated at 700 ��C for different growth time are shown in Fig. 1. It can be seen that as-grown samples show typical wurtzite structure of ZnO and there is no obvious difference among all samples. The peaks at 2��=31.88��, 34.41��, 36.26��, 47.52��, 56.60��, 62.81��, 66.50��, 68.03�� and 69.10�� are corresponding to the (100), (002), (101), (102), (110), (103), (200), (112) and (201) plane of ZnO,respectively. From the XRD pattern, the lattice constants of the ZnO whiskers were calculated as a=3.25 ? and c=5.21 ?, which agrees well with the reported values (a=3.249 82 ? and c=5.206 61 ?) in JCPDS card No.36-1451. And no characteristic peaks from other impurities, such as Zn, were detected. The high intensity and narrow width of ZnO diffraction peaks indicate that the as-grown ZnO whiskers have been highly crystallized.
The surface morphologies of the samples grown at 700 ��C for the growth time of 8, 12, 16 and 20 min are shown in Fig. 2, respectively. It can be seen that all the samples consist of large quantity of tetrapod-like ZnO whiskers. No spherical or other morphologies are observed. Each tetrapod-like whisker is self-assembled with four regular and uniform pillar-like ZnO arms. The length of arm whose diameter gradually tapers down from 0.5 to 0.4 ?m is in the range of 20-45 ?m and each arm shows a tip in the end. The length and diameter of the arms of ZnO whiskers increase obviously with the increase of growth time. Arms have smooth surface and no evident dislocations or stacking faults.
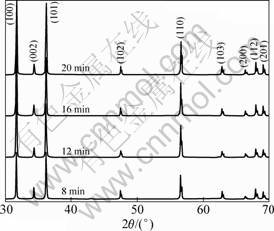
Fig. 1 XRD patterns of tetrapod-like ZnO whiskers obtained at 700 ��C with different growth time

Fig. 2 SEM images of tetrapod-like ZnO whiskers grown at 700 ��C with different growth time: (a) 8 min; (b) 12 min; (c) 16 min; (d) 20 min
Exact growth mechanism of tetrapod-like ZnO whiskers is debatable up to now. However, the most widely accepted theory is the Vapor�Csolid (VS) mechanisms, which was proposed to explain growth of 1D micro- and nanostructures without any catalyst [17-18]. It was believed that tetrapod-like ZnO whiskers could also be explained by the VS mechanism. It was reported that the low oxidation rates of Zn vapor on the surface of Zn particles were necessary for preparing ZnO with 1D nanostructure [16]. According to the principle of gas expanding, the oxygen concentration at 700 ��C is much lower than that at room temperature in a fixed volume furnace, which is the necessary condition for the generation of tetrapod-like ZnO whiskers. According to VS mechanisms the growth process of tetrapod-like ZnO whiskers can be divided into two stages: the formation of nucleation and the growth of succeeding arms. In the first stage, the supersaturation zinc vapor was mass-generated from the Zn particles at 700 ��C. The gaseous Zn atoms reacted with O atoms to form ZnO nuclei and then formed heterostructure with octahedron zinc on the surface of Zn particles. Simultaneously, the internal stresses were generated in ZnO nuclei due to the deficiency of oxygen as the reaction proceeded [19]. In the second stage, these ZnO nuclei were expelled out of the grain boundary when they became unable to resist the internal stress. The emerged octahedral crystals began to develop their arms on their alternative faces to form large and uniform tetrapod-like crystals in the vapor of zinc and oxygen. The results were similar to the growth mode of CdTe tetrapod whiskers [20]. Otherwise, with the increase of the growth time of ZnO whiskers in the range of 8-20 min, on one hand, the collision probability of Zn atoms and oxygen atoms raised greatly, which provided more reaction chance for each other to form longer and thicker arms; on the other hand, due to the deficiency of oxygen and the low oxidation rate of Zn vapor, which provided enough time for atoms to move on surface to look for appropriate sites. Both processes result in the increased crystallinity of tetrapod-like ZnO whiskers with the increase of the growth time of ZnO whiskers in the range of 8-20 min. This means that the tetrapod-like ZnO whiskers obtained at 700 ��C with the growth time of 20 min has the best microstructure.
3.2 Photoluminescence characteristics of whiskers
Figure 3 shows the room temperature PL spectra of the tetrapod-like ZnO whiskers grown at 700 ��C with the growth time of 8, 12, 16 and 20 min, respectively. It can be seen that all samples show a strong single UV emission centering about 385-391 nm without any accompanying deep-level emission. The intensity of UV emission increases markedly with the increase of growth time up to 20 min.
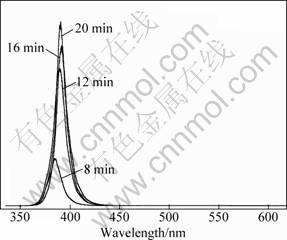
Fig. 3 Room temperature PL spectra of tetrapod-like ZnO whiskers grown at 700 ��C with different growth time
Figure 4 shows the measured FWHM of the UV emission peak from the PL spectra varying with growth time. It is found that the FWHM of the sample obtained with the growth time of 20 min is the narrowest. The FWHM is 90 meV (or 10 nm), which is narrower than that (120 meV) of the ZnO nanotubes obtained by vapor phase growth reported by XING et al [21]. It is suggested that both the intensity and FWHM of the UV PL spectra of tetrapod-like ZnO whiskers depend strongly on the microcrystalline structure. In other words, good photoluminescence property of ZnO largely depends on its structure and the above results show that the tetrapod-like ZnO whiskers have very high crystallinity. Relatively large length and thick diameter arms are also in favor of UV PL.
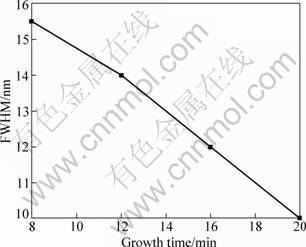
Fig. 4 FWHM of UV emission of room temperature PL spectra of tetrapod-like ZnO whiskers grown at 700 ��C with different growth time
As for the origin of the UV emission of PL spectra, the commonly cited opinion [22-23] is that the UV emission should correspond to the near band edge exciton emission of the wide band gap of ZnO. It can also be explained by the recombination of free excitons through an exciton-exciton collision process. The sharp and strong single UV emission in the room temperature PL spectra was attributed to the high purity and crystallinity of as-grown tetrapod-like ZnO whiskers. This crystalline is produced by the change of the diameter of tetrapod-like arms which are produced due to the low oxidation rates.
It is very interesting that there is no deep level emission in the room temperature PL spectra of as-grown sample. It was reported that the deep level emission of the ZnO is relative to the intrinsic defect centers [24-27]. According to our previous studies, the deep level emission of ZnO is mainly relative to the electronics transition from the bottom of conduction band to the antisite OZn levels [24]. In our experiments, no deep level emission peaks were detected, which implies that the concentration of the antisite OZn defects is very low (even may be negligible) in the tetrapod-like ZnO whiskers. Because the experiment proceeded under the conditions of the supersaturation zinc vapors and a lower oxygen concentration at 700 ��C, the continuous supply of O atoms is insufficient to form ZnO with Zn ions and there may be residual Zn ions, which may result in plentiful interstitial zinc (Zni) and oxygen vacancy VO defects in the tetrapod-like ZnO whiskers. It is well known that more Zni and more VO all depress the concentration of the antisite defect OZn in the tetrapod-like ZnO whiskers. As a result, the strong single UV emission without deep level emission is observed from as-grown samples. Therefore, it can be deduced that the optical properties of ZnO are mainly governed by crystal quality and intrinsic defects. By our knowledge, this single UV emission (without any other emission) from tetrapod-like ZnO whiskers had never been reported. For the non-single UV emission (with green emission), different explanations were proposed in recent years. ZHAO et al [25] found that the increased surface area of multipod ZnO whiskers and a great fraction of oxygen vacancies defects in the nucleus of multipod ZnO were responsible for the strong green emission with 516 nm wavelength. LIN et al [26] deduced from their experimental result of green emission with 521 nm, that the oxygen antisite OZn defects could also induce green emission from ZnO. DAI et al [17] thought that the green emission originates from the radioactive recombination of a photo-generated hole with an electron occupying the oxygen vacancy. Experimentally, the optical properties without green emission in the present tetrapod-like ZnO whiskers are similar to those reported in Ref. [26].
4 Conclusions
1) All samples comprise the uniform tetrapod-like ZnO whiskers. The length and diameter of the arms of ZnO whiskers increase obviously with the increase of the growth time of ZnO whiskers.
2) As-grown samples show the strong single UV emission centering 385-391 nm without any accompanying deep-level emission. With the increase of growth time up to 20 min, the intensity of UV emission increases markedly. Excellent photoluminescence property of the tetrapod-like ZnO whiskers was observed in the sample which grew at 700 ��C for 20 min.
3) The strong single UV emission without deep level emission is attributed to the high purity and perfect crystallinity and the lower concentration of antisite OZn defects of as-grown tetrapod-like ZnO whiskers.
References
[1] MINNE S C, MANALIS S R, QUATE C F. Parallel atomic force microscopy using cantilevers with integrated piezoresistive sensors and integrated piezoelectric actuators [J]. Applied Physics Letters, 1995, 67: 3918-3920.
[2] CHEN S J, LIU Y C, SHAO C L, MU R, LU Y M, ZHANG J Y, SHEN D Z, FAN X W. Structural and optical properties of uniform ZnO nanosheets [J]. Advanced Materials, 2005, 17: 586-590.
[3] SATIO N, HANEDA H, SEKIGUCHI T, OHASHI N, SAKAGUCHI I, KOUMOTO K. Low-temperature fabrication of light-emitting zinc oxide micropatterns using self-assembled monolayers [J]. Advanced Materials, 2002, 14: 418-421.
[4] GOLEGO N, STUDENIKIN S A, COCIVERA M. Sensor photoresponse of thin-film oxides of zinc and titanium to oxygen gas [J]. Journal of the Electrochemical Society, 2000, 147: 1592-1594.
[5] CHEN Y J, CAO M S, WANG T H, WAN Q. Photonic bandgaps in patterned waveguides of silicon-rich silicon dioxide [J]. Applied Physics Letters, 2004, 84: 2415-2417.
[6] LI F, LI Z, JIN F J. Fabrication and characterization of ZnO micro and nanostructures prepared by thermal evaporation[J]. Physica B, 2008, 403: 664-669.
[7] WU J J, LIU S C, WU C T, CHEN K H, CHEN L C. Heterostructures of ZnO-Zn coaxial nanocables and ZnO nanotubes [J]. Applied Physics Letters, 2002, 81: 1312-1314.
[8] ZHAO Y N, CAO M S, LI J G, CHEN Y J. A novel and simple combustion route towards long legs nanotetrapod ZnO [J]. Materials Research Bulletin, 2005, 40: 1745-1750.
[9] HU J Q, BANDO Y. Growth and optical properties of single-crystal tubular ZnO whiskers [J]. Applied Physics Letters, 2003, 82: 1401-1403.
[10] HUANG M H, WU Y, FEICK H, TRAN N, WEBER E , YANG P. Catalytic growth of zinc oxide nanowires by vapor transport [J]. Advanced Materials, 2001, 13: 113-116.
[11] PARK W I, KIM D H, JUNG S W, YI G C. Metalorganic vapor-phase epitaxial growth of vertically well-aligned ZnO nanorods [J]. Applied Physics Letters, 2002, 80: 4232-4234.
[12] ZHENG M J, ZHANG L D, LI G H, SHEN W Z. Fabrication and optical properties of large-scale uniform zinc oxide nanowire arrays by one-step electrochemical deposition technique [J]. Chemical Physics Letters, 2002, 363: 123-128.
[13] LI Y, MENG G W, ZHANG L D, PHILLIPP F. Ordered semiconductor ZnO nanowire arrays and their photoluminescence properties [J]. Applied Physics Letters, 2000, 76: 2011-2013.
[14] ZHOU Z W, PENG W M, KE S Y, DENG H. Tetrapod-shaped ZnO whisker and its composites [J].Journal of Materials Processing Technology, 1999, 89: 415-418.
[15] LEE C J, LEE T J, LYU S C, ZHANG Y, RUH H, LEE H. Field emission from well-aligned zinc oxide nanowires grown at low temperature [J]. Applied Physics Letters, 2002, 81: 3648-3650.
[16] ZHANG J, YU W, ZHANG L. Fabrication of semiconducting ZnO nanobelts using a halide source and their photoluminescence properties [J]. Physics Letters A, 2002, 299: 276-281.
[17] DAI Y, ZHANG Y, LI Q K, NAN C W. Synthesis and optical properties of tetrapod-like zinc oxide nanorods [J]. Chemical Physics Letters, 2002, 358: 83-86.
[18] YAN H, HR R, PHAM J, YANG P. Morphogenesis of one-dimensional ZnO nano-and microcrystals [J]. Advanced Materials, 2003, 15: 402-405.
[19] KITANO M, HAMABE T, MAEDA S, OKABE T. Growth of large tetrapod-like ZnO crystals II. Morphological considerations on growth mechanism[J]. Journal of Crystal Growth, 1991, 108: 277-284.
[20] MANNA L, MILLIRON D J, MEISEL A, SCHER E C, ALIVISATORS A P. Controlled growth of tetrapod-branched inorganic nanocrystals [J]. Nature Materials, 2003, 2: 382-385.
[21] XING Y J, XI Z H, XUE Z Q, ZHANG X D. Optical properties of the ZnO nanotubes synthesized via vapor phase growth [J]. Applied Physics Letters, 2003, 83: 1689-1691.
[22] KONG Y C, YU D P, ZHANG B, FANG W, FENG S Q. Ultraviolet-emitting ZnO nanowires synthesized by a physical vapor deposition approach [J]. Applied Physics Letters, 2001, 78: 407-409.
[23] LI C, FANG G J, FU Q, SU F H, LI G H, WU X G, ZHAO X Z. Effect of substrate temperature on the growth and photoluminescence properties of vertically aligned ZnO nanostructures[J]. Journal of Crystal Growth, 2006, 292: 19-25.
[24] FAN Xi-mei, LIAN Jian-she, GUO Zuo-xing, JIANG Qing. Surface morphology and photoluminescence properties of ZnO thin films obtained by PLD [J]. Transactions of Nonferrous Metals Society of China, 2005, 15(3): 519-523.
[25] ZHAO Y N, CAO M S, JIN H B, ZHANG L, QIU C J. Catalyst-free synthesis, growth mechanism and optical properties of multipod ZnO with nanonail-like legs[J].Scripta Materialia, 2006, 54: 2057-2061.
[26] LIN B, FU Z, YIA Y. Green luminescent center in undoped zinc oxide films deposited on silicon substrates [J]. Applied Physics Letters, 2001, 79: 943-945.
[27] ZHAO Lei, LIAN Jian-she, LIU Yu-hua, JIANG Qing. Influence of preparation methods on photoluminescence properties of ZnO films on quartz glass [J]. Transactions of Nonferrous Metals Society of China, 2008 18(1): 145-149.
��ϣ÷���������� 漣��� ��
���Ͻ�ͨ��ѧ ���Ͽ�ѧ�빤��ѧԺ�������Ƚ������������ص�ʵ���ң��ɶ� 610031
ժ Ҫ���Խ���пΪԭ�ϲ������κδ�������700 ��Cʱͨ��ƽ���������Ʒ��ϳ�����״����п���롣�о��˳ɳ�ʱ�����Ʒ�ṹ�ͷ������ܵ�Ӱ�졣����������Ʊ�����Ʒ��������״����п��ɵģ�����״����п��ij��Ⱥ�ֱ����ɳ�ʱ����ӳ������ӣ�����Ʒ�����·������У���385~391 nm������ǿ������ⷢ�䣬��û���κ���ˮƽ���䣬�����ⷢ���ǿ������Ʒ�ɳ�ʱ����ӳ�����ǿ��
�ؼ��ʣ�����п�����룻��������������
(Edited by LI Xiang-qun)
Foundation item: Project (2009AA03Z427) supported by the National High-tech Research and Development Program of China; Project (2006z02-006-3) supported by the Science Foundation of Sichuan Province, China
Corresponding author: FAN Xi-mei; Tel: +86-28-87602714; Fax: +86-28-87600454; E-mail: fanximei@126.com
DOI: 10.1016/S1003-6326(11)60972-2

