
Aging precipitation and recrystallization in
high-nitrogen austenitic stainless steel
SHI Feng(石 锋)1,2, WANG Li-jun(王立军)3, CUI Wen-fang(崔文芳)3, QI Yang(祁 阳)1,3, LIU Chun-ming(刘春明)2
1. School of Science, Northeastern University, Shenyang 110004, China;
2. School of Materials and Metallurgy, Northeastern University, Shenyang 110004, China;
3. Key Laboratory for Anisotropy and Texture of Materials, Ministry of Education,
Northeastern University, Shenyang 110004, China
Received 10 August 2009; accepted 15 September 2009
Abstract: The interaction between precipitation and recrystallization in cold deformed Fe-18Cr-12Mn-0.48N high-nitrogen austenitic stainless steel was investigated by means of hardness test, optical microscopy (OM) and transmission electron microscopy (TEM). The results show that the recrystallization of the steel begins at about 750℃. When aging at 750℃, the precipitation occurs prior to recrystallization. Large numbers of the second phases nucleate in dislocation, grain boundary and subgrain boundary. Precipitation of the second-phase particles hinders the formation of recrystallization nucleus.
Key words: high-nitrogen austenitic stainless steel; aging; precipitation; recrystallization
1 Introduction
As an interstitial element, N added to austenitic stainless steel (ASS) is beneficial for increasing its strength without significant loss of toughness and ductility when it is dissolved in matrix[1-2]. In addition, the addition of N can reduce materials cost through a replacement of expensive Ni because of its strong austenite-stabilizing effect similarly to Ni, and can improve the localized corrosion resistance of ASS[1, 3-7]. Therefore, a great deal of attention is now focused on high-nitrogen austenitic stainless steels (HNASS) containing more than 0.4% N (mass fraction)[8]. The aging reactions at elevated temperatures may induce the precipitation of carbides, nitrides, and carbonitrides etc in HNASS. Cold work can accelerate the precipitation of the second phases and induce the precipitation of intermetallics[9]. During aging treatment of cold deformed HNASS, the two processes affect each other unless recrystallization is complete before precipitation begins[10]. The interaction between precipitation and recrystallization will have effects on the microstructures and properties of steel. The research on this aspect is proceeding like a raging fire in Al alloy, nickel-based alloy and Cu alloy etc[11-16], but there are few reports on HNASS. In this work, the interaction between precipitation and recrystallization is investigated in a newly developed HNASS which is cold deformed to obtain some rich information and to play a role in the extensive application for this type of steel.
2 Experimental
The steel used in present study was melted in induction furnace and electroslag remelting furnace both filled with N2. The chemical composition of the steel is 18.76% Cr, 11.8% Mn, 0.48% N, 0.041% C, 0.02% Al, 0.16% Si and balance Fe(mass fraction). The ingot was hot forged into 25 mm×25 mm square bars. The square bars were solid solution treated at 1 150 ℃ for 1 h and followed by water quenching. Then, they were cut into rod bar specimens with 6 mm in diameter and 10 mm in length and cold compressed by 30% in thickness. After cold compression, some specimens were aged for 1 h from 600 ℃ to 900 ℃ with a temperature interval of 50 ℃ and other specimens were aged for 30 min and 1 h at 750 ℃.
After being etched using a solution of HCl + picric acid + alcohol, microstructures were observed by
Olympus GX 71 optical microscopy (OM). Thin foils for transmission electron microscopy (TEM) were prepared using a twin-jet electrolytic polisher, operated in an electrolyte of perchloric acid and alcohol at 50 V and -30 ℃. The micro-hardness was measured using 401 MVDTM micro-Vickers hardness tester under a 50 g load and 10 s holding time.
3 Results and discussion
3.1 Confirmation of recrystallization temperature
Fig.1 shows the variation of micro-hardness of the cold compressed steel with aging temperature. The steel begins to remarkably soften above 700 ℃. The softening may result from the mutual effect of recrystallization and precipitation. However, the experimental steel is not one of the steels to be strengthened by aging. Therefore, the softening should be caused by the recrystallization. If the temperature corresponding to half of sum of micro-hardness of the cold compressed steel and the completely softened steel is defined as the beginning temperature of recrystallization, it is about 750 ℃ according to the microhardness test.
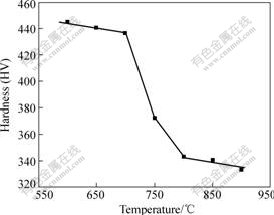
Fig.1 Variation of micro-hardness of cold compressed steel with aging temperature
Fig.2 shows the microstructures of the cold compressed specimens aged at 700, 750 and 800 ℃ for 1 h. No recrystallized grains but the obvious precipitates were observed in the specimen aged at 700 ℃ for 1 h, as shown in Fig.2(a). Recrystallization began in the specimen aged at 750 ℃ for 1 h, as shown in Fig.2(b), and was quite obvious in the specimen aged at 800 ℃ for 1 h, as shown in Fig.2(c). The microstructural results are coincident with those of micro-hardness test shown in Fig.1.
3.2 Interaction between precipitation and recrystalli- zation
Fig.3 shows the OM and TEM observations of

Fig.2 Microstructures of cold compressed specimens aged at 700 ℃ (a), 750 ℃ (b) and 800 ℃ (c) for 1 h
specimens aged at 750 ℃ for 30 min and 1 h. In the specimen aged at 750 ℃ for 30 min, the recrystallization did not happen as shown in Fig.3(a), but the precipitation inside the grain and at the grain boundary took place as shown in Figs.3(b) and (c). All the precipitates are identified as s phase with bct structure, and their lattice parameters were determined to be a=0.880 0 nm and c=0.454 4 nm from the electron diffraction pattern of the precipitate. The bigger precipitates coarsen and smaller precipitates dissolve with prolonging the aging time, which makes the ability of precipitate to pin the dislocations and the grain boundaries become lower. The recrystallization nucleus was observed in the specimen aged at 750 ℃ for 1 h, as shown in Fig.3(d). In conclusion, the precipitation occurs prior to recrystallization at 750 ℃.
In the specimen aged at 750 ℃ for 30 min, various deformation microstructures were observed, as shown in Figs.4(a)-(c). The deformation mode includes slipping

Fig.3 Microstructures of cold compressed specimens aged at 750 ℃ for 30 min (OM (a) and TEM (b, c)) to show precipitates and 1 h (TEM (d)) to show recrystallization nucleus

Fig.4 TEM microstructures aged at 750 ℃ for 30 min after cold-deformation by 30%: (a) Slipping bands; (b) Deformation twins; (c) Dislocation tangle; (d) Subgrain; (e) Precipitates inside subgrain and at subgrain boundary
(Fig.4(a)) and twinning (Fig.4(b)), and high density dislocation appears in deformed specimen, as shown in Fig.4(c). These high density dislocations provide favorite nucleation sites for precipitation of second-phase particles. Large numbers of second-phase particles precipitate in dislocation (Figs.3(b) and (c)). These precipitates could pin dislocation and hinder its motion and rearrange, thus inhibit the stage of recovery to a certain extent. In this specimen, the formation of the subgrain structure was also observed, as shown in Fig.4(d). Grain boundary, dislocation and subgrain boundary are all the advantaged sites for precipitation of second-phase particles in deformed specimen. After subgrain formation, the precipitates could nucleate in subgrain boundary immediately if conditions are satisfied. Fig.4(e) shows the precipitates inside the subgrain and subgrain boundary. Thus, they inevitably affects the growth of subgrain once the precipitation occurs during recovery. These precipitates pinning subgrain boundary could hinder the growth of subgrain, further hindering the formation of recrystallization nucleus by polygonization, subgrain growth and migration of subgrain boundary. This can be explained by the following equation[17]:
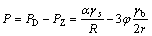 (1)
(1)
where P is the driving force for subgrain growth; PD is the driving force causing by interface energy of subgrain; PZ is Zener resistance causing by pinning of the second-phase particles; α is a shape factor; R and r are radii of subgrain and the second-phase particle, respectively; φ is the volume fraction of the second-phase particles; and gs and gb are the interface energy of subgrain boundary and interface energy affecting pinning of the second-phase particles, respectively. The precipitates preferentially nucleate in defect sites such as dislocation and subgrain boundary in deformed microstructure, making pinning pressure PZ rapidly augment, which results in the fact that the driving force of subgrain growth P, that is, the driving force of recrystallization reduces and hinders the formation of recrystallization nucleus. So the precipitation in the specimen aged at 750 ℃ for 30 min delays the occurrence of recrystallization.
4 Conclusions
1) After 30% cold deformation, the recrystallization of the experimental steel begins at about 750 ℃.
2) When aging at 750 ℃, the precipitation occurs prior to recrystallization. The precipitation of these second-phase particles in dislocation, grain boundary and subgrain grain boundary hinders the formation of recrystallization nucleus.
References
[1] DI SCHINO A, KENNY J M. Effect of grain size on the corrosion resistance of a high nitrogen -low nickel austenitic stainless steel [J]. Journal of Materials Science Letters, 2002, 21(24): 1969-1971.
[2] VANDERSCHAEVE F, TAILLARD R, FOCT J. Discontinuous precipitation of Cr2N in a high nitrogen, chromium-manganese austenitic stainless steel [J]. Journal of Materials Science, 1995, 30(23): 6035-6046.
[3] BANDY R, LU Y C, NEWMAN R C, CLAYTON C R. Role of nitrogen in improving resistance to localized corrosion in austenitic stainless steels [C]//Proceedings-the Electrochemical Society. NJ: Electrochemical Soc Inc, 1984: 471-481.
[4] BABA H, KODAMA T, KATADA Y. Role of nitrogen on the corrosion behavior of austenitic stainless steels [J]. Corrosion Science, 2002, 44: 2393-2407.
[5] KURODA D, HIROMOTO S, HANAWA T, KATADA Y. Corrosion behavior of nickel-free high nitrogen austenitic stainless steel in simulated biological environments [J]. Materials Transactions, 2002, 43(12): 3100-3104.
[6] BREQLIOZZI G, DI SCHINO A, HAEFKE H, KENNY J M. Cavitation erosion resistance of a high nitrogen austenitic stainless steel as a function of its grain size [J]. Journal of Materials Science Letters, 2003, 22(13): 981-983.
[7] BAYOUMI F M, GHANEM W A. Effect of nitrogen on the corrosion behavior of austenitic stainless steel in chloride solutions [J]. Materials Letters, 2005, 59: 3311-3314.
[8] SIMMONS J W. Overview: high-nitrogen alloying of stainless steels [J]. Materials Science and Engineering A, 1996, 207(2): 159-169.
[9] SHI Feng, WANG Li-jun, CUI Wen-fang, LIU Chun-ming. Precipitates in an isothermally aged Fe-18Cr-12Mn-0.04C-0.48N high-nitrogen austenitic stainless steel [J]. Rare Metals, 2007, 26(Spec. Issue): 185-190.
[10] MARTIN J W, DOHERTY R D. Stability of microstructure in metallic systems [M]. London: Cambridge University Press, 1976: 148-150.
[11] ZHANG Xin-ming, WU Wen-xiang, LIU Sheng-dan, JIAN Xiong, TANG Jian-guo. Precipitation and recrystallization of AA3003 aluminium alloy during annealing [J]. Journal of Central South University: Science and Technology, 2006, 37(1): 1-5.(in Chinese)
[12] KREJCI J, KARMAZIN L. Phase transformations and recrystallization in Ni-33at.% Cr alloy [J]. Materials Science and Engineering A, 1994, 188(1/2): 185-191.
[13] LIU Ping, CAO Xing-guo, KANG Bu-xi, HUANG Jin-liang, GU Hai-cheng. The mutual action of aging precipitation and recrystallization and its effects on the properties of rapidly solidified Cu-Cr-Zr-Mg alloy [J]. Journal of Function Materials, 2000, 31(2): 167-169. (in Chinese)
[14] KLIBER J, SCHINDLER I. Recrystallization/precipitation behaviour in microalloyed steels [J]. Journal of Materials Processing Technology, 1996, 60: 597-602.
[15] RYU J H, LEE D N. The effect of precipitation on the evolution of recrystallization texture in AA8011 aluminum alloy sheet [J]. Materials Science and Engineering A, 2002, 336: 225-232.
[16] LEE K J. Recrystallization and precipitation interaction in Nb-containing steels [J]. Scripta Materialia, 1999, 40(7): 837-843.
[17] HUMPHREYS F J, HATHERLY M. Recrystallization and related annealing phenomena [M]. Oxford: Elsevier Science Ltd, 1995: 164-166.
(Edited by YUAN Sai-qian)
Foundation item: Project(2004CB619103) supported by the National Basic Research Program of China; Project(50534010) supported by the National Natural Science Foundation of China
Corresponding author: LIU Chun-ming; Tel: +86-24-83687554; E-mail: cmliu@mail.neu.edu.cn