用于固态照明的红色荧光粉Lu2MoO6:Eu3+的发光性质
来源期刊:中国有色金属学报(英文版)2016年第6期
论文作者:李丽 沈君 周贤菊 潘雨 常文轩 何琪伟 韦先涛
文章页码:1670 - 1675
关键词:Lu2MoO6:Eu3+;发光性质;红色荧光粉
Key words:Lu2MoO6:Eu3+; luminescent properties; red phosphor
摘 要:采用高温固相法制得Eu3+掺杂的Lu2MoO6荧光粉,通过X射线衍射(XRD)及激发、发射光谱和衰减寿命等手段对样品的结构和发光性质进行了表征。XRD结果表明:制备的荧光粉均为单斜结构。实验结果表明该样品在可见光谱范围内能够被近紫外光有效地吸收,该吸收来自Mo6+–O2-吸收带。在掺杂10% Eu3+的情况下,发光最强。详细地研究最佳临界距离Rc和能量机制。Lu2MoO6:Eu3+ 红色荧光粉是一种可应用于近紫外激发白光LED用的新型红色荧光粉。
Abstract: The Eu3+ activated Lu2MoO6 phosphors were synthesized by high-temperature solid-state reaction method. The X-ray diffraction (XRD), excitation spectra, emission spectra and decay lifetime of the phosphors were measured to characterize the structure and luminescent properties. The XRD results show that all the prepared phosphors can be assigned to the monoclinic structure. The experimental results indicate efficient absorption of near ultraviolet light from the Mo6+-O2- group followed by intensive emission in the visible spectral range. The optimal content of Eu3+ is 10% (mole fraction). The critical distance Rc and energy transfer mechanism were also discussed in detail. This red emitting material may be applied as a promising red phosphor for the near ultraviolet excited white light emitting diodes.
Trans. Nonferrous Met. Soc. China 26(2016) 1670-1675
Li LI1, Jun SHEN1, Xian-ju ZHOU1, Yu PAN1, Wen-xuan CHANG1, Qi-wei HE1, Xian-tao WEI2
1. College of Science, Chongqing University of Posts and Telecommunications, Chongqing 400065, China;
2. Department of Physics, University of Science and Technology of China, Hefei 230026, China
Received 14 July 2015; accepted 3 March 2016
Abstract: The Eu3+ activated Lu2MoO6 phosphors were synthesized by high-temperature solid-state reaction method. The X-ray diffraction (XRD), excitation spectra, emission spectra and decay lifetime of the phosphors were measured to characterize the structure and luminescent properties. The XRD results show that all the prepared phosphors can be assigned to the monoclinic structure. The experimental results indicate efficient absorption of near ultraviolet light from the Mo6+-O2- group followed by intensive emission in the visible spectral range. The optimal content of Eu3+ is 10% (mole fraction). The critical distance Rc and energy transfer mechanism were also discussed in detail. This red emitting material may be applied as a promising red phosphor for the near ultraviolet excited white light emitting diodes.
Key words: Lu2MoO6:Eu3+; luminescent properties; red phosphor
1 Introduction
White light-emitting diodes (WLED) have recently attracted great attention as promising candidates for next-generation lighting due to its low energy consumption, high efficiency, excellent reliability, long lifetime, etc [1,2]. In order to generate white light using LEDs, one approach is to combine a blue LED chip emitting at 465 nm with a broad band yellow Y3Al5O12:Ce3+ (YAG:Ce3+) phosphor. However, it exhibits low color rendering index (CRI) due to lacking of red component in the emission spectra, which limits its application. Another approach is to combine a near-ultraviolet (NUV) InGaN-based LED chip (350-420 nm) with the red, green and blue phosphors. In this approach, the phosphors are excited by the NUV chips, and the tricolor emissions make an excellent white light, which usually offer better color rendering performance [3]. Unfortunately, the efficiency of currently commercially used red phosphor Y2O2S:Eu3+ is much lower than that of green and blue phosphors [4]. Therefore, more efforts should be devoted to searching for efficient red phosphors that can efficiently absorb light in the NUV region (350-420 nm).
Trivalent europium (Eu3+) ions activated phosphors are considered as ideal red sources for WLED because of the sharp emission lines in the red spectral region [5]. For applications in NUV-based WLED, the phosphors should be able to efficiently absorb the emission from the LED chips. However, the f-f excitation of Eu3+ ions is narrow in bandwidth and weak in intensity due to the parity forbidden nature of the f-f transitions, which leads to the low efficiency. In order to enhance light absorption, sensitizers are introduced to absorb the excitation light and transfer the excitation energy to the Eu3+ ions, which is a feasible method to overcome this shortage and obtain promising red phosphors [6].
The Eu3+-doped molybdates and tungstates were investigated because the MoO4 and WO4 groups can efficiently absorb ultraviolet light through the excitation of the Mo-O and W-O charge transfer states (CTS), respectively. This excitation is then followed by a transfer of the absorbed energy to Eu3+ ions for red emission [7,8]. Nevertheless, most of these materials exhibit low efficient absorption in the NUV region. Recently, much attention has been drawn to the Eu3+- doped molybdate and tungstate R2MO6 (R=Y, Gd, La; M=Mo, W) for their remarkable properties such as excellent thermal and chemical stabilities and a broad excitation band [9-12]. However, little attention has been attracted to investigate the Eu3+-doped Lu2MoO6 phosphors. LI et al [13] have synthesized the Lu2MoO6:Eu3+ phosphors by sol-gel method and investigated the structure and photoluminescence properties of such phosphors. However, the structural, luminescent properties and energy transfer mechanism of Eu3+-doped Lu2MoO6 obtained by solid state reaction method were not studied. In this work, Eu3+-doped Lu2MoO6 phosphors were synthesized using the high-temperature solid-state reaction method. The effect of Eu3+ molar concentration on the structure, luminescence and decay lifetime of the Lu2MoO6:Eu3+ phosphors was investigated. The energy transfer mechanism of Eu3+ in Lu2MoO6 host was also discussed in detail.
2 Experimental
2.1 Sample preparation
The Lu2MoO6:Eu3+ phosphors were synthesized by the high-temperature solid-state method. The starting materials are analytical reagent (AR) grade molybdenum trioxide (MoO3), lutetium oxide (Lu2O3) (99.99%) and europium oxide (Eu2O3) (99.99%). The stoichiometric amounts of reactants were ground thoroughly in an agate mortar and preheated at 600 °C for 1 h. Subsequently, the products were removed from the muffle furnace, cooled, finely ground and sintered at 1200 °C for 4 h in air. Finally, the products were cooled to room temperature and then ground into white powder to form the final products.
2.2 Characterization
The crystal structures were analyzed by X-ray diffractometer (Persee, XD-2, Beijing Purkinje General Instrument Co., Ltd, Beijing, China) with Cu Kα radiation (l=0.15406 nm). The excitation, emission spectra and the decay curves were measured by FLSP920 (Edinburgh Instrument Ltd, Livingston, UK) fluorescence spectrophotometer equipped with 450 W xenon lamp or a pulse xenon lamp as light sources and Shimidazu R9287 (Hamamatsu Photonics K.K., Hamamatsu, Japan) photomultiplier (200-900 nm) along with a liquid nitrogen-cooled InGaAs (Hamamatsu Photonics K.K.) (800-1700 nm) as the detectors. All spectra were collected at room temperature under identical experimental conditions so that the emission intensities of the samples with different Eu3+ doping concentrations can be compared.
3 Results and discussion
3.1 Structure analysis of Lu2MoO6:Eu3+ phosphors
Figure 1 shows unit cell of Lu2MoO6 drawn with VESTA [14]. This material is crystallized in the I2/a space group (No.15). The Lu3+ ions occupy three nonequivalent crystallographic sites, namely, 4e, 4e (with C2 site symmetry) and 8f (with C1 site symmetry), and all are coordinated to eight O atoms, whereas the Mo atoms are coordinated to five O atoms with four of which at a short distance and the other at a long distance [15].
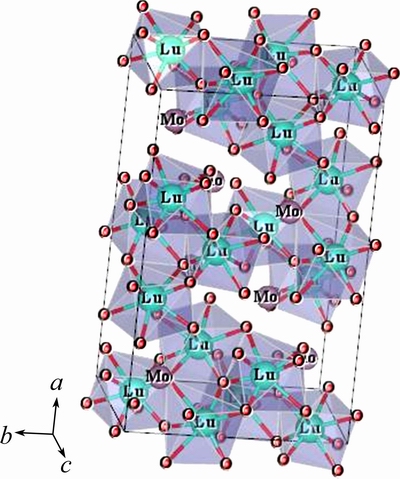
Fig. 1 One unit cell of Lu2MoO6 with 8-fold coordination of Lu3+ ions
Figure 2 shows the XRD patterns of Lu2MoO6 phosphors doped with xEu3+ (x=1%, 3%, 5%, 10%, 20%, 40%). It indicates that all the diffraction peaks coincide well with the data from the JCPDS card No. 25–0972 (Tm2MoO6), and Lu2MoO6 can be assigned to the monoclinic structure [13]. No additional peaks of other phases have been found, indicating that the Eu3+ ions are effectively doped into the host lattice. Additionally, the diffraction peaks of Lu2MoO6:Eu3+ samples are found to shift a little to lower angles with the increase of Eu3+ concentration, this is because the radius of Eu3+ (0.95 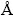 ) is larger than that of Lu3+ (0.85
) is larger than that of Lu3+ (0.85 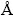 ) in Lu2MoO6 host. When Lu3+ is substituted by Eu3+, the interplanar distance d increases, the diffraction angles decrease according to the Bragg equation: 2dsinθ=λ, where d is the interplanar distance, θ is diffraction angle of peak, and λ is the X-ray wavelength (0.15405 nm). Therefore, it also demonstrates that Eu3+ ions are effectively doped into the Lu2MoO6 host to replace Lu3+ sites.
) in Lu2MoO6 host. When Lu3+ is substituted by Eu3+, the interplanar distance d increases, the diffraction angles decrease according to the Bragg equation: 2dsinθ=λ, where d is the interplanar distance, θ is diffraction angle of peak, and λ is the X-ray wavelength (0.15405 nm). Therefore, it also demonstrates that Eu3+ ions are effectively doped into the Lu2MoO6 host to replace Lu3+ sites.
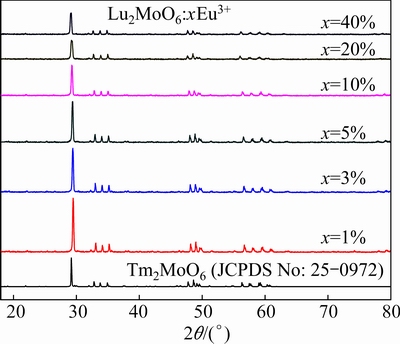
Fig. 2 X-ray diffraction patterns of Lu2MoO6:xEu3+ phosphor contrasted with standard pattern of JCPDS 25-0972
3.2 Photoluminescent properties and luminescence decay lifetime of Lu2MoO6:Eu3+ phosphors
Figure 3 shows the emission spectra of Lu2MoO6:xEu3+ phosphors under 363 nm excitation as a function of the Eu3+ concentration. The emission spectra of the Lu2MoO6:x%Eu3+ phosphors with similar shape consist of sharp lines with wavelength from 550 to 720 nm. The dominant red emission centered at 610 nm is assigned to the 5D0→7F2 transition of Eu3+ ions, while other weak emission located at 578 nm, 585 nm, 657 nm and 706 nm are attributed to the following transitions: 5D0→7F0, 5D0→7F1, 5D0→7F3 and 5D0→7F4, respectively. It is well-known that Eu3+ is an excellent probe ion, because the 5D0→7F2 transition (electric dipole allowed) is very hypersensitive to the chemical environment, while the 5D0→7F1 transition (magnetic dipole allowed) is insensitive to the environment. According to the Judd-Ofelt theory, the magnetic dipole transition is allowed. The electric dipole transition is permitted when the Eu3+ occupies the crystallographic sites without inversion symmetry [16]. Consequently, when the Eu3+ ions occupy the inversion center sites, the 5D0→7F1 transition should be relatively strong, while the 5D0→7F2 transition is very weak. The above experimental results indicate that the Eu3+ mainly occupies the nonequivalent crystallographic sites (both the C1 and C2) without inversion symmetry in the Lu2MoO6 lattice, which is consistent with the reported crystal structure of Lu2MoO6.
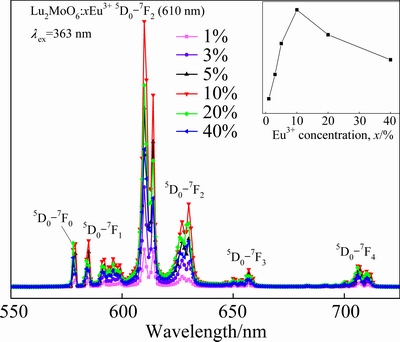
Fig. 3 Emission spectra of Lu2MoO6:xEu3+ phosphors (Inset represents emission intensity as function of Eu3+ concentration)
From the inset in Fig. 3, it is noticed that the emission intensity of 610 nm first increases and approaches a maximum at a doping concentration of 10% and then decreases with the increase of Eu3+ concentration. With the increase of Eu3+ concentration, the luminescence centers of Eu3+ increase, which results in increasing the emission intensity of Eu3+. Whereas, when Eu3+ concentration is very high, the concentration quenching occurs, thus reducing emission intensity. The optimal doping concentration of Eu3+ is 10%.
In order to obtain the concentration quenching mechanism of Eu3+ under high doping concentration, the emission intensity of 5D0→7F2 transition (610 nm) is integrated under 363 nm according to the DEXTER theory [17,18], and its relationship with doping Eu3+ concentration (x) can be expressed as
 (2)
(2)
where s stands for the series of electronic multidipole interaction, s=3, 6, 8, 10 denote the exchange interactions of ions, dipole-dipole, dipole-quadrupole, quadrupole-quadrupole interactions, respectively, and b is a constant [19]. The value of s can be deduced from the slope -s/3 of the linear line in Fig. 4, which plots lg(I/x) vs lg x on a logarithmic scale of I/x. The slope -s/3 can be obtained as -2.1038 by fitting the data. Subsequently, the value of s is calculated as approximately 6. Thus, the results indicate that the concentration quenching mechanism of Eu3+ emission in the Lu2MoO6 host is attributed to the dipole-dipole interaction.
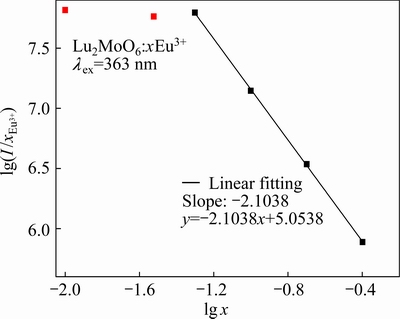
Fig. 4 Logarithmic plots for emission intensity per activator Eu3+ ion (I/x) as function of Eu3+ concentration (x) in Lu2MoO6:Eu3+ phosphors
Moreover, in terms of the Blasse’s concentration quenching theory in inorganic phosphors [20], the critical distance Rc in Lu2MoO6:%Eu3+ phosphors can be estimated from the following equation:
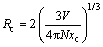 (1)
(1)
where V is the volume of the unit cell of Lu2MoO6 (V=879.749  3), xc is the critical concentration of Eu3+ (xc=0.1), and N is the number of host cations per unit cell (N=8) [13]. From Eq. (1), it can be calculated that the value of Rc is approximately equal to 12.8
3), xc is the critical concentration of Eu3+ (xc=0.1), and N is the number of host cations per unit cell (N=8) [13]. From Eq. (1), it can be calculated that the value of Rc is approximately equal to 12.8 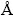 . The results show the different values estimated from the other Eu3+-doped phosphors [21,22], which emphasizes the influence of crystal structure on the luminescent properties of the Eu3+ ions. From the presented experimental results, it is seen that the Lu2MoO6:Eu3+ phosphors exhibit a strong red light with high color purity, which can be utilized to improve the color rendering property of white LEDs.
. The results show the different values estimated from the other Eu3+-doped phosphors [21,22], which emphasizes the influence of crystal structure on the luminescent properties of the Eu3+ ions. From the presented experimental results, it is seen that the Lu2MoO6:Eu3+ phosphors exhibit a strong red light with high color purity, which can be utilized to improve the color rendering property of white LEDs.
Figure 5 shows the excitation spectra of Lu2MoO6:xEu3+ phosphors monitoring the 5D0→7F2 emission at 610 nm. As shown in Fig. 5, the excitation spectra Lu2MoO6:xEu3+ phosphors all have the same profile, while the intensity declines with the increase of the Eu3+ concentration. The excitation spectra consist of two parts: the narrow excitation peaks between 450 nm and 550 nm and the intense broad band ranging from 250 nm to 440 nm. The broad band centered at 363 nm is attributed to the overlap of the Mo6+-O2- and Eu3+-O2 charge transfer states [23]. The narrow peaks at 462 nm and 530 nm correspond to the 7F0→5D2 and 7F0→5D1 transitions of Eu3+, respectively. Therefore, the Lu2MoO6:Eu3+ phosphors can effectively absorb the emission in the 370-400 nm range from near-ultraviolet LED chips, which may be a potential candidate for application in near-ultraviolet excited WLED. Additionally, the observation of the Mo6+-O2- charge transfer state in the excitation spectra of Eu3+ indicates that energy transfer from the MoO66- groups to the emitting Eu3+ ions is present.
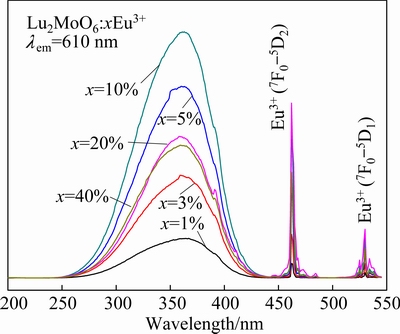
Fig. 5 Excitation spectra of Lu2MoO6:xEu3+ phosphors
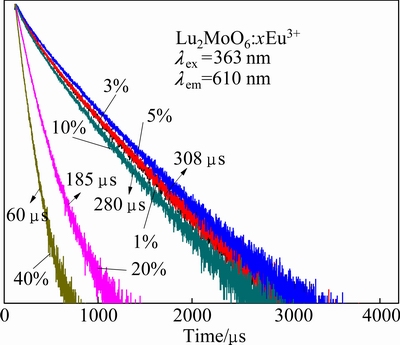
Fig. 6 Luminescence decay curves of Eu3+ (lex=363 nm, lem= 610 nm) in Lu2MoO6:xEu3+ phosphors
Figure 6 shows the luminescence decay curves of red emission (610 nm) for Lu2MoO6:xEu3+ phosphors under 363 nm excitation. The decay curves of phosphors show nonexponential characteristics and a mean decay lifetime (τm) can be evaluated by
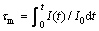 (3)
(3)
where I(t) is the luminescence intensity at the time t, and I0 represents the initial intensity at t=0 [24]. The calculated lifetimes are shown in the inset in Fig. 6. When the concentration of Eu3+(x) increases from 0 to 5%, there is no apparent change in the decay curve. As x reaches 10%, 20% and 40%, the faster decay was observed.
The emission spectra of the optimal Lu2MoO6: 10%Eu3+ and commercial Y2O2S:5%Eu3+ phosphors under 363 nm excitation are shown in Fig. 7. It is obvious that both the spectra show the characteristic emission of the Eu3+ ions. However, the spectral distributions are different as a result of different site symmetry for the Eu3+ ions in these two host lattices [5]. Moreover, the emission intensity of Lu2MoO6:10%Eu3+ under 363 nm excitation is about 2.2 times higher than that of Y2O2S:5%Eu3+ by comparing the two emission spectra. This result suggests that the Lu2MoO6:Eu3+ phosphors are potential red phosphors for near ultraviolet based solid state lighting.
Figure 8 shows the CIE (Commission Internationale de L’Eclairage) chromaticity diagram of Lu2MoO6: 10%Eu3+ phosphor under 363 nm excitation [25]. The Lu2MoO6:10%Eu3+ phosphors exhibit strong red emission. The corresponding chromaticity coordinates were calculated to be (0.65, 0.35), which are marked with Point 1 in the red luminescent region of Fig. 8. Expected values of the commercial Y2O2S:Eu3+ phosphors and the red phosphors are (0.62, 0.35) and (0.67, 0.33), which are shown with Points 2 and 3, respectively [26]. Based on our results, we suggest that Lu2MoO6:Eu3+ phosphors have met the commercial requirement in solid-state lighting.
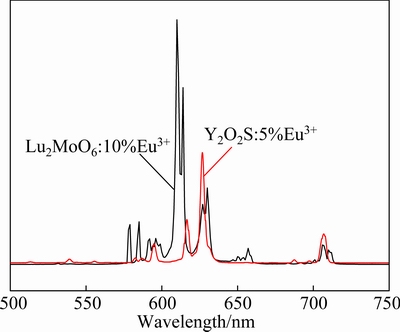
Fig. 7 Emission spectra of Lu2MoO6:10%Eu3+ and Y2O2S: 5%Eu3+ phosphors under 363 nm excitation
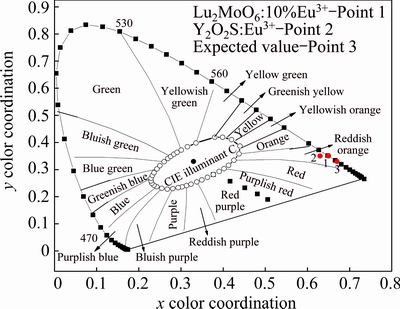
Fig. 8 CIE chromaticity diagrams of Lu2MoO6:10%Eu3+ phosphors under 363 nm excitation
3.3 Energy transfer mechanism
According to the investigation above, the energy level diagrams involved in the energy transfer process from the host to Eu3+ in Lu2MoO6 are presented in Fig. 9. Under ultra-violet light excitation, the MoO66- group is excited owing to charge transfer transition from the O 2p states in the host’s valence band to the Mo 4d states in the host’s conduction band. The absorption energy can be transferred to the Eu3+ ions followed by red emission from the 5D0→7FJ transitions as shown in Fig. 3, because the resonant nonradiative energy transfer will occur, as illustrated in Fig. 9.
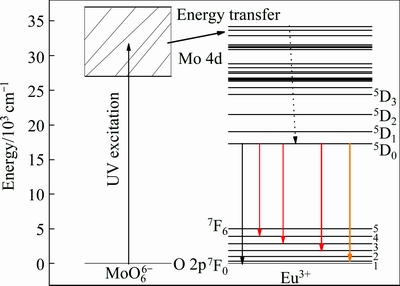
Fig. 9 Energy level diagram of MoO66- and Eu3+ with energy transfer process
4 Conclusions
1) The Eu3+ activated Lu2MoO6 phosphors were synthesized by the high-temperature solid-state reaction method. The XRD results indicate that Lu2MoO6 can be assigned to the monoclinic structure.
2) The excitation and emission spectra show that Lu2MoO6:Eu3+ phosphors can be effectively excited by the near ultraviolet light (363 nm) and exhibit a strong red emission at 610 nm. The optimal Eu3+-doped concentration and critical distance are 10% and 12.8 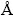 , respectively. The concentration quenching mechanism of Eu3+ emission in the Lu2MoO6 host is ascribed to the dipole-dipole interaction.
, respectively. The concentration quenching mechanism of Eu3+ emission in the Lu2MoO6 host is ascribed to the dipole-dipole interaction.
3) The experimental spectroscopic results presented in this work show the Lu2MoO6:Eu3+ phosphor as an excellent candidate for getting red emission for near ultraviolet based solid state lighting.
References
[1] LIN C C, LIU R S. Advances in phosphors for light-emitting diodes [J]. Journal of Physical Chemistry Letters, 2011, 2: 1268-1277.
[2] YE S, XIAO F, PAN Y X, MA Y Y, ZHANG Q Y. Phosphors in phosphor-converted white light-emitting diodes: Recent advances in materials, techniques and properties [J]. Materials Science and Engineering R, 2010, 71: 1-34.
[3] LIU R S, LIU YH, BAGKAR N C, HU S F. Enhanced luminescence of SrSi2O2N2:Eu2+ phosphors by codoping with Ce3+, Mn2+, and Dy3+ions [J]. Applied Physics Letters, 2007, 91: 061119.
[4] KUTTY T R N, NAG A. Role of interface states associated with transitional nanophase precipitates in the photoluminescence enhancement of SrTiO3:Pr3+, Al3+ [J]. Journal of Materials Chemistry, 2003,13: 2271-2278.
[5] DENG K M, GONG T, CHEN Y H, DUAN CK, YIN M. Efficient red-emitting phosphor for near-ultraviolet-based solid-state lighting [J]. Optics Letters, 2011, 36: 4470-4472.
[6] HAN B, ZHANG J, WANG Z M, LIU YY, SHI H Z. Investigation on the concentration quenching and energy transfer of red-light-emitting phosphor Y2MoO6:Eu3+ [J]. Journal of Luminescence, 2014, 149: 150-154.
[7] ZHOU L Y, WEI J S, YI L H, GONG F Z, HUANG J L, WANG W. A promising red phosphor MgMoO4:Eu3+ for white light emitting diodes [J]. Materials Research Bulletin, 2009, 44: 1411-1414.
[8] WANG Z J, ZHONG J P, LIANG H B, WANG J. Luminescence properties of lutetium based redemitting phosphor NaLu(WO4)2:Eu3+ [J]. Optical Materials Express, 2013, 3: 418-425.
[9] WANG M F, ZHANG H, LI L, LIU X G, HONG F, LI R, SONG H J, GUI M X, SHEN J R, ZHU W H, WANG J B, ZHOU L Q, JEONG J H. Charge transfer bands of Mo-O and photoluminescence propertiesof micro-material Y2MoO6:Eu3+ red phosphor [J]. Journal of Alloys and Compounds, 2014, 585: 138-145.
[10] PANG M L, LIU X M. Luminescence properties of R2MoO6:Eu3+ (R=Gd, Y, La) phosphors prepared by Pechini sol-gel process [J]. Journal of Materials Research, 2005, 20: 2676-2681.
[11] LI H Y, NOH H M, MOON B K, CHOI B C, JEONG J H, LEE H S, YI S S. Wide-band excited Y6(W Mo)0.5O12:Eu red phosphor for white light emitting diode: Structure evolution, photoluminescence properties, and energy transfer mechanisms involved [J]. Inorganic Chemistry, 2013, 52: 11210-11217.
[12] LEI F, YAN B, CHEN H H. Solid-state synthesis, characterization and luminescent properties of Eu3+-doped gadolinium tungstate and molybdate phosphors:Gd(2-x)MO6:Eux3+ (M=W, Mo) [J]. Journal of Solid State Chemistry, 2008,181: 2845-2851.
[13] LI H Y, YANG H K, JEONG J H, JANG K, LEE H S, YI S S. Sol-gel synthesis, structure and photoluminescence properties of nanocrystalline Lu2MoO6:Eu [J]. Materials Research Bulletin, 2011, 46: 1352-1358.
[14] MOMMA K, IZUMI F. VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data [J]. Journal of Applied Crystallography, 2011, 44: 1272-1276.
[15] ALONSO J A, RIVILLAS F, MARTINEZ-LOPE M J, POMJAKUSHIN V. Preparation and structural study from neutron diffraction data of R2MoO6 (R=Dy, Ho, Er, Tm, Yb, Y) [J]. Journal of Solid State Chemistry, 2004, 177: 2470-2476.
[16] HAN B, LIANG H B, NI H Y, SU Q, YANG G T, SHI J Y, ZHANG G B. Intense red light emission of Eu3+ doped LiGd(PO3)4 for mercury-free lamps and plasma display panels application [J]. Optics Express, 2009, 17: 7138-7144.
[17] DEXTER D L. A theory of sensitized luminescence in solids [J]. Journal of Chemical Physics, 1953, 21: 836-850.
[18] DEXTER D L, JAMES H S. Theory of concentration quenching in inorganic phosphors [J]. Journal of Chemical Physics, 1954, 22: 1063-1070.
[19] van UITERT L G. Characterization of energy transfer interactions between rare earth ions [J]. Journal of the Electrochemical Society, 1967, 114: 1048-1053.
[20] BLASSE G. Energy transfer in oxidic phosphors [J]. Physics Letters A, 1968, 28: 444-445.
[21] YU R J, NOH H M, MOON B K, CHOI B C, JEONG J H, JANG K, YI S S, JANG J K. Synthesis and luminescence properties of a novel red-emitting phosphor Ba3La(PO4)3:Eu3+ for solid-state lighting [J]. Journal of Alloys and Compounds, 2013, 576: 236-241.
[22] GUO H, ZHANG H, WEI R F, ZHENG M D, ZHANG L H. Preparation, structural and luminescent properties of Ba2Gd2Si4O13:Eu3+ for white LEDs [J]. Optics Express, 2011, 19: A201-A206.
[23] BLASSE G, CORSMIT A F. Electronic and vibrational spectra of ordered perovskites [J]. Journal of Solid State Chemistry, 1973, 6: 513-518.
[24] AN Y T, LABBE C, CARDIN J, MORALES M, GOURBILLEAU F. Highly efficient infrared quantum cutting in Tb3+-Yb3+ codoped silicon oxynitride for solar cell applications [J]. Advanced Optical Materials, 2013, 1: 855-862.
[25] CHO H, HWANG S M, LEE J B, KA D H, KIM T W, LEE B S, LEE J Y, LEE J I, RYU J H. White luminescence of Ho3+/Tm3+/Yb3+- codoped CaWO4 synthesized via citrate complex route assisted by microwave irradiation[J]. Transactions of Nonferrous Metals Society of China, 2014, 24(S1): s134-s140.
[26] WANG Z J, LI P L, YANG Z P, GUO Q L. A novel red phosphor BaZn2(PO4)2:Sm3+, R+ (R= Li, Na, K) [J]. Journal of Luminescence, 2012, 132: 1944-1948.
李 丽1,沈 君1,周贤菊1,潘 雨1,常文轩1,何琪伟1,韦先涛2
1. 重庆邮电大学 理学院,重庆 400065;
2. 中国科学技术大学 物理系,合肥 230026
摘 要:采用高温固相法制得Eu3+掺杂的Lu2MoO6荧光粉,通过X射线衍射(XRD)及激发、发射光谱和衰减寿命等手段对样品的结构和发光性质进行了表征。XRD结果表明:制备的荧光粉均为单斜结构。实验结果表明该样品在可见光谱范围内能够被近紫外光有效地吸收,该吸收来自Mo6+–O2-吸收带。在掺杂10% Eu3+的情况下,发光最强。详细地研究最佳临界距离Rc和能量机制。Lu2MoO6:Eu3+ 红色荧光粉是一种可应用于近紫外激发白光LED用的新型红色荧光粉。
关键词:Lu2MoO6:Eu3+;发光性质;红色荧光粉
(Edited by Yun-bin HE)
Foundation item: Project (11404047) supported by the National Natural Science Foundation of China; Projects (CSTC2015jcyjA50005, CSTC2014JCYJA50034) supported by the Natural Science Foundation Project of Chongqing, China; Project (KJ1500412, KJ1500409) supported by Scientific and Technological Research Program of Chongqing Municipal Education Commission, China
Corresponding author: Li LI; Tel: +86-23-62471721; E-mail: lilic@cqupt.edu.cn
DOI: 10.1016/S1003-6326(16)64276-0

