Trans. Nonferrous Met. Soc. China 23(2013) 406-411
Effects of nano-additive TiO2 on performance of micro-arc oxidation coatings formed on 6063 aluminum alloy
Hong-xia LI1, Ren-guo SONG2, Zhen-guo JI1
1. College of Materials and Environmental Engineering, Hangzhou Dianzi University, Hangzhou 310018, China;
2. Key Laboratory of Advanced Metal Materials of Changzhou City, Changzhou University, Changzhou 213164, China
Received 18 November 2011; accepted 18 April 2012
Abstract: Ceramic coatings were fabricated on aluminum alloy substrates by micro-arc oxidation (MAO) in silicate electrolytes doped with different concentrations of TiO2 nano-additive. Effects of nano-additive concentration on the structural and mechanical properties of the MAO coatings were analyzed. The results revealed that some nano-particle were incorporated into the resulting coating during the MAO process, while there was a reasonable concentration for the TiO2 nano-additive. With increasing the nano-additive concentration to 3.2 g/L, the adhesion value increased, while mean friction coefficient and mass loss decreased. A further increase of nano-additive deteriorated the adhesion and mean friction coefficient values, which was consistent with the micro-hardness tests.
Key words: Al alloy; micro-arc oxidation; nano-additive; micro-hardness; friction and wear
1 Introduction
Micro-arc oxidation (MAO) has attracted much interest as an effective technique to improve the hardness and wear resistance of aluminum alloys by forming a relatively thick and hard ceramic coating on aluminum alloys through plasma discharging under high voltage [1-3]. Thus, the technique shows a wide prospect for its application [4,5]. However, the ceramic coatings generally possess a foam-like structure with high bulk porosity and relatively poor mechanical properties [6,7], which restrict them from even wider technical applications [8]. Up to now, researches mainly focused on the effects of the processing parameters [9-12], such as current density [13]; however, nano-additive doping in the electrolyte also had great influence on the properties of the ceramic coatings [14,15]. JIN et al [8] found that the MAO Al2O3 coatings with Fe micro-grains were much denser and harder, and the wear resistance was also improved significantly since ceramic coatings became denser with a lower population of the micro-pores [8]. SHI et al [16] fabricated coatings in Na2SiO3�CNa3PO4 solution system by doping two kinds of additives (Na2B4O7 and EDTA). The results revealed that doping of additives had little effect on the elemental composition, while it influenced the morphological feature of the coating. However, few data can be available concerning on the effect of nano-additive concentrations on the properties of the prepared MAO coatings.
In this work, in order to improve the structural and mechanical properties of ceramic coatings prepared by MAO method, a new electrolyte based on silicate solution with different concentrations of TiO2 nano-additive was used to fabricate ceramic coatings on 6063 alloy. Effects of different nano-additive concentrations on the structural and mechanical properties of the MAO coatings were analyzed.
2 Experimental
The substrate materials selected for this study were commercial 6063 aluminum alloys (0.45%-0.90% Mg, 0.2%-0.6% Si, 0.35% Fe, 0.10% Cu, 0.10% Mn, 0.10% Cr, 0.10% Zn, 0.10% Ti and balance Al in mass fraction). The samples with dimensions of 30 mm��25 mm��3 mm were polished with abrasive paper and degreased with acetone followed by rinsing with distilled water before coating formation. The electrolyte was a dilute aqueous solution of 1 g/L KOH and 10 g/L Na2SiO3. Rutile TiO2 nano-additive with size about 10 nm was dispersed evenly in the solution and the concentration of nano-additives was in a range of 0.8-4.0 g/L. The electrolyte was agitated with a mechanical stirrer. A five- litter bath from the stainless steel was used. The specimen was served as the anode and the bath wall was served as the contrary electrode. The system was cooled by cold water pumped through double walls of the bath. The electrolyte temperature was controlled at 25-30 ��C throughout the process. The power supply was pulsed DC MAO equipment (DSM30IM) with the maximum voltage amplitude of 1000 V. During the MAO process, the current density was fixed at 15 A/dm2 by controlling the voltage amplitude and the pulse width was 5000 ��s. Time of coating formation was 1 h according to our experimental experience, under which the prepared coatings had optimal properties. After the treatment, the samples were rinsed in distilled water and dried in air.
The crystallographic characteristics of the coatings were investigated using a Thermo ARL X�� TRA X-ray diffractometer (XRD) (Cu K�� radiation) with step size of 0.04��. The X-ray generator settings were 45 kV and 40 mA, respectively. The surface morphology of the coatings was characterized by a Hitachi S-4700 scanning electron microscope (SEM). Energy dispersive X-ray spectroscopy (EDX) was used for qualitative elemental chemical analysis. The coating hardness was evaluated using a HMV-IT micro-hardness tester with Vickers indenter under a load of 1.96 N (200 g). The adhesion of the coatings to substrate was carried out on a conventional WS-2005 scratch tester at a constant linear velocity of 4 mm/min and a loading rate of 100 N/min. The corresponding critical load represented the adhesion to substrate for the coatings. The tribological properties of the coatings were performed on a WTM-2E ball-on-disk tribometer with a rotational speed of 336 r/min. The coating was served as the disk, and the counterpart was a Si3N4 ceramic ball (4 mm in diameter, HV1550 in hardness). The tests were carried out at a normal load of 9.8 N with a wear track radius of 15 mm. The abrasion loss was measured after 1 h friction measurement using an electronic direct reading balance (LJBROR L-200, readability 0.01 mg).
3 Results and discussion
3.1 Phase analysis
Figure 1 shows XRD patterns of the ceramic coatings prepared in silicate electrolyte with different concentrations of TiO2 nano-additive. For the coating prepared in silicate solution without nano-additives, it can be seen that the prepared ceramic coatings consist of two crystal phases of ��-Al2O3 and ��-Al2O3 in addition to some diffraction peaks of 6063 alloy. It is similar for the coatings prepared in silicate solution with TiO2 nano-additive; however, the peak intensity at 46�� with addition of TiO2 nano-powder is increased because of the appearance of TiO2 phase, which indicates that some nano-particles have entered into the prepared ceramic coatings. As well known, continuous and long time discharge at 15 A/dm2 results in a high temperature in the discharge channels, which is propitious to the deposition of TiO2 nano-particles [15]. So TiO2 peaks at 46�� can be detected in the coating. However, titanate compound was not formed during the process since no corresponding diffraction peaks were found.
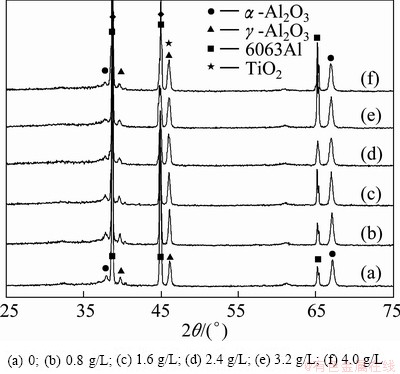
Fig. 1 XRD patterns of ceramic coatings prepared with different TiO2 nano-additive concentrations
Generally, the in situ growth of the Al2O3 film on the aluminum alloy by MAO technique includes the following processes.
Cathode reaction:
2H++2e��H2�� (1)
Anode reaction:
4OH-��2H2O+O2��+4e (2)
Al��Al3++3e (3)
Al3++3OH-��Al(OH)3 (4)
2Al(OH)3��Al2O3+3H2O (5)
2Al3++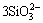 ��3SiO2+Al2O3 (6)
��3SiO2+Al2O3 (6)
2Al3++3O2-��Al2O3 (7)
Due to the high temperature and upper voltage caused by discharge sparks, Al3+ from the substrate combines with OH- and O2- in the electrolyte and forms molten Al2O3. Simultaneously, the molten Al2O3 erupts from the discharge channels and forms the ceramic coatings by quenching of electrolyte with a high cooling rate.
3.2 SEM observation and EDX analysis
Figure 2 represents the morphology of the coatings prepared in silicate electrolyte with different TiO2 nano-additive concentrations. It was found that the surface of the MAO coating was porous and loose (Fig. 2(a)), while it changed greatly for the coatings prepared with nano-additives in comparison with that without nano-additives. Compositions and structures of the prepared coatings combine both substrates and electrolytes during MAO process. With increasing the concentration of TiO2 nano-additives from 0.8 g/L to 4 g/L, the coating surface became denser and smoother and the number of pores decreased. It can be concluded that addition of nano-powder plays an essential role in fabricating ceramic coatings with a lower porosity. For the composite oxide coatings, TiO2 nano-particles could embed into the micro-arc discharge channels through the diffusion by the introduction of TiO2 nano-additive in the electrolyte during MAO. When the TiO2 nano powders were added into the electrolyte, they were homogeneously distributed under vigorous stirring. During MAO, when the molten Al2O3 erupted from the discharge channels, TiO2 particles would be mixed within the Al2O3 coating. We thought that more and more dispersed nano particles entered into the pores with increasing the nano-additive concentrations, so the coating surface became denser and smoother. The coating color was darkened from grey white to blue black. By this method, high quality coatings with high micro-hardness, strong adhesion and excellent wear resistance could be synthesized.
The EDX spectrum collected from polished cross- sections of ceramic coatings with TiO2 nano-additive concentration of 4 g/L shows the elemental composition and element concentration (Fig. 3). It can be seen that the prepared coating mainly consists of elements Al, O, Si and Ti, which confirms that some TiO2 nano-additive has melted into the resulting coating. Figure 4 shows the major elemental concentrations of ceramic coatings prepared with different TiO2 nano-additive concentrations. From Fig. 4, it can be seen that the content of O changed little, while the content of Al decreased and the content of Ti increased with increasing nano-additive concentration. The mass fraction of Ti was 10.29% for the coating prepared in silicate solution with nano-additive concentration of 4.0 g/L, which increased 2 times more than compared with that prepared with nano-additive concentration of 0.8 g/L.
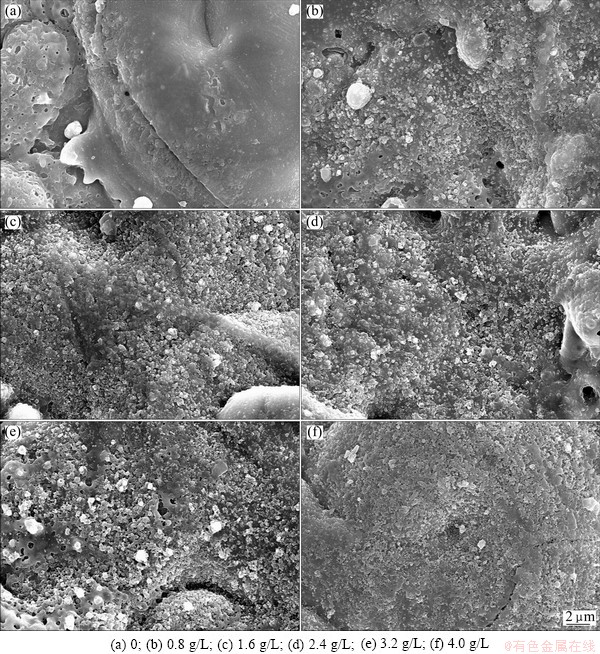
Fig. 2 SEM images of ceramic coatings with different TiO2 nano-additive concentrations
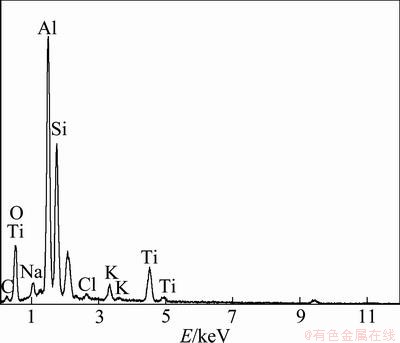
Fig. 3 EDX spectrum of ceramic coating with nano-additive concentration of 4 g/L
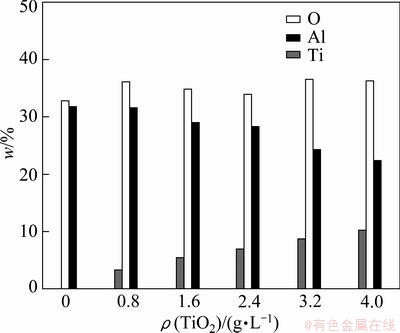
Fig. 4 Elemental concentrations of ceramic coatings prepared with different TiO2 nano-additive concentrations
3.3 Micro-hardness tests
Micro-hardness tests were carried out on the samples prepared with different nano-additive concentrations, which are shown in Fig. 5. To improve the accuracy of the micro-hardness value, several-point hardness tests were utilized to get an average hardness value. For the coating prepared without nano-additive, the average micro-hardness value is about HV1356.5. It increases with increasing TiO2 nano-additive concentration, and is about HV1620 for the coating prepared with nano-additive concentration of 3.2 g/L, which increased by 19% compared with that prepared without nano-additive. The value decreased to HV1585 when the nano-additive concentration was further increased to 4.0 g/L, which indicates that there is a reasonable concentration of TiO2 nano-additive for the silicate solution. From above analysis, we can see that each micro-hardness value of the ceramic coating prepared with nano-additive is higher than that without nano-additive. Such a considerable increase in hardness is attributed to the better crystallization and smoother surface morphology with fewer defects. However, when the nano-additive concentration was further increased to 4.0 g/L, the concentration of TiO2 embedded in the coatings becomes higher. The decrease of hardness value may be caused by the too much TiO2 nano-additive incorporation since the hardness of TiO2 is lower than that of ceramic Al2O3 coating.
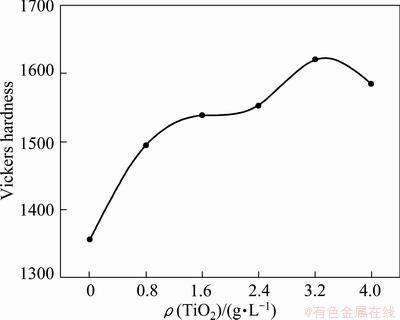
Fig. 5 Average value of micro-hardness of ceramic coatings prepared with different TiO2 nano-additive concentrations
3.4 Adhesion and friction tests
Adhesion values of the coatings prepared with different TiO2 nano-additive concentrations are shown in Fig. 6. In this work, the addition of nano-powder to electrolyte led to a remarkable improvement of adhesion values. Adhesion value increased with increasing the nano-additive concentrations, and reached the highest value for the coating prepared with nano-additive concentration of 3.2 g/L. A further increase of nano-additive concentration deteriorated the bond of the coating to aluminum substrate, which is consistent with the micro-hardness tests.
Figure 6 also shows mean friction coefficients with test duration for the coatings prepared with different nano-additive concentrations. The addition of nano-powder to electrolyte led to reduction of friction coefficient. The coating prepared with nano-additive concentration of 3.2 g/L exhibited the lowest mean friction coefficient of 0.673 and the value was more stable during the tests. The mass loss of the coatings prepared with different nano-additive concentrations was measured after 1 h fiction test (Fig. 6). The value was decreased to 0.09 mg for the ceramic coating prepared with nano-additive concentration of 3.2 g/L, which indicates excellent wear resistance. It is mainly attributed to its dense surface, high hardness and relatively low friction coefficient. However, no obvious change was found when the nano-additive concentration was increased to 4.0 g/L.
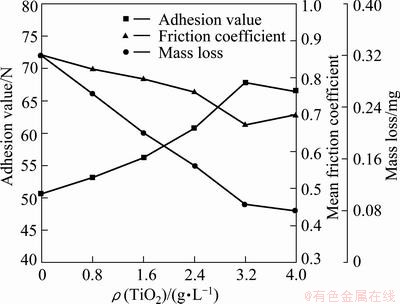
Fig. 6 Adhesion value, average friction coefficient and mass loss of ceramic coatings prepared with different TiO2 nano-additive concentrations
4 Conclusions
1) Ceramic coatings were synthesized on 6063 aluminum alloy substrates in silicate electrolyte with different TiO2 nano-additive concentrations by MAO technique.
2) XRD and EDX analyses showed that TiO2 nano-particles were melted into the prepared ceramic coatings. The surface morphology of the coatings with nano-additive changed greatly in comparison with that without nano-additive.
3) The average micro-hardness value and adhesion value of the ceramic coatings increased with increasing nano-additive concentration to 3.2 g/L, while the values decreased for the coating prepared with nano-additive concentration of 4.0 g/L. The lowest mean friction coefficient of 0.673 and excellent wear resistance were measured for the coating prepared with nano-additive concentration of 3.2 g/L, which was thought the reasonable concentration of TiO2 nano-additive in the silicate solution with current density of 15 A/dm2 and oxidation time of 1 h. The effects of other fabrication parameters during MAO process on the performance of TiO2 doped Al2O3 coatings will be discussed next.
References
[1]  Wei-ling, CHEN Ti-jun, MA Ying, XU Wei-jun, YANG Jian, HAO Yuan. Effects of increase extent of voltage on wear and corrosion resistance of micro-arc oxidation coatings on AZ91D alloy [J]. Transactions of Nonferrous Metals Society of China, 2008, 18(s): s354-s360.
Wei-ling, CHEN Ti-jun, MA Ying, XU Wei-jun, YANG Jian, HAO Yuan. Effects of increase extent of voltage on wear and corrosion resistance of micro-arc oxidation coatings on AZ91D alloy [J]. Transactions of Nonferrous Metals Society of China, 2008, 18(s): s354-s360.
[2] LI Hong-xia, RUDNEV V S, ZHENG Xiao-hua, YAROVAYA T P, SONG Ren-guo. Characterization of Al2O3 ceramic coatings on 6063 aluminum alloy prepared in borate electrolytes by micro-arc oxidation [J]. J Alloys Compound, 2008, 462: 99-102.
[3] WANG Chang-zheng, ZHANG Dong, JIANG Yong-feng. Growth process and wear resistance for ceramic coatings formed on Al�CCu�CMg alloy by micro-arc oxidation [J]. Applied Surface Science, 2006, 253: 674-678.
[4] GUO Hong-fei, AN Mao-zhong. Effect of surfactants on surface morphology of ceramic coatings fabricated on magnesium alloys by micro-arc oxidation [J]. Thin Solid Films, 2006, 500: 186-189.
[5] XIN Shi-gang, SONG Li-xin, ZHAO Rong-gen, HU Xing-fan. Influence of cathodic current on composition, structure and properties of Al2O3 coatings on aluminum alloy prepared by micro-arc oxidation process [J]. Thin Solid Films, 2006, 515: 326-332.
[6] BAI A. Optimization of anti-corrosion ability of micro-arc oxide coating on AZ91D alloy using experimental strategies [J]. Surface & Coatings Technology, 2010, 204: 1856-1862.
[7] FORNO A D, BESTETTI M. Effect of the electrolytic solution composition on the performance of micro-arc anodic oxidation films formed on AM60B magnesium alloy [J]. Surface & Coatings Technology, 2010, 205: 1783-1788.
[8] JIN Fan-ya, CHU P K, TONG Hong-hui, ZHAO Jun. Improvement of surface porosity and properties of alumina films by incorporation of Fe micrograins in micro-arc oxidation [J]. Applied Surface Science, 2006, 253: 863-868.
[9] ZHOU Fei, WANG Yuan, DING Hong-yan, WANG Mei-ling, YU Min, DAI Zhen-dong. Friction characteristic of micro-arc oxidative Al2O3 coatings sliding against Si3N4 balls in various environments [J]. Surface & Coatings Technology, 2008, 202: 3808�C3814.
[10] YAN Yuan-yuan, HAN Yong, LI Di-chen, HUANG Juan-juan, LIAN Qin. Effect of NaAlO2 concentrations on microstructure and corrosion resistance of Al2O3/ZrO2 coatings formed on zirconium by micro-arc oxidation [J]. Applied Surface Science, 2010, 256: 6359-6366.
[11] OH Y J, MUN J I, KIM J H. Effects of alloying elements on microstructure and protective properties of Al2O3 coatings formed on aluminum alloy substrates by plasma electrolysis [J]. Surface & Coatings Technology, 2009, 204: 141-148.
[12] CAI Jing-shun, CAO Fa-he, CHANG Lin-rong, ZHENG Jun-jun, ZHANG Jian-qing, CAO Chu-nan. The preparation and corrosion behaviors of MAO coating on AZ91D with rare earth conversion precursor film [J]. Applied Surface Science, 2011, 257: 3804-3811.
[13] YANG Yue, WU Hua. Effect of current density on corrosion resistance of micro-arc oxide coatings on magnesium alloy [J]. Transactions of Nonferrous Metals Society of China, 2010, 20: s688-s692.
[14] WU Xiang-qing, XIE Fa-qin, HU Zong-chun, WANG Li. Effects of additives on corrosion and wear resistance of micro-arc oxidation coatings on TiAl alloy [J]. Transactions of Nonferrous Metals Society of China, 2010, 20: 1032-1036.
[15] YANG Yue, LIU Yao-hui. Effects of current density on the microstructure and the corrosion resistance of alumina coatings embedded with SiC nano-particles produced by micro-arc oxidation [J]. Journal of Materials Science and Technology, 2010, 26(11): 1016-1020.
[16] SHI Ling-ling, XU Yong-jun, LI Kang, YAO Zhong-ping, WU Song-quan. Effect of additives on structure and corrosion resistance of ceramic coatings on Mg�CLi alloy by micro-arc oxidation [J]. Current Applied Physics, 2010, 10: 719-723.
TiO2�������Ӽ���6063���Ͻ��������մ�Ϳ�����ܵ�Ӱ��
���ϼ1�����ʹ�2�������1
1. ���ݵ��ӿƼ���ѧ �����뻷������ѧԺ������ 310018��
2. ���ݴ�ѧ �Ƚ��������ϳ������ص�ʵ���ң����� 213164
ժ Ҫ��ͨ���ڹ����ε��Һ�м���TiO2�������Ӽ����о��������Ӽ�Ũ�ȵı仯��6063���Ͻ��������մ�Ϳ�����ܵ�Ӱ�졣����������������Ӽ����뵽�մ�Ϳ���У������Ӽ�Ũ�ȵ�ѡȡ��һ���Ϻ����ķ�Χ�������������Ӽ���Ũ�����ӵ�3.2 g/L��Ϳ��Ľ����������ƽ��Ħ�����������������С����Ũ�����ӵ�4.0 g/Lʱ��Ϳ��Ľ������������ƽ��Ħ���������ӣ�����Ϳ����Ӳ�ȵIJ��Խ��һ�¡�
�ؼ��ʣ����Ͻ����������������Ӽ�����Ӳ�ȣ�Ħ��ĥ��
(Edited by Hua YANG)
Corresponding author: Hong-xia LI; Tel: +86-571-86878609; E-mail: lhx19781226@163.com
DOI: 10.1016/S1003-6326(13)62477-2