����ʽ���ʯ�ᾧ������ͭ����Ϸ�ˮ
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2017���2��
�������ߣ�����Ԫ ���� ��С�� �Ƴ�� ������ ���� �ž� Mohammad ALI
����ҳ�룺457 - 466
�ؼ��ʣ����ʯ��ͭ�����������ѳ����ؽ�����ˮ������
Key words��struvite; copper-ammonia complex; ammonia removal; heavy metal wastewater; precipitation
ժ Ҫ��ͨ����ϳ�ͭ����Ϸ�Һ������þ����Mg2+����������� ������ʵ��������ӵĽ��룬�Ӷ�ʵ��ͭ�백����ͬ���ѳ����ݴ˽���������ʽ���ʯ�ᾧ��(TSSF)������ȥ��ͭ�����������У���һ������90%ҩ��(MgCl2 �� Na2HPO4)���ڶ�������ʣ��10%ҩ����������Դ��þԴ��������pH����Ӧʱ���ͭ������ȥ���ʵ�Ӱ�졣���������TSSF����ͭ������ȥ����������һ��ʽ����������n(NH3-N):n(Mg):n(P)=1:1.2:1.5 (Ħ����)��pH=9����Ӧʱ��30 min����£�ͭ��������ȥ���ʷֱ�Ϊ98.9%��99.96%������ͭ������������ʯ�����ľ������ƽ�����TSSF����ͭ�����ĸ�Чȥ���������û��ڼ��㼰������ͼ������ѧ�����ֶν�һ��������ͭ��������ӵĽ�����ơ����⣬X�����������(XRD)��������(EDS)���Ҳ֤ʵ�����ʯ���γɼ�ͭ�ij�������ʾ��ͭ��������Һ�ൽ�����ת����̡�
Abstract: Mg2+ and PO43+ were added into the synthetic wastewater, leading to the dissociation of the complex ions in the wastewater, and resulting in removal of copper and ammonia therein. The effects of agents addition amount, pH, and reaction time on the removal efficiency of copper and ammonia were investigated. In particular, two-sectional struvite formation (TSSF) process was established for copper and ammonia removal. MgCl2 and Na2HPO4 were added by following 90% addition in the first section and remained 10% in the second during the TSSF process. Compared with one sectional struvite formation, TSSF possessed much better performance. Under condition of n(NH3-N):n(Mg):n(P)=1:1.2:1.5 (molar ratio), pH=9, and reaction time of 30 min, the removal efficiencies of copper and ammonia were 98.9% and 99.96%, respectively. The enhanced performance of TSSF is explained by the competition of ammonia by copper-ammonia complexes and struvite. The dissociation of copper-ammonia complexes is further demonstrated by thermodynamic equilibrium analysis, on the basis of calculations and establishment of predominance phases diagram. Moreover, XRD and EDS analyses further confirmed the formation of struvite and precipitation of copper, which prove the transmission of copper and ammonia from liquid phase into solid phase.
Trans. Nonferrous Met. Soc. China 27(2017) 457-466
Li-yuan CHAI1,2, Cong PENG1,2, Xiao-bo MIN1,2, Chong-jian TANG1,2, Yu-xia SONG1,2, Yang ZHANG1,2, Jing ZHANG1,2, Mohammad ALI1,2
1. School of Metallurgy and Environment, Central South University, Changsha 410083, China;
2. Chinese National Engineering Research Centre for Control and Treatment of Heavy Metal Pollution, Central South University, Changsha 410083, China
Received 30 May 2016; accepted 6 January 2017
Abstract: Mg2+ and PO43+ were added into the synthetic wastewater, leading to the dissociation of the complex ions in the wastewater, and resulting in removal of copper and ammonia therein. The effects of agents addition amount, pH, and reaction time on the removal efficiency of copper and ammonia were investigated. In particular, two-sectional struvite formation (TSSF) process was established for copper and ammonia removal. MgCl2 and Na2HPO4 were added by following 90% addition in the first section and remained 10% in the second during the TSSF process. Compared with one sectional struvite formation, TSSF possessed much better performance. Under condition of n(NH3-N):n(Mg):n(P)=1:1.2:1.5 (molar ratio), pH=9, and reaction time of 30 min, the removal efficiencies of copper and ammonia were 98.9% and 99.96%, respectively. The enhanced performance of TSSF is explained by the competition of ammonia by copper-ammonia complexes and struvite. The dissociation of copper-ammonia complexes is further demonstrated by thermodynamic equilibrium analysis, on the basis of calculations and establishment of predominance phases diagram. Moreover, XRD and EDS analyses further confirmed the formation of struvite and precipitation of copper, which prove the transmission of copper and ammonia from liquid phase into solid phase.
Key words: struvite; copper-ammonia complex; ammonia removal; heavy metal wastewater; precipitation
1 Introduction
Ammonia is one essential nutrient that can lead to water eutrophication when it is present in excess [1-4]. Meanwhile, ammonia is a versatile ligand capable of forming complex compounds with many metal ions such as 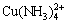 ,
, 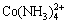 and
and 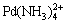 [5,6]. For example, the wastewater from mineral extraction industries contains heavy metals and ammonia [7-9]. Copper and ammonia are frequently found in the effluents of tannery, explosives, and timber industries [10,11], with ammonia present at concentration ranging from 0.4 to 380 mg/L [12]. Spent etching solution with a high concentration of both ammonia (500-2000 mg/L) and copper (100-3000 mg/L) is formed after printed circuit board (PCB) etching [13,14]. The chelated metal-ammonia ions are quite stable in solution and thus are difficult to decompose, which imposes a great challenge for efficient as well as simultaneous removal of metal and ammonia from the complex wastewater. Previous studies mainly focused on removal of either heavy metals [15-17] or ammonia [18]. Only a few researches investigated simultaneous removal of heavy metal and ammonia from their chelated solution. For example, SUN et al [19] studied the simultaneous removal of beryllium and ammonia nitrogen by biological aerated filter. PRESSMAN et al [20] used nitrifying biofilm to remove ammonia and copper simultaneously. ZHANG et al [21] used a dual chamber microbial fuel cell to recover ammonia-copper complex from wastewater. However, these biological methods either had no satisfactory efficiency, or had long treatment period arisen from the microbial activity inhibition caused by heavy metals.
[5,6]. For example, the wastewater from mineral extraction industries contains heavy metals and ammonia [7-9]. Copper and ammonia are frequently found in the effluents of tannery, explosives, and timber industries [10,11], with ammonia present at concentration ranging from 0.4 to 380 mg/L [12]. Spent etching solution with a high concentration of both ammonia (500-2000 mg/L) and copper (100-3000 mg/L) is formed after printed circuit board (PCB) etching [13,14]. The chelated metal-ammonia ions are quite stable in solution and thus are difficult to decompose, which imposes a great challenge for efficient as well as simultaneous removal of metal and ammonia from the complex wastewater. Previous studies mainly focused on removal of either heavy metals [15-17] or ammonia [18]. Only a few researches investigated simultaneous removal of heavy metal and ammonia from their chelated solution. For example, SUN et al [19] studied the simultaneous removal of beryllium and ammonia nitrogen by biological aerated filter. PRESSMAN et al [20] used nitrifying biofilm to remove ammonia and copper simultaneously. ZHANG et al [21] used a dual chamber microbial fuel cell to recover ammonia-copper complex from wastewater. However, these biological methods either had no satisfactory efficiency, or had long treatment period arisen from the microbial activity inhibition caused by heavy metals.
Struvite formation method is a kind of chemical precipitation method to remove ammonia from wastewater, which is typically realized by precipitation of magnesium ammonia phosphate hexahydrate (MgNH4PO4��6H2O, also denoted as MAP) by adding magnesium salts and phosphates to wastewater [22-24]. Generally, the chemical struvite formation method could efficiently avoid the inhibition caused by heavy metals. However, there is seldom report about the simultaneous removal of ammonia and heavy metals by struvite formation method, which might be due to the cost of extra addition of magnesium and phosphate. Nonetheless, the struvite is not so thermostable thus it could be thermalized, so the magnesium and phosphate could be recycled for further precipitation of ammonia, which was confirmed by many researchers [25,26]. From this perspective, struvite formation method can actually be an economical method for heavy metal and ammonia removal from wastewater. So, it is quite meaningful to study the struvite formation method as a promising way to remove heavy metal and ammonia from wastewater.
The objectives of this study are to establish the struvite formation method for treatment of copper- ammonia complex wastewater and to dissolve the tricky problem in dissociating copper-ammonia complex. Firstly, the effects of the influential factors including pH, agent dosage, and reaction time on removal efficiency were investigated. Secondly, the thermodynamic analysis toward the struvite formation process was conducted based on the detection of the precipitates by SEM, XRD and EDS analyses in order to elucidate the transmission of copper and ammonia from solution to solids. Especially, the two-sectional struvite formation (TSSF) process was further established and investigated with much better performance.
2 Experimental
2.1 Materials
All the reagents were of analytical grade and were used without further purification. Ultrapure water (18 M����cm) was used in all experiments. Concentrated sulfuric acid (H2SO4), ammonium chloride (NH4Cl), magnesium chloride hexahydrate (MgCl2��6H2O), disodium hydrogen phosphate (Na2HPO4), copper sulfate (CuSO4) and sodium hydroxide (NaOH) were purchased from the Sinopharm Group Chemical Reagent Co., Ltd.
2.2 Synthetic wastewater
The copper-ammonia complex wastewater in the present study was in-situ prepared with the total copper of 200 mg/L and ammonia of 500 mg/L, namely by adding CuSO4 (containing 0.08 g Cu) reagent into NH4Cl solution (containing 0.2 g N), and then diluting it with ultrapure water to 400 mL, and the pH of the wastewater was adjusted to 9.0 by dosing 0.2 mol/L NaOH solution. Results showed that the dark blue solution was formed under alkaline condition (pH 9.0).
2.3 Procedures
The 400 mL synthetic wastewater was measured and introduced into 500 mL-scale beaker. The MgCl2 and Na2HPO4 reagents were separately dissolved to volume of 30 mL. The reagent solutions were added by peristaltic pump at a velocity of 50 mL/min, and the addition volume was precisely controlled by the peristaltic pump. The addition for the first section was 90%, while the remaining 10% for the subsequent second section. pH adjustment was conducted when addition of MgCl2 and Na2HPO4 reagents began. After addition of reagent and pH adjustment, the time for precipitation process was precisely controlled. The second section was continued after accomplishment of the first section, and the remaining 10% reagent solutions were added into the beaker. Other conditions were the same with that in the first section. Effects of pH, addition amount of MgCl2 and Na2HPO4, and reaction time were investigated. The pH values from 3-14 were selected, and addition amount of MgCl2 was chosen in accordance with molar ratio n(MgCl2):n(N) from 0.8:1 to 1.5:1, based on n(Na2HPO4):n(N) at 1.1:1, while the addition amount of Na2HPO4 for investigation was selected according to molar ratio n(MgCl2):n(N) from 0.8:1 to 1.5:1, based on n(MgCl2):n(N) at 1.2:1. Moreover, the time for TSSF method was investigated from 5 to 40 min.
2.4 Analytical methods
The determination of ammonium concentration was carried out following the standard methods (APHA, 1998) [27]. Copper concentration was tested by inductively coupled plasma atomic emission spectrometry (ICP-AES, PerkinElmer Optima 5300 DV).
The precipitates obtained after filtration were dried for further experiments. The surface characteristics of the precipitate were examined by a JSM-6360LV scanning electron microscope (SEM, JEOL Ltd., Japan). The elements in the precipitate and their distribution condition in the precipitate were evaluated by EDS spectra on the JSM-6360 scanning electron microscope equipped with an energy-dispersive X-ray spectrometer. The X-ray diffraction (XRD) patterns were obtained using Rigaku D/Max-RB diffractometer with Cu K�� radiation (��=0.15406 nm, 35 kV, 40 mA).
3 Results and discussion
3.1 Performance of TSSF process
3.1.1 Effect of MgCl2 addition amount on ammonia and copper removal
MgCl2 and NaHPO4 were added into the prepared wastewater followed the molar ratio of n(NH3-N): n(Mg):n(P)=1:x:1.1. The x therein was set at 0.8, 0.9, 1.0, 1.1, 1.2, 1.3, 1.4 and 1.5, respectively. pH 9.0 was chosen preferentially, as would be further discussed in the following thermodynamic equilibrium analysis. The reaction time for both the first and second sections was controlled at 20 min.
The residual concentrations of copper and ammonia nitrogen are shown in Fig. 1(a). With increasing addition amount of MgCl2, the residual concentration of ammonia nitrogen and copper decreased firstly, and increased afterwards. For n(NH3-N):n(Mg)=1:1.2, namely ��(MgCl2��6H2O)=8.70 g/L (mass concentration), it was the minimum point of the residual concentration. At this point, the residual ammonia was 60.8 mg/L, with removal efficiency of 87.84%, and the residual copper was 16.40 mg/L, with removal efficiency of 91.80%. The trends of copper and ammonia nitrogen removal were basically similar. The slight decrease in removal when further increasing addition amount of MgCl2 was probably caused by acceleration of magnesium ions combination with phosphate in the solution to form newberyite (MgHPO4��3H2O), bobierrite (Mg3(PO4)2��8H2O), cattiite (Mg3(PO4)2��22H2O), and so forth [28,29].
3.1.2 Effect of Na2HPO4 addition amount on ammonia and copper removal
Based on the optimal point of MgCl2 addition amount mentioned above, ��(Mg2+��of 1.03 g/L with n(NH3-N):n(Mg)=1:1.2 was selected. The pH and reaction time were maintained at 9.0 and 20 min, respectively, which were the same with the former condition. The addition amount of Na2HPO4 was a variable, with molar ratio of n(NH3-N):n(Mg):n(P)= 1:1.2:x, and x therein was maintained at 0.8, 0.9, 1.0, 1.1, 1.2, 1.3, 1.4 and 1.5, respectively. The results are shown in Fig. 1(b). The residual concentrations of ammonia and copper continuously decreased as the addition amount of Na2HPO4 increased. When n(NH3-N):n(P)=1:1.5, namely 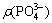 =5.14 g/L, the residual ammonia and copper concentrations were 4.6 mg/L and 0.02 mg/L respectively at this point, which was far lower than the limit value in Chinese discharge standard of pollutant for municipal wastewater treatment plant (GB18918-2002). With continuously adding Na2HPO4, the residual concentration of ammonia decreased quite slowly.
=5.14 g/L, the residual ammonia and copper concentrations were 4.6 mg/L and 0.02 mg/L respectively at this point, which was far lower than the limit value in Chinese discharge standard of pollutant for municipal wastewater treatment plant (GB18918-2002). With continuously adding Na2HPO4, the residual concentration of ammonia decreased quite slowly.
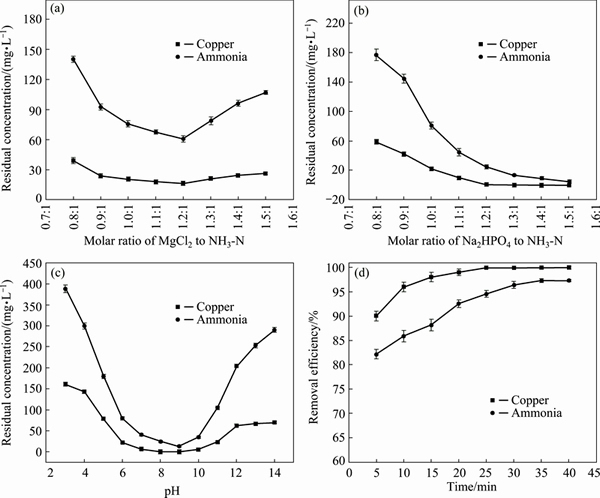
Fig. 1 Effect of addition of Mg2+ (a), 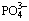 (b), pH value (c), and reaction time (d) on copper and ammonia removal performance of TSSF method
(b), pH value (c), and reaction time (d) on copper and ammonia removal performance of TSSF method
3.1.3 Effect of pH on ammonia and copper removal
The addition amounts of 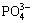 and Mg2+ were set at 4.46 g/L and 1.03 g/L, respectively, and reaction time was maintained at 20 min. pH value was functioned as variable, to investigate the removal efficiency of copper and ammonia under different pH values. The results are shown in Fig. 1(c). Under pH value of 8-10, favorable removal efficiency of copper and ammonia can be achieved. It had the best removal at pH 9.0, with the residual ammonia and copper concentration of 13.6 mg/L and 0.21 mg/L and corresponding removal efficiency of 97.28% and 99.90%, respectively. However, when pH>10.0 or pH<8.0, the removal of copper and ammonia decreased. This could be arisen from the decrease of MgNH4PO4��6H2O formed in the solution, under these two kinds of pH conditions. This demonstrated that the optimal pH value was around 9.0.
and Mg2+ were set at 4.46 g/L and 1.03 g/L, respectively, and reaction time was maintained at 20 min. pH value was functioned as variable, to investigate the removal efficiency of copper and ammonia under different pH values. The results are shown in Fig. 1(c). Under pH value of 8-10, favorable removal efficiency of copper and ammonia can be achieved. It had the best removal at pH 9.0, with the residual ammonia and copper concentration of 13.6 mg/L and 0.21 mg/L and corresponding removal efficiency of 97.28% and 99.90%, respectively. However, when pH>10.0 or pH<8.0, the removal of copper and ammonia decreased. This could be arisen from the decrease of MgNH4PO4��6H2O formed in the solution, under these two kinds of pH conditions. This demonstrated that the optimal pH value was around 9.0.
3.1.4 Effect of reaction time on ammonia and copper removal
The removal performance of copper and ammonia under different reaction time was measured by maintaining 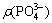 =4.46 g/L and ��(Mg2+)=1.03 g/L.
=4.46 g/L and ��(Mg2+)=1.03 g/L.
The results are shown in Fig. 1(d). The removal of copper and ammonia nitrogen increased as time passed. The ammonia nitrogen removal efficiency reached the peak value after 35 min, while copper removal reached earlier: it came to peak value at 25 min. These results showed that the removal efficiency of copper was greater than that of ammonia. This indicated that the low concentration of ammonia in the solution hardly had any chelation with copper under TSSF process. So, from this perspective it suggested that TSSF method was quite effective in the dissociation of copper-ammonia complexes.
3.2 Optimized performance of TSSF
On the basis of what mentioned above, it was concluded that the optimal conditions of the facile TSSF process were as follows: pH 9.0, the molar ratio n(Mg):n(P):n(NH3) of 1.2:1.5:1, and reaction time of 30 min. Under these conditions, experiments upon the treatment of wastewater samples containing different initial copper and ammonia concentrations were carried out (Table 1).
As seen in Table 1, the TSSF method had great performance in simultaneously removing copper and ammonia from wastewater, under optimal conditions. Although the residual concentrations of copper and ammonia slightly increased under relatively high initial concentrations, the results showed that they were still less than the prescribed limits in the standards. For example, for the water sample with initial copper and ammonia concentration of 500 mg/L and 1500 mg/L, the results after treatment of TSSF method showed their residual concentration of 0.18 mg/L and 7.7 mg/L respectively, which were satisfactory for wastewater treatment.
Table 1 Performance of TSSF process upon different initial copper and ammonia concentrations under optimal condition
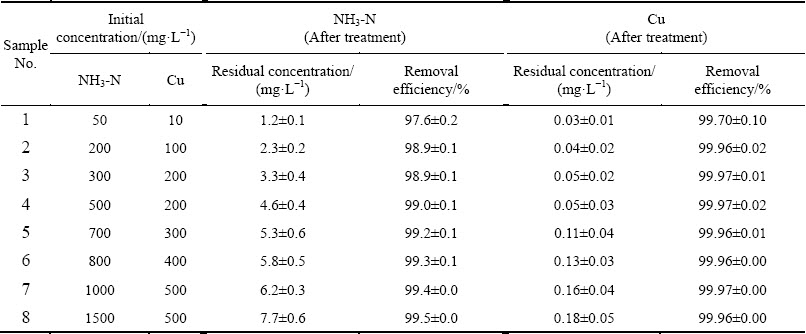
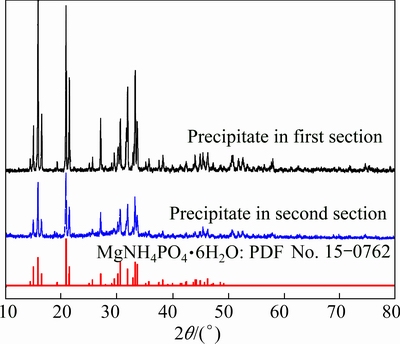
Fig. 2 XRD pattern of precipitate from first section and second section in TSSF
3.3 XRD, SEM and EDS characterization of precipitates
Figure 2 shows the comparison of XRD patterns (including first section and second section) of the precipitate from TSSF method and MgNH4PO4��6H2O powder from the Joint Committee on Powder Diffraction Standards (JCPDS) card (No. 15-0762). By comparison, all the relative diffraction peaks can be indexed to MgNH4PO4��6H2O (struvite). It can be seen from the XRD pattern that precipitate MgNH4PO4��6H2O had several intensive diffraction peaks that were sharp and symmetrical, which indicated that the precipitate possessed high degree of crystallinity and relatively great crystallite size [30,31]. In SEM and EDS observations, two types of precipitates were identified, namely the big prism-shaped materials and the fine particles attached on them (Figs. 3(a) and (b)). Mg and P dominating in the prism-shaped material with low copper content (Fig. 3(c)) confirmed the struvite crystal, which was also in accordance with the hypothesis based on the XRD analysis that the struvite possessed high degree of crystallinity and relatively great crystallite size, as mentioned above. Whereas, the fine particles attached on the struvite crystals contained a strong peak of copper (Fig. 3(d)). Combined with the thermodynamic equilibrium analysis in the following text, it could be deduced that the fine particles mainly consisted of copper hydroxide. Besides, the comparison of XRD pattern with the first section precipitate showed that the second section precipitate had lower peak intensity (Fig. 2), indicating the probably lower degree of crystallization, which can be further illustrated by SEM (Fig. 3(e)) that there were more relatively small and irregular crystals. It could be arisen from the relatively much low ammonia concentration in the second section of TSSF, which might influence the crystallization property of struvite. It can still be clearly seen in Figs. 3(f) and (g) that there was no significant copper detected by EDS, and no small copper hydroxide particles attached on the struvite (Fig. 3(e)). This suggested that the copper was almost removed in the first section. For example, for the optimum condition of n(Mg):n(P):n(NH3-N) of 1.2:1.5:1, and original ammonia and copper concentrations of 500 mg/L and 200 mg/L, the residual ammonia concentration in the first section was 32.6 mg/L, while the copper concentration was only 0.4 mg/L, indicating that the struvite formation has great potential in decomposition of [Cu(NH3)4]2+, which leads to the further precipitation of copper.
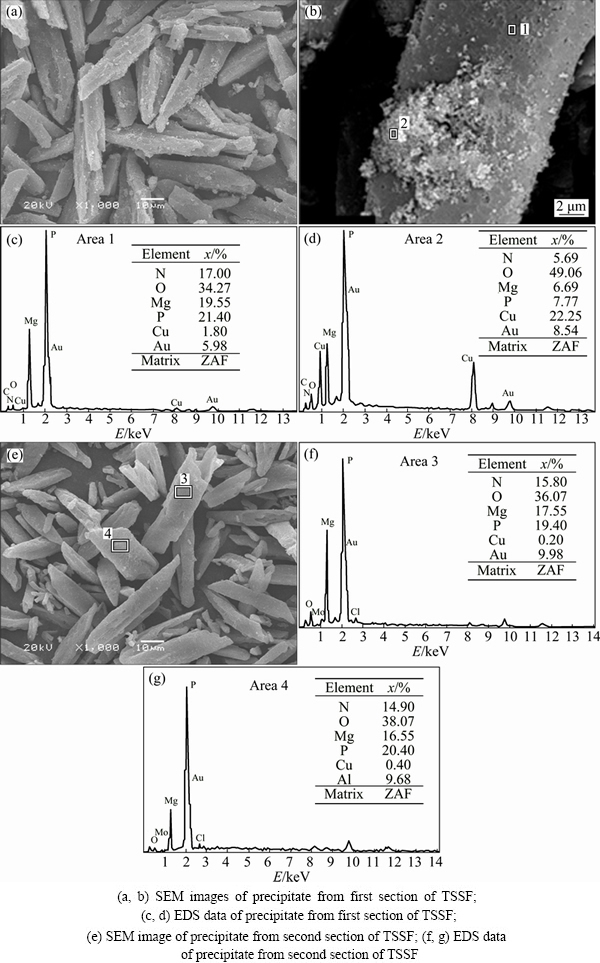
Fig. 3 SEM images and EDS results of precipitate from TSSF
The above observations elucidated the transformation of copper and ammonia from solution phase to solid phase after adding Mg2+ and 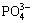 , and it further confirmed the dissociation of copper ammonia complex under pH 9.0. The addition of Mg2+ and
, and it further confirmed the dissociation of copper ammonia complex under pH 9.0. The addition of Mg2+ and 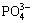 enhanced the dissociation of ammonia ion from the complex in the solution, and it was captured to form precipitate MgNH4PO4��6H2O (Eq. (1)). Thus, the released Cu2+ was hydrolyzed to form copper hydroxide under the alkaline atmosphere of the solution (Eq. (2)). Finally, the copper hydroxide co-precipitated with struvite under certain interactions between them, resulting in the high removal of both copper and ammonia. Further investigations on the co-precipitation interactions and mechanism of the simultaneously high copper and ammonia removal performance are still being conducted.
enhanced the dissociation of ammonia ion from the complex in the solution, and it was captured to form precipitate MgNH4PO4��6H2O (Eq. (1)). Thus, the released Cu2+ was hydrolyzed to form copper hydroxide under the alkaline atmosphere of the solution (Eq. (2)). Finally, the copper hydroxide co-precipitated with struvite under certain interactions between them, resulting in the high removal of both copper and ammonia. Further investigations on the co-precipitation interactions and mechanism of the simultaneously high copper and ammonia removal performance are still being conducted.
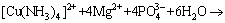
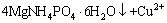 (1)
(1)
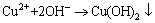 (2)
(2)
3.4 Removal performance without struvite formation
From the aforementioned performance results, it was confirmed that the ammonia and copper could be efficiently removed from their chelated wastewater by adding Mg2+ and 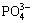 into the system. The copper and ammonia removal seemed to be quite relevant to the decomposition of copper ammonia complexes in the solution. In order to elucidate this point, some investigations have been conducted, including the studies on the removal efficiency without addition of Mg2+ and
into the system. The copper and ammonia removal seemed to be quite relevant to the decomposition of copper ammonia complexes in the solution. In order to elucidate this point, some investigations have been conducted, including the studies on the removal efficiency without addition of Mg2+ and 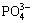 , and the thermodynamic equilibrium analysis upon the system after addition.
, and the thermodynamic equilibrium analysis upon the system after addition.
The residual copper content was tested under different pH values, based on no addition of MgCl2 and NaHPO4. CuSO4 solution was selected as the control group, and the test results are shown in Fig. 4.
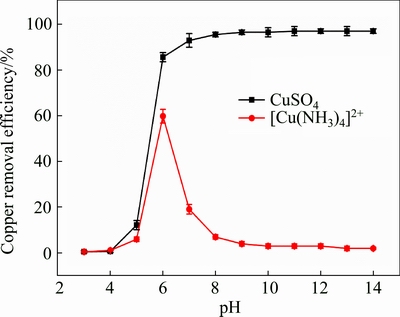
Fig. 4 Effect of pH on copper removal in CuSO4 solution and in copper-ammonia complex wastewater in complex solution at pH of 6
As shown in Fig. 4, the copper removal efficiency in copper ammonia complex wastewater was quite low, only except for the condition at pH=6.0 (with efficiency of 58.1%). As pH increased, the copper removal slightly decreased when pH was lower than 6. There was no obvious copper removal when pH was higher than 9.0, suggesting that the copper still existed in the form of complex in the solution. As to CuSO4, the copper removal efficiency reached 83.4% under pH=6, and it slightly rose up as pH increased. The removal efficiency reached 96.1% when pH was 9.0. As to the complex wastewater, the performance in pH 6.0 seemed to be abnormal, but it can be explained by stability decreasing of copper ammonia complex in relatively low pH values. The stability of copper-ammonia complex declines as pH decreases, so it is less stable in the pH range of 5-6 than under pH higher than 7.0 [32]. As a result, the copper hydroxide would be formed to precipitate the re-ionized copper. Thus, 58.1% of copper removal in CuSO4 solution and in the copper-ammonia complex wastewater was realized in the complex solution under pH of 6.0. It can be concluded from the results that a large content of copper existed in the form of complex, which was enhanced as pH increased. pH adjustment could not enhance the removal of copper-ammonia complex ions, but even enhance the stability of complex under higher pH values. After comparison, it is clear that the addition of Mg2+ and 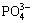 is the key point for the treatment efficiency. For further confirmation, thermodynamic equilibrium calculations were conducted subsequently.
is the key point for the treatment efficiency. For further confirmation, thermodynamic equilibrium calculations were conducted subsequently.
3.5 Thermodynamic equilibrium analysis
The species possibly existing in the system mainly included Cu2+, NH4+, [Cu(NH3)4]2+, Mg2+, 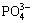 , H+, OH-,
, H+, OH-, 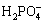 ,
, 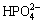 ,
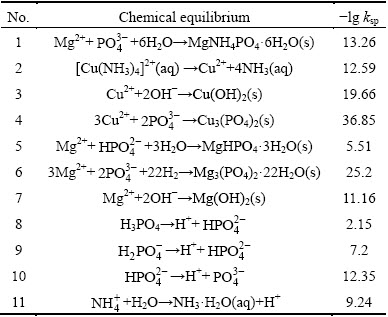
, 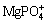 , NH3��H2O, MgHPO4, Mg3(PO4)2, MgNH4PO4, Cu(OH)2, Cu3(PO4)2, and Mg(OH)2. The equilibrium relations between the ions and the solids are listed in Table 2 [33,34].
, NH3��H2O, MgHPO4, Mg3(PO4)2, MgNH4PO4, Cu(OH)2, Cu3(PO4)2, and Mg(OH)2. The equilibrium relations between the ions and the solids are listed in Table 2 [33,34].
Table 2 Thermodynamic equilibrium data for struvite formation system
In order to simplify the thermodynamic calculations, ksp1, ksp2, ksp3, ksp4, kN are used as follows:
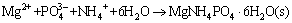
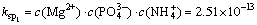 (3)
(3)
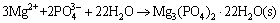
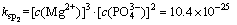 (4)
(4)
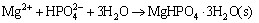
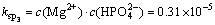 (5)
(5)
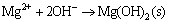
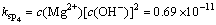 (6)
(6)
Ammonia in the water has the following chemical equilibrium (kN) [35]:
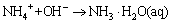
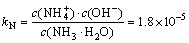 (7)
(7)
It can be deduced from Table 2 that
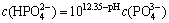 (8)
(8)
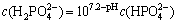 (9)
(9)
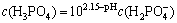 (10)
(10)
It can be drawn from the formulas listed above that 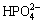 is the main form existing in the solution within pH of 8-11.
is the main form existing in the solution within pH of 8-11.
3.5.1 Analysis of existence of MgHPO4
Assuming that the precipitates contained MgHPO4, the solubility equilibrium can be expressed as follows:
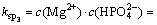
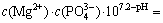
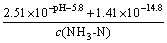 (11)
(11)
Then, 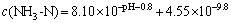 .
.
Thus, the following conclusion can be drawn from the above equation: when pH ��5, c(NH3-N)��1.28��10-5 mol/L, and when pH=9.0, c(NH3-N)=2.0��10-9 mol/L. Then, this conclusion absolutely failed to conform with the fact that the c(NH3-N) adopted in our study was 3.57��10-2 mol/L. As a consequence, there will be no MgHPO4 in the precipitates, when pH��5.0.
3.5.2 Analysis of existence of Mg(OH)2
Assuming that there existed precipitate of Mg(OH)2, according to the aforementioned equations, it is easy to draw the following formula in assuming condition pH<9.5:
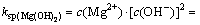
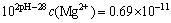 (12)
(12)
Then, 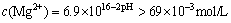 .
.
But this value is far larger than concentration of Mg2+ in the solution of our study, so there will have no precipitation of Mg(OH)2 when pH<9.5.
3.5.3 Analysis of existence of Mg3(PO4)2
Assuming that there was only MgNH4PO4��6H2O in the precipitation (namely precipitation of Mg3(PO4)2 is excluded), and supposing that x mol/L ammonia entering into precipitation MgNH4PO4��6H2O, we can deduce the following formulas:
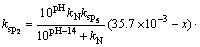
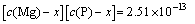 (13)
(13)
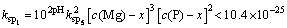 (14)
(14)
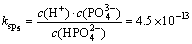 (15)
(15)
where 35.7��10-3 mol/L indicated the initial ammonia concentration.
It can be inferred from Eqs. (13), (14) and (15) that c(Mg)< 35.7��10-3 mol/L.
The calculation results indicated that the molar ratio of Mg2+ to ammonia should be lower than 1. But for the actual situation, the molar ratio of n(Mg):n(NH3-N) was always kept higher than 1, demonstrating that the above- mentioned conclusion should be false. In other words, MgNH4PO4��6H2O and Mg3(PO4)2 coexisted in the precipitates under the tested conditions. This indicates that when Mg2+ and 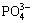 were simultaneously added into the solution, formation of Mg3(PO4)2 would inevitably occur. This phenomenon was considered to be not favorable for struvite precipitation since the effective source of Mg and P for struvite precipitation could be decreased. Thus, the performance of struvite formation could be negatively influenced when the co-reaction and precipitation of Mg3(PO4)2 occurred.
were simultaneously added into the solution, formation of Mg3(PO4)2 would inevitably occur. This phenomenon was considered to be not favorable for struvite precipitation since the effective source of Mg and P for struvite precipitation could be decreased. Thus, the performance of struvite formation could be negatively influenced when the co-reaction and precipitation of Mg3(PO4)2 occurred.
3.5.4 Predominance diagrams at optimal pH value of 9.0
Based on the analysis above, existence possibility of some species can be excluded from the system, and MgNH4PO4��6H2O, Mg3(PO4)2, [Cu(NH3)4]2+, Cu(OH)2(s), Cu3PO4(s) were taken for further predominance species analysis. The predominance diagram of copper compounds has been drawn at different Mg and P concentrations. The pH value was 9.0, which was optimal for struvite formation method as mentioned above. The results are shown in Fig. 5.
As shown in Fig. 5, predominance diagram had shown three predominance areas, including Cu(OH)2, [Cu(NH3)4]2+, Cu(OH)2 and [Cu(NH3)4]2+. The specific predominance area of Cu(OH)2 and [Cu(NH3)4]2+ means the pure existence. For circumstance without addition of Mg and P (Fig. 5(a)), there was a wide predominance area for [Cu(NH3)4]2+, and the single Cu(OH)2 can exist only under very low ammonia content (with high -lgcT,N as shown in Fig, 5(a)). The predominance diagrams varied significantly when Mg and P were added into the system. The molar concentration from 1.0��10-3 mol/L to 1.0��10-1 mol/L would create regular changes to the diagrams. With increasing molar concentrations of Mg and P (-lg cMg+P decreased as shown in Fig. 5), the predominance area for single Cu(OH)2 remarkably increased, and the area of single [Cu(NH3)4]2+ was significantly reduced. This phase diagram analysis suggested that the addition of Mg and P would probably decompose copper ammonia complex, and transform the copper therein into Cu(OH)2. In particular, the predominance area for [Cu(NH3)4]2+ is quite small (Fig. 5(d)) under 1.0��10-1 mol/L of Mg and P, suggesting that the addition of Mg and P could possess favorable treatment ability of copper ammonia complex.
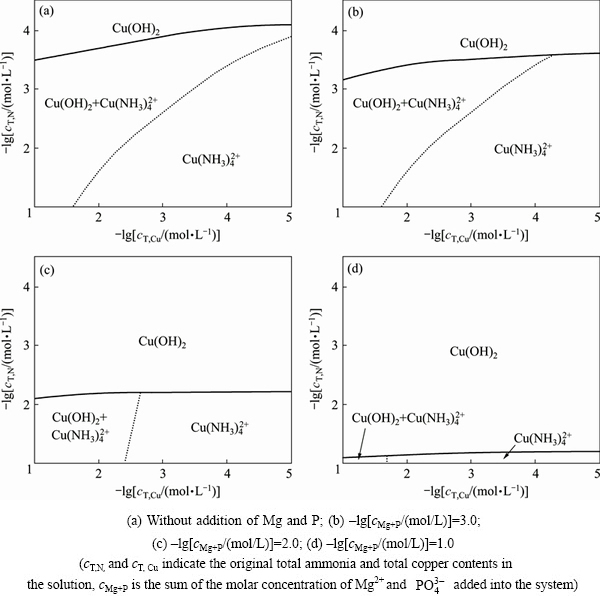
Fig. 5 Predominance diagram for addition of Mg and P at pH 9.0
4 Conclusions
1) TSSF method has great efficiency in removal of copper and ammonia from copper-ammonia complex wastewater.
2) Compared with single sectional struvite formation method, TSSF method can enhance the removal effect. Factor experiments were conducted and the experiment under optimal conditions was also conducted. It was found that the maximum removal efficiencies could reach 99.0% and 99.97% for ammonia and copper, respectively, which indicates that TSSF method is quite a promising way in the treatment of copper-ammonia wastewater.
3) The thermodynamic equilibrium calculations were done to further confirm the dissociation of copper ammonia ions in the solution. Precipitate characterization including SEM, XRD, and EDS further prove the transmission of copper and ammonia from solution into solids.
4) The superiority of TSSF method is explained by competition of Mg3(PO4)2 and MgNH4PO4.
References
[1] KASSOTAKI E, BUTTIGLIERI G, CLIMENT L F, RODA I R, PIJUAN M. Enhanced sulfamethoxazole degradation through ammonia oxidizing bacteria co-metabolism and fate of transformation products [J]. Water Research, 2016, 94: 111-119.
[2] MANSER N D, WANG M, ERGAS S J, MIHELCIC J R, MULDER A, VOSSENBERG J V D, LIER J B V, STEEN P V D. Biological nitrogen removal in a photosequencing batch reactor with an algal-nitrifying bacterial consortium and anammox granules [J]. Environmental Science & Technology, 2016, 3(4): 175-179.
[3] TANG C J, ZHENG P, CHAI L Y, MIN X B. Characterization and quantification of anammox start-up in UASB reactors seeded with conventional activated sludge [J]. International Biodeterioration & Biodegradation, 2013, 82: 141-148.
[4] MOHAMMAD A, CHAI L Y, MIN X B, TANG C J, AFRIN S, LIAO Q, WANG H Y, PENG C, SONG Y X, ZHENG P. Performance and characteristics of a nitritation air-lift reactor under long-term HRT shortening [J]. International Biodeterioration & Biodegradation, 2016, 111: 45-53.
[5] KRAVTSOV V I, ASTAKHOVA R K, TSVENTARNYI E G, KURTOVA O Y, PEGANOVA, N A. Electroreduction of ammonia and hydroxyammonia complexes of divalent metals: Effect of the ligand concentration and the EDL structure [J]. Russian Journal of Electrochemistry, 2002, 38(2): 157-164.
[6] SMITH S R, ZHOU C Q, BARON J Y, CHOI Y, LIPKOWSKI J. Elucidating the interfacial interactions of copper and ammonia with the sulfur passive layer during thiosulfate mediated gold leaching [J]. Electrochimica Acta, 2016, 210: 925-934.
[7] MIN X B, ZHOU M, CHAI L Y, WANG Y Y, SHU Y D. Treatment of nickel-ammonia complex ion-containing ammonia nitrogen wastewater [J]. Transactions of Nonferrous Metals Society of China, 2009, 19(5): 1360-1364.
[8] MIN X B, XE X D, CHAI L Y, LIANG Y J, LI M, KE Y. Environmental availability and ecological risk assessment of heavy metals in zinc leaching residue [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(1): 208-218.
[9] CHAI L Y, WU J X, WU Y J, TANG C B, YANG W C. Environmental risk assessment on slag and iron-rich matte produced from reducing-matting smelting of lead-bearing wastes and iron-rich wastes [J]. Transactions of Nonferrous Metals Society of China, 2015, 25(10): 3429-3435.
[10] LEE L Y, ONG S L, NG H Y, HU J Y, KOH Y N. Simultaneous ammonium-nitrogen and copper removal, and copper recovery using nitrifying biofilm from the ultra-compact biofilm reactor [J]. Bioresource Technology, 2008, 99(14): 6614-6620.
[11] LUO S L, YUAN L, CHAI L Y, MIN X B, WANG Y Y, FANG Y, WANG P. Biosorption behaviors of Cu2+, Zn2+, Cd2+ and mixture by waste activated sludge [J]. Transactions of Nonferrous Metals Society of China, 2006, 16(6): 1431-1435.
[12] TURAKHIA M H, COOKSEY K E, CHARACKLIS W G. Influence of a calcium-specific chelant on biofilm removal [J]. Applied and Environmental Microbiology, 1983, 46(5): 1236-1238.
[13] KOCHERGINSKY N M, YANG Q. Big Carrousel mechanism of copper removal from ammoniacal wastewater through supported liquid membrane [J]. Separation and Purification Technology, 2007, 54: 104-116.
[14] ZENG X X, CHAI L Y, TANG J X, LIU X D, YANG Z H. Taxonomy characterization and biosorption of fungus strain [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(9): 2759-2765.
[15] NUSSINOVITCH A, DAGAN O. Hydrocolloid liquid-core capsules for the removal of heavy-metal cations from water [J]. Journal of Hazardous Materials, 2015, 299: 122-131.
[16] YANG J, CHAI L Y, WANG Y Y, HE X W, WANG J L. Transportation and distribution of chromium in the anaerobic sludge treating the chromium-containing wastewater [J]. International Journal of Environment & Pollution, 2010, 38(3): 256-266.
[17] WANG T, ZHANG L Y, LI C F, YANG W C, SONG T T, TANG C J, MENG Y, DAI S, WANG H Y, CHAI L Y, LUO J. Synthesis of core-shell magnetic Fe3O4@poly(m-phenylenediamine) particles for chromium reduction and adsorption [J]. Environmental Science & Technology, 2015, 49(9): 5654-5662.
[18] ZHANG B, SUN B S, JI M, LIU H N, LIU X H. Quantification and comparison of ammonia-oxidizing bacterial communities in MBRs treating various types of wastewater [J]. Bioresource Technology, 2010, 101(9): 3054-3059.
[19] SUN F, SUN W L. A simultaneous removal of beryllium and ammonium�Cnitrogen from smelting wastewater in bench-and pilot-scale biological aerated filter [J]. Chemical Engineering Journal, 2012, 210: 263-270.
[20] PRESSMAN J G, LEE W H, BISHOP P L, WAHMAN D G. Effect of free ammonia concentration on monochloramine penetration within a nitrifying biofilm and its effect on activity, viability, and recovery [J]. Water Research, 2012, 46(3): 882-894.
[21] ZHANG L J, TAO H C, WEI X Y, LEI T, LI J B, WANG A J, WU W M. Bioelectrochemical recovery of ammonia-copper(II) complexes from wastewater using a dual chamber microbial fuel cell [J]. Chemosphere, 2012, 89: 1177-1182.
[22] CAPDEVIELLE A,  F, DAUMER M L. Optimization of struvite precipitation in synthetic biologically treated swine wastewater��Determination of the optimal process parameters [J]. Journal of Hazardous Materials, 2013, 244: 357-369.
F, DAUMER M L. Optimization of struvite precipitation in synthetic biologically treated swine wastewater��Determination of the optimal process parameters [J]. Journal of Hazardous Materials, 2013, 244: 357-369.
[23] BI W, LI Y Y, HU Y Y. Recovery of phosphorus and nitrogen from alkaline hydrolysis supernatant of excess sludge by magnesium ammonium phosphate [J]. Bioresource Technology, 2014, 166: 1-8.
[24] PENG C, CHAI L Y, TANG C J, MIN X B, SONG Y X, DUAN C S, YU C. Study on the mechanism of copper-ammonia complex decomposition in struvite formation process and enhanced ammonia and copper removal [J]. Journal of Environmental Sciences, 2017, 51: 222-233.
[25] YU R, GENG J, REN H, WANG Y, XU K. Struvite pyrolysate recycling combined with dry pyrolysis for ammonium removal from wastewater [J]. Bioresource Technology, 2013, 132(2): 154-159.
[26] HUANG H, HUANG L, ZHANG Q, JIANG Y, DING L. Chlorination decomposition of struvite and recycling of its product for the removal of ammonium-nitrogen from landfill leachate [J]. Chemosphere, 2015, 136(5): 995-1002.
[27] American Public Health Association. Standard methods for the examination of water and wastewater [S]. 17th edition, 1989.
[28] TAMIMI F, NIHOUANNEN D L, BASSETT D C, IBASCO S. Biocompatibility of magnesium phosphate minerals and their stability under physiological conditions [J]. Acta Biomaterialia, 2011, 7: 2678-2685.
[29] ZHANG C, MIN X B, ZHANG J Q, WANG M, ZHOU B S, SHEN C. Reductive acid leaching of cadmium from zinc neutral leaching residue using hydrazine sulfate [J]. Transactions of Nonferrous Metals Society of China, 2015, 25(12): 4175-4182.
[30] BUSHROA A R, RAHBARI R G, MASJUKI H H, MUHAMAD M R. Approximation of crystallite size and microstrain via XRD line broadening analysis in TiSiN thin films [J]. Vacuum, 2012, 86(8): 1107-1112.
[31] CHAI L Y, YAN X, LI Q Z, YANG B T, WANG Q W. A comparative study of abiological granular sludge (ABGS) formation in different processes for zinc removal from wastewater [J]. Environmental Science and Pollution Research, 2014, 21: 12436-12444.
[32] ZHANG L, TAO H, WEI X, LEI T, LI J, WANG A, WU W. Bioelectrochemical recovery of ammonia-copper(II) complexes from wastewater using a dual chamber microbial fuel cell [J]. Chemosphere, 2012, 89: 1177-1182.
[33] OHLINGER K N, YOUNG T M, SCHROEDER E D. Predicting struvite formation in digestion [J]. Water Research, 1998, 32(12): 3607-3614.
[34] MICHALOWSKI T, PIETRZYK A. A thermodynamic study of struvite water system [J]. Talanta, 2006, 68(3): 599-601.
[35] JIANG K, ZHOU K G, PENG J L. Thermodynamics of solid-liquid equilibrium in NH4+-Mg2+-PO43-H+-H2O system [J]. Journal of Central South University, 2009, 40(5):1178-1182.
����Ԫ1,2���� ��1,2����С��1,2���Ƴ��1,2��������1,2���� ��1,2���� ��1,2��Mohammad ALI1,2
1. ���ϴ�ѧ ұ���뻷��ѧԺ����ɳ 410083��
2. ���ϴ�ѧ �����ؽ�����Ⱦ���ι��̼����о����ģ���ɳ 410083
ժ Ҫ��ͨ����ϳ�ͭ����Ϸ�Һ������þ����Mg2+�����������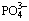 ������ʵ��������ӵĽ��룬�Ӷ�ʵ��ͭ�백����ͬ���ѳ����ݴ˽���������ʽ���ʯ�ᾧ��(TSSF)������ȥ��ͭ�����������У���һ������90%ҩ��(MgCl2 �� Na2HPO4)���ڶ�������ʣ��10%ҩ����������Դ��þԴ��������pH����Ӧʱ���ͭ������ȥ���ʵ�Ӱ�졣���������TSSF����ͭ������ȥ����������һ��ʽ����������n(NH3-N):n(Mg):n(P)=1:1.2:1.5 (Ħ����)��pH=9����Ӧʱ��30 min����£�ͭ��������ȥ���ʷֱ�Ϊ98.9%��99.96%������ͭ������������ʯ�����ľ������ƽ�����TSSF����ͭ�����ĸ�Чȥ���������û��ڼ��㼰������ͼ������ѧ�����ֶν�һ��������ͭ��������ӵĽ�����ơ����⣬X�����������(XRD)��������(EDS)���Ҳ֤ʵ�����ʯ���γɼ�ͭ�ij�������ʾ��ͭ��������Һ�ൽ�����ת����̡�
������ʵ��������ӵĽ��룬�Ӷ�ʵ��ͭ�백����ͬ���ѳ����ݴ˽���������ʽ���ʯ�ᾧ��(TSSF)������ȥ��ͭ�����������У���һ������90%ҩ��(MgCl2 �� Na2HPO4)���ڶ�������ʣ��10%ҩ����������Դ��þԴ��������pH����Ӧʱ���ͭ������ȥ���ʵ�Ӱ�졣���������TSSF����ͭ������ȥ����������һ��ʽ����������n(NH3-N):n(Mg):n(P)=1:1.2:1.5 (Ħ����)��pH=9����Ӧʱ��30 min����£�ͭ��������ȥ���ʷֱ�Ϊ98.9%��99.96%������ͭ������������ʯ�����ľ������ƽ�����TSSF����ͭ�����ĸ�Чȥ���������û��ڼ��㼰������ͼ������ѧ�����ֶν�һ��������ͭ��������ӵĽ�����ơ����⣬X�����������(XRD)��������(EDS)���Ҳ֤ʵ�����ʯ���γɼ�ͭ�ij�������ʾ��ͭ��������Һ�ൽ�����ת����̡�
�ؼ��ʣ����ʯ��ͭ�����������ѳ����ؽ�����ˮ������
(Edited by Xiang-qun LI)
Foundation item: Project (51674305) supported by the National Natural Science Foundation of China; Project (2013WK2007) supported by the Key Project of Science and Technology of Hunan Province, China; Project (2015CX001) supported by the Innovation Stimulating Program of Central South University, China; Key Project (1602FKDC007) supported by Science and Technology Program of Gansu Province, China
Corresponding author: Chong-jian TANG; Tel: +86-731-88830511; Fax: +86-731-88710171; E-mail: chjtang@csu.edu.cn
DOI: 10.1016/S1003-6326(17)60052-9

