前处理工艺对Li[Ni1/3Co1/3Mn1/3]O2正极材料结构、形貌和电化学性能的影响
来源期刊:中国有色金属学报(英文版)2011年第9期
论文作者:杨顺毅 王先友 刘子玲 陈权启 杨秀康 魏启亮
文章页码:1995 - 2001
关键词:锂离子电池;Li[Ni1/3Co1/3Mn1/3]O2;碳酸盐共沉淀法;前处理工艺;电化学性能
Key words:lithium ion batteries; Li[Ni1/3Co1/3Mn1/3]O2; carbonate co-precipitation method; pretreatment process; electrochemical characteristics
摘 要:以[Ni1/3Co1/3Mn1/3]3O4和氢氧化锂为原料,分别采用球磨法和液相法前处理工艺制备层状正极材料Li[Ni1/3Mn1/3Co1/3]O2。采用X-射线衍射(XRD)、场发射扫描电镜(FESEM)、恒流充放电等手段对材料的物理和电化学性能进行表征。结果表明:采用不同前处理工艺制备出的Li[Ni1/3Mn1/3Co1/3]O2材料在结构、形貌和电化学性能上有较大差异;与球磨处理法制备的材料相比,采用液相法前处理工艺制备的Li[Ni1/3Mn1/3Co1/3]O2不但保持了前驱体较好的球形形貌,同时还具有较好的循环稳定性和倍率性能;该样品在20 mA/g电流密度下,首次放电容量为178 mA・h/g,50次循环后,容量保持率达98.7%;在1 000 mA/g电流密度下,样品容量为135 mA・h/g。
Abstract:
The layered Li[Ni1/3Mn1/3Co1/3]O2 was separately synthesized by pretreatment process of ball mill method and solution phase route, using [Ni1/3Co1/3Mn1/3]3O4 and lithium hydroxide as raw materials. The physical and electrochemical behaviors of Li[Ni1/3Mn1/3Co1/3]O2 were characterized by X-ray diffraction (XRD), scanning electron microscopy (SEM), field emission scanning electron microscopy (FESEM) and electrochemical charge/discharge cycling tests. The results show that the difference in pretreatment process results in the difference in compound Li[Ni1/3Co1/3Mn1/3]O2 structure, morphology and the electrochemical characteristics. The Li[Ni1/3Mn1/3Co1/3]O2 prepared by solution phase route maintains the uniform spherical morphology of the [Ni1/3Co1/3Mn1/3]3O4, and it exhibits a higher capacity retention and better rate capability than that prepared by ball mill method. The initial discharge capacity of this sample reaches 178 mA・h/g and the capacity retention after 50 cycles is 98.7% at a current density of 20 mA/g. Moreover, it delivers high discharge capacity of 135 mA・h/g at a current density of 1 000 mA/g.

YANG Shun-yi, WANG Xian-you, LIU Zi-ling, CHEN Quan-qi, YANG Xiu-kang, WEI Qi-liang
Key Laboratory of Environmentally Friendly Chemistry and Applications of Ministry of Education,
School of Chemistry, Xiangtan University, Xiangtan 411105, China
Received 28 September 2010; accepted 20 December 2010
Abstract: The layered Li[Ni1/3Mn1/3Co1/3]O2 was separately synthesized by pretreatment process of ball mill method and solution phase route, using [Ni1/3Co1/3Mn1/3]3O4 and lithium hydroxide as raw materials. The physical and electrochemical behaviors of Li[Ni1/3Mn1/3Co1/3]O2 were characterized by X-ray diffraction (XRD), scanning electron microscopy (SEM), field emission scanning electron microscopy (FESEM) and electrochemical charge/discharge cycling tests. The results show that the difference in pretreatment process results in the difference in compound Li[Ni1/3Co1/3Mn1/3]O2 structure, morphology and the electrochemical characteristics. The Li[Ni1/3Mn1/3Co1/3]O2 prepared by solution phase route maintains the uniform spherical morphology of the [Ni1/3Co1/3Mn1/3]3O4, and it exhibits a higher capacity retention and better rate capability than that prepared by ball mill method. The initial discharge capacity of this sample reaches 178 mA?h/g and the capacity retention after 50 cycles is 98.7% at a current density of 20 mA/g. Moreover, it delivers high discharge capacity of 135 mA?h/g at a current density of 1 000 mA/g.
Key words: lithium ion batteries; Li[Ni1/3Co1/3Mn1/3]O2; carbonate co-precipitation method; pretreatment process; electrochemical characteristics
1 Introduction
In recent years, there is a great deal of interest in using lithium transition metal oxide, Li[Ni1/3Co1/3Mn1/3]O2, as the positive electrode material for high-energy and high-power Li-ion secondary batteries. This material attracts significant interest because the combination of nickel, cobalt and manganese can provide advantages such as higher reversible capacity with mild thermal stability at charged state, lower cost and less toxicity compared with LiCoO2 [1-3]. Unfortunately, it is difficult to prepare this complicated material, and this material shows low rate capability depending on the synthetic route.
To synthesize the ideal Li[Ni1/3Co1/3Mn1/3]O2 cathode material, many researchers suggested using co-precipitation method to prepare the precursor firstly [4-6]. The hydroxide co-precipitation route is one of the most often used methods and can easily obtain homogeneous precursor [Ni1/3Co1/3Mn1/3](OH)2. However, Mn(OH)2(Mn2+) in the reaction solution can easily be oxidized to be MnOOH (Mn3+) or MnO2(Mn4+) upon precipitation conditions, which results in the mixed transition metal hydroxide [Ni1/3Co1/3Mn1/3](OH)2 unstable [7]. By contrast, the carbonate co-precipitation route can form a homogeneous precursor of [Ni1/3Co1/3Mn1/3]CO3, and the manganese remains as Mn2+ in the carbonate precursor. It was proved to be an efficient technology to synthesize homogeneous and pure Li[Ni1/3Co1/3Mn1/3]O2 with high electrochemical performance.
In general, the preparation of Li[Ni1/3Co1/3Mn1/3]O2 by carbonate co-precipitation method could be divided into three steps. The first step is a co-precipitation of transition metal salts in a continuous-flow stirred tank reactor (CSTR). The second step consists of mixing the precipitate or pre-calcined precursor with the stoichiometric amount of lithium salt, leading to good dispersion of lithium salt and precursor in the mixture. The last step is to heat the mixture at a given temperature in order to form well-order layered Li[Ni1/3Co1/3Mn1/3]O2 cathode materials. During the synthesis process, each step could influence the properties of Li[Ni1/3Co1/3Mn1/3]O2, such as structure, composition, morphology, and electrochemical performance. In recent years, many works focused on optimizing the preparation conditions of the first step and the third step [8]. Nevertheless, it has been found that the pretreatment processes in the second step will influence the electrochemical properties of materials, but to our best knowledge, the works have not been seen so far.
In this work, using NH4HCO3 as the complexing agent, the spherical [Ni1/3Co1/3Mn1/3]CO3 was firstly synthesized by carbonate co-precipitation method adopting Na2CO3-NH4HCO3 route. Then the [Ni1/3Co1/3Mn1/3]CO3 was fired at 500 °C for 5 h to decompose the carbonate into an oxide compound [Ni1/3Co1/3Mn1/3]3O4 as precursor. Two different pretreatment processes between the precursor and lithium hydroxide were used to synthesize the layered Li[Ni1/3Mn1/3Co1/3]O2. The effects of the pretreatment process on the structure, morphology and electrochemical properties of the layered Li[Ni1/3Co1/3Mn1/3]O2 were studied.
2 Experimental
2.1 Chemical preparation
Spherical [Ni1/3Co1/3Mn1/3]CO3 was firstly synthesized by carbonate co-precipitation process. A 1.8 mol/L aqueous solution of NiSO4, CoSO4, and MnSO4 with n(Co)?n(Ni)?n(Mn) of 1?1?1 was pumped into a CSTR reactor with a capacity of 3 L. At the same time, a 1.8 mol/L aqueous solution of Na2CO3 and desired amount of precipitant agent NH4HCO3 were independently fed into the reactor. The solution was maintained at 60 °C for 12 h while accurately monitoring the pH at 7.5 of the mixture and a stirring speed of 1 000 r/min. Then, the spherical powder was filtered, washed, and vacuum-dried inside an oven at 40 °C for several hours. Thus prepared powder was further dried at 110 °C to remove the adsorbed water and to obtain carbonate powder [Ni1/3Co1/3Mn1/3]CO3. The [Ni1/3Co1/3Mn1/3]CO3 was fired at 500 °C for 5 h to obtain the precursor [Ni1/3Co1/3Mn1/3]3O4.
Two different pretreatment processes between the [Ni1/3Co1/3Mn1/3]3O4 precursor and lithium hydroxide (LiOH・H2O) were used to synthesize Li[Ni1/3Co1/3Mn1/3]O2. The first process is ball mill method. Stoichiometric amount of precursor [Ni1/3Co1/3Mn1/3]3O4 and lithium hydroxide were mixed with a mole ratio of Li to M (M=Ni, Co, Mn) of 1.05:1 in the presence of ethyl alcohol, and the mixture was ground for 4 h at a stirring speed of 300 r/min using a ND-2L ball miller (NDDZ, China). Then, the ground mixture was dried at 80 °C in air to evaporate ethyl alcohol.
The second process is solution phase route. LiOH・H2O was slowly dissolved in distilled water to get a homogenous lithium hydroxide solution; [Ni1/3Co1/3Mn1/3]3O4 precursor was added to the above solution with a mole ratio of 1.05:1 of Li to M (M=Ni, Co, Mn) and mixed thoroughly for 2 h to obtain black suspension. The suspension was continuously stirred at 80 °C until the slurry was formed. Then a desired amount of ethyl alcohol was added to the slurry and continuously stirred for 4 h. To evaporate ethyl alcohol, the slurry was dried at 80 °C in air.
The above two mixtures were pre-calcined at 500 °C for 5 h in air to melt lithium hydroxide and then calcined at 900 °C for 12 h in air to obtain the final product Li[Ni1/3Co1/3Mn1/3]O2.
2.2 Instruments and measurements
The phase identification of the samples were performed with a D/Max-3C diffractometer (Rigaku, Japan) using Cu Kα radiation (λ=1.541 78 ?) and a graphite monochromator at 36 kV, 20 mA. The scanning rate was 8 (°)/min and the scanning range of diffraction angle (2θ) was 10°≤2θ≤80°. The surface morphologies of the [Ni1/3Co1/3Mn1/3]CO3 powder and [Ni1/3Co1/3Mn1/3]3O4 precursor were observed using the Gemini 1525 high resolution field emission scanning electron microscope (FESEM) (LEO, Germany). The mixtures and the final product Li[Ni1/3Co1/3Mn1/3]O2 were observed using the JSM-5600LV SEM (JEOL, Japan).
2.3 Cell preparation and electrochemical tests
The electrochemical tests of LiNi1/3Co1/3Mn1/3O2 were carried out using coin cells assembled in an argon-filled glove box. In all cells, the cathode consisted of a mixture of active material (80%, mass fraction, the same below), acetylene black (10%), graphite (5%) and polyvinylidene fluoride (PVDF) as binder agent (5%), and lithium served as counter and reference electrodes; a Celgard 2400 was used as separator; the electrolyte was a 1 mol/L LiPF6 solution in ethylene carbonate (EC)- dimethyl carbonate (DMC) (1:1, V/V). Galvanostatic charge/discharge measurements were carried out in BTS-XWJ-6.44S-00052 Neware battery test system (Newell, China).
3 Results and discussion
3.1 Structure and morphology analysis
Through the co-precipitation process, brown colored [Ni1/3Co1/3Mn1/3]CO3 powder was obtained. The X-ray diffraction pattern of the [Ni1/3Co1/3Mn1/3]CO3 powder is presented in Fig. 1(a). The co-precipitated carbonate [Ni1/3Co1/3Mn1/3]CO3 is a single-phase and has typical structures corresponding to those of NiCO3 (JCPDS No.12-0771), CoCO3 (JCPDS No. 11-0692) and MnCO3 (JCPDS No. 44-1472), all of which have divalent transition metals in their formal charge. This structure was indexed to a hexagonal structure with a space group of R-3c. High resolution field emission scanning electron micrograph of the [Ni1/3Co1/3Mn1/3]CO3 is given in Fig. 2(a). The particle morphology of the carbonate is spherical with average secondary particle diameter of about 15 ?m.
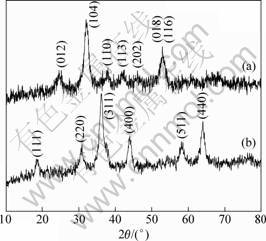
Fig. 1 X-ray diffraction patterns of [Ni1/3Co1/3Mn1/3]CO3 (a) and [Ni1/3Co1/3Mn1/3]3O4 (b)

Fig. 2 High resolution field emission scanning electron micrographs of [Ni1/3Co1/3Mn1/3]CO3 (a) and [Ni1/3Co1/3Mn1/3]3O4 (b)
The carbonate [Ni1/3Co1/3Mn1/3]CO3 powder was fired to form [Ni1/3Co1/3Mn1/3]3O4. After firing at 500 °C for 5 h, the hexagonal carbonate structure changed to a cubic spinel Co3O4 structure as seen from Fig. 1(b), the calculated lattice constant is a=8.245 0 ?. As can be seen in Fig. 2(b), the [Ni1/3Co1/3Mn1/3]3O4 particles maintain the basic morphology from [Ni1/3Co1/3Mn1/3]CO3.
SEM was used to compare the morphologies of the mixtures synthesized by different pretreatment processes. As shown in Figs. 3(a) and (b), the [Ni1/3Co1/3Mn1/3]3O4 sphere is crushed into many small particles with irregular shape and mixed randomly with lithium hydroxide after ball milling. By contrast, the mixture prepared by solution phase route could retain the spherical morphology of [Ni1/3Co1/3Mn1/3]3O4 precursor, and the surface of sphere becomes rough since the lithium hydroxide particles attach to the surface of the [Ni1/3Co1/3Mn1/3]3O4 ball, as shown in Figs. 3(c) and (d).
Figure 4 shows the SEM images of the final product Li[Ni1/3Co1/3 Mn1/3]O2. It can be seen from Figs. 4(a) and (b) that the secondary particle of the sample prepared by ball mill method has an irregular shape, and it is an agglomeration of particles with small grain size. However, the sample synthesized by the solution phase route keeps the morphology of the precursor [Ni1/3Co1/3Mn1/3]3O4 after the high temperature calcinations, as shown in Figs. 4(c) and (d). Each of spherical particles is made up of a large number of primary grains with less than 1 μm in diameter. Usually, this kind of spherical morphology could favor to enhance high rate capability of the material [8].
Figure 5 shows the XRD patterns of the Li[Ni1/3Co1/3Mn1/3]O2 prepared by different pretreatment processes. Both the samples have typical hexagonal α-NaFeO2 structure (space group: 166,![]() ), no secondary phase was observed in the figure. The narrow diffraction peaks indicate the high crystalline of both Li[Ni1/3Co1/3Mn1/3]O2 powders. Clear peak splits of (006)/(102) and (018)/(110) can be observed in Fig. 5, which indicates the formation of highly ordered layered structure for both Li[Ni1/3Co1/3Mn1/3]O2 powders [9]. According to Ref. [10], the rI-factor defined by (I006+I102)/I101 is an indicator of the hexagonal ordering, the lower the rI, the better the hexagonal ordering. In addition, the I003/I104 is also a parameter to characterize the cation mixing and the decrease of this ratio indicates the increase of the structural deviation from hexagonal to cubic symmetry. It was reported that the undesirable cation mixing would appear when the I003/I104 is smaller than 1.2 [9]. The lattice parameters obtained by fitting the (hkl) and 2θ values for the compound are summarized in Table 1. The Li[Ni1/3Co1/3Mn1/3]O2 sample prepared by solution phase route exhibits a lower rI=(I006+I102)/I101 value and higher I003/I104 value than the sample prepared by ball mill method. This reveals that the former has a better layered structure than the latter, which implies that it might have better electrochemical performance [11-12].
), no secondary phase was observed in the figure. The narrow diffraction peaks indicate the high crystalline of both Li[Ni1/3Co1/3Mn1/3]O2 powders. Clear peak splits of (006)/(102) and (018)/(110) can be observed in Fig. 5, which indicates the formation of highly ordered layered structure for both Li[Ni1/3Co1/3Mn1/3]O2 powders [9]. According to Ref. [10], the rI-factor defined by (I006+I102)/I101 is an indicator of the hexagonal ordering, the lower the rI, the better the hexagonal ordering. In addition, the I003/I104 is also a parameter to characterize the cation mixing and the decrease of this ratio indicates the increase of the structural deviation from hexagonal to cubic symmetry. It was reported that the undesirable cation mixing would appear when the I003/I104 is smaller than 1.2 [9]. The lattice parameters obtained by fitting the (hkl) and 2θ values for the compound are summarized in Table 1. The Li[Ni1/3Co1/3Mn1/3]O2 sample prepared by solution phase route exhibits a lower rI=(I006+I102)/I101 value and higher I003/I104 value than the sample prepared by ball mill method. This reveals that the former has a better layered structure than the latter, which implies that it might have better electrochemical performance [11-12].

Fig. 3 SEM images of mixture of [Ni1/3Co1/3Mn1/3]3O4 and lithium hydroxide prepared by ball mill method (a), (b) and prepared by solution phase route (c), (d) at different magnifications

Fig. 4 SEM images of Li[Ni1/3Co1/3Mn1/3]O2 samples prepared by ball mill method (a), (b) and prepared by solution phase route (c), (d) at different magnifications
Table 1 Lattice parameters of Li[Ni1/3Co1/3Mn1/3]O2 calculated by unit cell package software

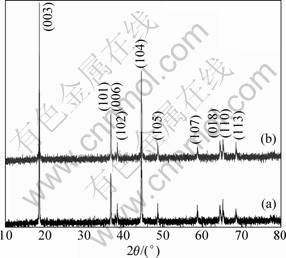
Fig. 5 XRD patterns of Li[Ni1/3Co1/3Mn1/3]O2 sample prepared by ball mill method (a) and solution phase method (b)
3.2 Electrochemical properties
Figure 6 shows the charge/discharge curves for both Li[Ni1/3Co1/3Mn1/3]O2 cathode materials vs Li metal by applying a constant current of 20 mA/g between 2.5 and 4.4 V. In Fig. 6(a), Li[Ni1/3Co1/3Mn1/3]O2 synthesized by ball mill method shows the typical potential plateaus at 3.75 V which originates from the Ni2+/Ni4+ redox couple [13], and it delivers the discharge capacity of 174 mA?h/g at the first cycle, continuous cycling results in severe capacity loss, the capacity retention rate is merely 86.5% after 50 cycles. It is important to note from Fig. 6(b) that the charge/discharge curves of Li[Ni1/3Co1/3Mn1/3]O2 prepared by solution phase route are very smooth and consistent, and have little change even after 50 cycles. It delivers an initial discharge capacity of approximately 178 mA?h/g, and the capacity retention remains 98.7% after 50 cycles.
Figure 7 shows the cycle performance of Li[Ni1/3Co1/3Mn1/3]O2 at 100 mA/g current density between 2.5 and 4.4 V. The Li[Ni1/3Co1/3Mn1/3]O2 sample synthesized by ball mill exhibits the initial discharge capacities of 159.7 mA?h/g, and the capacity retention after 100 cycles is only 80.0%. However, the sample prepared by solution phase route has discharge capacity of 171.8 mA?h/g at the first cycle and excellent cycle performance. The capacity retention reaches 93.7% after 100 cycles.
To realize lithium-ion batteries for high power applications, it requires the electrode materials with good rate capability, which means that the electrode can maintain a large amount of its full capacity when discharged at a high rate. The rate capabilities of the Li[Ni1/3Co1/3Mn1/3]O2 materials synthesized by different pretreatment processes are shown in Fig. 8. The cells were charged galvanostatically with a 0.2C (40 mA/g) before each discharge testing, then discharged at different rates from 0.2C to 5C stepwise, and finally returned to 0.2C. For the sample prepared by ball mill method, it can be seen that the discharge capacity reduces quickly with the increase of the rate. At 5C, the delivered capacity was only 50-60 mA?h/g. The lowering of discharge curve at high rate is due to the polarization of the cell induced by the cell resistance, which is mainly contributed by the Li ion transport into and through the electrode [14-15]. However, the capacity of the sample synthesized by solution phase route is obviously higher than that of sample prepared by ball mill method. It delivers a discharge capacity of 169.9 mA?h/g at 0.5C (the capacity retention rate is about 97.6 % of that at 0.2C), 163.8 mA?h/g at 1C (the capacity retention rate is about 93.1% of that at 0.2C) and 150.6 mA?h/g at 2C (the capacity retention rate is about 86.5% of that at 0.2C). Even at 5C (1 000 mA/g), the capacity of sample is still as high as 135 mA?h/g and the capacity retention rate is about 78.1% of that at 0.2C rate. This excellent capacity retention at a high current density is mainly attributed to its highly ordered layer structure, spherical morphology and good packing properties of the secondary micro-spheres accumulated by primary particles.
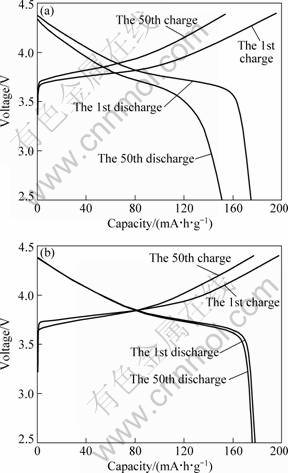
Fig. 6 Charge and discharge curves of Li[Ni1/3Co1/3Mn1/3]O2 cathode materials prepared by different pretreatment processes at current density of 20 mA/g between 2.5 and 4.4 V: (a) Ball mill method; (b) Solution phase route

Fig. 7 Cycle performance of Li[Ni1/3Co1/3Mn1/3]O2 prepared by different pretreatment processes at current density of 100 mA/g between 2.5 and 4.4 V
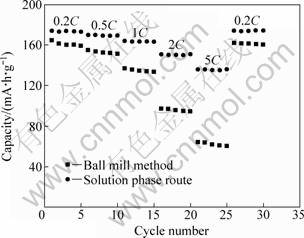
Fig. 8 Cycle performance of Li/Li[Ni1/3Co1/3Mn1/3]O2 cell at various current densities between 2.5 and 4.4 V
4 Conclusions
1) Li[Ni1/3Co1/3Mn1/3]O2 materials with different morphology and electrochemical performance were obtained by the ball mill method and solution phase route, respectively.
2) The sample prepared by ball mill method shows an irregular shape, which is different from the precursor, and displays poor electrochemical performance.
3) Li[Ni1/3Co1/3Mn1/3]O2 material synthesized by solution phase route has a well-ordered layer structure and shows nearly the same spherical shape as the precursor, and it reveals a higher discharge capacity, better cycle performance and rate capability than the sample prepared by ball mill method.
4) It is believed that the pretreatment process between the precursor and lithium hydroxide plays an important role in controlling the morphology and electrochemical characteristics of the Li[Ni1/3Co1/3Mn1/3]O2 during the synthesis process, and the solution phase route is an efficient way to prepare the uniform and spherical Li[Ni1/3Co1/3Mn1/3]O2 material with excellent electrochemical performance.
References
[1] OHZUKU T, MAKIMURA Y. Layered lithium insertion material of LiCo1/3Ni1/3Mn1/3O2 for lithium-ion batteries [J]. Chem Lett, 2001, 30(7): 642-643.
[2] PARK S H, SHIN H S, MYUNG S T, YOON C S, AMINE K, SUN Y K. Synthesis of nanostructured Li[Ni1/3Co1/3Mn1/3]O2 via a modified carbonate process [J]. Chem Mater, 2005, 17(1): 6-8.
[3] SUN Y K, MYUNG S T, PARK B C, PRAKASH J, BELHAROUAK I, AMINE K. High-energy cathode material for long-life and safe lithium batteries [J]. Nat Mater, 2009, 8(4): 320-324.
[4] ZHANG X Y, JIANG W J, MAUGER A, LU Q, GENDROND F, JULIEN C M. Minimization of the cation mixing in Li1+x(NMC)1-xO2 as cathode material [J]. J Power Sources, 2010, 195(5): 1292-1301.
[5] ZHANG S. Characterization of high tap density Li[Ni1/3Co1/3Mn1/3]O2 cathode material synthesized via hydroxide co-precipitation [J]. Electrochim Acta, 2007, 52(25): 7337-7342.
[6] DENG C, LIU L, ZHOU W, SUN K, SUN D. Effect of synthesis condition on the structure and electrochemical properties of Li[Ni1/3Mn1/3Co1/3]O2 prepared by hydroxide co-precipitation method [J]. Electrochim Acta, 2008, 53(1): 2441-2447.
[7] DENG C, ZHANG S, FU B L, YANG S Y, MA L. Synthetic optimization of nanostructured Li[Ni1/3Mn1/3Co1/3]O2 cathode material prepared by hydroxide coprecipitation at 273 K [J]. J Alloys Compd, 2010, 496(1-2): 521-527.
[8] CHO T H, PARK S M, YOSHIO M, HIRAI T, HIDESHIMA Y. Effect of synthesis condition on the structural and electrochemical properties of Li[Ni1/3Mn1/3Co1/3]O2 prepared by carbonate co-precipitation method [J]. J Power Sources, 2005, 142(1-2): 306-312.
[9] OHZUKU T, UEDA A, NAGAYAMA M. Electrochemistry and structure chemistry of LiNiO2 (![]() ) for 4 volt secondary lithium cells [J]. J Electrochem Soc, 1993, 140(7): 1862-1870.
) for 4 volt secondary lithium cells [J]. J Electrochem Soc, 1993, 140(7): 1862-1870.
[10] REIMERS J N, ROSSEN E, JONES C D, DAHN J R. Structure and electrochemistry of LixFeyNi1-yO2 [J]. Solid State Ionics, 1993, 61(4): 335-344.
[11] HU Chuan-yue, GUO Jun, WANG Xing-yan. Synthesis and electrochemical properties of layered Li(Ni3/8Co3/8Mn2/8)O2 cathode material for lithium ion batteries [J]. The Chinese Journal of Nonferrous Metals, 2008, 18(9): 1711-1726. (in Chinese)
[12] GUO Hua-jun, ZHANG Ming, LI Xin-hai, ZHANG Xin-ming, WANG Zhi-xing, PENG Wen-jie, HU Min. Synthesis and characterization of LiNi0.45Co0.1Mn0.45O2 cathode for lithium ion batteries [J]. Transactions of Nonferrous Metals Society of China, 2005, 15(5): 1185-1189.
[13] BELHAROUAK I, SUN Y K, LIU J, AMINE K. Li(Ni1/3Co1/3Mn1/3)O2 as a suitable cathode for high power applications [J]. J Power Sources, 2003, 123(2): 247-252.
[14] YU Xiao-yuan, HU Guo-rong, LIU Ye-xiang. Modification of layered LiNi1/3Co1/3Mn1/3O2 cathode materials with doping multiple components [J]. The Chinese Journal of Nonferrous Metals, 2010, 20(6): 1170-1176. (in Chinese)
[15] SU Zhi, WENG Zhi-wang, SHEN Zhong. Surface modification and electrochemical properties of LiMnO2 as cathode materials for lithium-ion batteries [J]. The Chinese Journal of Nonferrous Metals, 2010, 20(6): 1183-1188. (in Chinese)
杨顺毅,王先友,刘子玲,陈权启,杨秀康,魏启亮
湘潭大学 化学学院,环境友好化学与应用教育部重点实验室,湘潭 411105
摘 要:以[Ni1/3Co1/3Mn1/3]3O4和氢氧化锂为原料,分别采用球磨法和液相法前处理工艺制备层状正极材料Li[Ni1/3Mn1/3Co1/3]O2。采用X-射线衍射(XRD)、场发射扫描电镜(FESEM)、恒流充放电等手段对材料的物理和电化学性能进行表征。结果表明:采用不同前处理工艺制备出的Li[Ni1/3Mn1/3Co1/3]O2材料在结构、形貌和电化学性能上有较大差异;与球磨处理法制备的材料相比,采用液相法前处理工艺制备的Li[Ni1/3Mn1/3Co1/3]O2不但保持了前驱体较好的球形形貌,同时还具有较好的循环稳定性和倍率性能;该样品在20 mA/g电流密度下,首次放电容量为178 mA?h/g,50次循环后,容量保持率达98.7%;在1 000 mA/g电流密度下,样品容量为135 mA?h/g。
关键词:锂离子电池;Li[Ni1/3Co1/3Mn1/3]O2;碳酸盐共沉淀法;前处理工艺;电化学性能
(Edited by LI Xiang-qun)
Foundation item: Project (20871101) supported by the National Natural Science Foundation of China; Project (2009WK2007) supported by Key Project of Science and Technology Department of Hunan Province, China; Project (CX2009B133) supported by Colleges and Universities in Hunan Province Plans to Graduate Research and Innovation, China
Corresponding author: WANG Xian-you Tel: +86-731-58292060; Fax: +86-731-58292061; E-mail: wxianyou@yahoo.com
DOI: 10.1016/S1003-6326(11)60962-X

