DOI: 10.11817/j.issn.1672-7207.2018.09.003
�Ȼ�����-�Ҷ��������ܼ���Sn2+/Sn�ĵ绯ѧ��Ϊ
�ղ�1�����1, 2����һ��1, 2�����Ӣ1, 2������1, 2�����ջ�1
(1. ����������ѧ ұ������Դ����ѧԺ������ ������650093��
2. ʡ������������ɫ������Դ������ù����ص�ʵ���ң����� ������650093)
ժҪ������ѭ���������ͼ�ʱ���෨�о����Ȼ�����-�Ҷ��������ܼ�SnCl2��2H2O/[1ChCl:2EG] DES�У�Sn2+/Sn�ĵ绯ѧ��Ϊ��Sn�ĵ�ᾧ�κ˻������о����������Sn�ĵ������Ҫһ���Ĺ���λ�������¶ȵ����ߣ�����Ĺ���λ��С�����323 Kʱ�Ĵ���ϵ����Ϊ0.35��Sn2+����ɢϵ��Ϊ1.628��10-6cm2/s����303~343 K�ڣ�Sn2+��SnCl2��2H2O/[1ChCl:2EG] DES�еĵ绹ԭ��Ӧ��������ɢ���Ƶ�������̣�������ʩ�ӵ�λ��С(����)ʱ��Sn�ڲ�̼�缫�ϵĵ�ᾧ����Ϊ����ɢ���Ƶ���ά˲ʱ�κˣ�������ʩ�ӵ�λ�ϴ�(�ϸ�)ʱ��Sn�ĵ�ᾧ���̸���������ά�����κˡ�
�ؼ��ʣ������ܼ����Ȼ�����-�Ҷ������绯ѧ����ᾧ���κ˻���
��ͼ����ţ�TF111.52 ���ױ�־�룺A ���±�ţ�1672-7207(2018)09-2129-08
Electrochemistry of Sn2+/Sn in choline chloride-glycol deep eutectic solvents
SU Bo1, LI Jian1, 2, HUA Yixin1, 2, XU Cunying1, 2, LI Yan1, 2, AI Ganghua1
(1. Faculty of Metallurgical and Energy Engineering, Kunming University of Science and Technology, Kunming 650093, China;
2. State Key Laboratory of Complex Nonferrous Metal Resources Cleaning Utilization, Kunming 650093, China)
Abstract: The electrochemical behaviors of Sn2+/Sn and the nucleation mechanism of the electrocrystallization of tin in choline chloride-glycol deep eutectic solvents SnCl2��2H2O/[1ChCl:2EG] DES were studied by means of cyclic voltammetry and chronoamperometry. The results show that a certain overpotential is required for tin electrodeposition and the required overpotential decreases with the increase of temperature. The transfer coefficient �� is calculated to be 0.35 and the diffusion coefficient of Sn2+ is estimated to be 1.628��10-6cm2/s at 323 K. The electroreduction of Sn2+ in SnCl2 ��2H2O/[1ChCl:2EG] DES is a quasi-reversible process controlled by diffusion at 303-343 K. When the applied potential on cathode is lower(more positive), the initial electrocrystallization process of Sn on a GC electrode is found to be a three-dimensional instantaneous nucleation under diffusion control by chronoamperometry. When the applied potential on cathode is larger(more negative), it is more likely to be three-dimensional progressive nucleation.
Key words: deep eutectic solvent; choline chloride-ethylene glycol; electrochemistry; electrocrystallization; nucleation mechanism
�����������(��ͭ��Ǧ����������������)���ᴿ����⾫����һ��ʮ����Ч�����㷺���õķ�������������������ԡ����õ������Լ���ʴ�Ե��ŵ㣬���㷺Ӧ���ڵ���ͨ�š�ʳƷ��װ����������������е��ԭ���ܼ����칤ҵ������[1]����ұ�������Ĵ����г���������ͭ���顢�ࡢǦ������������ʣ���ͨ���������⾫�������ѳ����������ȡ�������⾫����Ҫ�������Ե��Һ������������-����-���ӻ���(��ӻ���)�����������-�����-����ȵ��Һ��ϵ�����Һ���н�ǿ�ĸ�ʴ�ԣ��ҵ��ĵ���Ч��Ҳ����������ȸ���Ӧ��������90%������Һ��(ionic liquid solvents��ILs)/�����ܼ�(deep eutectic solvents��DESs)�������нϿ��ĵ绯ѧ���ڣ����ҵͶ��ɽ��⡢���ӷ������ȶ��Խϸ�[2]�������ܼ���������ѧ����������Һ�弫Ϊ���ƣ���ˣ��䱻��Ϊһ����������Һ���������Һ��[3-4]��һ�����Ȼ�����������������Ļ�����(�����ء��Ҷ������������������)��Ϲ��۶���[5]�������Ȼ�����ĵ����ܼ���Ϊ����ʣ��ѳɹ����ڲ�ͬ�����ϵ�������ֽ����ͺϽ�[6]���������Լ������������Ӱ��[7]�����ҿɵ������������������۽����Լ�������뵼��[8]���������Ϊһ�����͵���ɫ�ܼ������������Ժ����Ȼ��������Ȼ�����-�Ҷ��������ܼ�(SnCl2��2H2O/[1ChCl:2EG] DES)Ϊ���Һ����Sn-Pb�Ͻ�(��Pb 5%~20%)���е����룬�����֦״����(Sn��������w(Sn)��99.95%)��ͨ��ѭ������������ʱ���෨�ȵ绯ѧ��������Sn2+�������ĵ绹ԭ�κ˻�����������о������ۡ�
1 ʵ�鲿��
1.1 �Լ������
�Ȼ�����(HOC2H4N(CH3)3Cl����дΪChCl��w(ChCl)��98%��AR����ҩ��ѧ�Լ���˾)���Ҷ���(HOC2H4OH����дΪEG��w(EG)��98%��AR����¤�����ɷ�����˾)����ˮ���Ȼ�����(SnCl2��2H2O��w(SnCl2��2H2O)��98.5%��AR���ɶ����������Լ���)����ˮ�Ҵ�(AR������з紬��ѧ�Լ��Ƽ�����˾)����-���������(w(Al2O3)��99%�������ɿƼ���չ����˾)��
1.2 ʵ���豸���������
RET basic���´������Ƚ�����(�¹�IKA��˾)��DZF-6090��ո�����(�Ϻ�����ʵ���豸����˾)��DU-20�͵�����ԡ��(�Ϻ�һ��Ƽ�����˾)��CHI760D�绯ѧ����վ(�Ϻ�������������˾)��FEI Quanta 200xɨ���������(����PEI��˾)��
1.3 ʵ�鷽��
�����ܼ����Ʊ��������Ȼ�������н�ǿ����ˮ�ԣ�ʵ��ǰ�Ƚ��Ȼ�����������ո������У���80 �����24 h��֮�������ʵ�����Ϊ1:2�ֱ��ȡһ���������Ȼ�������Ҷ���������ƿ���ʵ���ϣ�����70 �����ԡ���м��Ƚ���ֱ����Ϊ��ɫ������Һ�弴Ϊ�Ȼ�����-�Ҷ��������ܼ�(1ChCl:2EG DES)�������õĵ����ܼ��ܷ⣬��70 ����ո������б��汸�ã���ȡһ��������SnCl2��2H2O���������Ʊ��õ�1ChCl:2EG DES�У���70 ����ԡ���ڳ�ֽ��衢�ܽ��γɾ���Һ�壬�õ�����һ��Ũ��SnCl2��2H2O��1ChCl:2EG DES��
�绯ѧ���ԣ��������缫��ϵ���Բ�̼(GC)�缫(ֱ��4 mm)Ϊ�����缫����Բ�̵缫(ֱ��4 mm)Ϊ�Ե缫����˿�缫(ֱ��1 mm)Ϊ�αȵ缫���ֱ��ڲ�ͬɨ��(20~100 mV/s)�Ͳ�ͬ�¶�(303~343 K)�½���ѭ������(CV)���ԣ����ú��λ��Ծ�����м�ʱ�������ԡ��缫��ÿ�β���ǰ���ý���ɰֽ��ĥ��Ȼ��������Ϊ0.5 ��m��Al2O3��۴�ĥ����������Է�ֹ����ЧӦ��Ȼ������ˮ�Ҵ�������ˮ��ϴ������á�
�����ʵ�飺ʯīƬ������(����������Ϊ40 mm��20 mm��2 mm)����ͭƬ������(����������Ϊ35 mm��20 mm��0.5 mm)�����������Ϊ20 mm���缫�ڽ���ʵ��ǰ�Ƚ���ĥ�⡢������پ���ˮ�Ҵ����ݳ���ȥ֬����ȥ����ˮ��ϴ����紵�ɺ��á�
2 ���������
2.1 ɨ�ٶ�SnCl2��2H2O/[1ChCl:2EG]DESѭ��������Ӱ��
���ò�ͬ�ĵ���ɨ���ٶȶ�0.1 mol/L SnCl2��2H2O/[1ChCl:2EG] DES����ѭ���������ԣ����ý����ͼ1��ʾ������������1��ʾ��
��ͼ1���Կ���������ɨ�ٵ����ӣ����������ķ�ֵ�����ܶ�ipc��ipa��֮����ͬʱ��������ֵ����Epc�Ͱ�����Epc/2���������Եĸ�ƫ�ƣ���������ֵ����������ƫ�ƣ����⣬������ֵ�����ܶ�ipc��ɨ�ٵ�ƽ����v1/2�����õ����Թ�ϵ�������缫��Ӧ����ɢ���ơ��ɱ�1��֪������ͬɨ�������£�ipc/ipa��1���ҵ��Ʋ��Ep��|Epc-Epa|Զ���ڿ�����̵ı�ֵ(2.3RT/(nF)��32 mV��323 Kʱ)��|Epc-Epc/2|��ɨ���������������ʹ�����ɨ���£���ֵҲԶ���ڿ�����̵ı�ֵ(2.20RT/(nF)��30.6 mV��323 K)�����Ͻ��������SnCl2��2H2O/ [1ChCl:2EG] DES��Sn2+�ĵ绯ѧ��ԭ����������ɢ���Ƶ��������[9]��
��1 0.1 mol/L SnCl2��2H2O/[1ChCl:2EG] DES�ڲ�ͬ����ɨ����ѭ���������ߵ��������(323 K)
Table 1 Values related to CV at different scan rates in 0.1 mol/L SnCl2��2H2O/[1ChCl:2EG] DES at 323 K


ͼ1 ɨ�ٶ�0.1 mol/L SnCl2��2H2O/[1ChCl:2EG] DESѭ��������Ӱ��(GC�缫��323 K)
Fig. 1 Effect of scan rate on cyclic voltammograms in 0.1 mol/L SnCl2��2H2O/[1ChCl:2EG] DES (GC electrode, 323 K)
��������ϵ�е缫��Ӧ��������ֵ�������Ӧ��ɨ��ƽ����v1/2�Ĺ�ϵͬ��������������ϵ������Randles-Sevick���̱�ʾ����[9]��
 (1)
(1)
ʽ�У�ipcΪ������ֵ�����ܶȣ�mA/cm2��nΪ������������FΪ�����ڳ�����96 484 C/mol��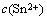 ΪSn2+��Ũ�ȣ�mol/L��
ΪSn2+��Ũ�ȣ�mol/L��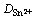 ΪSn2+����ɢϵ����cm2/s����Ϊ����ϵ����n��Ϊ���ʾ��������еĵ���ת������vΪɨ�٣�V/s��RΪ���峣����8.314 kJ/mol��TΪ����ѧ�¶ȣ�K��
ΪSn2+����ɢϵ����cm2/s����Ϊ����ϵ����n��Ϊ���ʾ��������еĵ���ת������vΪɨ�٣�V/s��RΪ���峣����8.314 kJ/mol��TΪ����ѧ�¶ȣ�K�� ����ʽ(2)[9]�õ�
����ʽ(2)[9]�õ�
 (2)
(2)
����ʽ(2)�ͱ�1��������ݿ�������¶�Ϊ323 Kʱ�Ĵ���ϵ�� Ϊ0.35��
Ϊ0.35��
2.2 �¶ȶ���ɢϵ��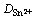 ��Ӱ��
��Ӱ��
��0.1 mol/L SnCl2��2H2O/[1ChCl:2EG] DES�ڲ�ͬ�¶��²��Ե�ѭ������������ͼ2��ʾ��
Ϊ���о��¶ȶ�������ֵ�����ܶ�ipc�ͷ����Epc��Ӱ�죬��ͼ2��ʾ���ߵ���ض���ѧ�������ڱ�2��

ͼ2 �¶ȶ�0.1 mol/L SnCl2��2H2O/[1ChCl:2EG] DES��ѭ��������Ӱ��(GC�缫��ɨ��20 mV/s)
Fig. 2 Effect of temperature on cyclic voltammograms in 0.1 mol/L SnCl2��2H2O/[1ChCl:2EG] DES (GC electrode, 20 mV/s)
��2 0.1 mol/L SnCl2��2H2O/[1ChCl:2EG] DES�ڲ�ͬ�¶���ѭ���������ߵ��������(20 mV/s)
Table 2 Values related to CV at different temperatures in 0.1 mol/L SnCl2��2H2O/[1ChCl:2EG] DES at 20 mV/s
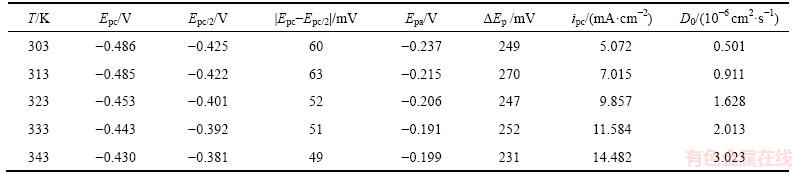
�ɱ�2��֪�������¶����ߣ����������Epc��������ƫ�ƣ�ͬʱ������ֵ�����ܶ�ipcҲ�����������¶����߿ɼ��ٽ������ĵ��������ͼ2�ľֲ��Ŵ�ͼ�ɼ���������������ɨ��֮������������������κ˵����������������κ���Ҫһ���Ĺ���λ�������¶ȵ����ߣ�������κ˹���λ������С������Ҫ����Ϊ�ڽϸ��¶��£�������ԭ��Ӧ�������������С�����¶����ߣ�����������ܶ�ipaҲ�����������������EpaҲ��������ƫ�ơ���Ep��|Epc-Epa| (284~315 mV)Զ���ڿ�����̵ı�ֵ2.3RT/(nF)[9]���ٴα�����SnCl2��2H2O/[1ChCl:2EG] DES�У�Sn2+�Ļ�ԭ����������̡�
����ͬ�¶��µ�����������ܶ�ipc��ɨ��ƽ������ͼ����ͼ3��ʾ��

ͼ3 ��ͬ�¶�������������ܶ�(ipc)��ɨ��ƽ����(v1/2)�Ĺ�ϵ
Fig. 3 Plot of cathodic peak ipc as a function of square root of scan rate v1/2 at different temperatures
��ʽ(1)��ͼ3��ò�ͬ�¶���Sn2+����ɢϵ������2���ɱ�2���Կ����������¶����ߣ�Sn2+���ӵ���ɢϵ������������Ϊ�¶�����ʹSnCl2��2H2O/ [1ChCl:2EG] DES���Ƚ��ͣ��Ӷ�ʹSn2+Ǩ��������������[10-11]������ͬ�¶ȵ���ɢϵ���Ķ��� ���¶ȵĵ���T-1��ͼ����ͼ4��ʾ��
���¶ȵĵ���T-1��ͼ����ͼ4��ʾ��

ͼ4 ��ɢϵ���Ķ���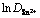 ���¶ȵ���T-1�Ĺ�ϵ
���¶ȵ���T-1�Ĺ�ϵ
Fig. 4 Plot of logarithm of diffusion coefficient against reciprocal value of absolute temperature T-1
against reciprocal value of absolute temperature T-1
��ͼ4���Կ����� ��T-1�����õ����Թ�ϵ�����ϰ�������˹��ʽ[12]��
��T-1�����õ����Թ�ϵ�����ϰ�������˹��ʽ[12]��
 (3)
(3)
��ʽ(3)���Եõ�Sn2+��������ɢ���ۻ��Ea=39.69 kJ/mol���ڱ��IJ��������º�0.1 mol/L SnCl2��2H2O/[1ChCl:2EG] DES�У��¶ȴ�303 K������343 K��Sn2+����ɢϵ��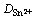 ��0.501��10-6 cm2/s��������3.023��10-6 cm2/s����323 Kʱ��Sn2+��1ChCl:2EG DES�е���ɢϵ��Ϊ1.628��10-6 cm2/s������ͬ�¶���Sn2+��1ChCl:2PG DES[13]�е���ɢϵ��(1.2��10-6 cm2/s)�Ƚ����������Sn2+��1ChCl:2urea DES�е���ɢϵ��(4.1��10-7 cm2/s)[13]��Լ4������Sn2+��1-����-1-���������f˫(��������)�ǰ���(BMPTFSI) ((8.6��0.4)��10-8 cm2/s)[14]�е�Ҫ��ö࣬��һ���������Һ����Ȳ���Լ�Sn2+�����е���λ����й�[15]��
��0.501��10-6 cm2/s��������3.023��10-6 cm2/s����323 Kʱ��Sn2+��1ChCl:2EG DES�е���ɢϵ��Ϊ1.628��10-6 cm2/s������ͬ�¶���Sn2+��1ChCl:2PG DES[13]�е���ɢϵ��(1.2��10-6 cm2/s)�Ƚ����������Sn2+��1ChCl:2urea DES�е���ɢϵ��(4.1��10-7 cm2/s)[13]��Լ4������Sn2+��1-����-1-���������f˫(��������)�ǰ���(BMPTFSI) ((8.6��0.4)��10-8 cm2/s)[14]�е�Ҫ��ö࣬��һ���������Һ����Ȳ���Լ�Sn2+�����е���λ����й�[15]��
2.3 ��ʱ���������������������κ���������
��ǰ����ѭ���������߿�֪��Sn2+��GC�缫�ϵij�����Ҫһ���Ĺ���λ��Ϊ��������Ϊ�˽�һ����ʾ����Sn��GC�缫�ϵ��κ˺��������������ú��λ��Ծ���ⶨ323 K�£�SnCl2��2H2O/[1ChCl:2EG] DES�н���Sn��������ڵ�ʱ��-������̬���ߣ���ͼ5��ʾ��
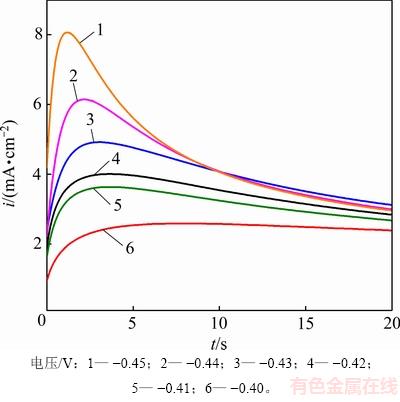
ͼ5 SnCl2��2H2O/[1ChCl:2EG] DES��Sn�������ʱ��-������̬����(323 K)
Fig. 5 Time-current transient curves of Sn electrodeposition in SnCl2��2H2O/[1ChCl:2EG] DES (323 K)
��ͼ5�ɼ�ʱ��-������̬���߳��ֳ�����ĵ���V���α仯���ƣ����ʱ�������ߵ���Ҫ����Ϊ���ڵ�������ڼ���ʱ���ڣ������ܶȼ�������������Ϊ�ڵ�ᾧ���ڣ����ž��������������Լ���������ɣ���������������������ӣ����µ����ܶ�Ѹ�����߲��ﵽ���ֵim���˺�������ɢ���ȵ����ӣ��缫����ĵ�������ʿ�ʼƶ���������ܶȻ���˥������������ƽ�ȣ�Sn�ĵ�ᾧ���̾������������ĵ���ʧ���������Ľ�����̣������Sn�ڵ�����ij���Ϊ����ɢ���Ƶ���ά�����κ˹���[16]���о������֣�����ʩ���ڵ缫����ĵ�λ���ӣ������ܶ����ֵimҲ���ﵽ�������ܶ������ʱ��tm�������̣�˵��Sn���κ˹��̲�δ��ѭ����Ŀ����ض�����[17]��Ŀǰ��2�ּ��˵��κ˻����ֱ��Ϊ��˲ʱ�κ�ģ�͡��͡������κ�ģ�͡������С�˲ʱ�κ�ģ�͡���ָ�ڵ�λ��Ծһ��ʼ�����еĻ���λ�㶼��������������κ�ģ�͡���������ʱ������ƣ�����λ�㱻�����ģ�͡�Ϊ������2���κ�ģʽ��SCHARIFKER��[18]����ʱ�������ʵ�������������������((i/im)2��t/tm�Ĺ�ϵ����)����Ӧ������бȽϣ���һ��������ϵõ���ά˲ʱ�κ�ģ�ͺ���ά�����κ�ģ�͵����۱���ʽ����ʽ���¡�
��ά˲ʱ�κˣ�
 (4)
(4)
��ά�����κˣ�
 (5)
(5)
����ͼ5�е����ݣ���(i/im)2 ��t/tm��ͼ���������۵���ά�κ�����ģ�����߽��жԱȣ������ͼ6��ʾ��
��ͼ6�ɼ�������Sn��GC�缫�ϵ��κ˳��ڣ���ʵ�����ݵ㶼������2�������κ�ģ������֮�䣻���缫����ʩ�ӵĵ�λ��С(-0.40~-0.43 V)ʱ��ʵ��������ά˲ʱ�κ�ģ�����ý���Ƚ��Ǻϣ������缫����ʩ�ӵĵ�λ�ϴ�(-0.44~-0.45 V)��t/tm��1ʱ��ʵ�����ӽ���ά˲ʱ�κ�ģ�ͣ�����t/tm��1ʱ��ʵ������ƫ����ά˲ʱ�κ�ģ���������ý����ת��ƫ����ά�����κ�ģ�����ߡ���˵���ڵ缫����ʩ�ӽϸ��ĵ�λʱ��������ʱ����ӳ����缫�����˲ʱ����λ�㱻����ռ�ݣ���ʼ����������������ά�����κ˻���λ�㡣

ͼ6 ʱ��-����ʵ����������һ������������ά˲ʱ/�����κ�ģ�����ߵĶԱ�
Fig. 6 Comparison of dimensionless experimental data dimensional normalization derived from current time-transients with theoretical models for 3D instantaneous and progressive nucleation
����ABYANEH��[19]���Ƶ���������̬�������ֵ��û�����淨���������ʳ��� ������ʽ���£�
������ʽ���£�
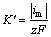 (6)
(6)
ʽ�У� Ϊ�����������ʳ�����mol/(cm2��s)��imΪ��̬���߷�ֵ�����ܶȣ�mA/cm2��zΪ���ת������FΪ�����ڳ�����C/mol��
Ϊ�����������ʳ�����mol/(cm2��s)��imΪ��̬���߷�ֵ�����ܶȣ�mA/cm2��zΪ���ת������FΪ�����ڳ�����C/mol��
��ʱ����������в�ͬ��Ծ��λ�µķ�ֵ��������ʽ(6)��� ����
����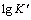 �Խ�Ծ��λE��ͼ����ͼ7��ʾ��
�Խ�Ծ��λE��ͼ����ͼ7��ʾ��
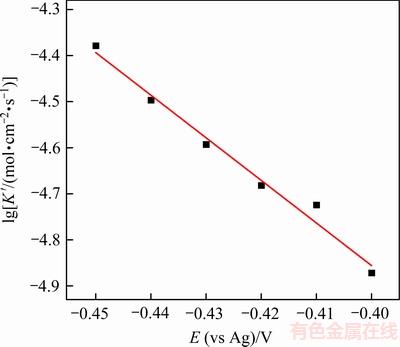
ͼ7 Sn�����������ʵĶ�������ӽ�Ծ��λ�Ĺ�ϵ
Fig. 7 Plot of logarithm of Sn crystal growth rate as a function of step potential
��ͼ7��֪��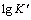 ��E�������õ����Թ�ϵ������������������Ծ��λ�ĸ��ƶ�����˵����ӵ�λ��Ϊ���������Ծ����������������Ĵٽ����á���һ��������ʽ[18, 20]����þ������ܶ�N��
��E�������õ����Թ�ϵ������������������Ծ��λ�ĸ��ƶ�����˵����ӵ�λ��Ϊ���������Ծ����������������Ĵٽ����á���һ��������ʽ[18, 20]����þ������ܶ�N��

 (7)
(7)
ʽ�У�iΪ�����ܶȣ�mA/cm2��MΪ�������Ħ��������kg/mol�� Ϊ��������ܶȣ�kg/m3��
Ϊ��������ܶȣ�kg/m3��
�� �Խ�Ծ��λE��ͼ����ͼ8��ʾ��
�Խ�Ծ��λE��ͼ����ͼ8��ʾ��

ͼ8 Sn�������ܶ�����ӽ�Ծ��λ�Ĺ�ϵ
Fig. 8 Plot of logarithm of number density of Sn nucleus N as a function of step potential
��ͼ8��֪���������ܶ� ���Ծ��λ�ĸ��Ƴ��������ӣ������ھ������Ļ���λ�������ģ���Ȼ��ӵ�λ��������������λ����Ŀ�����ӣ�������������Χ�ڻ���λ����Ŀ�������ޣ�����ʱ������ƣ��������������õ�λ�㽫����ռ�ݣ���ˣ�����ӵ�λ����һ���̶Ⱥ������ܶȽ����ٷ������Ա仯��
���Ծ��λ�ĸ��Ƴ��������ӣ������ھ������Ļ���λ�������ģ���Ȼ��ӵ�λ��������������λ����Ŀ�����ӣ�������������Χ�ڻ���λ����Ŀ�������ޣ�����ʱ������ƣ��������������õ�λ�㽫����ռ�ݣ���ˣ�����ӵ�λ����һ���̶Ⱥ������ܶȽ����ٷ������Ա仯��
�Դ�ͭƬΪ���壬�ֱ�ʩ��-0.43 V��-0.45 V��λ���е����60 s��������������������SEM�����������ͼ9��ʾ����ͼ9�ɼ�����ӵ�λ�Ծ������ܶȺ;�����С�н����Ե�Ӱ�졣
����ӵ�λΪ-0.43Vʱ(ͼ9(a))��Sn�����ֲ����ȣ���ò���Դ���ǵ�С����ֱ��������1 ��m���ң�������������д���δ��ռ�ݵ�λ�㣻����ӵ�λ������-0.45 Vʱ(ͼ9(b))ʱ������λ�㱻����������ܶ��������ӣ�ͬʱ����ֱ��������С����������ʱ��-����������Ľ��һ�¡��������λ���ƣ�Sn2+�����绹ԭ�����������ӣ�ʹ���������κ��ٶȼӿ죬����λ�㼸��ȫ����ռ�ݣ�����ʱ����ӳ������ھ��˱ؽ�����£�ͽ������Ӷ������ž�����[21]���ڱ��ĵ�ʵ�������£�ͭ����������GC�缫�ľ�����Խϴֲڣ��ڼ�����ʼ��˲�����λ�����ת��Ϊ���ˣ��ڳ��ڵ��κ˳ɳ������еõ�������������������״������������������˲ʱ�κ˻��������

ͼ9 ��ͬ��ӵ�λ��Sn��ͭ�����ϵ����������ò��SEMͼ
Fig. 9 SEM micrograph of Sn deposits initial stage on Cu substrate at different potentials
3 ����
1) ѭ���������߳���1�����Ե�������ԭ�壬˵��Sn2+/Sn��GC�缫���淢���绯ѧ��ԭ/�������̣�ͬʱ����1ChCl:2EG DES����Ϊ���������Sn�ĵ������ʹ����ĵ�⾫����
2) ��303~343 K�ڣ�SnCl2��2H2O/[1ChCl:2EG] DES��Sn2+�ĵ绯��ԭ��Ӧ��������ɢ���Ƶ�������̣�Sn2+����ɢϵ��Ϊ(0.501~3.023)��10-6 cm2/s��������Һ�¶ȵ�����������
3) ��ʩ���ڵ缫����ĵ�λ��Сʱ������Sn���κ�������ά˲ʱ�κˣ�����ʩ�ӵ�λ����ʱ���κ˹������ɵ���������ά�����κˣ��ر��ǵ�����ʱ���ӳ�ʱ��������һ���ɡ�
�ο����ף�
[1] SEKAR R, EAGAMMAI C, JAYAKRISHNAN S. Effect of additives on electrodeposition of tin and its structural and corrosion behaviour[J]. Journal of Applied Electrochemistry, 2010, 17(1): 87-97.
[2] ����ȫ. ����Һ��: ���ʡ��Ʊ���Ӧ��[M]. ����: �й�ʯ��������, 2006: 377-384.
DENG Youquan. Ionic liquids: properties, preparation and applications[M]. Beijing: Sinopec Press, 2006: 377-384.
[3] ABBOTT A P, FRISCH G, HARTLEY J, et al��Processing of metals and metal oxides using ionic liquids[J]. Green Chemistry, 2011, 13(3): 471-481.
[4] ABBOTT A P, CAPPER G, DAVIES D L, et al. Preparation of novel, moisture-stable, Lewis-acidic ionic liquids containing quaternary ammonium salts with functional side chains electronic supplementary information (ESI) available: plot of conductivity vs. temperature for the ionic liquid formed from zinc chloride and choline chloride (2:1)[J]. Chemical Communications, 2001, 19: 2010-2011.
[5] ABBOTT A P, CAPPER G, MCKENZIE K J, et al. Electrodeposition of zinc-tin alloys from deep eutectic solvents based on choline chloride[J]. Journal of Electroanalytical Chemistry, 2007, 599(2): 288-294.
[6] ANICAI L, PETICA A, COSTOVICI S, et al. Electrodeposition of Sn and NiSn alloys coatings using choline chloride based ionic liquids: evaluation of corrosion behavior[J]. Electrochimica Acta, 2013, 114: 868-877.
[7] WHITEHEAD A H, P LZLER M, GOLLAS B. Zinc electrodeposition from a deep eutectic system containing choline chloride and ethylene glycol[J]. Journal of the Electrochemical Society, 2010, 157(6): D328-D334.
LZLER M, GOLLAS B. Zinc electrodeposition from a deep eutectic system containing choline chloride and ethylene glycol[J]. Journal of the Electrochemical Society, 2010, 157(6): D328-D334.
[8] DALE P J, SAMANTILLEKE A P, SHIVAGAN D D, et al. Synthesis of cadmium and zinc semiconductor compounds from an ionic liquid containing choline chloride and urea[J]. Thin Solid Films, 2007, 515(15): 5751-5754.
[9] BARD A J, FAULKNER L R. Electrochemical methods: fundamentals and applications[M]. New York: J Wiley & Sons, 2000: 230-232.
[10] KUZNETSOV S A, GAUNE-ESCARD M. Kinetics of electrode processes and thermodynamic properties of europium chlorides dissolved in alkali chloride melts[J]. Journal of Electroanalytical Chemistry, 2006, 595(1): 11-22.
[11] GUO Wujie, HOU Yucui, REN Shuhang, et al. Formation of deep eutectic solvents by phenols and choline chloride and their physical properties[J]. Journal of Chemical and Engineering Data, 2013, 58(4): 866-872.
[12] ��һ��. ұ����̶���ѧ����[M]. ����: ұ��ҵ������, 2004: 27-29.
HUA Yixin. The dynamics of metallurgical process[M]. Beijing: Metallurgical Industry Press, 2004: 27-29.
[13] SALOM S, PEREIRA N M, FERREIRA E S, et al. Tin electrodeposition from choline chloride based solvent: influence of the hydrogen bond donors[J]. Journal of Electroanalytical Chemistry, 2013, 703(16): 80-87.
S, PEREIRA N M, FERREIRA E S, et al. Tin electrodeposition from choline chloride based solvent: influence of the hydrogen bond donors[J]. Journal of Electroanalytical Chemistry, 2013, 703(16): 80-87.
[14] TACHIKAWA N, SERIZAWA N, KATAYAMA Y, et al. Electrochemistry of Sn(��)/Sn in a hydrophobic room- temperature ionic liquid[J]. Electrochimica Acta, 2008, 53(22): 6530-6534.
[15] YAMAGATA M, TACHIKAWA N, KATAYAMA Y, et al. Electrochemical behavior of several iron complexes in hydrophobic room-temperature ionic liquids[J]. Electrochimica Acta, 2007, 52(9): 3317-3322.
[16] ZHANG Qibo, HUA Yixin. Influence of [B-mim]HSO4 on the nucleation and growth of zinc on aluminum from acidic sulphate bath[J]. Asian Journal of Chemistry, 2013, 25(2): 701-707.
[17] RADISIC A, LONG J G, HOFFMANN P M, et al. Nucleation and growth of copper on TiN from pyrophosphate solution[J]. Journal of the Electrochemical Society, 2001, 148: C41-C46.
[18] SCHARIFKER B, HILLS G. Theoretical and experimental studies of multiple nucleation[J]. Electrochimica Acta, 1983, 28(7): 879-889.
[19] ABYANEH M Y, HENDRIKX J, VISSCHER W, et al. The electrocrystallization of zinc from alkaline media[J]. Journal of the Electrochemical Society, 1982, 129(12): 2654-2659.
[20] SUN JIE, MING Tingyun, QIAN Huixuan, et al. Electrochemical behavior of copper electrodeposition in BMIMPF6Ionic liquid[J]. Chemical Journal of Chinese Universities, 2018, 39(7): 1497-1502.
[21] NAN Tianxiang, YANG Jianguang, CHEN Bing. Electrochemical mechanism of tin membrane electrodeposition under ultrasonic waves[J]. Ultrasonics Sonochemistry, 2018, 42: 731-737.
(�༭ ����ΰ)
�ո����ڣ�2017-09-13�������ڣ�2017-11-02
������Ŀ(Foundation item)�������ص�����о���չ�ƻ�(973�ƻ�)��Ŀ(2014CB643404)��������Ȼ��ѧ����������Ŀ(51504112) (Project(2014CB643404) supported by the National Basic Research Development Program (973 Program) of China; Project(51504112) supported by the National Natural Science Foundation of China)
ͨ�����ߣ���ᣬ���ڣ�˶ʿ����ʦ��������ɫ����ұ���о���E-mail: kglj1010@163.com