Acidianus manzaensis�Բ�ͬ����ṹ��ͭ�����������
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2019���3��
�������ߣ������� ������ ����� ����� �Բ��� �Ŷ��� ��ޱ ������ �Ľ���
����ҳ�룺617 - 624
�ؼ��ʣ���ͭ����ṹ�����������Acidianus manzaensis��SR-XRD��XANES
Key words��chalcopyrite; crystal structure; bioleaching; Acidianus manzaensis; SR-XRD; XANES
ժ Ҫ������ͬ������X��������(SR-XRD)����K��X�������ս��߽ṹ(XANES)����ѧ�ȼ������Ƚ��о� A. manzaensis�Բ�ͬ����ṹ��ͭ��(���ࡢ����ͦ���)�Ľ�����ͨ����583��773��848 K�ȴ���ԭʼ��ͭ��æ��ࡢ����ͦ����ͭ����������Ľ������������10 d��������������ࡢ���ࡢ�����ԭʼ��ͭ�����Һ��[Cu2+]�ֱ�Ϊ1.27��1.86��1.43��1.13 g/L����������Ļ�ͭ����������͵Ļ�ͭ������ױ�A. manzaensis������SR-XRD��XANES�Ľ����������4�����ͻ�ͭ����������IJ�����Ҫ���ɻƼ������ͻ�ͭ����ɣ�����������������ij��ڲ����������ڦ���ͦ����ͭ������������̶��ԣ���ͭ���ڽ����ij�ʼ�β����������ڵ�6��ת��Ϊ��ͭ��
Abstract: Bioleaching of chalcopyrite with different crystal structures (��-phase, ��-phase and ��-phase) by Acidianus manzaensis was comparatively studied by synchrotron radiation based X-ray diffraction (SR-XRD) and S K-edge X-ray absorption near edge structure (XANES) spectroscopy. The ��-phase, ��-phase and ��-phase chalcopyrite was prepared by heating original chalcopyrite at 583, 773 and 848 K, respectively. Bioleaching results showed that [Cu2+] in the leaching solution of ��-phase, ��-phase, ��-phase and original chalcopyrite after 10 days of bioleaching was 1.27, 1.86, 1.43 and 1.13 g/L, respectively, suggesting that ��-phase had a better leaching kinetics than others. SR-XRD and XANES results indicated that jarosite and chalcopyrite were the main components in the leaching residues in all cases, and elemental sulfur formed in the early stage of bioleaching. While for ��-phase and ��-phase chalcopyrite during bioleaching, bornite was produced in the initial stage of leaching, and turned into chalcocite on day 6.
Trans. Nonferrous Met. Soc. China 29(2019) 617-624
Zhen-yuan NIE1, Wei-wei ZHANG1, Hong-chang LIU1,2, Hong-rui ZHU1, Chang-hui ZHAO1, Duo-rui ZHANG1, Wei ZHU3, Chen-yan MA4, Jin-lan XIA1
1. Key Lab of Biometallurgy of Ministry of Education of China, School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China;
2. CAS Key Laboratory of Mineralogy and Metallogeny, Guangzhou Institute of Geochemistry, Chinese Academy of Sciences, Guangzhou 510640, China;
3. College of Science, Hunan University of Technology, Zhuzhou 412000, China;
4. Beijing Synchrotron Radiation Facility, Institute of High Energy Physics, Chinese Academy of Sciences, Beijing 100049, China
Received 6 December 2017; accepted 17 May 2018
Abstract: Bioleaching of chalcopyrite with different crystal structures (��-phase, ��-phase and ��-phase) by Acidianus manzaensis was comparatively studied by synchrotron radiation based X-ray diffraction (SR-XRD) and S K-edge X-ray absorption near edge structure (XANES) spectroscopy. The ��-phase, ��-phase and ��-phase chalcopyrite was prepared by heating original chalcopyrite at 583, 773 and 848 K, respectively. Bioleaching results showed that [Cu2+] in the leaching solution of ��-phase, ��-phase, ��-phase and original chalcopyrite after 10 days of bioleaching was 1.27, 1.86, 1.43 and 1.13 g/L, respectively, suggesting that ��-phase had a better leaching kinetics than others. SR-XRD and XANES results indicated that jarosite and chalcopyrite were the main components in the leaching residues in all cases, and elemental sulfur formed in the early stage of bioleaching. While for ��-phase and ��-phase chalcopyrite during bioleaching, bornite was produced in the initial stage of leaching, and turned into chalcocite on day 6.
Key words: chalcopyrite; crystal structure; bioleaching; Acidianus manzaensis; SR-XRD; XANES
1 Introduction
Chalcopyrite is a typical cupriferous mineral. In recent decades, it has become an attractive target as a model compound of metal sulfide and stimulated the joint research efforts from metallurgists and chemists for its relatively complicate structure and industrial importance. Bio-hydrometallurgical treatment of low- grade sulfide ores is an attractive alternative to conventional pyrometallurgical routes, which provides benefits in both economic and environmental aspects [1].
However, the application of chalcopyrite in bioleaching practice is still unsatisfactory, because of its low leaching kinetics. So far, numerous studies have been taken to investigate the influencing factors on chalcopyrite leaching kinetics, for the purpose of finding out a better bioleaching strategy. These studies mostly concerned on the typical bacteria, environmental parameters and catalysts [2-4]. The crystal structure of chalcopyrite could also lead to different bioleaching behaviors [5]; however, such an aspect is far from clearly-evaluated.
In many cases, bioleaching process is mineral- selective and restricted to crystal boundaries [6]. Some authors pointed out that pyrite exhibited a better attachment and leaching efficiency in the presence of Leptospirillum ferriphilum compared with chalcopyrite [7], probably due to the structural differences between the minerals. Previous study also indicated that the leaching rate of chalcopyrite decreased with time in the presence of Sulfobacillus thermosulfidooxidans, which was caused by the different leachabilities of different bonding forms in the structural motif of chalcopyrite cell, while the same bonding form of atoms in the pyrite cell led to a basic steady leaching rate [8]. For chalcopyrite at low temperature, it shows tetragonal structure, namely ��-phase. In an inert atmosphere, the thermal decomposition of chalcopyrite occurs over a certain temperature range, which produces ��- and ��-phases chalcopyrite with sulfur deficiency at higher temperatures. These structural changes may alter the leachability of chalcopyrite.
The crystal phase of chalcopyrite could be also related to different geological genesis of copper deposits. It has been found that the leaching efficiency of chalcopyrite concentrate from Hanaoka (Japan) was better than that from New Mexico [9]. Same result was found by NAOKI et al [10] that, under the promotion effect of ferrous iron, chalcopyrite concentrate from Akenobe (Japan) had better leaching efficiency than that from Zhezkent (Kazakhstan).
To the best of our knowledge, there is less report on the bioleaching of chalcopyrite with different crystal structures in the presence of thermoacidophilic microorganisms. Therefore, the aim of the present study is to elucidate the crystal phase-dependence of chalcopyrite leaching mechanisms in the presence of thermoacidophilic Acidianus manzaensis.
One difficulty for analyzing the surface products during bioleaching of chalcopyrite is that some of the metal-deficient secondary minerals are less abundant and unstable [11]. By combining synchrotron radiation based X-ray diffraction (SR-XRD) and S K-edge X-ray absorption near edge structure (XANES) spectroscopy, the surface chemical species could be well analyzed [12,13].
In the present study, by using SR-XRD and XANES, the influence of chalcopyrite crystal structure and composition during bioleaching in the presence of A. manzaensis was investigated, which could be helpful to understanding the crystal phase-dependence of chalcopyrite bioleaching.
2 Experimental
2.1 Microorganism and metal sulfides
The thermoacidophilic Archaea strain A. manzaensis YN-25 (accession number of 16S rDNA in GeneBank: EF522787) was provided by the Key Laboratory of Biometallurgy of Ministry of Education of China, Changsha, China. The basal medium for A. manzaensis cultivation consisted of the following components: 3.0 g/L (NH4)2SO4, 0.5 g/L MgSO4��7H2O, 0.5 g/L K2HPO4, 0.1 g/L KCl, 0.01 g/L Ca(NO3)2, and 0.2 g/L yeast extracts [14]. A. manzaensis was cultured in the basal salt medium supplemented with chalcopyrite (10 g/L), and the initial pH was adjusted to 1.5 with 1 mol/L H2SO4.
The pure chalcopyrite (CuFeS2), covellite (CuS), chalcocite (Cu2S) and jarosite (KFe3(SO4)2(OH)6) samples used in this study were provided by School of Minerals Processing and Bioengineering, Central South University, Changsha, China. The mineralogical composition tests (by XRD) indicated that the original chalcopyrite mineral is mainly chalcopyrite. X-ray fluorescence spectroscopic analysis showed that the original chalcopyrite contained (wt.%): Cu 35.84, Fe 31.03 and S 29.25. The mineral was ground to fine powder, guaranteeing that the particle size was 37-75 ��m.
2.2 TG-DSC
Phase transition of chalcopyrite was studied by thermo gravimetric/differential scanning calorimetry (TG-DSC) in nitrogen from room temperature to 1173 K (10 K/min). The temperature points of stable phases were determined by finding the peak of temperature gradients. In order to obtain ��-phase, ��-phase and ��-phase of chalcopyrite, the original chalcopyrite was heated respectively to 583, 773 and 848 K with N2 (99.9999%) ventilated, maintained for 12 h, and naturally cooled to room temperature.
2.3 Bioleaching experiment
For leaching experiments, A. manzaensis cells were inoculated into 250 mL flasks containing 100 mL sterilized culture medium and 1 g chalcopyrite (the initial cell concentration was 1.0��107 cells/mL) at 338 K with 170 r/min shaking. Parallel experiments (without cells, but the same mixed culture medium and chalcopyrite) were prepared as sterile control.
During leaching of chalcopyrite, solution samples were taken out at 2 day intervals to monitor the leaching characteristics, i.e., cell density, redox potential (ORP) value, [Fe2+], [Fe3+] and [Cu2+]. The cell density was determined by direct counting with a blood corpuscle counter. The ORP value was measured with a platinum (Pt) electrode, using a calomel electrode (Hg/Hg2Cl2) as reference. [Fe2+] and [Fe3+] were determined by 5-sulfosalicylic acid spectrophotometry, and [Cu2+] was determined by bis-(cyclohexanone)oxalyldihydrazone spectrophotometry according to previous studies [15,16]. Triplicate leaching experiments were performed under identical conditions. In order to analyze the intermediate compounds, the mineral samples were prepared at 2 day intervals until test.
2.4 SR-XRD and XANES spectroscopy
The analysis of SR-XRD was performed at 14B beamline in Shanghai Synchrotron Radiation Facility (SSRF), Shanghai Institute of Applied Physics, Shanghai, China. The measurement was made with a step of 0.01��, and a dwell of 5 s. The energy and the size of X-ray spot for XRD analysis was 11 keV and 0.5 mm �� 0.5 mm, respectively. XANES of S K-edge analysis was performed at 4B7A beamline (medium X-ray beamline, 2100�C6000 eV) at the Beijing Synchrotron Radiation Facility (BSRF), Institute of High Energy Physics of China, Beijing, China. The S K-edge XANES data were recorded in fluorescence mode at ambient temperature and scanned at step width of 0.2 eV between 2450 and 2512 eV across the S K-edge. The possible intermediate compounds such as covellite, chalcopyrite, chalcocite, jarosite and elemental sulfur (S0) were chosen as reference compounds. The XANES spectra were normalized to the maximum of the absorption jump, and fitted by their linear combinations using the reference spectra with IFEFFIT program [17].
3 Results and discussion
3.1 TG-DSC
Heat treatment enables effective phase transition of CuFeS2, which is not possible to achieve in its normal state. It can be seen from Fig. 1 that three obvious endothermic peaks at 583, 767 and 840 K appeared, which was consistent with the results of BALAZ et al [18].
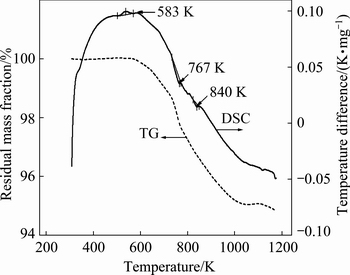
Fig. 1 TG-DSC analysis result of original chalcopyrite
The first weak peak at 583 K was preliminary phase transition of original chalcopyrite, corresponding to the ��-phase. The peak at 767 K was generated by the desulfurization of chalcopyrite, which showed maximum temperature difference rate of 0.01451 K��min/mg, corresponding to the ��-phase [19]. The peak at 840 K was the secondary desulfurization of chalcopyrite, corresponding to the ��-phase. Based on the above results, the temperatures of 583, 773 and 848 K were chosen to prepare ��-phase, ��-phase and ��-phase chalcopyrite, respectively, as described as Eq. (1) [18]:
Original chalcoyrite ��-phase
��-phase ��-phase
��-phase ��-phase (1)
��-phase (1)
3.2 Leaching characteristics of chalcopyrite by A. manzaensis
The growth characteristics of A. manzaensis and the leaching curves of chalcopyrite in the presence or absence of microorganism are shown in Fig. 2.
Bioleaching experiment showed that the leaching characteristics changed over time. The cell density in the bioleaching of ��-phase and original chalcopyrite reached a maximum value on day 6, and then decreased, while A. manzaensis grown on ��-phase chalcopyrite showed a lower growth (Fig. 2(a)). The cell density in the bioleaching of ��-phase reached a maximum value on the 8th day, and then decreased, but still much higher than that of other phases of chalcopyrite (Fig. 2(a)). The ORP value during bioleaching of ��-phase, ��-phase, ��-phase and original chalcopyrite showed no significant difference, which increased rapidly in the first two days and then stayed stable (at (620��5) mV) (Fig. 2(b)). The leaching efficiency of chalcopyrite is dependent on the ORP in solution and it is enhanced when leaching is performed in appropriate range of ORP [20]. The ORP values of leaching solution during chalcopyrite bioleaching depend on the ferric to ferrous ion concentration ratio ([Fe3+]/[Fe2+]) [21-23]. On the contrary, in the sterile control experiment, OPR values changed little for the original chalcopyrite and ��-, ��-, ��-phase chalcopyrite (Fig. 2(b)).
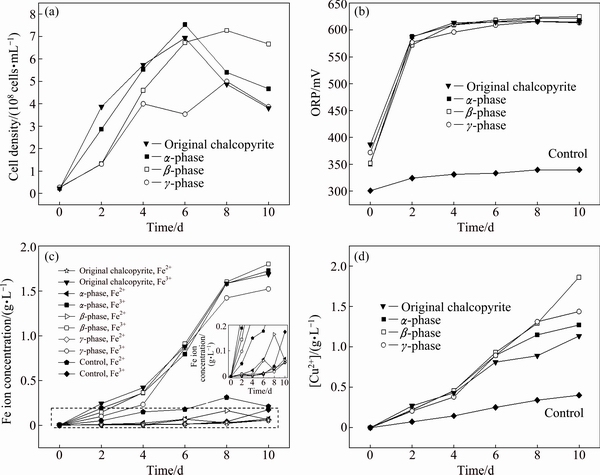
Fig. 2 Curves of cell density (a), ORP (b), iron (c) and copper (d) concentrations of ��-phase, ��-phase, ��-phase and original chalcopyrite bioleached with A. manzaensis and in sterile control experiment
Chalcopyrite dissolved according to Eqs. (2) and (3). During bioleaching of ��-phase, ��-phase, ��-phase and original chalcopyrite by A. manzaensis, the [Fe3+] increased rapidly from day 4 to day 8 before reaching a stable state (Fig. 2(c)). The [Fe2+] was basically zero, which was due to the rapid oxidation of ferrous ion to ferric ion by A. manzaensis. Compared to bioleaching experiment, [Fe3+] in the solution of the sterile control experiment was basically zero (Fig. 2(c)), due to the lack of sufficient oxidant. The [Cu2+] curves showed that copper leaching rate of ��-phase, ��-phase and original chalcopyrite persistently increased before slowing down on day 6. On the other hand, the leaching rate of ��-phase increased over the whole experiment. After 10-day bioleaching, the [Cu2+] values of ��-phase, ��-phase, ��-phase and original chalcopyrite were 1.27, 1.86, 1.43 and 1.13 g/L, corresponding to 35.72%, 52.40%, 40.28% and 31.83% of copper extraction, respectively (Fig. 2(d)). It should be noted that [Cu2+] at days 6-10 was lower than that of [Fe3+], suggesting that more iron was dissolved than copper, which was thought to be the reason of forming iron deficient secondary minerals [24,25]. While for the sterile control experiment, [Cu2+] was only ~0.40 g/L after 10 days of chemical leaching, and there was no significant difference between original chalcopyrite and ��-, ��-, ��-phase chalcopyrite (Fig. 2(d)):
4Fe2++O2+4H+��4Fe3++2H2O (2)
4Fe3++CuFeS2+O2+4H+��5Fe2++2S0+Cu2++H2O (3)
3.3 SR-XRD
The SR-XRD patterns of different crystal phase chalcopyrite leached at 65 ��C are shown in Fig. 3. The bioleaching residues were composed mostly of chalcopyrite and jarosite, but there were differences on the formation of other secondary products. For the ��-phase chalcopyrite leached for 2 days, only cubanite was detected (Fig. 3(a)), while massive jarosite appeared after 10 days (Fig. 3(b)). Combined with XRD results, the low copper leaching rate of ��-phase may be explained by crystal structure [18], where the high lattice energy caused by ordered atomic structure of ��-phase chalcopyrite made it difficult to dissolve.
In the bioleaching of ��-phase chalcopyrite, isocubanite was detected on day 2 and then transferred into haycockite. The production of unstable secondary sulfides could be explained by the disordered structure of ��-phase chalcopyrite which belonged to cubic system and jarosite precipitation occurred much earlier than that during bioleaching of ��-phase chalcopyrite [26].
The SR-XRD results of ��-phase residues indicated that more nonstoichiometric metal sulfides (Cu2.33Fe2.33S4 and Cu1.1Fe1.1S2) appeared which were caused by destroyed and mutable crystal form of ��-phase [18]. It was proposed that the high lattice energy of minerals, metal-deficient products and jarosite mainly resulted in the weak leaching performance during bioleaching of ��-phase chalcopyrite [27].
3.4 S K-edge XANES spectroscopy
The S K-edge XANES spectroscopy was used to detect sulfur species changes during bioleaching of chalcopyrite with different phases. The S K-edge XANES spectra of reference samples showed clear differences in their peak positions, peak intensities and the absorption edges (Fig. 4).
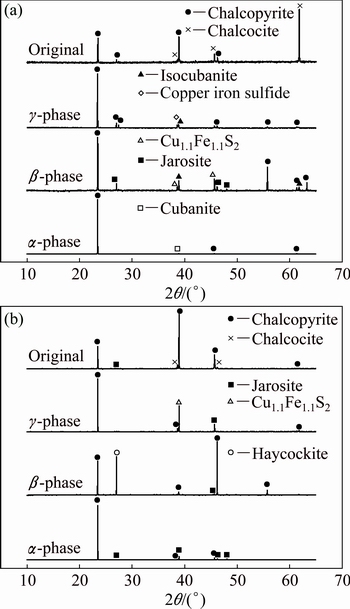
Fig. 3 SR-XRD patterns of ��-phase, ��-phase, ��-phase and original chalcopyrite residues leached by A. manzaensis after 2 days (a) and after 10 days (b)
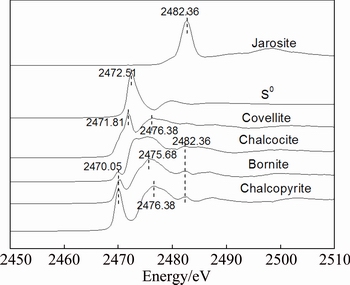
Fig. 4 Normalized S K-edge XANES spectra of standard samples chalcopyrite (CuFeS2), bornite (Cu5FeS4), covellite (CuS), chalcocite (Cu2S), S0 and jarosite (RFe3(SO4)2(OH)6)
The S K-edge XANES spectra of chalcopyrite with different phases during bioleaching by A. manzaensis (Figs. 5(a-c)) changed over time. Results in Figs. 5(a, d) showed that the intensity of the peaks at 2482.36 eV became stronger over time. Results in Figs. 5(b, c) showed that the intensity of the peaks at 2482.36 eV became stronger during first 8 days and became weaker after that.
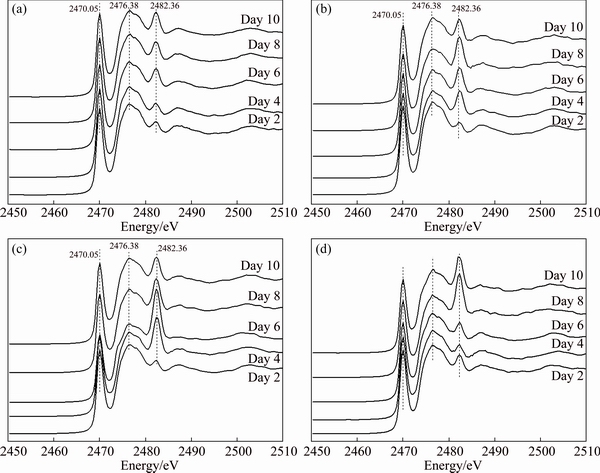
Fig. 5 Normalized S K-edge XANES spectra of ��-phase (a), ��-phase (b), ��-phase (c) and original (d) chalcopyrite leached with A. manzaensis
The S K-edge XANES spectra of leaching residues (Fig. 5) were fitted using the linear combination with reference spectra (Fig. 4). The fitted results of S K-edge XANES spectra of ��-phase, ��-phase, ��-phase and original chalcopyrite during bioleaching are given in Table 1, 2, 3 and 4, respectively.
Table 1 Fitted results of S K-edge XANES spectra of bioleaching residues from ��-phase chalcopyrite with different reference spectra
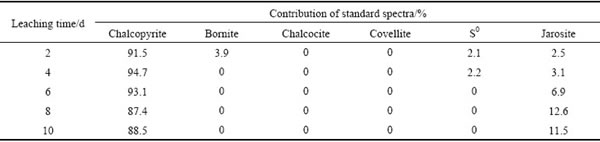
Table 2 Fitted results of S K-edge XANES spectra of bioleaching residues from ��-phase chalcopyrite with different reference spectra
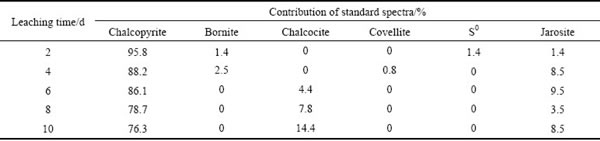
Table 3 Fitted results of S K-edge XANES spectra of bioleaching residues from ��-phase chalcopyrite with different reference spectra
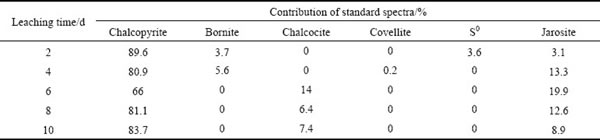
Table 4 Fitted results of S K-edge XANES spectra of bioleaching residues from original chalcopyrite with different reference spectra
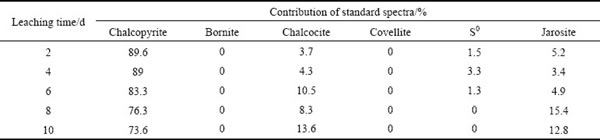
The fitted results of S K-edge XANES spectra of ��-phase chalcopyrite during bioleaching showed that the residues were composed of 94.7% of chalcopyrite, 2.2% of S0 and 3.1% of jarosite at day 4; from day 2 to day 8, chalcopyrite gradually decreased and jarosite gradually increased (Table 1). However, other secondary products like bornite and chalcocite were not found, which were consistent with the SR-XRD results (Figs. 3(a, b)).
The fitted results of S K-edge XANES spectra of ��-phase chalcopyrite during bioleaching (Table 2) showed that chalcopyrite consistently decreased from day 2 to day 10. Bornite (Cu5FeS4) appeared at day 2 and day 4, as shown in Eq. (4) [12]. After 4 days, the bornite disappeared and chalcocite gradually increased with time from day 6 to day 10. The detected chalcocite on the surface of chalcopyrite could probably be produced with the increase of [Cu2+] in the bioleaching solution, and also could be converted from bornite following Eqs. (5) and (6) [12,13]. The synergistic effect of chalcopyrite and bornite accelerated the dissolution of chalcopyrite [28]:
5CuFeS2+12H+��Cu5FeS4+6H2S+4Fe2+ (4)
CuFeS2+3Cu2++3Fe2+��2Cu2S+4Fe3+ (5)
2CuFeS2+6H+��5Cu2S+3H2S+2Fe2+ (6)
The fitted results of S K-edge XANES spectra of ��-phase chalcopyrite during bioleaching (Table 3) showed similar products with those of ��-phase and ��-phase during bioleaching, but more jarosite was produced, especially after 6 days. In addition, covellite and S0 were detected during initial stage of bioleaching of ��-phase and ��-phase chalcopyrite by extreme thermophiles [29]. However, there was only S0 detected at day 2 and day 4. By contrast, the fitted results of original chalcopyrite indicated that chalcocite increased in the first 6 days and then decreased. S0 was produced in the first 6 days while jarosite gradually increased. Based on the above results, it can be proposed that the secondary mineral formed during the leaching process is phase-dependent.
4 Conclusions
(1) Bioleaching results showed that the [Cu2+] in the leaching solution of ��-phase, ��-phase, ��-phase and original chalcopyrite after 10 d of bioleaching were 1.27, 1.86, 1.43 and 1.13 g/L corresponding to 35.72%, 52.40%, 40.28% and 31.83% of copper extraction, respectively.
(2) The results of SR-XRD and S K-edge XANES spectroscopy indicated that jarosite and chalcopyrite were the main components in the leaching residue, and the highest amount of nonstoichiometric metal sulfides was detected in the leaching residue from ��-phase chalcopyrite. Bornite was produced in the initial stage and turned into chalcocite during bioleaching of ��-phase and ��-phase chalcopyrite.
(3) It was indicated that, due to more disorder bonding forms of chalcopyrite crystal structures, ��-phase had a better leaching kinetic than ��-phase, ��-phase and original chalcopyrite.
References
[1] KHOSHKHOO M, DOPSON M, SHCHUKAREV A, SANDSTROM A. Chalcopyrite leaching and bioleaching: An X-ray photoelectron spectroscopic (XPS) investigation on the nature of hindered dissolution [J]. Hydrometallurgy, 2014, 149: 220-227.
[2] ACRES R G, HARMER S L, BEATTIE D A. Synchrotron XPS studies of solution exposed chalcopyrite, bornite, and heterogeneous chalcopyrite with bornite [J]. International Journal of Mineral Processing, 2010, 94(1): 43-51.
[3] BEVILAQUA D, GARCIA O, TUOVINEN O. Oxidative dissolution of bornite by Acidithiobacillus ferrooxidans [J]. Process Biochemistry, 2010, 45(1): 101-106.
[4] LEE J, ACAR S, DOERR D L, BRIERLEY J A. Comparative bioleaching and mineralogy of composited sulfide ores containing enargite, covellite and chalcocite by mesophilic and thermophilic microorganisms [J]. Hydrometallurgy, 2011, 105(3): 213-221.
[5] BAI Jing, WEN Jian-kang, HUANG Song-tao, WU Biao, YAO Guo-cheng. Review on bioleaching of chaicopyrite with different mineralization [J]. Chinese Journal of Rare Metals, 2012, 36(4): 644-650. (in Chinese)
[6] HARNEIT K, GOKSEL A, KOCK D, KLOCK JH, GEHRKE T, SAND W. Adhesion to metal sulfide surfaces by cells of Acidithiobacillus ferrooxidans, Acidithiobacillus thiooxidans and Leptospirillum ferrooxidans [J]. Hydrometallurgy, 2006, 83(1): 245-254.
[7] AFRICA C J, van HILLE R P, HARRISON S T. Attachment of Acidithiobacillus ferrooxidans and Leptospirillum ferriphilum cultured under varying conditions to pyrite, chalcopyrite, low-grade ore and quartz in a packed column reactor [J]. Applied Microbiology and Biotechnology, 2013, 97(3): 1317-1324.
[8] MA Jun, WANG Ju-xiang, WU Biao, WEN Jian-kang, SHANG He, WU Ming-lin. Effects of crystal structure on differences of chalcopyrite and pyrite bioleaching [J]. The Chinese Journal of Nonferrous Metals, 2015, 25(10): 2898-2904. (in Chinese)
[9] YASUHIRO K, MASAHIKO T, SATORU ASAI T S. Copper recovery from chalcopyrite concentrate by acidophilic thermophile Acidianus brierleyi in batch and continuous-flow stirred tank reactors [J]. Hydrometallurgy, 2001, 59: 271-282.
[10] NAOKI H, MASAHIKO H, TSUYOSHI H, MASAMI T. A case of ferrous sulfate addition enhancing chalcopyrite leaching [J]. Hydrometallurgy, 1997, 47: 37-45.
[11] KLAUBER C. A critical review of the surface chemistry of acidic ferric sulphate dissolution of chalcopyrite with regards to hindered dissolution [J]. International Journal of Mineral Processing, 2008, 86(1): 1-17.
[12] LIANG C L, XIA J L, YANG Y, NIE Z Y, ZHAO X J, ZHENG L, MA C Y, ZHAO Y D. Characterization of the thermo-reduction process of chalcopyrite at 65 ��C by cyclic voltammetry and XANES spectroscopy [J]. Hydrometallurgy, 2011, 107(1): 13-21.
[13] LIU H C, NIE Z Y, XIA J L, ZHU H R, YANG Y, ZHAO C H, ZHENG L, ZHAO Y D. Investigation of copper, iron and sulfur speciation during bioleaching of chalcopyrite by moderate thermophile Sulfobacillus thermosulfidooxidans [J]. International Journal of Mineral Processing, 2015, 137: 1-8.
[14] HE H, XIA J L, YANG Y, JIANG H C, XIAO C Q, ZHENG L, ZHAO Y D, QIU G Z. Sulfur speciation on the surface of chalcopyrite leached by Acidianus manzaensis [J]. Hydrometallurgy, 2009, 99(1-2): 45-50.
[15] KARAMANEV D, NIKOLOV L, MAMATARKOVA V. Rapid simultaneous quantitative determination of ferric and ferrous ions in drainage waters and similar solutions [J]. Minerals Engineering, 2002, 15(5): 341-346.
[16] QI Yan-shan, YE Tao, WU Chen-guang, GAO Can-zhu. Determination of Cu2+ in zincate solution by biscyclohexanone oxalyldihydrazone spectrophotometry [J]. Materials Protection, 2011, 44(4): 72-74. (in Chinese)
[17] RAVEL B, NEWVILLE M. ATHENA, ARTEMIS, HEPHAESTUS: data analysis for X-ray absorption spectroscopy using IFEFFIT [J]. Journal of Synchrotron Radiation, 2005, 12(4): 537-541.
[18] BALAZ P, TKACOVA K, AVVAKUMOV E. The effect of mechanical activation on the thermal decomposition of chalcopyrite [J]. Journal of Thermal Analysis and Calorimetry, 1989, 35(5): 1325-1330.
[19] BLOISE A, CATALANO M, BARRESE E, GUALTIERI A F, GANDOLFI N B, CAPELLA S, BELLUSO E. TG/DSC study of the thermal behaviour of hazardous mineral fibres [J]. Journal of Thermal Analysis and Calorimetry, 2016, 123(3): 2225-2239.
[20] HE H, XIA J L, HONG F F, TAO X X, LENG Y W, ZHAO Y D. Analysis of sulfur speciation on chalcopyrite surface bioleached with Acidithiobacillus ferrooxidans [J]. Minerals Engineering, 2012, 27: 60-64.
[21] OWUSU C, FORNASIERO D, ADDAI-MENSAH J, ZANIN M. Influence of pulp aeration on the flotation of chalcopyrite with xanthate in chalcopyrite/pyrite mixtures [J]. International Journal of Mineral Processing, 2015, 134: 50-57.
[22] MORENO M E, CASILLAS N, LARIOS D E, CRUZ R, LARA R, CARREON A A, GUTIERREZ B A, BARCENA S M, PEDROZA T M. Electrochemical assessment accounting for the interaction of chalcopyrite/xanthate system [J]. International Journal of Electrochemical Science, 2015, 10: 10619-10630.
[23] AHMADI A, SCHAFFIE M, MANAFI Z, RANJBAR M. Electrochemical bioleaching of high grade chalcopyrite flotation concentrates in a stirred bioreactor [J]. Hydrometallurgy, 2010, 104(1): 99-105.
[24] LIU Hong-chang, XIA Jin-lan, NIE Zhen-yuan, WEN Wen, YANG Yi, MA Chen-yan, ZHENG Lei, ZHAO Yi-dong. Formation and evolution of secondary minerals during bioleaching of chalcopyrite by thermoacidophilic Archaea Acidianus manzaensis [J]. Transactions of Nonferrous Metals Society of China, 2016, 26(9): 2485-2494.
[25] MA Ya-long, LIU Hong-chang, XIA Jin-lan, NIE Zhen-yuan, ZHU Hong-rui, ZHAO Yi-dong, MA Chen-yan, ZHENG Lei, HONG Cai-hao, WEN Wen. Relatedness between catalytic effect of activated carbon and passivation phenomenon during chalcopyrite bioleaching by mixed thermophilic archaea at 65 ��C [J]. Transactions of Nonferrous Metals Society of China, 2017, 27(6): 1374-1384.
[26] XIE H Z, HUANG H G, JIANG C, LIN H H, SUN P, LUO J S, ZOU L C, CHEN J H. A study on copper extraction from chalcopyrite concentrate by acidic hot-pressure oxidation [J]. Mining and Mentallurgical Engineering, 2003, 23: 54-59.
[27] YANG Y, LIU W, CHEN M. XANES and XRD study of the effect of ferrous and ferric ions on chalcopyrite bioleaching at 30 ��C and 48 ��C [J]. Minerals Engineering, 2015, 70: 99-108.
[28] ZHAO H B, WANG J, HU M H, QIN W, ZHANG Y S, QIU G Z. Synergistic bioleaching of chalcopyrite and bornite in the presence of Acidithiobacillus ferrooxidans [J]. Bioresource Technology, 2013, 149: 71-76.
[29] ZHU W, XIA J L, YANG Y, NIE Z Y, ZHENG L, MA C Y, ZHANG R Y, PENG A A, TANG L, QIU G Z. Sulfur oxidation activities of pure and mixed thermophiles and sulfur speciation in bioleaching of chalcopyrite [J]. BioresourceTechnology, 2011, 102(4): 3877-3882.
������1��������1�������1,2�������1���Բ���1���Ŷ���1, �� ޱ3��������4���Ľ���1
1. ���ϴ�ѧ ��Դ�ӹ������﹤��ѧԺ ����ұ��������ص�ʵ���ң���ɳ 410083��
2. �й���ѧԺ ���ݵ���ѧ�о��� �й���ѧԺ�ɿ�ѧ�����ѧ�ص�ʵ���ң����� 510640��
3. ���Ϲ�ҵ��ѧ ��ѧԺ������ 412000��
4. �й���ѧԺ ���������о��� ����ͬ������װ�ã����� 100049
ժ Ҫ������ͬ������X��������(SR-XRD)����K��X�������ս��߽ṹ(XANES)����ѧ�ȼ������Ƚ��о� A. manzaensis�Բ�ͬ����ṹ��ͭ��(���ࡢ����ͦ���)�Ľ�����ͨ����583��773��848 K�ȴ���ԭʼ��ͭ��æ��ࡢ����ͦ����ͭ����������Ľ������������10 d��������������ࡢ���ࡢ�����ԭʼ��ͭ�����Һ��[Cu2+]�ֱ�Ϊ1.27��1.86��1.43��1.13 g/L����������Ļ�ͭ����������͵Ļ�ͭ������ױ�A. manzaensis������SR-XRD��XANES�Ľ����������4�����ͻ�ͭ����������IJ�����Ҫ���ɻƼ������ͻ�ͭ����ɣ�����������������ij��ڲ����������ڦ���ͦ����ͭ������������̶��ԣ���ͭ���ڽ����ij�ʼ�β����������ڵ�6��ת��Ϊ��ͭ��
�ؼ��ʣ���ͭ����ṹ�����������Acidianus manzaensis��SR-XRD��XANES
(Edited by Bing YANG)
Foundation item: Projects (51774342, 51404104) supported by the National Natural Science Foundation of China; Project (2017A030313219) supported by the Natural Science Foundation of Guangdong Province, China; Project (2015JJ3062) supported by Science Foundation for Youths of Hunan Province, China; Projects (2017-BEPC-PT-001052, 2016-BEPC-PT-000887) supported by Beijing Synchrotron Radiation Facility Public User Program, China; Project (2016-SSRF-PT-004969) supported by the Open Funds of Shanghai Synchrotron Radiation Facility, China
Corresponding author: Hong-chang LIU, Tel: +86-20-85290341, E-mail: liuhongchang@gig.ac.cn;
Jin-lan XIA, Tel: +86-731-88836944, E-mail: jlxia@csu.edu.cn
DOI: 10.1016/S1003-6326(19)64971-X

