烧结温度对LiNi0.8Co0.1Mn0.1O2材料倍率性能和循环性能的影响
来源期刊:中国有色金属学报(英文版)2014年第12期
论文作者:李向群 熊训辉 王志兴 陈启元
文章页码:4023 - 4029
关键词:锂离子电池;LiNi0.8Co0.1Mn0.1O2;煅烧温度;循环性能;倍率性能
Key words:lithium ion battery; LiNi0.8Co0.1Mn0.1O2; sintering temperature; cycling performance; rate performance
摘 要:以共沉淀法制备的球形Ni0.8Co0.1Mn0.1(OH)2和LiOH・H2O为原料,研究烧结温度对LiNi0.8Co0.1Mn0.1O2 材料形貌、结构以及材料循环性能和倍率性能的影响。SEM和XRD结果表明:温度对材料形貌和结构有较大的影响,控制适当温度既能保证材料具有良好的形貌,也能抑制材料中锂镍的混排。电化学测试结果显示,当烧结温度从700 °C升高至750 °C时,材料性能逐渐提高,但是温度过高会恶化材料的性能。750 °C和780 °C烧结材料的循环性能几乎一致,200次循环后容量保持率为71.9%,但780 °C烧结材料的倍率性能低于750 °C材料的,其原因归结于温度过高,锂镍的混排加剧。在小电流充放电时,对材料性能影响有限,但是在大电流充放电时,3a位的Ni2+将严重阻碍锂离子的扩散。
Abstract: LiNi0.8Co0.1Mn0.1O2 powder was prepared by mixing LiOH・H2O and co-precipitated Ni0.8Co0.1Mn0.1(OH)2 at a molar ratio of 1:1.05, followed by sintering at different temperatures. The effects of temperature on the morphology, structure and electrochemical performance were extensively studied. SEM and XRD results demonstrate that the sintering temperature has large influence on the morphology and structure and suitable temperature is very important to obtain spherical materials and suppresses the ionic distribution. The charge-discharge tests show that the electrochemical performance of LiNi0.8Co0.1Mn0.1O2 powders becomes better with the increase of temperature from 700 °C to 750 °C and higher temperature will deteriorate the performance. Although both of materials obtained at 750 °C and 780 °C demonstrate almost identical cyclic stability at 2C rate, which delivers 71.9% retention after 200 cycles, the rate performance of powder calcined at 780 °C is much poorer than that at 750 °C. The XRD results demonstrate that the poor performance is ascribed to more severe ionic distribution caused by higher temperature.
Trans. Nonferrous Met. Soc. China 24(2014) 4023-4029
Xiang-qun LI1, Xun-hui XIONG2, Zhi-xing WANG2, Qi-yuan CHEN1
1. School of Chemistry and Chemical Engineering, Changsha 410083, China;
2. School of Metallurgy and Environment, Central South University, Changsha 410083, China
Received 8 January 2014; accepted 20 October 2014
Abstract: LiNi0.8Co0.1Mn0.1O2 powder was prepared by mixing LiOH・H2O and co-precipitated Ni0.8Co0.1Mn0.1(OH)2 at a molar ratio of 1:1.05, followed by sintering at different temperatures. The effects of temperature on the morphology, structure and electrochemical performance were extensively studied. SEM and XRD results demonstrate that the sintering temperature has large influence on the morphology and structure and suitable temperature is very important to obtain spherical materials and suppresses the ionic distribution. The charge-discharge tests show that the electrochemical performance of LiNi0.8Co0.1Mn0.1O2 powders becomes better with the increase of temperature from 700 °C to 750 °C and higher temperature will deteriorate the performance. Although both of materials obtained at 750 °C and 780 °C demonstrate almost identical cyclic stability at 2C rate, which delivers 71.9% retention after 200 cycles, the rate performance of powder calcined at 780 °C is much poorer than that at 750 °C. The XRD results demonstrate that the poor performance is ascribed to more severe ionic distribution caused by higher temperature.
Key words: lithium ion battery; LiNi0.8Co0.1Mn0.1O2; sintering temperature; cycling performance; rate performance
1 Introduction
Due to the advantages including high operation voltage, high energy density, long cycle life, light mass, small volume, low self-discharge rate, non-memory effect and environment-friendly, lithium ion batteries are considered the most attractive choice among rechargeable batteries. During the past decades, lithium ion batteries have been widely used in the small electronic devices, including cell phones, laptop computers and video cameras [1,2]. LiCoO2 layered cathode material has been extensively used as a promising candidate for commercial applications. However, its further development is limited by the toxicity and high expense of cobalt [3]. Due to the Jahn-Teller effect, LiMn2O4 suffers from the problems such as the fast decrease of electric capacity and the poor cycling performance [4,5]. Similar with LiCoO2, LiNiO2 also exhibits layered structures. In spite of the low-cost, low toxicity and large electric capacity, the serious cation mixing Li+/Ni2+ has remarkable influence on the electrochemical performance of lithium ion batteries. Besides, the safety problem of LiNiO2 still needs to overcome [6,7]. The cathode material LiNi1-x-yCoxMnyO2 with layed structure not only inherits its good cycling performance of LiCoO2, high specific electric capacity of LiNiO2 and thermal stability of LiMn2O4, but also shows low-toxicity and low-cost [8]. Owing to its high specific capacity (>200 mA・h/g) and good cycling stability, LiNi0.8Co0.1Mn0.1O2 possesses a great potential to replace LiCoO2 as the cathode material of high-capacity batteries used in electric vehicles and hybrid electric vehicles. Thus, it has attracted increasing attention in recent years [9,10].
In order to improve the electrochemical properties of Ni-rich materials, the parameters of the synthesis process have been extensively investigated. CHEN et al [11] found that the spherical Ni0.8Co0.1Mn0.1(OH)2 precursor with average size about 0.5 μm could be prepared by controlling the speed of co-precipitation, and the obtained LiNi0.8Co0.1Mn0.1O2 showed excellent cycling performance. HU et al [12] evaluated the effect of stirring speed, pH value, concentration of ammonia and reaction time in co-precipitation process on the morphology, size distribution and tap density of the Ni0.8Co0.2(OH)2 precursor. Besides, the effects of sintering parameters on the electrochemical performance of Ni0.8Co0.2(OH)2 were investigated in the optimum precursor fabrication process. ZHONG et al [13] prepared Ni0.8Co0.2(OH)2 with metal nitrate using maleic acid and ethanol as solution and the specific capacitance showed an increasing tendency instead of decreasing after 25 cycles. LI et al [14] produced LiNi0.8Co0.1Mn0.1- O2 via the rapid coprecipitation technique with metal chloride. The results showed that the materials fabricated by rapid coprecipitation exhibited low cation mixing and good electrochemical properties.
Sintering temperature and sintering time are the most important parameters for both conventional high temperature solid-state reaction and wet chemical pre-treated solid-state reactions including sol-gel and coprecipitation method. The migration of Li+ and the active energy needed for lattice reconstruction determine that the reaction must happen at a certain temperature. The distance between ions and the complexity of the lattice determine that the reaction should completely finish at a certain period. Accordingly, in the present work, the effects of sintering temperature on the morphology, microstructure and electrochemical performance, especially rate performance of LiNi0.8Co0.1Mn0.1O2 were investigated.
2 Experimental
2.1 Sample preparation
LiOH・H2O (excess coefficient=1.05) and Ni0.8Co0.1Mn0.1(OH)2 were weighed according to the stoichiometric ratio, followed by mixing and grinding in a mortar for 40 min in order to obtain a homogeneous mixture. The mixture was then sintered in a tube furnace under oxygen flow at 700, 725, 750 and 800 °C for 15 h, respectively. After cooling down to room temperature, LiNi0.8Co0.1Mn0.1O2 was fabricated.
2.2 Physical characterization
The phases of the samples were characterized by X-ray diffraction (XRD), which was conducted on a Rigaku diffractometer (Rigaku Corporation, Japan) at 40 kV and 50 mA. The step width, scanning speed and the scanning range of diffraction angle were 0.02°, 10 (°)/min and 10°-85°, respectively. A scanning electron microscope (SEM) (JSM-5600LV, JEOL) was used for microstructural examination.
2.3 Fabrication of battery and electrochemical property tests
Electrochemical charge-discharge tests were performed using the cathode with a mixture of 80% cathode material, 10% super P carbon black and 10% polyvinylidene fluoride (PVDF). The electrolyte was 1.0 mol/L LiPF6/EC+DMC+EMC (1:1:1, volume ratio). Preliminary cell tests were done using 2025 coin-type cell adopting Li metal as an anode with a voltage window of 2.8-4.3 V (versus Li/Li+) at room temperature. The Neware BTS-5 V/10 mA battery testing system was applied to the charge-discharge cycling tests at different rates. The voltage range was 2.8-4.3 V. The AC impedance experiments were conducted on the US CHI660 electrochemical workstation with the testing frequency ranging from 0.01 Hz to 100 kHz.
3 Results and discussion
3.1 DSC-TG analyses
In order to determine the sintering parameters of LiNi0.8Co0.1Mn0.1O2, the homogeneous mixture of Ni0.8Co0.1Mn0.1(OH)2 precursor and LiOH・H2O was analyzed by DSC-TG technique as shown in Fig. 1. It is obvious that the reaction consists of four steps. First, from room temperature to 280 °C, the absorbed water and crystal water lost, accompanied by huge heat-absorption and apparent mass loss. The physically absorbed water evaporated from room temperature to 100 °C. The next step was between 100 °C and 230 °C, which is ascribed to the crystal water. The third step was the decomposition of the Ni0.8Co0.1Mn0.1(OH)2 precursor and the melting of LiOH, and following process was the crystallization of LiNi0.8Co0.1Mn0.1O2. After reaching 820 °C, a slight but continuous mass loss was observed, probably due to the decomposition of the crystals. It has been reported that the over-heated sintering leads to the formation of nonstoichiometric Li1-xMO2-y accompanied by precipitation of O and Li [15,16]. Therefore, in order to make sure of the full reaction, the sintering temperature should be controlled in the range of 600-820 °C. Besides, during the sintering process, the samples should be kept around 500 °C for a while, so that the melted LiOH can completely permeate into Ni0.8Co0.1Mn0.1(OH)2.

Fig. 1 DSC-TG curves for precursors
3.2 Morphology and microstructure analyses
Figure 2 shows the SEM images of the Ni0.8Co0.1- Mn0.1(OH)2 precursor and the LiNi0.8Co0.1Mn0.1O2 sintered at different temperatures. Figures 2(a) and (b) show that the precursor samples are spherical with the diameter of 10-12 μm, formed by the agglomeration of tiny rod-like particles. During sintering at 700-750 °C, the precursors are still in regular spheres with the diameter of 10-12 μm with clear boundaries, and the agglomeration is still not apparent. From 700 to 750 °C, the primary particles tend to grow, leading to higher porosity. However, when reaching 780 °C, the primary particles grow significantly, resulting in an obvious morphology change. For the sake of the good mobility for better processability of the electrode fabrication and the high energy density of the lithium ion battery provided by the high tap density of spherical particles, the sintering temperature should be below 780 °C.
Figure 3 shows the XRD patterns of LiNi0.8Co0.1Mn0.1O2 sintered at different temperatures. It is obvious that all samples synthesized at different temperatures exhibit the same α-NaFeO2 structure, in which Li and transition metals occupied 3a and 3b positions, respectively. There are no impurity peaks in the XRD patterns. The c/a of the structure is relevant to the stability of the layered structure and when c/a>4.899, which means that the crystallization of the material is high. The I(003)/I(104) demonstrates the cation disorder of Li and Ni, namely higher I(003)/I(104) and lower cation disorder [17,18]. Table 1 lists the lattice parameters and I(003)/I(104) values of the LiNi0.8Co0.1Mn0.1O2 calculated by the software Jade. It is clear that the diffraction peak intensity and lattice parameter are strongly dependant on the temperature. From 700 °C to 750 °C, c/a increases. This is because with increasing temperature, the formed layer structures tend to be perfect. Besides, higher temperature leads to higher crystallinity, lower lattice stress and larger grain size. Due to the close diameter of Li+ and Ni2+, they can be easily mixed during sintering. With increasing temperature, the I(003)/I(104) decreases, which is attributed to higher temperature leading to the oxidation of Ni2+ to Ni3+ and decrease of cation disorder. However, when the temperature is too high, the cation disorder exhibits a further increase. According to the thermodynamic property, Ni3+ tends to transform into Ni2+ at a much higher temperature. Furthermore, based on the kinetic rate equation, the evaporation speed of Li is proportional to the exponent of the sintering temperature. The accelerated evaporation of Li results in more significant inhomogeneity. As a consequence, it is necessary to further investigate the influence of sintering temperature on the structure of material [9].

Fig. 2 Typical SEM images of Ni0.8Co0.1Mn0.1(OH)2 precursor (a, b) and LiNi0.8Co0.1Mn0.1O2 sintered at 700 °C (c), 725 °C (d), 750 °C (e) and 780 °C (f)

Fig. 3 XRD patterns of LiNi0.8Co0.1Mn0.1O2 sintered at different temperatures
Table 1 Lattice parameters of LiNi0.8Co0.1Mn0.1O2 calcined at different temperatures
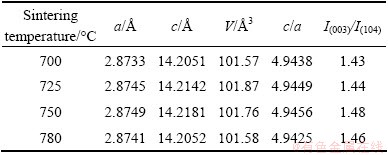

Fig. 4 Initial charging-discharging curves (a) and cycling performance at 2C (b) for LiNi0.8Co0.1Mn0.1O2 calcined at different temperatures
Figure 4 shows the initial charge-discharge curves and the cycling performance at 2C for LiNi0.8Co0.1Mn0.1O2 calcined at different temperatures. When the temperature increases from 700 °C to 750 °C, the initial discharging specific capacity increases and the cycling property is improved. The materials calcined at 750 °C possess higher initial discharging specific capacity (195.4 mA・h/g), and the capacity after 200 cycles at 2C is 113.7 mA・h/g with a retention of 71.9%. The XRD patterns reveal that the sample calcined at 750 °C exhibits good crystallinity and low cation disorder, leading to the best electrochemical property. However, the initial discharging performance and the cycling properties at 750 °C and 780 °C with 2C were similar, which is not in accordance with the XRD and SEM results. Thus, the reason needs further investigation.
Figure 5 presents the first discharge curves at different rates and rate capability of electrodes as a function of rate after 5 cycles. Due to the low sintering temperatures, the samples sintered at 700 °C and 725 °C exhibit incomplete crystals, large amount of residue Ni2+, severe inhomogeneity and poor rate capability. The results are in accordance with the XRD and cycling performance results. The materials calcined at 750 °C and 780 °C show almost the same performance at the rate lower than 5C. But the capacity of the latter material is far below the former one when charging-discharging at the rate between 5C and 10C. Such a result should be attributed to the transformation of Ni3+ into Ni2+ and higher evaporation speed of Li salt, which will lead more Ni2+ to 3a position [20,21]. At low current, Ni2+ at 3a position does not have significant influence on the specific capacity and cycling performance. However, when charging-discharging at a high rate, it inhibits the diffusion of Li+, resulting in lower rate capability. Figure 5 reveals that the sample calcined at 750 °C shows the best rate capability. The capability at 10C is 131.3 mA・h/g, 67.4 % of that at 0.1C.

Fig. 5 First discharge curves of different rates obtained from four samples after being calcined at 700 °C (a), 725 °C (b), 750 °C, 7801°C (d) and discharge capacity of first 5 cycles at different rates (e)
In order to better understand the influence of the two temperatures of 750 °C and 780 °C on the electrochemical performance, XRD was used to characterize the materials sintered at the two temperatures with the scanning angle of 10°-120° and scanning speed of 2 (°)/min. Rietveld method (using the software Fullprof) was applied for the data refinement. Pseudo-Voigt function was used to simulate the diffraction peaks and over 30 parameters including the atomic position, atom occupancies, lattice parameter and isotropic thermal parameters were refined. Figure 6 presents the XRD patterns and Rietveld refinements of LiNi0.8Co0.1Mn0.1O2 obtained at 750 °C and 780 °C. As shown in Fig. 6, the neglectable difference between experimental data and fitted results indicates that the refined results were reliable. The refinement results indicate that the ratios of cation mixing of the materials processed at 750 °C and 780 °C are 1.6 % and 2.3 %, respectively. Higher cation disorder means more Ni2+ at 3a position, leading to the restrained diffusion of Li ion at 3a position. Thus, the material sintered at 780 °C shows poor electrochemical property, especially at high rates. The results are consistent with the data in Table 2.

Fig. 6 XRD patterns (a) and Rietveld refinements (b) of LiNi0.8Co0.1Mn0.1O2 obtained at 750 °C and 780 °C
4 Conclusions
1) The influence of sintering temperature on the morphology, structure and electrochemical performance is significant. A proper sintering temperature can not only keep the morphology of spherical particles, but also inhibit the inhomogeneity of Li+ ions and Ni+ ions. The LiNi0.8Co0.1Mn0.1O2 sintered at 750 °C shows the best electrochemical performance.
2) LiNi0.8Co0.1Mn0.1O2 sintered at 750 °C shows good cycling performance and rate capability. Its capacity retention ratio is 71.9 % after charging- discharging 200 times at 2C. Its capability at 10C is 131.3 mA・h/g, which is about 67.4 % of that at 0.1C.
References
[1] FERGUS J W. Recent developments in cathode materials for lithium ion batteries [J]. Journal of Power Sources, 2010, 195(4): 939-954.
[2] SCROSATI B. Recent advances in lithium ion battery materials [J]. Electrochimica Acta, 2000, 45(15-16): 2461-2466.
[3] LUO Wen-bin, ZHOU Fu, ZHAO Xue-mei, LU Zhong-hua, LI Xin-hai, DAHN J R. Synthesis, characterization and thermal stability of LiNi1/3Mn1/3Co1/3-zMgzO2, LiNi1/3-zMn1/3Co1/3MgzO2 and LiNi1/3Mn1/3-zCo1/3MgzO2 [J]. Chemistry Materials, 2010, 22: 1164-1172.
[4] BLYR A, SIGALA C, AMATUCCI G, GUYOMARD D, CHABRE Y, TARASCON J M. Self-discharge of LiMn2O4/C Li-ion cells in their discharged state: Uderstanding by means of three-electrode measurements [J]. Journal of the Electrochemical Society, 1998, 145(1): 194-209.
[5] AOSHIMA T, OKAHARA K, KIYOHARA C, SHIZUKA K. Mechanisms of manganese spinels dissolution and capacity fade at high temperature [J]. Journal of Power Sources, 2001, 97-98: 377-380.
[6] DAHN J R, SACKEN U, MICHAL C A, Structure and electrochemistry of Li1±yNiO2 and a new Li2NiO2 phase with the Ni(OH)2 structure [J]. Solid State Ionics, 1990, 44(1-2): 87-97.
[7] DAHN J R, SACKEN U V, JUZKOW M W, AL-JANABY H, Rechargeable LiNiO2/carbon cells [J]. Journal of the Electrochemical Society, 1991, 138(8): 2207-2211
[8] KIM M H, SHIN H S, SHIN D, SUN Y K. Synthesis and electrochemical properties of Li[Ni0.8Co0.1Mn0.1]O2 and Li[Ni0.8Co0.2]O2 via co-precipitation [J]. Journal of Power Sources, 2006, 159: 1328-1333.
[9] SHIM J, KOSTECKI R, RICHARDSON T, SONG X, STRIEBEL K A. Electrochemical analysis for cycle performance and capacity fading of a lithium-ion battery cycled at elevated temperature [J]. Journal of Power Sources, 2002, 112: 222-230.
[10] XIONG Xun-hui, WANG Zhi-xing, GUO Hua-jun, ZHANG Qian, LI Xin-hai, Enhanced electrochemical properties of lithium-reactive V2O5 coated on the LiNi0.8Co0.1Mn0.1O2 cathode material for lithium ion batteries at 60 °C [J]. Journal of Materials Chemistry A, 2013, 1: 1284-1288.
[11] CHEN Wei, LI Xin-hai, WANG Zhi-xing, GUO Hua-jun, YUE Peng, LI Ling-jun. Influence of feeding rate on performance of LiNi0.8Co0.1Mn0.1O2 cathode materials [J]. The Chinese Journal of Nonferrous Metals, 2012, 22(7): 1956-1962. (in Chinese)
[12] HU Guo-rong, LIU Yan-jun, PENG Zhong-dong, DU Ke, GAO Xu-guang. Synthesize and properties of spherical cathode materials LiNi0.8Co0.2O2 by controlled crystallization method [J]. The Chinese Journal of Nonferrous Metals, 2007, 17(1): 59-67. (in Chinese)
[13] ZHONG Y D, ZHAO X B, CAO G S, TU T P, ZHU T J. Characterization of particulate sol-gel synthesis of LiNi0.8C00.2O2 via maleic acid assistance with different solvents [J]. Journal of Alloys and Compounds, 2006, 420(1): 298-305.
[14] LI Ling-jun, LI Xin-hai, WANG Zhi-xing, GUO Hua-jun, YUE Peng, CHEN Wei, WU Ling. A simple and effective method to synthesize layered LiNi0.8Co0.1Mn0.1O2 cathode materials for lithium ion battery [J]. Powder Technology, 2004, 206: 353-357.
[15] HWANG B J, SANTHANAM R, CHEN C H. Effect of synthesis conditions on electrochemical properties of LiNi1-yCoyO2 cathode for lithium rechargeable batteries [J]. Journal of Power Sources, 2002, 114: 244-252.
[16] WANG G X, ZHONG S, BRADHURST D H, DOU S X, LIU H K. Synthesis and characterization of LiNiO2 compounds as cathodes for rechargeable lithium batteries [J]. Journal of Power Sources, 1998, 76: 141-146.
[17] LI W, REIMERS J N, DAHN J R. In situ X-ray diffraction and electrochemical studies of Li1-xNiO2 [J]. Solid State Ionics, 1993, 67(1-2): 123-130.
[18] OHZUKU T, UEDE A, NAGAYAMA M. Electrochemistry and structural chemistry of LiNiO2 (R3-m) for 4 volt secondary lithium cells [J]. Journal of the Electrochemical Society, 1993, 140(7): 1862-1870.
[19] MOSHTEV R, ZLATILOVA P, VASILEV S, BAKALOVA I, KOZAWA A. Synthesis, XRD characterization and electrochemical performance of over lithiated LiNiO2 [J]. Journal of Power Sources, 1999, 81-82: 434-441.
[20] ALCANTARA R, LAVELA P, TIRADO J L, STOYANOVA R, ZHECHEVA E. Changes in structure and cathode performance with composition and preparation temperature of lithium cobalt nickel oxide [J]. Journal of the Electrochemical Society, 1998, 145(3): 730-736.
[21] COVER R K B, KANNO R, MITCHELL B J, YONEMURA M, KAWAMOTO Y. Effects of sintering temperature on the structure of the layered phase Lix(Ni0.8Co0.2)O2 [J]. Journal of the Electrochemical Society, 2000, 147(11): 4045-4051.
李向群1,熊训辉2,王志兴2,陈启元1
1. 中南大学 化学化工学院,长沙 410083;
2. 中南大学 冶金与环境学院,长沙 410083
摘 要:以共沉淀法制备的球形Ni0.8Co0.1Mn0.1(OH)2和LiOH・H2O为原料,研究烧结温度对LiNi0.8Co0.1Mn0.1O2 材料形貌、结构以及材料循环性能和倍率性能的影响。SEM和XRD结果表明:温度对材料形貌和结构有较大的影响,控制适当温度既能保证材料具有良好的形貌,也能抑制材料中锂镍的混排。电化学测试结果显示,当烧结温度从700 °C升高至750 °C时,材料性能逐渐提高,但是温度过高会恶化材料的性能。750 °C和780 °C烧结材料的循环性能几乎一致,200次循环后容量保持率为71.9%,但780 °C烧结材料的倍率性能低于750 °C材料的,其原因归结于温度过高,锂镍的混排加剧。在小电流充放电时,对材料性能影响有限,但是在大电流充放电时,3a位的Ni2+将严重阻碍锂离子的扩散。
关键词:锂离子电池;LiNi0.8Co0.1Mn0.1O2;煅烧温度;循环性能;倍率性能
(Edited by Hua YANG)
Foundation item: Project (2014CB643406) supported by the National Basic Research Program of China
Corresponding author: Zhi-xing WANG; Tel: +86-731-88836633; E-mail: zxwang21@16.3com
DOI: 10.1016/S1003-6326(14)63564-0

