Trans. Nonferrous Met. Soc. China 23(2013) 266-270
Thermodynamics study on leaching process of gibbsitic bauxite by hydrochloric acid
Ai-chun ZHAO, Yan LIU, Ting-an ZHANG, Guo-zhi  , Zhi-he DOU
, Zhi-he DOU
Key Laboratory of Ecological Utilization of Multi-metal Intergrown Ores of Ministry of Education, School of Materials and Metallurgy, Northeastern University, Shenyang 110004, China
Received 17 November 2011; accepted 27 July 2012
Abstract: For the low-grade gibbsitic bauxite, the leaching rate of alumina is very low during the Bayer process. The acid leaching method is attracting more attention, and the hydrochloric acid leaching was developed rapidly. The mineral composition and chemical composition were investigated by X-ray diffraction analysis and semi-quantitative analysis. The thermodynamics of leaching process was analyzed. The results show that the major minerals in the bauxite are gibbsite, secondly goethite and quartz, anatase and so on. The acid leaching reactions of the bauxite would be thermodynamically easy and completed. Under the conditions that ore granularity is less than -55 μm, the L/S ratio is 100:7, and the leaching temperature is 373-383 K, the leaching time is 120 min and the concentration of HCl is 10%, both the leaching rates of Al and Fe are over 95%. The main composition of leaching slag is SiO2 which is easy for comprehensive utilization.
Key words: alumina; bauxite; gibbsitic bauxite; acid leaching; thermodynamics
1 Introduction
Bauxite is the main raw material for producing alumina [1-6]. With the rapid development of both alumina industry and the other bauxite-required industries, the shortage supplement of high-quality bauxite resources is more serious in recent years. Researchers have done a lot of studies on the development and utilization of all kinds of middle and low grade bauxite and achieved some progresses. The Bayer process is the main method of producing alumina [7-9]. The dressing-Bayer process [10] and Bayer process after roasting pretreatment for desulphurization [11-13] are separately developed according to the refractory low grade diasporic bauxite and the high sulfur bauxite. To the middle-low grade gibbsitic bauxite, desiliconizing is treated by washing bauxite or the floatation firstly, then the Bayer process is used for leaching alumina.
The acid process for alumina production has been given great attention in recent years, and some achievements have been reached [14-16]. However, the thermodynamics study on the extraction of alumina from the bauxite in the acid system has not been reported.
The gibbsite is also named as the water aluminite or aluminium-oxygen, with Al(OH)3 as its structural formula and with Al2O3・3H2O as its molecular formula. It belongs to monoclinic crystal system, which has complete crystallization of hexagonal plates. The hydroxide ion with a hexagonal close-packing array displays octahedral coordination with the aluminum ion which is filled in 2/3 of octahedral interstices of the two layers adjacent of hydroxide ions. It has ionic bond between layers and molecular bond of interlayer structure. Gibbsite is associated with kaolinite, goethite, hematite and illite.
Low-grade gibbsitic bauxite with high content of iron oxide was used as raw material by hydrochloric acid leaching. The thermodynamics reactions of the main valuable metals were studied in the acid leaching process and verified by the experimental results.
2 Experimental
The applied raw material of gibbsite was from Australia. The chemical composition and mineral composition of the gibbsite samples are listed in Tables 1 and 2. The XRD pattern is shown in Fig. 1.
Table 1 Chemical composition of gibbsite sample (mass fraction, %)

Table 2 Mineralogical composition of gibbsite sample (in mass fraction, %)


Fig. 1 XRD pattern of gibbsite sample
As shown in Table 2, the bauxite sample mainly exists in the forms of gibbsite, secondly goethite and quartz, anatase and so on. The mineral features with the low content of aluminum, the relatively low aluminum-silicon ratio and the high content of iron indicated that it is a kind of typical refractory ore. The absolute dissolution rate of alumina could only reach 51.75% if the Bayer process for alumina digestion is used. The ore is more suitable as the raw material of bauxite for acid leaching.
3 Thermodynamic analysis
Table 2 demonstrates that the Australia gibbsite ores are mainly composed of gibbsite, secondly goethite and quartz, anatase and so on. Based on the thermodynamics analysis, the leaching process of Australia gibbsite ore is relatively complex. The minerals don’t only contain gibbsite and goethite and so on. Meanwhile, the reacted unconventional medium solution is a kind of strong electrolyte solution. Thus it forms a complex multiphase system. Only when the main mineral dissolves, aluminum which is encapsulated and filled could be extracted.
For convenience of the study, it is assumed that the ore only contains the two kinds of minerals named gibbsite and goethite. By the thermodynamic data of Al(OH)3 and FeO(OH), calculations of the related reactions are carried out. The thermodynamic calculations and analysis are the qualitative analysis and quantitative estimations which still have the certain reference value on the study of leaching process. According to the principles of chemical thermodynamics [17], the calculation formulas are as follows:
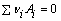 (1)
(1)
 (2)
(2)
 (3)
(3)
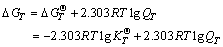
 (4)
(4)
where Ai is reactants or products; vi is measuring coefficient and it is negative for reactants and it is positive for products; T is the thermodynamic temperature; QT is reaction quotient at temperature T;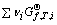 is standard molar Gibbs free energy at temperature T for reactants or products;
is standard molar Gibbs free energy at temperature T for reactants or products;  is standard molar Gibbs free energy change in the chemical-reaction at temperature T;
is standard molar Gibbs free energy change in the chemical-reaction at temperature T;  is standard equilibrium constant of chemical reactions at temperature T; R is mole gas constant, 8.314 J/(K・mol).
is standard equilibrium constant of chemical reactions at temperature T; R is mole gas constant, 8.314 J/(K・mol).
1) The chemical reaction formula of gibbsite and hydrochloric acid is shown as follows:
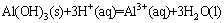 (5)
(5)
The Gibbs free energy values of each substance at different temperatures are cited from Ref. [18] and the calculation results are listed in Table 3.
The results show that the free energy change in the chemical-reaction of gibbsite and hydrochloric acid decreases gradually as the temperature rises from 298 K to 423 K. It is explained that increasing temperature is in favor of acid leaching reaction of gibbsite. At the same time, the value of  is far less than 0 and the calculated value of
is far less than 0 and the calculated value of 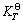 is great (all are over 1030 orders of magnitude). So the reaction happens thoroughly. Standard equilibrium constants of
is great (all are over 1030 orders of magnitude). So the reaction happens thoroughly. Standard equilibrium constants of 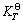 and QT are both dimensionless. For the above reaction of gibbsite and hydrochloric acid, the formula is as follows:
and QT are both dimensionless. For the above reaction of gibbsite and hydrochloric acid, the formula is as follows:
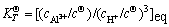 (6)
(6)
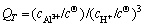 (7)
(7)
where c is volume molar concentration of related substances; cΘ is standard concentration; eq is the abbreviation of equilibrium. According to the thermodynamic criterion, the forward reaction can proceed spontaneously until reaching equilibrium (in another word  ) when QT is less than
) when QT is less than 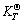 . The calculation results show that
. The calculation results show that 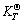 of acid leaching reaction of gibbsite is very large and decreases with increasing temperature. Therefore, with about 2.74% diluted hydrochloric acid (about 1 mol/L), Al3+ concentration must reach 5.69×1024 mol/L(T=423 K, the result is shown in Table 3) when cH+ in the leaching system is reduced to 1×10-2 mol/L(pH=2, the highest pH of leaching process in experimental study) and
of acid leaching reaction of gibbsite is very large and decreases with increasing temperature. Therefore, with about 2.74% diluted hydrochloric acid (about 1 mol/L), Al3+ concentration must reach 5.69×1024 mol/L(T=423 K, the result is shown in Table 3) when cH+ in the leaching system is reduced to 1×10-2 mol/L(pH=2, the highest pH of leaching process in experimental study) and  . But even all of aluminum in the gibbsite is leached, Al3+ concentration is not likely to reach such a high value. From the theoretical point of view, aluminum in the gibbsite could be digested with about 2.74% diluted hydrochloric acid thoroughly.
. But even all of aluminum in the gibbsite is leached, Al3+ concentration is not likely to reach such a high value. From the theoretical point of view, aluminum in the gibbsite could be digested with about 2.74% diluted hydrochloric acid thoroughly.
2) The chemical reaction equation of goethite with the diluted acid can be expressed as
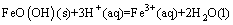 (8)
(8)
 (9)
(9)
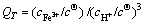 (10)
(10)
It is obtained from Table 4 that 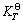 of acid leaching reaction of goethite is very large and decreases with the rise of temperature. But when the temperature rises to 423 K,
of acid leaching reaction of goethite is very large and decreases with the rise of temperature. But when the temperature rises to 423 K,  is over the order of magnitude of 107. This hints that the reaction of goethite and the diluted acid happens easily. Fe3+ concentration must reach 26.1 mol/L(T=423 K, the result is shown in Table 4) when pH of the leaching system is 2 and
is over the order of magnitude of 107. This hints that the reaction of goethite and the diluted acid happens easily. Fe3+ concentration must reach 26.1 mol/L(T=423 K, the result is shown in Table 4) when pH of the leaching system is 2 and  . But even all of the iron in the gibbsite is digested, Fe3+ concentration is impossible to achieve such a high value. This suggests that the above reaction could not reach balance. In other words, iron in the gibbsite could be digested with the very diluted hydrochloric acid thoroughly.
. But even all of the iron in the gibbsite is digested, Fe3+ concentration is impossible to achieve such a high value. This suggests that the above reaction could not reach balance. In other words, iron in the gibbsite could be digested with the very diluted hydrochloric acid thoroughly.
4 Results and discussion
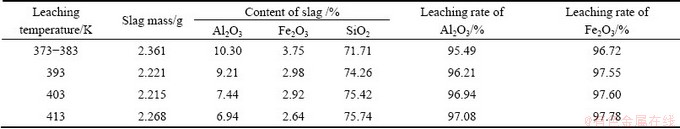
The leaching experiment was carried out in a high pressure reactor of WHFS-2L. The effects of leaching temperature on the leaching performance were studied at 373-413 K, liquid-solid ratio 100:7, leaching time 2 h, ore granularity less than 55 μm, ore mass 14 g, concentration of hydrochloric acid 10% and volume of hydrochloric acid 200 mL. The results are summarized in Table 5.
Table 3 Thermodynamic calculation for reaction of Al(OH)3 with diluted acid
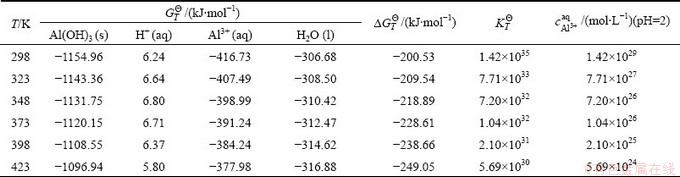
Table 4 Thermodynamic calculation for reaction of FeO(OH) with diluted acid

Table 5 Effects of leaching temperature on leaching performance
The calculation formulas of leaching rate are
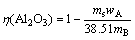 (11)
(11)
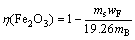 (12)
(12)
where η(Al2O3) is the leaching rate of alumina; η(Fe2O3) is the leaching rate of iron oxide; ms is the dried solid mass after solid-liquid separation; mB is the experimental ore mass; wA is the content of alumina in the leaching slag, %; wF is the content of iron oxide in the leaching slag; 38.51 is the content of alumina in the raw ore in %; 19.26 is the content of iron oxide in the raw ore in %.
From Table 5, the results are found that increasing the temperature is beneficial to the acid leaching reactions of gibbsite and goethite and the reactions tend to be more complete and agree well with the thermodynamic calculations.
5 Conclusions
From the thermodynamic calculations, the values of  of the main minerals gibbsite and goethite in the bauxite by diluted acid leaching are far less than 0 and all of the values of calculated
of the main minerals gibbsite and goethite in the bauxite by diluted acid leaching are far less than 0 and all of the values of calculated 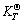 are large. So the reactions can proceed very thoroughly. With a liquid-solid ratio 100:7, leaching time 2 h, ore granularity less than 55 μm, concentration of hydrochloric acid 10% and leaching temperature 373-383 K, both the leaching rates of Fe and Al are over 95% and the main composition of leaching slag is SiO2 which is easy for comprehensive utilization.
are large. So the reactions can proceed very thoroughly. With a liquid-solid ratio 100:7, leaching time 2 h, ore granularity less than 55 μm, concentration of hydrochloric acid 10% and leaching temperature 373-383 K, both the leaching rates of Fe and Al are over 95% and the main composition of leaching slag is SiO2 which is easy for comprehensive utilization.
References
[1] BARDOSSY G, BOURKE D J. An assessment of world bauxite deposit as sources for greenfield alumina plant developments [J]. Aluminum, 1993, 69: 888-894.
[2] CHEN Qi, GUAN Hui-qin, XIONG Hui. World aluminum industry resources-exploration and utilization state of bauxite and alumina [J]. Mining and Investment, 2007(1): 27-33. (in Chinese)
[3] MU Wen-ning, ZHAI Yu-chun, WU Yan, SHI Shuang-zhi. Leaching alumina from bauxite by sulfuric acid [J]. Multipurpose Utilization of Mineral Resources, 2008, 6(3): 18-20. (in Chinese)
[4] FANG Qi-xue, HUANG Guo-zhi, GE Chang-li, LIAO Xin-tai. Characteristics and problems and countermeasures of bauxite resources in our country [J]. Light Metal Mines, 2000(10): 8-11. (in Chinese)
[5] BI Shi-wen, YU Hai-yan, YANG Yi-hong, ZHAO Fu-hui, YIN Zhong-lin, ZHAI Xiu-jing. Alumina production by Bayer method [M]. Beijing: Metallurgical Industry Press, 2007: 1-2. (in Chinese)
[6] YANG Chong-yu. Alumina technology [M]. Beijing: Metallurgical Industry Press, 1982: 2. (in Chinese)
[7] LIU Y, LIN C X, WU Y G. Characterization of red mud derived from a combined Bayer process and bauxite calcination method [J]. Journal of Hazardous Materials, 2007, 146(1-2): 255-261.
[8] EYER S I, BHARGAVA S, SUMICH M. Removal of organic from Bayer liquor and wet oxidation [J]. Light Metals, 2000: 45-51.
[9] PIGA L, POCHETTI F, STOPPA L. Application of thermal analysis techniques to a sample of red mud-A by-product of the Bayer process for magnetic separation [J]. Thermochimica Acta, 1995, 254: 337-345.
[10] ZENG Ke-wen, LIU Jun-xing, ZHOU Kai, ZHENG Gui-bing, ZHANG Yun-hai, REN Ai-jun, LIU Shui-hong. Mineral dressing test research of bauxite with low alumina silica ratio [J]. Nonferrous Metals, 2008(5): 1-7. (in Chinese)
[11]  Guo-zhi, ZHANG Ting-an, BAO Li, ZHANG Wei-guang, DOU Zhi-he. Desulphurization of high sulphur bauxite by fluosolid roasting and its effect on digestion performance [J]. Mining and Metallurgical Engineering, 2008, 28(6): 58-61. (in Chinese)
Guo-zhi, ZHANG Ting-an, BAO Li, ZHANG Wei-guang, DOU Zhi-he. Desulphurization of high sulphur bauxite by fluosolid roasting and its effect on digestion performance [J]. Mining and Metallurgical Engineering, 2008, 28(6): 58-61. (in Chinese)
[12]  Guo-zhi, ZHANG Ting-an, BAO Li, DOU Zhi-he, ZHAO Ai-chun, QU Hai-cui, NI Pei-yuan. Roasting pretreatment of high-sulfur bauxite and digestion performance of roasted ore [J]. The Chinese Journal of Nonferrous Metals, 2009, 19(9): 1684-1689. (in Chinese)
Guo-zhi, ZHANG Ting-an, BAO Li, DOU Zhi-he, ZHAO Ai-chun, QU Hai-cui, NI Pei-yuan. Roasting pretreatment of high-sulfur bauxite and digestion performance of roasted ore [J]. The Chinese Journal of Nonferrous Metals, 2009, 19(9): 1684-1689. (in Chinese)
[13]  Guo-zhi, ZHANG Ting-an, BAO Li, DOU Zhi-he. On the settling performance of red mud from preroasted high-sulfur bauxite [J]. Journal of Northeastern University: Natural Science, 2009, 30(2): 242-245. (in Chinese)
Guo-zhi, ZHANG Ting-an, BAO Li, DOU Zhi-he. On the settling performance of red mud from preroasted high-sulfur bauxite [J]. Journal of Northeastern University: Natural Science, 2009, 30(2): 242-245. (in Chinese)
[14] MENDELOVICI E. Acid and thermal treatments of lateritic bauxites [J]. Journal of Thermal Analysis and Calorimetry, 2004, 75: 957-964.
[15] HAZEK M N EL, AHMED F Y, KASABY M A EL, ATTIA R M. Sulfuric acid leaching of polymetallic abu Zeneima gibbsite-shale [J]. Hydrometallurgy, 2008, 90: 34-39.
[16] REDDY B R, MISHRA S K, BANERJEE G N. Kinetics of leaching of a gibbsitic bauxite with hydrochloric acid [J]. Hydrometallurgy, 1999, 51: 131-138.
[17] LIANG Ying-jiao. Physical chemistry [M]. Beijing: Metallurgical Industry Press, 1998: 69-90. (in Chinese)
[18] LIANG Ying-jiao, CHE Yin-chang. Thermodynamic databook of inorganic substance [M]. Shenyang: Northeastern University Press, 1993. (in Chinese).
三水型铝土矿盐酸浸出过程的热力学研究
赵爱春,刘 燕,张廷安,吕国志,豆志河
东北大学 材料与冶金学院 多金属共生矿生态化利用教育部重点实验室,沈阳 110004
摘 要:针对低品位三水型铝土矿使用拜耳法提取氧化铝存在溶出率低的情况,采用盐酸法浸出,利用X衍射分析和半定量分析手段研究铝土矿的矿物组成和化学组成,并对矿石中主要有价金属的浸出反应进行热力学分析。结果表明:铝土矿的主要成分为三水铝石,其次是针铁矿和石英,以及锐钛矿等,其主要矿物三水铝石和针铁矿的酸浸反应很容易进行,在配矿液固比100:7、浸出时间2 h、矿石粒度小于55 μm、盐酸浓度10%、浸出温度373~383 K的条件下矿物中铁和铝的浸出率均在95%以上,浸出渣主要成分为SiO2,易于综合利用。
关键词:铝土矿;氧化铝;三水型铝土矿;酸浸;热力学
(Edited by Hua YANG)
Foundation item: Projects (50974035, 51074047, 51004033) supported by the National Natural Science Foundation of China; Project (2008BAB34B01) supported by the National Science and Technology Pillar Program of China during the Eleventh Five-Year Plan Period; Project (N100302005) supported by the National Higher-education Institution General Research and Development Funding, China; Project (2010AA03A405) supported by the Hi-tech Research and Development Program of China
Corresponding author: Yan LIU; Tel: +86-13109876098; E-mail: shanqibao2000@163.com
DOI: 10.1016/S1003-6326(13)62455-3