J. Cent. South Univ. Technol. (2008) 15: 763-767
DOI: 10.1007/s11771-008-0141-5
Solvothermal preparation and characteristics of Sn-doped In2O3
GU Ying-ying(��ӳӨ)1, QIN Li-ping(����ƽ)1, 2, WANG Man-juan(������)1, LIU Xue-ying(��ѩӱ)2
(1. School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China;
2. Science and Technology Department, Guangxi University of Technology, Liuzhou 545006, China)
Abstract: Sn-doped In2O3 (ITO) nanopowders were prepared in ethanol solvent by solvothermal process. The effects of the solvothermal temperature, coprecipitation pH value and SnO2 content on the products phase and microwave absorption were investigated by X-ray diffractometry and microwave reflectance. ITO nanopowders with cubic structure can be respectively prepared at 250 and 270 �� for 6 h. The prepared product is InOOH or the mixture of InOOH and In3Sn4O12 when the solvothermal temperature is below 250��. With rising solvothermal temperature and prolonging time, the absorption of the ITO powders gradually decreases. The products are ITO nanopowders by coprecipitating at pH=9 or 11, but ITO powders with Sn3O4 at pH=6. The absorption of powders prepared at pH=6 is better than that at any other pH value. The products are all ITO nanopowders and crystal size reduces with increasing SnO2 content. The microwave absorption of ITO nanopowders with SnO2 content of 8% (mass fraction) is the best among samples with different SnO2 contents.
Key words: Sn-doped In2O3 (ITO); solvothermal process; microwave absorption; preparation
1 Introduction
Sn-doped In2O3 (ITO) is one kind of N-type semiconductor material with a wide band gap of 3.5 eV. The main features of it are as follows: 1) high free carrier density and good carrier mobility; 2) suitable doping and oxygen vacancies; and 3) regular ITO film crystal structure.
Wide band gap and high carrier concentration make ITO have a series of specific optical properties besides excellent electrical conductivity, such as transparency under visible light, absorbency to ultraviolet light, reflection to infrared light[1-2]. Due to these characteristics, ITO is widely used in electronics, transparent electrode, solar cells, electro-irradiance, and especially in screen display[3-11].
There are many reports about the preparation of ITO nano-powders worldwide[12-14], such as chemical coprecipition, sol-gel process, inorganic coprecipition, atomizing-combustion technique and hydrothermal- calcining. But how to prepare ITO nanocrystals with perfect properties by simple process is always of great importance.
Recently solvothermal reaction has been widely used in material field[15-16]. Due to the physico-chemical properties of the organic solvents such as polarity, viscosity and softness, new synthetic routes can be worked out according to different solvents and aim products. LEE and CHOI[17] prepared blue ITO nanopowders by solvothermal process in stainless steel pressure vessels equipped with magnetic stirring. The results show that ITO powders are in spherical shape by using ethylene as solvent and in square shape by using high viscosity polyethylene glycol 600 as solvent. They did not study the microwave absorption of ITO powders. The reports about the microwave absorption of ITO nanopowders are few at home and abroad. Therefore, in this work, ITO powders with cubic structure were prepared in ethylene solvent and a Teflon-lined stainless steel vessel (100 mL) by a solvothermal process. Meanwhile, the microwave absorption of the powders was investigated.
2 Experimental
2.1 Solvothermal preparation
In2(SO4)3 solution was gained by dissolving indium in H2SO4 solution (4 mol/L). In2(SO4)3 and SnCl4 solutions were mixed as the molar ratio of In to Sn was 9?1. Ammonia (25%-28��) was added into the solution under stirring to adjust the pH value in coprecipitate, and continued stirring for 40 min. The solution was filtrated and washed until no Cl- and SO42- were examined, and then dried at 80 ��. The solvothermal precursor was obtained. The obtained precursor after rubbing was solvothermally treated at a certain temperature in a Teflon-lined stainless steel vessel (100 mL) with ethylene solvent. After reaction, the products were filtrated and repeatedly washed with ethanol and dried at 80 ��.
2.2 Analysis of products
The products were analyzed by X-ray diffraction (XRD, Bruit advanced D8) using Cu K�� radiation. XRD patterns of powders were obtained in the 2�� range between 0? and 80? with a step of 0.02? and a scan speed of 2 (?)/min. The crystal sizes of the ITO powders were calculated from the line broadening of the (222) diffraction line according to the Scherrer equation:
D= 0.9��/(��cos��)
where D is the crystal size, �� is the wavelength of Cu K�� radiation, and �� is the corrected half-width of the diffraction peak.
The microwave absorption of powders was measured by measuring microwave reflectance in frequency range of 8-12 GHz by swept amplitude analyzer. The method of measuring ITO powders microwave adsorption performance was to join ITO powders in molten olefin at the mass ratio of 8?3, after stirring, directly pouring in flange, and then measuring microwave reflectance in frequency range of 8-12 GHz by swept amplitude analyzer to investigate the effect of preparation condition on ITO powders microwave adsorption performance.
3 Results and discussion
3.1 Effects of solvothermal temperature and time on products
3.1.1 Effects of solvothermal temperature and time on product phase
Fig.1 shows XRD patterns of the samples prepared by the same precursor at different solvothermal temperatures and time using ethylene as solvent.

Fig.1 XRD patterns of samples prepared at different conditions: (a) 210 ��, 6 h, InOOH; (b) 230 ��, 6 h, In4Sn3O12, InOOH; (c) 230 ��, 10 h, In4Sn3O12, InOOH; (d) 250 ��, 6 h, In2O3; (e) 270 ��, 6 h, In2O3
It can be seen that the samples prepared by solvothermal process at 210 �� are poorly crystallized InOOH (PDF71-2284) (Fig.1(a)). The sample obtained at 230 �� for 6 h (Fig.1(b)) is the mixture of In4Sn3O12 (PDF88-0773) and InOOH. From Figs.1(b) and (c), it is found that reaction time has little influence on the phase of the products.
The product obtained at 250 �� for 6 h (Fig.1(d)) is cubic structural In2O3 (PDF 65-3170), no SnO2 appears, and the diffraction peaks of the sample shift towards smaller angles, indicating that in the process of heat dealing Sn dopes into In2O3 and Sn atoms occupy the sites of In atoms. This results in solution of Sn-doped In2O3 powders[18-19].
When the solvothermal temperature is 270 ��, the sample is also cubic structural In2O3 crystal (Fig.1(e)) and has high crystallization.
The crystal sizes of the ITO powders were calculated from the Scherrer equation. According to the equation, the crystal sizes of samples prepared at 250 and 270 �� are 40.5 and 50.3 nm, respectively, which shows that with increasing temperature, the crystal size of product increases.
Fig.2 shows the TEM images of ITO powders prepared at different temperatures in ethylene. It reveals that the average sizes of the particles corresponding to Figs.2(a) and (b) are approximately 50 and 60 nm, respectively, indicating that the ITO particle size increases as the solvothermal temperature increases, which is in agreement with the analytical result of XRD.

Fig.2 TEM images of ITO powders prepared at different temperatures: (a) 250 ��; (b) 270 ��
3.1.2 Effects of solvothermal temperature on microwave absorption of products
Fig.3 shows the microwave absorption of powders prepared by coprecipitating at pH=9 and different solvothermal temperatures for 6 h. When the temperature is 250��, the prepared sample has the maximal microwave reflectance of -24 dB in the frequency range of 7-12 GHz and the frequency ranges of microwave reflectance under -10 dB are 8.3-9.2 GHz and 9.45- 10.30 GHz, which shows that the prepared sample has a wide microwave absorption range of 1.75 GHz. When the temperature is 270 ��, the prepared sample has the maximal microwave reflectance of -15 dB, which is lower than that at 250 ��, and the frequency range of microwave reflectance under -10 dB is 1.0 GHz. Clearly the sample��s microwave absorption prepared at 250 �� is better than that at 270 ��. When the temperature is 250 ��, the powder becomes good crystallite ITO, and it is of small size, large specific surface area, high surface atom rate and many hanging bonds, which result in the increase of the surface polarization velocity, multiply dispersion and powders microwave absorption. When the temperature increases, the crystal size increases and hanging bonds decrease, which result in the decrease of the surface polarization velocity, multiply dispersion and powders microwave absorption.
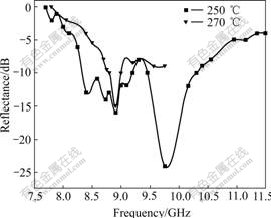
Fig.3 Microwave absorption of powders prepared at different solvothermal temperatures
3.1.3 Effects of solvothermal time on microwave absorp- tion of products
Fig.4 shows the effects of solvothermal time on the microwave absorption of powders prepared by coprecipitating at pH=9 and solvothermal temperature of 250 ��. When the time is 6 h, the prepared sample has wider frequency ranges of 8.3-9.2 GHz and 9.45-10.30 GHz with reflectance under -10 dB. The breadth is 1.75 GHz. When the time is 12 h, the maximal microwave reflectance of the prepared sample is -28 dB, but the frequency range is narrower than that for 6 h. When the time is 15 h, the microwave absorption of the prepared sample is clearly the weakest.

Fig.4 Microwave absorption of powders prepared at different solvothermal time
It can be seen that in conditions of the same precursor and solvothermal temperature, the microwave absorption of ITO nanopowders gradually increases and then decreases with prolonging solvothermal time. When the time is 12 h, the sample has the maximal microwave reflectance but a narrow microwave absorption range. This is because with prolonging solvothermal time, the crystallization and size of the well crystallized crystals increase. The increasing trend of size is greater than crystal��s crystallization, so microwave absorption of the powder reduces as a whole.
3.2 Effects of coprecipitation pH value on products
3.2.1 Effects of coprecipitation pH value on product phase
Fig.5 shows XRD patterns of samples prepared by solvothermal process at different pH values and the same solvothermal temperature and time (250 ��, 6 h). From Fig.5, it can be summarized that the product is cubic structural ITO at pH=9 and pH=11, and cubic ITO with impurity Sn3O4 at pH=6, respectively. Table 1 summarizes the crystal size of the ITO powders prepared at different pH values according to the Scherrer equation.

Fig.5 XRD patterns of samples prepared at different coprecipitation pH values
Table 1 Crystal sizes of samples prepared at different pH values

From Table 1, it is clear that the crystal size of samples gradually reduces with increasing coprecipita- tion pH value.
3.2.2 Effects of pH value on microwave absorption of products
Fig.6 shows the microwave absorption of powders prepared at solvothermal temperature of 250 �� for 6 h and different pH values. When the pH value is 6, the prepared sample has the maximal microwave reflectance of -32 dB in a frequency range of 7-12 GHz, and the frequency range with microwave reflectance under -10 dB is 8.8-11.2 GHz, which shows that it has a wide microwave absorption range of 2.4 GHz. The microwave absorption of sample prepared at pH=6 is clearly more excellent than that prepared at any other pH values. The microwave absorption of samples reduces with increasing pH value. When the pH value is 6, the sample is ITO powder with impurity of Sn3O4. This shows that it may be propitious to microwave absorption when ITO contains small amount of other components.
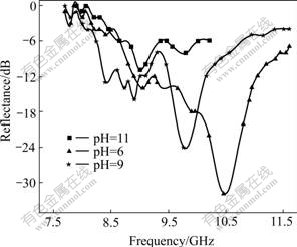
Fig.6 Microwave absorption of powders prepared at different pH values
3.3 Effects of SnO2 content on products
3.3.1 Effects of SnO2 content on product phase
In this work, the SnO2 content, m(SnO2)/ [m(SnO2)+m(In2O3)], was calculated as the comparative ratio of In2(SO4)3 and SnCl4 raw materials.
Fig.7 shows XRD patterns of samples prepared by solvothermal process at different SnO2 content and the same solvothermal condition (250 ��, 6 h, pH=9). From Fig.7, it can be summarized that the products are all well crystallized ITO powders even though SnO2 content is 15% (mass fraction), which shows Sn ion can dope In2O3 crystal perfectly.

Fig.7 XRD patterns of samples prepared at different SnO2 contents
Table 2 summarizes the crystal sizes of the ITO powders prepared at different SnO2 contents, which are calculated from the (222) diffraction line according to the Scherrer equation.
Table 2 Crystal sizes of samples at different SnO2 contents according to XRD

From Table 2, it is clear that the crystal size of samples gradually reduces with increasing SnO2 content. This shows that doping of Sn ion restrains In2O3 to grow. LEITE et al[20] considered that the doped ion reduced particles mobility and driving power of growth, so restrained particles to grow.
3.3.2 Effects of SnO2 content on microwave absorption of products
Fig.8 shows the microwave absorption of powders prepared at 250 �� for 6 h and pH=9 but different SnO2 contents. Microwave absorption of ITO nanopowders gradually increases and then decreases with rising SnO2 content. When the SnO2 content reaches 8%, the sample has the best microwave absorption. This is because when the SnO2 content is low, the concentration of free carrier is low and the surface polarizing rate decreases, which decreases the microwave absorption of ITO powders. With increasing SnO2 content, the concentration of free carrier and the surface polarization velocity increase, leading to the increase of powders microwave absorption. With continuously increasing SnO2 content, doping reaches saturation. When the concentration of free carrier increases, the carrier mobility reduces, thus resulting in the decrease of powders microwave absorption.

Fig.8 Microwave absorption of powders prepared at different SnO2 contents
4 Conclusions
1) ITO nanopowders with perfect crystal and cubic structure are prepared at 250 �� or above in ethylene solvent by solvothermal process. With increasing solvothermal temperature and time, the microwave absorption of powders decreases.
2) When the coprecipitation pH value is 6, the product is ITO powder with Sn3O4. When pH value equals 9 or 11, the product is single phase ITO. ITO powders prepared at pH value of 6 are of good microwave absorption compared with those prepared at pH values of 9 and 11.
3) With increasing SnO2 content, the crystal size of samples is gradually reduced. When SnO2 content is 8%, the powders have better microwave absorption property.
4) When the solvothermal temperature is 250 ��, pH is 6 and SnO2 content is 8%, the powders prepared are of good microwave absorption.
References
[1] JIANG Xie-chang, HU Yong. Prospect and industrial production of the transparent and conductive glass with ITO film [J]. Vacuum, 1995, 12(4): 1-8.
[2] Vossen J L. Transparent conducting films [J]. Physical Thin Films, 1997(9): 171-173.
[3] Kim K Y, Park S B. Preparation and property control of nano-sized indium tin oxide particle [J]. Mater Chem Phys, 2004, 86(1): 210-221.
[4] Zhang Y F, Zhang J X. Preparation of nano-ITO powder by co-precipitation method and structure token [J]. Funct Mater, 2003, 34(5): 573-574. (in Chinese)
[5] Hedia M, Moncef S, Srahim B. Screen-printed tin-doped indium oxide (ITO) films for NH3 gas sensing [J]. Materials Science and Engineering C, 2006, C26: 500-504.
[6] Zhong Yi, Wang Da-jian, Liu Rong-pei, GUO Yu-zhong. Preparation technologies and applications of the indium tin oxide (ITO) targets [J]. Journal of Kunming University of Science and Technology, 1997, 22(1): 66-70. (in Chinese)
[7] CHEN Fei-xia, FU Jin-dong, WEI Ya-bin, ZHAO Shi-lin. Preparation of nano indium/tin oxide transparent heat insulation coatings and its performance characterization [J]. Paint and Coatings Industry, 2004, 34(2): 48-51. (in Chinese)
[8] DUAN Xue-chen, YANG Xiang-ping. Application and development of new material-ITO films [J]. Rare Metals and Cemented carbides, 1999, 138(3): 58-60. (in Chinese)
[9] LIU Shi-you. The productive situation and application of indium-doped tin dioxide films [J]. Materials Science and Engineering, 1997, 17(2): 98-100. (in Chinese)
[10] Ivanovskaya M, Gurlo A, Bogdanov P. Mechanism of O3 and NO2 detection and sensitivity of In2O3 sensors [J]. Sensors and Actuators B, 2001, B77: 264-267.
[11] ZHOU Ke-sheng, LIU Gui, YIN Li-song, KONG De-ming. Microwave absorbing property of nano-Fe3O4/BaTiO3 composite system [J]. Journal of Central South University: Science and Technology, 2005, 36(5): 872-876.
[12] TANG S C, YAO J S, CHEN J, LUO J G. Preparation of indium tin oxide (ITO) with a single-phase structure [J]. J Mater Process Technol, 2003, 137(1/3): 82-85.
[13] XU H R, ZHU G S, ZHOU H Y, YU A B. Preparation of monodispersed tin-doped indium oxide nano-powders under moderate conditions [J]. Mater Lett, 2005, 59(1): 19-21.
[14] YANAGISAWA K, UDAWATTE C P, NASU S. Preparation and characterization of cine indium tin oxide (ITO) powders by a hydrothermal treatment and post annealing method [J]. J Mater Res, 2000, 15(6): 1404-1408.
[15] LI J, CHEN Z, WANG R J, PROSERPIO D M. Low temperature route towards new materials: Solvothermal synthesis of metal chalcogenides in ethylenediamine [J]. Coordination Chemistry Reviews, 1999, 190/192: 707-735.
[16] CHEN Z, ZHENG X, CHEN R Y, CHENG W, ZHANG Y K. Solvothermal synthesis of metal chalcogenides and its employment [J]. Chinese J Mater Res, 2001,15(2): 151-158. (in Chinese)
[17] LEE J S, CHOI S C. Solvent effect on synthesis of indium tin oxide nano-powders by a solvothermal process [J]. J Eur Ceram Soc, 2005, 25: 3307-3314.
[18] Nadaud N, Lequeux N, Nanot M. Structural studies of tin-doped indium oxide (ITO) and In4Sn3O12 [J]. J Solid State Chem, 1998, 135(1): 140-148.
[19] Chen M, Pei Z L, Bai X D, HHANG R F, WEN L S. X-ray photoelectron spectroscopy studies of ITO thin films [J]. J Inorg Mater, 2000, 15(1): 188-192.
[20] LEITE E R, MACIEL A P, LONG E, WEBER I T, LISBOIZ- FILHO P N, PAIVA-SANTOS C O, ANDRADE A V C, PALOSCIMAS C A, MANIETTE Y, SCHREINER W H. Development of metal oxide nanoparticles with high stability against particle growth using a metastable solid solution [J]. Adv Mater, 2002, 14(12): 905-908.
Foundation item: Project(2001BA901A09) supported by the National Western Development and Technique Foundation during the 10th Five-Year Plan Period
Received date: 2008-03-27; Accepted date: 2008-05-08
Corresponding author: GU Ying-ying, Professor; Tel: +86-13467517387; E-mail: guyy02@163.com
(Edited by CHEN Wei-ping)