
Adsorption behavior of gel-type weak acid resin (110-H) for Pb2+
XIONG Chun-hua(�ܴ���), YAO Cai-ping(Ҧ��Ƽ)
Department of Applied Chemistry, Zhejiang Gongshang University, Hangzhou 310035, China
Received 2 January 2008; accepted 2 June 2008
Abstract: The adsorption properties of a novel gel-type weak acid resin (110-H) for Pb2+ were investigated using chemical methods and IR spectrometry. The optimal adsorption condition of 110-H for Pb2+ is at pH=6.49 in HAc-NaAc medium and the statically saturated adsorption capacity is 485 mg/g at 298 K. Pb2+ adsorbed on 110-H resin can be eluted with 0.025 mol/L HCl quantificationally. The adsorption rate constants determined under the temperatures of 288, 298, 308 and 318 K are 2.46��10-5, 3.82��10-5, 4.46��10-5 and 5.71��10-5 s-1, respectively. The apparent activation energy, Ea, is 18.1 kJ/mol and the thermodynamic parameters of adsorption, ��H=20.9 kJ/mol, ��S=161 J/(mol?K) and ��G298 K =-48.0 kJ/mol, respectively. The adsorption behavior of 110-H resin for Pb2+ accords with the Langmuir isotherm. Infrared spectra show that the oxygen atoms of the functional group of resin coordinate with Pb2+ to form the coordination bands.
Key words: gel-type weak acid resin; adsorption; lead; mechanism
1 Introduction
Heavy metal contamination in various water resources is of great concern because of the toxic effect on the human beings, and other animals and plants. Lead is among the most toxic elements and it is of particular interest because of its toxicity and widespread presence in the environment[1]. Methods like precipitation, membrane separation, electrochemical reduction[2-4] are available for treating water contaminated with heavy metals. Most of these methods involve high capital cost with recurring expenses, which are not suitable for small-scale industries. Studies on treatment of effluents bearing heavy metals have revealed that adsorption is a highly effective, cheap and easy method among the physicochemical treatment processes. A variety of adsorbents have been used for removal of heavy metals from metal-contaminated effluents. However, adsorbents such as activated carbon and oxides are generally expensive in spite of their high metal adsorption capacities[5]. Efforts are being directed towards finding other alternative low-cost effective adsorbents. And polymeric materials have been demonstrated to be such kind of adsorbents[6-8].
Because nucleophilic atoms such as oxygen, nitrogen, sulphur and phosphorus atoms can coordinate directly to metal atoms, the functional polymer that combines those atoms has high affinity towards heavy metal ions and can be used as a sorbent for adsorbing metal ions. The synthesis and adsorption property of polymeric materials have been reported by the author and other researchers in recent years[9-17]. The adsorption ability of functional polymer used to enrich metal ion is strong and the operation of the functional polymer is convenient. The polymer adsorbed metal ion can be recovered by using acid or alkaline solution and the recovered polymeric material can be reused. The adsorption ability of the polymer with various functional groups is different for distinct metals.
In this work, the adsorption of lead ion on gel-type weak acid resin (110-H) that contains a functional group [��COOH] is investigated systematically. The adsorption mechanism of 110-H for Pb(��) is examined by infrared spectrometry and chemical analysis. Some of basic adsorption parameters are determined.
2 Experimental
2.1 Materials
The gel-type weak acid resin (110-H) was provided by Nankai University, China. Standard solutions of lead (��) was prepared from Pb(NO3)2, with a purity of AR grade. Buffer solution of pH 3.00-6.49 was prepared by mixing HAc-NaAc. Other reagents were of AR grade.
2.2 Instruments
Shimadzu UV-2550 UV-Visible spectrophotometer; DSHZ-300A temperature constant shaking machine; THZ-C-1 temperature constant shaking machine; Mettler Toledo Delta 320 pH meter; SK5200LH ultrasonic cleaning instrument; and IR Nicolet 380 were used.
2.3 Experimental method
2.3.1 Adsorption equilibrium experiment
A desired amount of treated resin was weighed and added into a conical flask, in which a desired volume of buffer solution with pH 5.4 was added. After 24 h, a required amount of standard solution of Pb(��) was added. The flask was shaken in a DSHZ-300A temperature constant shaking machine at the constant equilibrium. The upper layer of clear solution was taken for analysis until adsorption equilibium. The adsorption capacity (Q) and distribution coefficient (D) were calculated by the following equations:

where ��0 and ��e are the initial and equilibrium contents of Pb2+ in solution (mg/mL), V is the total volume of solution (mL), and m is the mass of the resin (g).
2.3.2 Analytical method
A solution containing less than 100 ��g of Pb2+ was accurately added into a 25 mL colorimetric tube, and then 2 mL of 0.1% xylenol orange solution and 10 mL pH 5.4 methenamine-HNO3 buffer solution were added. After the addition of redistilled water to 25 mL, the absorbency was determined in a 1 cm cell on a Shimadzu UV-2550 UV-VISIBLE spectrophotometer at the wavelength of 560 nm and compared with the blank sample[18].
2.3.3 Elution test
A desired amount of resin was added into a mixed solution composed of pH 6.49 buffer solution and desired amount of standard solution of Pb2+. After the equilibrium, the concentration of Pb2+ in the aqueous phase was determined, and then the adsorption capacity of the resin for Pb2+ was obtained. The resin separated from aqueous phase was washed three times with pH 6.49 buffer solution. The resin adsorbed Pb2+ was shaken with eluant. After reaching equilibrium, the concentration of Pb2+ in aqueous phase was determined and then the percentage of elution was obtained.
3 Results and discussion
3.1 Influence of pH on distribution coefficient (D)
The adsorption behavior of lead onto the 110-H resin in HAc-NaAc medium was studied in the pH range of 3.40-6.49 and the results are shown in Fig.1. The results indicate that the distribution coefficient D increases with the increase of the pH value. The maximum adsorption is attained at pH 6.49, and then the adsorption capacity almost keeps constant when pH is over 6.49. In order to prevent Pb2+ from hydrolyzing, all the following experiments were carried out at pH 6.49 ([Pb2+] [OH-]2��Ksp[Pb(OH)2]).

Fig.1 Influence of pH on adsorption capacity of Pb2+ (resin 20.0 mg, T=298 K, ��0=0.4 mg/mL, r=100 r/min)
3.2 Isotherm adsorption curve
Five samples of 20.0 mg resin were added into a mixed solution composed of pH 6.49 buffer solution individually. After 24 h, [Pb2+]0=0.33, 0.40, 0.47, 0.53 and 0.60 mg/mL of standard solutions were added. When the adsorption equilibrium arrived, the concentration of Pb2+ was determined.
The Langmuir isotherms are represented by the following equation:
��e/Q=��e/Q0+1/(Q0b) (3)
where ��e is equilibrium content of metal ion, Q is the adsorbing capacity in equilibrium state, Q0 is the saturated sorbing capacity and b is the Langmuir constant.
The plots of ��e/Q vs ��e give a straight line. The results shown in Fig.2 and Table 1 indicate that the Langmuir-type adsorption isotherm is suitable for equilibrium studies.
Table 1 Linearity relation of ��e/Q (y) and ��e (x)


Fig.2 Langmuir isotherm curve (resin 20.0 mg, r=100 r/min, pH=6.49)
3.3 Determination of adsorption rate constant at different temperatures and apparent activation energy
The tests were carried out with 40.0 mg resin under conditions of T=288, 298, 308, 318 K, [Pb2+]=0.4 mg/ mL by the method mentioned above. 0.2 mL of the upper-layer clear solution was taken out at intervals for the determination of remaining concentrations. The results are shown in Fig.3.

Fig.3 Adsorption rate curve of Pb2+ ([Pb2+]=0.4 mg/mL, resin 40.0 mg, pH=6.49)
According to Brykina method, the adsorption rate constant k can be calculated from -ln(1-F)=kt, where F=Qt/Q��, Qt and Q�� are the adsorption capacity at certain time and at equilibrium, respectively. The experimental results accord with the equation and a straight line is obtained by plotting -ln(1-F) vs t (shown in Fig.4), and the adsorption rate constant of 110-H resin for Pb2+ can be found from the slope of the straight line, which are 2.46��10-5 , 3.82��10-5, 4.46��10-5 and 5.71��10-5 s-1 at 288, 298, 308 and 318 K, respectively. According to Boyd equation, it can be deduced from the linear relationship of -ln(1-F) vs t that the liquid film spreading is the controlling step in the adsorption process.

Fig.4 Determination of adsorption rate constant ([Pb2+]0=0.4 mg/mL, resin 40.0 mg, pH=6.49)
According to the formula of Arrhenius lgk= �CEa/(2.30RT)+lgA, the apparent activation energy of Ea=18.1 kJ/mol can be calculated (Fig.5). It can be seen from the rate constant that the adsorption speed accelerates as the temperature increases in the experimental temperature range.

Fig.5 Relationship between lg k and 1/T ([Pb2+]0=0.4 mg/mL, resin 40.0 mg, pH=6.49)
3.4 Influence of adsorption temperature on distribution ratio and determination of thermodynamic parameters
The distribution coefficient of the resin for Pb2+ in the temperature range of 288-318 K was measured, and the result is shown in Fig.6. A straight line was obtained by plotting lg D against 1/T with a correlation coefficient of 0.966 6. The result obviously indicates that it is favorable for the adsorption with the temperature increasing. It implies that the adsorption process is endothermic. So the adsorption reaction is a chemical adsorption. By the following equation,

��H=20.9 kJ/mol, and then ��S =161 J/(mol?K) can be obtained from the slope and the intercept of the line. In the light of ��G=��H-T��S, ��G288K=-46.4 kJ/mol, ��G298K=-48.0 kJ/mol, ��G308K=-49.6 kJ/mo1 and ��G318K=-51.2 kJ/mol were obtained. The values reveal that the adsorption reaction is a spontaneous process under the experimental condition.

Fig.6 Influence of temperature on distribution ratio ([Pb2+]0=0.4 mg/mL, resin 40.0 mg, pH=6.49)
3.5 Effect of Na+ on adsorption capacity of 110-H resin
Six samples of 20.0 mg resin were put into conical flasks individually, then 20.0 mL of 0.4 mol/L HAc-NaAc buffer solution with pH 6.49 was added. After 24 h, 5.0 mL standard solution of Pb2+ was added individually. The flasks were shaken with different NaCl concentrations, as shown in Table 2. The clear upper-layer solution was taken for analysis until adsorption equilibrium. The adsorption capacity is calculated and shown in Table 2 also. The results indicate that the capacity of the resin for Pb2+ changes little with the increase of Na+ concentration under the experimental condition.
Table 2 Effect of Na+ on adsorption capacity of 110-H resin

3.6 Elution
The test was carried out by the above-mentioned method. 30.0 mL eluant was added. The results are listed in Table 3. It can be seen that the elution is different with the change in the concentration of HCl. Considering the environmental pollution, 0.025 mol/L HCl is favourable.
Table 3 Elution test results of Pb (��)

3.7 Dynamic adsorption and desorption
3.7.1 Dynamic adsorption curve
The dynamic adsorption test was conducted using a glass column packed with 300 mg newly prepared 110-H resin at room temperature. After the column was conditioned, a solution containing 0.200 mg/mL Pb2+ was continuously fed into the column at a constant flow rate of 0.25 mL/min. The effluents from the column were analyzed quantitatively using the above-mentioned method. Breakthrough data were acquired by plotting the volume of solution passing through the column vs the concentration of Pb2+ in the effluent. When the Pb2+ concentration from the bottom of the column (��) is equal to that at the top of the resin bed (��0), the experiment was terminated, and a plot of ��/��0 vs the volume of effluent gives a typical breakthrough curve, as shown in Fig.7.
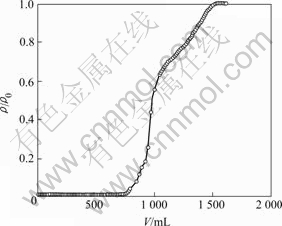
Fig.7 Dynamic adsorption curve (��o=0.200 mg/mL, flow-rate 0.25 mL/min)
3.7.2 Dynamic desorption curve
The dynamic desorption curves of 110-H resin for Pb2+ were obtained based on the volume of desorption solution and the Pb2+ concentration in the desorption solution. HCl of 0.025 mol/L was employed to strip Pb2+ from 110-H resin. The elution of Pb2+ from the column was at a flow rate of 0.25 mL/min. The results are shown in Fig.8.

Fig.8 Dynamic desorption curve (flow rate 5.4 BV/h)
3.8 Analysis of infrared spectra
It is deduced that the adsorption of Pb2+ by 110-H (��H��0) belongs to a chemical process. Therefore, the functional groups of 110-H, C=O, C��OH and Pb2+ are supposed to form chemical bonds. It is found from Fig.9 that the characteristic adsorption peak of the bond C=O (1 712 cm-1) disappears, and a new peak at 1 562 cm-1 occurs. The characteristic peak of the bond C��OH shifts from 1 392 cm-1 to 1 396 cm-1. These results show that there are coordination bonds between oxygen atoms and Pb2+, and H of C��OH is exchanged as a complex compound forms.

Fig.9 IR spectra of 110-H resin: (a) Before adsorption; (b) After adsorption
4 Conclusions
1) Pb2+ can be adsorbed on 110-H resin in the HAc-NaAc system with the pH value of 6.49, and the statically saturated adsorption capacity is 485 mg/g at 298 K. The Pb2+ adsorbed on 110-H resin can be eluted by 0.025 mol/L HCl quantitatively.
2) The adsorption behavior of 110-H resin for Pb2+ obeys the Langmiur isotherm. The apparent adsorption rate constant k298 K=3.82��10-5. The apparent activation energy Ea= 18.1 kJ/mol. The thermodynamic parameters of the adsorption are ��H=20.9 kJ/mol, ��S=161 J/(mol��K) and ��G298 K=-48.0 kJ/mol, respectively.
3) Infrared spectra show that the oxygen atoms of the functional group of resin coordinate to Pb2+ to form the coordination bands.
References
[1] ABDEL-HALIM S H, SHEHATA A M A, EL-SHAHAT M F. Removal of lead ions from industrial waste water by different types of natural materials [J]. Water Research, 2003, 37(7): 1678-1683.
[2] MAUCHAUFFEE S, MEUX E. Use of sodium decanoate for selective precipitation of metals contained in industrial wastewater [J]. Chemosphere, 2007, 69(5): 763-768.
[3] MELITA L, POPESCU M. Removal of Cr (��) from industrial water effluents and surface waters using activated composite membranes[J]. Journal of Membrane Science, 2008, 312(1/2): 157-162.
[4] BASHA C A, BHADRINARAYANA N S, ANANTHARAMAN N, MEERA SHERIFFA BEGUM K M. Heavy metal removal from copper smelting effluent using electrochemical cylindrical flow reactor [J]. Journal of Hazardous Materials, 2008, 152(1): 71-78.
[5] OZCAN A S, ERDEM B, OZCAN A. Adsorption of acid blue 193 from aqueous solutions onto Na-bentonite and DTMA-bentonite[J]. Journal of Colloid and Interface Science, 2004, 280(1): 44-54.
[6] JI Chun-nuan, QU Rong-jun, WANG Chun-hua, HOU Chen, SUN Chang-mei, XU Qiang, SUN Yan-zhi, WEI Chao. A chelating resin with bis[2-(2-benzothiazolylthioethyl)sulfoxide]: Synthesis, characterization and properties for the removal of trace heavy metal ion in water samples[J]. Talanta, 2007, 73(2): 195-201.
[7] DONIA A M, ATIA A A, HENIESH A M. Efficient removal of Hg(��) using magnetic chelating resin derived from copolymerization of bisthiourea/thiourea/glutaraldehyde[J]. Separation and Purification Technology, 2008, 60(1): 46-53.
[8] RAJESH N, JALAN R K, HOTWANY P. Solid phase extraction of chromium(��) from aqueous solutions by adsorption of its diphenylcarbazide complex on an Amberlite XAD-4 resin column [J]. Journal of Hazardous Materials, 2008, 150(3): 723-727.
[9] CHEN Yi-yong, CHAO Yan. Synthesis and characterization of polyacrylonitrile-2-amino-2-thiazoline resin and its adsorption behavior for noble metal ions [J]. Reactive Polymers, 2003, 55(1): 89-98.
[10] XIONG Chun-hua, SHU Zeng-nian. Adsorption behavior and mechanism of 4-amino-1,2,4-triazole resin for molybdenum(��)[J]. Nonferrous Metals, 2000, 52(2): 61-64. (in Chinese)
[11] GODE F, PEHLIVAN E. Removal of chromium (��) from aqueous solutions using Lewatit S100: The effect of pH, time, metal concentration and temperature [J]. Journal of Hazardous Materials, 2006, B136(2): 330-337.
[12] ATIA A A, DONIA A M, YOUSIF A M. Synthesis of amine and thio chelating resins and study of theirinteraction with zinc(��), cadmium(��) and mercury(��) ions in their aqueous solutions [J]. Reactive & Functional Polymers, 2003, 56(1): 75-82.
[13] BAKER H, KHALILI F. Analysis of the removal of lead (��) from aqueous solutions by adsorption onto insolubilized humic acid: Temperature and pH dependence [J]. Analytica Chimica Acta, 2004, 516(1/2): 179-186.
[14] XIONG Chun-hua, SHU Zeng-nian, WANG Yong-jiang. Adsorption of Mo(��) by 4-amino-pyridine resin[J]. Journal of Chemical Industry and Engineering (China), 2005, 56(7): 1267-1270. (in Chinese)
[15] XIONG Chun-hua, YAO Cai-ping, WANG Yong-jiang. Sorption behaviour and mechanism of ytterbium (��) on imino-diacetic acid resin[J]. Hydrometallurgy, 2006, 82(3/4): 190-194.
[16] XIONG Chun-hua, YAO Cai-ping, WU Xiang-mei. Adsorption of rhenjum(��) on 4-amino-1,2,4-triazole resin [J]. Hydrometallurgy, 2008, 90(2/4): 221-226.
[17] SHU Zeng-nian, XlONG Chun-hua, WANG Xu. Adsorption behavior and mechanism of amino methylene phosphonic acid resin for Ag (��) [J]. Trans Nonferrous Met Soc China, 2006, 16(3): 700-704.
[18] MARCZENKO Z. Spectrophotometric determination of elements [M]. ZHENG Yong-xi, REN Qi-yu, FENG Ke-cong, et al transl. Beijing: Geological Publishing House, 1983: 283.
Foundation item: Project (Y404279) supported by the Natural Science Foundation of Zhejiang Province, China
Corresponding author: XIONG Chun-hua; Tel: +86-571-88932083; E-mail: xiongch@163.com
(Edited by YNAN Sai-qian)