
Morphology control of ultrafine cuprous oxide powder and its growth mechanism
CAO Yan(曹 艳), WANG Yue-jun(王岳俊), ZHOU Kang-gen(周康根), BI Zhen(毕 贞)
School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China
Received 6 July 2009; accepted 30 December 2009
_____________________________________________________________________________________________________
Abstract: Four shapes of Cu2O particles as sphere, cube, truncated octahedron and octahedron were prepared via glucose reduction of Cu(Ⅱ) under alkaline condition. The products were characterized by XRD and SEM. The effects of the precursor (CuO, Cu(OH)2), reaction temperature and glucose concentration on morphology of Cu2O particles were investigated, and the mechanism of morphology control was discussed on the basic theory of crystal nucleation and growth. It is found that the Cu+ supersaturation is remarkably influenced by the precursor kind, reaction temperature and glucose concentration, and the morphology of Cu2O particles can be controlled by the Cu+ supersaturation.
Key words: cuprous oxide; morphology control; supersaturation; growth mechanism
_____________________________________________________________________________________________________
1 Introduction
Cu2O is widely used as the additive of corrosion-proof coating, colorant of glass and bactericide. Cu2O is a p-type semiconductor with a small band gap of about 2 eV, which makes it a promising material in the application of solar energy conversion[1-5]. Because of the strong correlation of the properties with the structure and shape, it is essential to investigate the shape- controlling of Cu2O for its industrial applications. Recently, the preparations of Cu2O with different crystallinity, particle size and morphology have been reported by several researchers. DONG et al[6] synthesized cubic and spherical Cu2O using hydrazine as the reducing agent and CTAB and glucose as the modifiers. WU et al[7] reported stellar Cu2O crystals synthesized under microwave irradiation in the presence of surfactant p-octyl polyethylene glycol phenylether. LUO et al[8] reported the synthesis of tubular, cubic and hollow cubic Cu2O particles in simple liquid-phase reduction systems using nonionic surfactant octylphenyl ether (Triton X-100) as solvent. LIANG and ZHU[9] synthesized uniform Cu2O crystals with star-like and flower-like morphologies. YANG et al[10] prepared cubic Cu2O in water/oil (W/O) microemulsions by γ-irradiation in the presence of ethylene glycol and discussed the morphology control mechanism. Spherical, cubic, polyhedral, star-like, and octahedral Cu2O particles were synthesized by ZHAO et al[11] via reducing Cu(OH)2 by glucose.
However, most of the methods mentioned above utilize organic additive as modifier, irradiation of microwave, poisonous hydrazine, or under hydrothermal conditions. Those processes are inconvenient and complex. In this work, Cu2O particles were prepared via the reduction of CuSO4 by glucose under alkaline condition, and four shapes and crystallinities of Cu2O particles were obtained through changing the reaction conditions without any template or surfactant. Furthermore, the mechanism concerning morphology control was discussed.
2 Experimental
2.1 Materials
Copper sulphate (CuSO4?5H2O), sodium hydroxide (NaOH), glucose (C6H12O6?H2O) and solvents are of analytical grade purity and were used without further purification.
In all the experiments, 1.0 mol/L CuSO4 and 5.0 mol/L NaOH were prepared respectively, and 2.0 mol/L glucose was prepared except the case of special explanation. The volumes of CuSO4, NaOH and glucose solutions were kept constant at 200, 100 and 200 mL for any experiment.
2.2 Preparation procedure of Cu2O particles
2.2.1 CuO precursor route
The basic route of preparing Cu2O by CuO precursor route was CuSO4→Cu(OH)2→CuO→Cu2O. The Cu(OH)2 was precipitated by mixing the CuSO4 and NaOH solutions in a 1 000 mL beaker, and transformed to CuO by aging it at 70 ℃ for 30 min. Then, CuO was reduced by glucose at a constant temperature. After the reaction being completed, the yielded powder was collected, washed with distilled water and ethanol, and then dried at 45 ℃ in vacuum.
2.2.2 Cu(OH)2 precursor route
The basic route of preparing Cu2O by Cu(OH)2 precursor route was CuSO4→Cu(OH)2→Cu2O. The difference from the CuO precursor route was that the glucose solution was added to the Cu(OH)2-suspended solution immediately after precipitating Cu(OH)2 in the Cu(OH)2 precursor route.
2.3 Characterization of Cu2O particles
The X-ray diffraction (XRD) of the powder was carried out on a Rigaku D/Max 2550 X-ray diffractometer, using Cu Kα radiation (λ=1.541 8 ?). The operation voltage and current were 40 kV and 300 mA, respectively. The morphology and particle size of Cu2O particles were investigated by SEM (JSEM-6360LV, Japan).
3 Results
3.1 Preparation of Cu2O particles via CuO precursor route
The effect of reaction temperature on morphology of Cu2O particles was investigated via CuO precursor route. Fig.1 shows the SEM images of Cu2O particles prepared with CuO at different temperatures. It can be seen from Fig.1 that the yielded particles are cubic when CuO is used as the precursor. Cu2O obtained at 50 ℃ is uniform cubic particles, but Cu2O obtained at 70 ℃ shows a wide particle size range.

Fig.1 SEM images of Cu2O particles prepared with CuO as precursor at 50 ℃ (a), 60 ℃ (b) and 70 ℃ (c)
3.2 Preparation of Cu2O particles via Cu(OH)2 precursor route
3.2.1 Effects of reaction temperature
The effect of reaction temperature on morphology of Cu2O particles was investigated via Cu(OH)2 precursor route. Fig.2 shows the SEM images of Cu2O particles prepared with Cu(OH)2 at different temperatures. It can be seen from Fig.2 that the Cu2O particles prepared at a relative low temperature (50 ℃, 60 ℃) are in a spherical shape with smooth surface as well as high dispersity, but both spherical and cubic Cu2O particles are obtained at 70 ℃.

Fig.2 SEM images of Cu2O particles prepared with Cu(OH) 2 as precursor at 50 ℃ (a), 60 ℃ (b) and 70 ℃ (c)
3.2.2 Effects of glucose concentration
The effect of glucose concentration on morphology of Cu2O particles was investigated at reaction temperature of 50 ℃ via Cu(OH)2 precursor route. The SEM images of products are shown in Fig.3 when the glucose concentration is 0.25, 0.35, and 0.45 mol/L, respectively. It can be seen from Fig.3 that the shape of Cu2O particles changes from octahedron, truncated octahedron, mixture of cube and sphere to sphere with increasing glucose concentration.

Fig.3 SEM images of Cu2O particles prepared with Cu(OH)2 as precursor at different glucose concentrations: (a) 0.25 mol/L; (b) 0.35 mol/L; (c) 0.45mol/L
3.3 XRD analysis of Cu2O with different morphologies
The XRD patterns of Cu2O with different shapes are shown in Fig.4. All the XRD patterns can be indentified to a single phase of crystalline Cu2O. But, it can be seen that the peak of CuO appears in the XRD pattern of octahedral Cu2O particles (Fig.4(a)). The result means that the reduction of Cu(OH)2 is uncompleted under the conditions of deficient glucose, then surplus Cu(OH)2 dehydrates into CuO. No other impurity phase is observed in the XRD patterns.
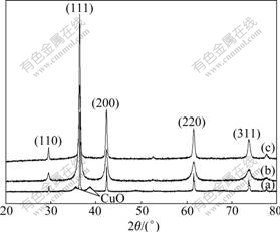
Fig.4 XRD patterns of Cu2O with octahedral (a), spherical (b) and cubic (c) shapes
4 Discussion
4.1 Shape-controlling mechanism
In this work, different shapes of Cu2O crystal particles were obtained via changing the reaction conditions such as the precursor, the glucose concentration and temperature. The change of Cu+ supersaturation resulted from these reaction conditions is the true reason of Cu2O shape variety.
When solution reaches a critical saturation condition, crystal embryos will overcome energy barrier to form nuclei. At an extremely high supersaturation, nucleation is an extraordinarily non-linear and fast process. As the results of effective collision among single-nuclei, molecule clusters, and primary particles under the actions of Brownian motion and fluid shear motion, aggregation becomes the main growth mode and spherical polycrystal particles finally form.
At an extremely low supersaturation, the solute supersaturation will rapidly reduce and nucleation potential will rapidly become weak as soon as the nucleation begins in the system. In this case, the solute will mainly precipitate on crystal in a manner of molecular scale growth and monocrystal particles finally form. In an aqueous solution system, the main growth modes of monocrystals are dislocation growth and two-dimensional growth[12], and the former is always priory to the latter[13]. The relation between the energy barrier (?G) of two-dimensional nucleation and supersaturation is as follows[14]:
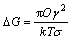 (1)
(1)
where γ is the step edge free energy; Ο is the area of single molecule; k is the constant of Boltzmann;T is the absolute temperature;σ is the supersaturation. It can be seen from Eq.(1), energy barrier of two- dimensional nucleation becomes small with increasing supersaturation. Thus, the two-dimensional growth only exists in a relatively high supersaturation system. In an extremely low supersaturation system, the crystal will grow up in dislocation mode.
Provided three-dimensional and two-dimensional critical supersaturations are σ1 and σ2 respectively, the relation of nucleation and growth mode to supersaturation σ is as follows:
1)As σ>σ1, the aggregation growth mode is dominant;
2) As σ2<σ<σ1 and σ is close to σ1, the two- dimensional growth mode is dominant;
3) As σ2<σ<σ1 and σ is close to σ2, the two- dimensional and dislocation growth modes are accompanied with each other;
4) As σ<σ2, the dislocation growth mode is dominant.
According to the crystal growth theory, the morphology of monocrystal is determined by the relative growth rate of different crystal faces[15]. For cubic Cu2O monocrystal, when face (111) grows relatively fast, the shape of Cu2O crystal tends to cube. When face (100) grows relatively fast, it tends to octahedron. When growth rate of face (111) is close to the growth rate of face (100), the shape of crystal tends to truncated octahedron [16]. Generally, face (111) must grow via the two-dimensional growth mechanism[13], so, different shapes of Cu2O crystal can be obtained via controlling the Cu+ supersaturation which determines the growth of face (111) and face (100).
4.2 Analysis of factors influencing morphology of Cu2O particles
During the glucose reductions of Cu(OH)2 or CuO to Cu2O particles, there are four reactions as follows:
Cu(OH)2=Cu2++2OH- (or CuO+H2O=Cu2++2OH-) (2)
2Cu2++3OH-+C6H12O6=2Cu++C6H11O7-+2H2O (3)
Cu++OH-=CuOH (4)
2CuOH=Cu2O+H2O (5)
Consequently, any factor that gives impacts to above reactions will affect the morphology of Cu2O particles.
4.2.1 Effects of precursor
As shown in Figs.1 and 2, the formation rate of Cu2+ is a key factor when glucose is excessive in the experiment. When CuO is used as the precursor, Cu+ supersaturation in the solution is also relatively low and situated at the two-dimensional growth mode because the formation rate of Cu2+ is slow, therefore, the Cu2O crystal is cube. When the precursor is Cu(OH)2, the formation rate of Cu2+ is very fast and Cu+ supersaturation in the solution is very high. In this case, the aggregation growth mode becomes dominant and so the Cu2O crystal is sphere.
4.2.2 Effects of reaction temperature
It can be seen from Figs.1 and 2 that the morphologies of the particles do not change at different temperatures except the case of Fig.2(c). This illustrates that the change of temperature does not change the growth mode of Cu2O. As shown in Fig.2(c), when Cu(OH)2 is used as the precursor at 70 ℃, both spherical and cubic particles are obtained. The reason is that when the temperature is 70 ℃, a part of Cu(OH)2 dehydrates into CuO immediately, and so both Cu(OH)2 and CuO work as the precursors together. In addition, as shown in Fig.1(c), the size distribution becomes wide. The reason is possibly that the reduction rate of Cu2+ and the Cu+ supersaturation rise with rising temperature, therefore, nucleation and growth progress simultaneously.
4.2.3 Effects of glucose concentration
With the glucose concentration rising, the reduction rate of Cu2+ is accelerated, and the Cu+ supersaturation rises gradually. The dominant growth mode of Cu2O crystal converts from dislocation growth to two- dimensional growth and aggregation growth. As a result, the morphology of Cu2O particles ranges from octahedron through truncated octahedron, mixture of cube and sphere to sphere.
5 Conclusions
1) When Cu(OH)2 is used as the precursor with excessive glucose, Cu+ supersaturation is high and Cu2O forms into spherical particles via aggregation growth mode.
2) When Cu(OH)2 is used as the precursor with deficient glucose, Cu+ supersaturation is low and Cu2O forms into octahedral particles with face (100) oriented growth via dislocation growth mode.
3) When CuO is used as the precursor with excessive glucose, the Cu+ supersaturation is moderate, and Cu2O forms into cubic particles with face (111) oriented growth.
4) When growth rate of face (111) is close to the growth rate of face (100), the shape of crystal tends to truncated octahedron.
References
[1] ?MITH M, GOTOVAC V, ALJINOVI? L J, LU?I?-LAV?EVI? M. An investigation of the electrochemical and photo-electrochemical properties of the cuprous oxide/liquid phase boundary [J]. Surface Science, 1995, 335: 171-176.
[2] JAYEWARDENA C, HEWAPARAMA K P, WIJEWARDENA D L A, GURUAGE H. Fabrication of n-Cu2O electrodes with higher energy conversion efficiency in a photo-electrochemical cell [J]. Solar Energy Materials & Solar Cells, 1998, 56: 29-33.
[3] WANG Xue-jun, LOU Tao. Photo degradation of dissolved organic matter and its impact on the biologic processes [J]. Photographic Science and Photochemistry, 2004, 22(4): 29-30.
[4] LI Jia-lin, LIU Li. Preparation of highly photocatalytic active nano-size TiO2-Cu2O particle composites with a novel electrochemical method [J]. Electrochemistry Communications, 2004, 6(9): 940-943.
[5] HE Ping, SHEN Xing-hai, GAO Hong-cheng. Size-controlled preparation of Cu2O octahedron nanocrystals and studies on their optical absorption [J]. Journal of Colloid and Interface Science, 2005, 284: 510-515.
[6] DONG Ya-jie, LI Ya-dong, WANG Cheng, CUI Ai-li, DENG Zhao-xiang. Preparation of cuprous oxide particles of different crystallinity [J]. Journal of Colloid and Interface Science, 2001, 243: 85-89.
[7] WU Zheng-cui, SHAO Ming-wang, ZHANG Wu, NI You-bao. Large-scale synthesis of uniform Cu2O stellar crystals via microwave-assisted route [J]. Journal of Crystal Growth, 2004, 260: 490-493.
[8] LUO Fang, WU Di, GAO Lei, LIAN Suo-yuan, WANG En-bo, KANG Zhen-hui, LAN Yang, XU Lin. Shape-controlled synthesis of Cu2O nanocrystals assisted by Triton X-100 [J]. Journal of Crystal Growth, 2005, 285: 534-540.
[9] LIANG Zhen-hua, ZHU Ying-jie. Synthesis of uniformly sized Cu2O crystals with star-like and flower-like morphologies [J]. Materials Letters, 2005, 59: 2423-2425.
[10] YANG Shi-guo, CHEN Qing-de, SHEN Xing-hai. The effect of ethylene glycol on the morphology of Cu2O nanoparticles synthesized in W/O microemulsion by γ-irradiation [J]. Spectroscopy and Spectral Analysis, 2007, 27(11): 2155-2159. (in Chinese)
[11] ZHAO Hua-tao, WANG Dong, ZHANG Lan-yue, BAI Zhi-ping, WU You-ting. A simple method for the preparation of Cu2O with different morphologies in high reaction concentration [J]. Chinese Journal of Inorganic Chemistry, 2009, 25(1): 142-146. (in Chinese)
[12] YU Xi-ling. Present-day development of the mechanism research of crystal growth [J]. Bulletin of National Natural Science Foundation of China, 2002, 16(4): 215-218. (in Chinese)
[13] WANG Ji-yang. Defect mechanism of crystal growth [J]. Physics, 2001, 30(6): 332-339.(in Chinese)
[14] LI Guang-hua, HAN Li, FANG Qi. Theory and application of crystal structure controlling its morphology [J]. Journal of Synthetic Crystals, 2005, 34(3): 546-549. (in Chinese)
[15] RAWLINGS J B, MILLER S M, WITKOWSKI W R. Model identification and control solution crystallization processes: A review [J]. Industrial & Engineering Chemistry Research, 1993, 32(7): 1275-1296.
[16] WU Hui-fang. Morphology and size control of silver oxide, silver and cuprous oxide micro-/nano-particles [D]. Xiamen: Xiamen University, 2007: 43-64. (in Chinese)
________________________
Corresponding author: ZHOU Kang-gen; Tel: +86-731-88836442; Fax: +86-731-88710171; E-mail: zhoukg63@yahoo.com.cn
(Edited by LI Xiang-qun)