Removal of SO2 from flue gas using Bayer red mud:Influence factors and mechanism
��Դ�ڿ������ϴ�ѧѧ��(Ӣ�İ�)2019���2��
�������ߣ���� ���� ��� ��� ��ѧǫ ��ƽ
����ҳ�룺467 - 478
Key words��Bayer red mud; flue gas desulfurization; wet catalytic oxidation; iron catalysis
Abstract: The absorbent composing of Bayer red mud and water was prepared and applied to removing SO2 from flue gas. Effects of the ratio of liquid to solid (L/S), the absorption temperature, the inlet SO2 concentration, the O2 concentration, SO42- and other different components of Bayer red mud on desulfurization were conducted. The mechanism of flue gas desulfurization was also established. The results indicated that L/S was the prominent factor, followed by the inlet SO2 concentration and the temperature was the least among them. The optimum condition was as follows: L/S, the temperature and the SO2 concentration were 20:1, 25 ��C and 1000 mg/m3, respectively, under the gas flow of 1.5 L/min. The desulfurization efficiency was not significantly influenced when O2 concentration was above 7%. The accumulation of SO42- inhibited the desulfurization efficiency. The alkali absorption and metal ions liquid catalytic oxidation were involved in the process, which accounted for 98.61%.
Cite this article as: TAO Lei, WU Heng, WANG Jie, LI Bin, WANG Xue-qian, NING Ping. Removal of SO2 from flue gas using Bayer red mud: Influence factors and mechanism [J]. Journal of Central South University, 2019, 26(2): 467�C478. DOI: https://doi.org/10.1007/s11771-019-4019-5.

ARTICLE
J. Cent. South Univ. (2019) 26: 467-478
DOI: https://doi.org/10.1007/s11771-019-4019-5
TAO Lei(����)1, WU Heng(���)1, WANG Jie(���)1, LI Bin(���)1,WANG Xue-qian(��ѧǫ)1, NING Ping(��ƽ)2
1. Faculty of Environmental Science and Engineering, Kunming University of Science and Technology, Kunming 650500, China;
2. National and Local Joint Engineering Research Center for Waste Gas Resource Utilization in Metallurgy and Chemical Industry, Kunming University of Science and Technology, Kunming 650500, China
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2019
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2019
Abstract: The absorbent composing of Bayer red mud and water was prepared and applied to removing SO2 from flue gas. Effects of the ratio of liquid to solid (L/S), the absorption temperature, the inlet SO2 concentration, the O2 concentration, SO42- and other different components of Bayer red mud on desulfurization were conducted. The mechanism of flue gas desulfurization was also established. The results indicated that L/S was the prominent factor, followed by the inlet SO2 concentration and the temperature was the least among them. The optimum condition was as follows: L/S, the temperature and the SO2 concentration were 20:1, 25 ��C and 1000 mg/m3, respectively, under the gas flow of 1.5 L/min. The desulfurization efficiency was not significantly influenced when O2 concentration was above 7%. The accumulation of SO42- inhibited the desulfurization efficiency. The alkali absorption and metal ions liquid catalytic oxidation were involved in the process, which accounted for 98.61%.
Key words: Bayer red mud; flue gas desulfurization; wet catalytic oxidation; iron catalysis
Cite this article as: TAO Lei, WU Heng, WANG Jie, LI Bin, WANG Xue-qian, NING Ping. Removal of SO2 from flue gas using Bayer red mud: Influence factors and mechanism [J]. Journal of Central South University, 2019, 26(2): 467�C478. DOI: https://doi.org/10.1007/s11771-019-4019-5.
1 Introduction
Sulfur dioxide (SO2), as one of the major air pollutants, has attracted wide attention in recent years. Thermal power plants and smelters are the major stationary emission sources of SO2 pollution [1]. Besides, the higher requirement on desulfurization methods has been put forward increasingly in many countries owing to the stricter SO2 emission standard [2]. However, the wet flue desulfurization that is widely used currently, including limestone-gypsum and ammonium process, still faces some problems. For example, the existing desulfurization by-product obtained is difficult to be used effectively but is easy to cause the potential secondary pollution [3]. Therefore, it is a hot research direction to establish a new environmentally friendly and inexpensive desulfurization method.
Red mud is the solid waste residue produced after the extraction of alumina from bauxite mine [4]. According to the production process, it is mainly divided into sintering red mud, Bayer red and combined red mud [5]. China is the largest producer of red mud in the world and about 1.0�C 1.8 t red mud is created for per ton of alumina, of which Bayer red mud accounts for a large amount because of the relative simple process, low lost and high-quality product [5�C8]. However, the Bayer red mud contains high alkali and low activity, thus the dealkalization is meaningful [9�C16]. The traditional methods such as landfill and stockpile not only need large site, but also likely lead to resource waste and pose secondary soil, groundwater, and air pollution [4, 17]. Therefore, the comprehensive utilization of red mud that is in the field of environment has been widely carried out, such as being used as adsorbent for removal of element mercury [6] and catalyst for enhanced nitrate reduction [18] owing to its alkalinity, composition and low costs. Some researches such as the flue gas desulfurization have been conducted [19, 20]. ELISABETTA et al [20] have studied the performance and liquid-side mass transfer of flue gas desulfurization by using the suspensions of Bayer red mud. However, there are few relevant studies on performance and mechanism about flue gas desulfurization with Bayer red mud.
In this study, SO2 absorbents were prepared using the Bayer red mud and water. The effects of several key factors including the ratio of solid to liquid (L/S), absorption temperature, the inlet SO2 concentration, the O2 concentration and SO42�C on desulfurization efficiency were researched. The chemical and physical properties of raw and spent Bayer red mud were investigated using characterization such as X-ray fluorescence (XRF), X-ray diffraction (XRD), X-ray photoelectron spectroscopy (XPS), scanning electron microscopy (SEM) and Brunauer-Emmett-Teller (BET). The mechanism of flue gas desulfurization was also established, especially the contribution of various components in the absorbent. The results will provide some guidance for resource utilization of Bayer red in the field of flue gas desulfurization.
2 Materials and methods
2.1 Materials
The Bayer red mud was provided by an aluminum plant of Yunnan Province, China, and the pH of which was approximately 12.3. The Bayer red mud slurry was prepared according to the mass ratio of solid to water, where the mass of Bayer red mud was fixed to 2 g. Chemicals including H2SO4, HCl and NaOH were analytical grade and without any further treatment.
2.2 Experiment setup and procedures
The flow chart of the experimental device is shown in Figure 1. SO2, O2 and N2 from the compressed cylinder (Messer Gas Co., China) was filled into the gas mixing chamber in a certain proportion controlled by three mass flowmeters (Sevenstar Co., China) and evenly mixed to simulate the industrial low concentration SO2 flue gas based on the requirement of the experiment. A certain amount of red mud slurry was added into the reaction tube and the temperature in the reactor was regulated by a constant temperature water bath (DF 101, China) beforehand. Then the flue gas was introduced into the self-made u-shaped glass bubbling reaction tube with an inner diameter of 20 mm and a height of 100 mm, where the aerator was evenly aerated with frosted head. The flue gas analyzer (ECOM J2KN, German) was adopted to measure the inlet and outlet SO2 concentration at the certain time. Thereafter, the off-gas was absorbed by NaOH solution.
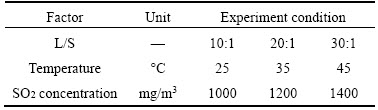
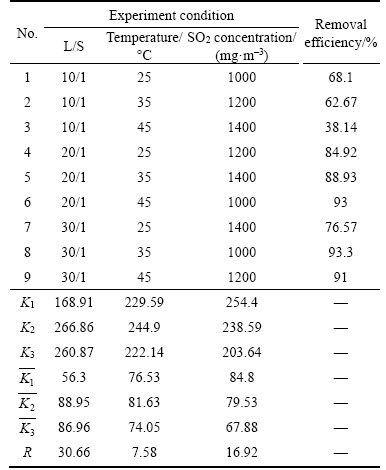
In the process of flue gas desulfurization, there are many factors affecting the desulfurization efficiency of Bayer red mud. Therefore, in order to obtain the impact of factors affecting the process of flue gas desulfurization and the optimum condition, orthogonal experiment (Table 1) was applied to studying the effects of the L/S, the absorption temperature and the inlet SO2 concentration. The desulfurization efficiency of two hours was selected as a test factor to find out the important order of factors and the optimal parameters in this process.
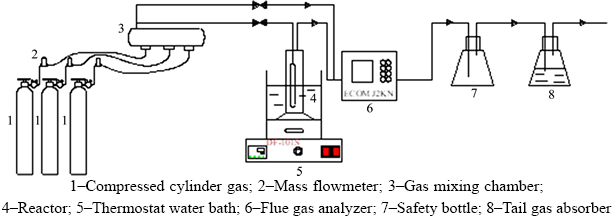
Figure 1 Flow chart of experimental device:
Table 1 Orthogonal experiment scheme of L9 (34) for flue gas desulfurization
The desulfurization efficiency was calculated by using Eq. (1).
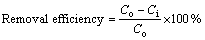 (1)
(1)
where Co and Ci are the inlet and outlet SO2 concentration, respectively, mg/m3.
In order to further prove the desulfurization mechanism of Bayer red mud slurry, the effects of SO42�C and different components including water, alkali and non-alkali on desulfurization were investigated by a series of experiments, of which, the former was conducted by using HCl and H2SO4 to adjust the red mud slurry pH to 3, where soluble alkali and solid alkali in the red mud were consumed by HCl and H2SO4, then the obtained acid red mud slurry was used for SO2 removal. The contribution of different components to desulfurization was carried out by using 40 mL water, 40 mL supernatant from the filtrate prepared by 2 g Bayer red mud and 40 mL water with 0.55 ��m membrane, slurry prepared by 40 mL water and the 2 g non-soluble alkali Bayer red mud, slurry prepared by 40 mL water and 2 g raw Bayer red mud as desulfurization agent, respectively. After the outlet SO2 concentration equals the inlet one, the total sulfur dioxide capacities were calculated according to Eq. (2), respectively. The titration of slurry prepared by 40 mL water and the 2 g non-soluble alkali Bayer red mud with 1.0 mol/L H2SO4 to pH 3.0 was used to calculate the contribution amount of solid alkali. Thereafter, the contribution of different components including water, soluble alkali, solid alkali and non-alkli were obtained by using Eq. (3).
Sulfur dioxide capacity=
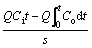 (2)
(2)
Contributions of component i= (3)
(3)
where Q refers to the total flue gas flow, mL/min; t represents the absorption time, min; s is the Bayer red mud mass, g; i=1�C4 represents the water, soluble alkali, solid alkali and non-alkli, respectively; Ti and Ts denote the SO2 capacity of water, soluble alkali, solid alkali, non-alkli and the total SO2 capacity of Bayer red mud, mg/g, respectively.
2.3 Characterization of experimental samples
The spent Bayer red mud was obtained by filtration after the outlet SO2 concentration reached to saturation in the process of the desulfurization. After that, the raw and the spent Bayer red mud were dried at 65 ��C in an oven for 48 h. Then, the dried samples were crushed evenly and sieved to less than 160 mesh size. The main elements of the samples were measured by using a XRF (Axios Max, Holland). The crystalline forms of the samples were performed by using a powder XRD (Riguka D/Mas 2200 diffractometer, Japan) with Cu K�� radiation at 40 kV and 40 mA. The element valences of the samples were detected by using a XPS (PHI 5000 Versaprobe, Japan) with Al K�� radiation and the C 1s peak was calibrated at 284.6 eV as the standard. The scanning electron microscope (VEDE 3 SBU, Czech) was applied to observing the microstructure of the samples. The BET surface areas of the samples were calculated by using the N2 adsorption/desorption (Micromeritics Tristar II 3020, USA) at �C196 ��C after the samples were degassed at 300 ��C for 3 h.
3 Results and discussions
3.1 Thermodynamic study
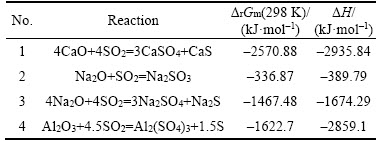
As described in the result of XRF and XRD, the main components of Bayer red mud are Fe2O3, Al2O3, CaO, SiO2, Na2O and TiO2. The basic thermodynamic parameters, such as standard free energy change (��G��) and standard enthalpy change (��H��) of the main components in the system were obtained from the related literature. Thereafter, the thermodynamic calculations including standard free energy change (��G��) and standard enthalpy change (��H��) were carried out to evaluate the thermodynamic feasibility of flue gas desulfurization by using Bayer red mud under the temperature of 298 K. The corresponding results are shown in Table 2. As Table 2 shows, the ��rGm(298 K) and ��H are all <0, indicating that these above reactions are spontaneous and exothermic [21]. Moreover, the values of ��rGm (298 K) imply that the order of reaction in the system follows the priority of calcium oxide.
Table 2 Thermodynamic results of flue gas desulfurization in studied system
3.2 Optimization of orthogonal experiment
Table 3 lists the results of orthogonal experiment, where 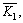
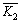 and
and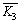 represent the average desulfurization efficiency at each factor level, reflecting the effect of each level of the same factor on the experiment results; R denotes the range, reflecting the influence of various factors on the results, of which the large range indicates that the level change of this factor has a great influence on the experiment results, and vice versa [22]. As Table 3 shows, L/S was the prominent factor, followed by the inlet SO2 concentration and the temperature was the least among them. The optimum desulfurization condition obtained by the orthogonal experiment is as follows: liquid�Csolid ratio of 20:1, the temperature of 35 ��C and the SO2 concentration of 1000 mg/m3 under the gas flow 1.5 L/min.
represent the average desulfurization efficiency at each factor level, reflecting the effect of each level of the same factor on the experiment results; R denotes the range, reflecting the influence of various factors on the results, of which the large range indicates that the level change of this factor has a great influence on the experiment results, and vice versa [22]. As Table 3 shows, L/S was the prominent factor, followed by the inlet SO2 concentration and the temperature was the least among them. The optimum desulfurization condition obtained by the orthogonal experiment is as follows: liquid�Csolid ratio of 20:1, the temperature of 35 ��C and the SO2 concentration of 1000 mg/m3 under the gas flow 1.5 L/min.
Table 3 Results of orthogonal test of desulfurization by Bayer red mud
3.3 Effects of process parameters on desulfurization
3.3.1 Effect of L/S
Figure 2 shows the effect of L/S on the SO2 removal. As the liquid�Csolid ratio increases in the range of 10:1�C30:1, the desulfurization efficiency increases. When liquid�Csolid ratio increases from 10:1 to 15:1, the desulfurization efficiency increases significantly; while when the liquid�Csolid ratio is higher than 20:1, with the increase of liquid�Csolid ratio, the desulfurization efficiency is steady at approximately 86%.
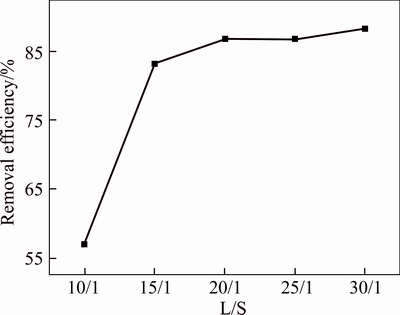
Figure 2 Effects of liquid�Csolid ratio on desulfurization efficiency (Inlet SO2 concentration, 1000 mg/m3; flue gas flow, 1.5 L/min; O2 concentration, 19.5%; mass of red mud, 2 g; reaction temperature, 25 ��C; reaction time, 4 h)
When the liquid�Csolid ratio is low, the slurry becomes thinner and the viscosity decreases with the increase of the liquid�Csolid ratio. According to the two-film theory, the decrease of viscosity is conducive to the increase of liquid film factor, and the alkaline components of red mud slurry are more likely to fully dissolve in the water, thus making SO2 be able to better react with the alkaline components in the red mud slurry, and then the desulfurization efficiency increases. When liquid�Csolid ratio is above 20:1, alkaline of red mud slurry has dissolved in water nearly completely, the increase of liquid�Csolid ratio will never increase the content of alkaline substances in the slurry; therefore, the desulfurization efficiency will no longer increase obviously. Moreover, the increase of liquid�Csolid ratio will increase the water and energy consumption. Thus, considering the economic factors and desulfurization efficiency, the liquid�Csolid ratio is selected as 20:1 in the desulfurization process.
3.3.2 Effect of absorption temperature
Figure 3 shows the effect of absorption temperature on the SO2 removal. As illustrated in Figure 3, the desulfurization efficiency of Bayer red mud slurry decreases with the increase of temperature. When the temperature is between 25 and 40 ��C, as the temperature increases, the desulfurization efficiency drops rapidly from 86.8% to 82%; When the temperature is above 40 ��C, the desulfurization efficiency drops slowly and is stable at around 80%. Previous thermodynamic calculations have shown that low temperature benefits the flue gas desulfurization with Bayer red mud. This result is different from the orthogonal experiment, which may be a consequence that low temperature is beneficial to the solubility of SO2 [23]. Thus, 25 ��C is chosen as the optimal absorption temperature.
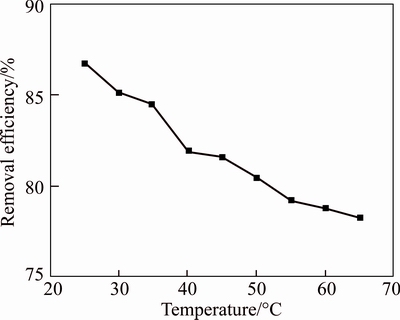
Figure 3 Effects of temperature on desulfurization efficiency (Inlet SO2 concentration, 1000 mg/m3; flue gas flow, 1.5 L/min; O2 concentration, 19.5%; volume of water, 40 mL; L/S, 20:1; reaction time, 4 h)
3.3.3 Effect of O2 concentration
Figure 4 displays the effects of O2 concentration on the desulfurization efficiency of red mud slurry. When the O2 concentration of flue gas increases from 0 to 7%, the desulfurization efficiency increases significantly. The reason is that the SO2 could generate H2SO3 and dissolve in water, the SO32�C is oxidized by O2 to form H2SO4 so as to accelerate the reaction process, thus promoting the desulfurization efficiency. When the O2 concentration is higher than 7%, the desulfurization efficiency of red mud slurry tends to be stable. In the actual situation, the O2 concentration of smelting flue gas can reach to 7% [24]; even some sections can reach 19.5%, which shows that O2 concentration is not a limiting factor for the flue gas desulfurization with Bayer red mud. Thus, the O2 concentration of the following experiments is controlled at 19.5% during the desulfurization process.
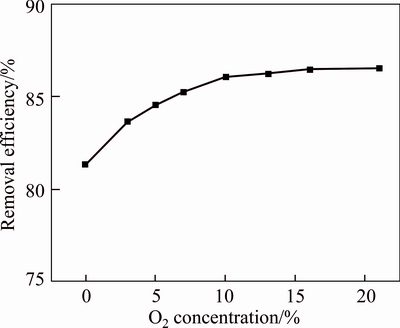
Figure 4 Effects of O2 concentration on desulfurization efficiency (Inlet SO2 concentration, 1000 mg/m3; flue gas flow, 1.5 L/min; volume of water, 40 mL; L/S, 20:1; reaction temperature, 25 ��C; reaction time, 4 h)
3.3.4 Effect of SO42�C and different components of Bayer red mud slurry
Figure 5 shows the desulfurization efficiency with absorption time with respect to water, the acid Bayer red mud slurry treated with H2SO4 or HCl, and raw Bayer red mud slurry. As the figure illustrates, the desulfurization efficiency of red mud slurry treated by H2SO4 solution is lower than that of red mud slurry treated by HCl. Specially, with the increase of absorption time, the desulfurization efficiency of acid Bayer red mud slurry treated by H2SO4 decreases continuously and it is less than 16.67% at 170 min. While the desulfurization efficiency with the red mud treated by HCl decreases slowly in a flat manner, the desulfurization efficiency still remains at 93.3% when the reaction time is 200 min, which may be interpreted by the following reasons. After the Bayer red mud is treated by H2SO4, the red mud slurry contains a large amount of SO42�C. The pre-reaction between the alkaline substances in the Bayer red mud and the sulfuric acid might be the reason why there is a decrease of alkaline substances content in the Bayer red mud, thus making the desulfurization efficiency become lower. Related research [25] shows that the increasing SO42�C and the generated hydroxyl Fe (II) ions would form spatial block effect, leading to the scenario that SO2 will no longer dissolve in the slurry and the reaction terminates, so that the desulfurization efficiency of red mud slurry is reduced. Thus, the excessive concentration of SO42�C in slurry is one of the main reasons that restricts the desulfurization.
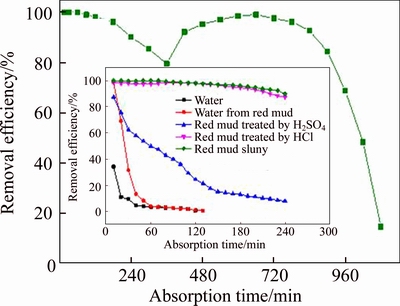
Figure 5 Effects of different components of Bayer red mud slurry on flue gas desulfurization (inlet SO2 concentration, 600 mg/m3; flue gas flow, 1.5 L/min; volume of water, 40 mL; L/S, 20:1; reaction temperature, 25 ��C)
Besides, compared with the results of flue gas desulfurization with Bayer red mud treated by HCl and H2SO4, it can be found that the treated red mud slurry could continue to remove SO2, so the other components except alkaline components of red mud slurry are expected to play a role in the desulfurization.
The contributions of different components,including water, soluble alkali, solid alkali and metal iron, in the Bayer red mud slurry to flue gas desulfurization are shown in Figure 6. As depicted in Figure 6, the contributions of different components to flue gas desulfurization are as following orders: solid alkali > soluble alkali > water, where solid alkali accounts for 79.65%, followed by soluble alkali of 1.67% and water is the least among them which accounts for 1.39%.
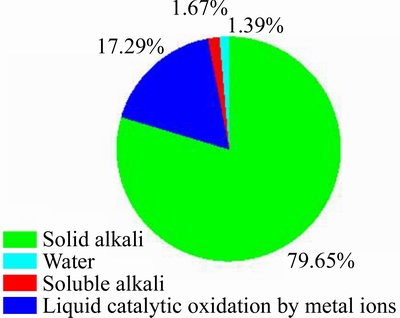
Figure 6 Contributions of different components of Bayer red mud slurry to flue gas desulfurization (Inlet SO2 concentration, 600 mg/m3; flue gas flow, 1.5 L/min; volume of water, 40 mL; L/S, 20:1; reaction temperature, 25 ��C)
3.4 Characterizations and reaction mechanisms
3.4.1 XRF analysis
Table 4 lists the XRF analysis of raw red mud. As shown in Table 4, the main components of Bayer red mud are Fe2O3, Al2O3, CaO, SiO2, Na2O and TiO2, which account for more than 88% of total components. Related results [26, 27] indicated that Al2O3, CaO, and Na2O could remove SO2.
Table 5 lists the XRF analysis of raw red mud and spent red mud. The content of S in the spent red mud increased from 0.379% to 13.517%, indicating that the desulfurization performance was better. The main elements including Fe, Al, Na and Ca in Bayer red mud decreased in the range of 1.241% to 8.026%, indicating the leaching of metal ions was occurred in the process. It should be mentioned that other elements such as Ni, Cu and Zn in the spent Bayer red mud are so low (<0.016 wt %) that the spent red mud could be disposed directly in the original way.
Table 4 XRF analysis of Bayer red mud

Table 5 XRF analysis of raw red mud and spent red mud (wt %)
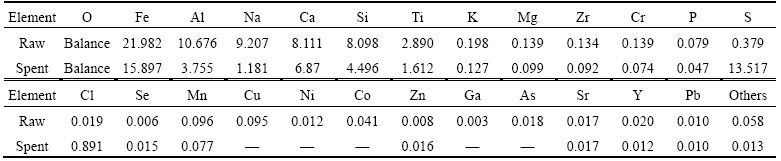
3.4.2 XRD analysis
Figure 7 depicts the XRD results of raw Bayer red mud. Alkaline substances such as hydrogarnet (3CaO��Al2O3��SiO2��4H2O), natrodavyne(3NaSiAlO4��
Na2CO3), sodium aluminosilicate hydrate (Na2O��
Al2O3��1.68SiO2��1.73H2O) and hematite (Fe2O3) exist in the Bayer red mud. Therefore, the Bayer red mud slurry exhibits high alkaline and could react with SO2.
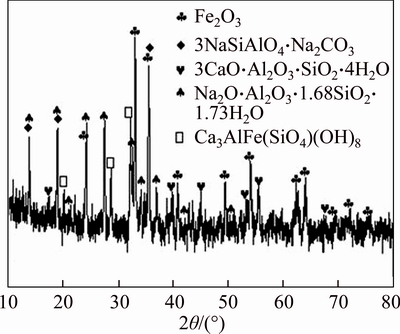
Figure 7 XRD pattern of raw Bayer red mud
Figure 8 shows the XRD patterns of the spent red mud samples after desulfurization. As shown in Figure 8(a), the solid alkaline components such as hydrogarnet and cancrinite have disappeared and the main products are CaSO4��2H2O and NaAl(SO4)2��6H2O in the XRD pattern after desulfurization, indicating that solid alkali has been consumed by acid. It is noticeably that the Fe2O3 still exists in the spent red mud, implying that Fe2O3 did not react in this system. Figure 8(b) shows the spectra of the spent Bay red mud in the absence of O2. There are dominating peaks ascribed to CaSO3��2H2O and some solid alkali still exists in the spent Bayer red mud, which is owing to the fact that O2 plays a vital role in flue gas desulfurization and the SO32�C could not be oxidized into SO42�C in the absence of O2.
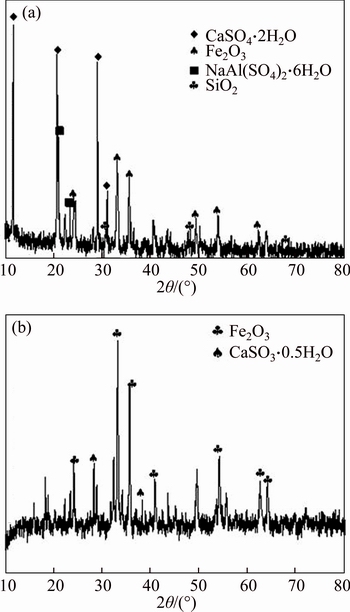
Figure 8 XRD patterns of spent Bayer red mud with O2 (a) and without O2(b)
3.4.3 XPS analysis
In order to verify the XRD results and discuss the mechanism of desulfurization, the XPS was carried out to determine the element states of the samples. Figure 9 displays the XPS spectra of the raw and spent Bayer red mud.
The results of XPS analysis for raw/spent Bayer red mud sample are shown in Figure 9(a). Results show that no obvious S peak was detected in the raw Bayer red mud, while the characteristic S 2p peak was identified after flue gas desulfurization, confirming that SO2 was absorbed in the red mud. Peak ascribed to Na was reduced, which is consistent with the XRF result.
Figure 9 XPS spectra of raw red mud and spent red mud for survey (a), O 1s (b), Fe 2p (c) and S 2p(d)
The results of XPS analysis of O 1s for raw/spent Bayer red mud sample are shown in Figure 9(b). The binding energy peak at 530.0 eV corresponds to the lattice oxygen. The binding energy peaks at 531.2 eV (or 531.9 eV) are assigned to the chemisorbed oxygen [28].
The results of XPS analysis of Fe 2p for raw/spent Bayer red mud sample are shown in Figure 9(c). The Fe 2p peaks can be split into three sub-peaks at 709.6 eV (or 711.0 eV), 713.4 eV (or 713.8 eV) and 723.5 eV (or 725.3 eV), which belong to Fe2O3 [29]. There is a shift of peak to high binding energy of iron in the spent red mud, indicating that Fe (II) was formed on the surface of red mud, which confirms the catalytic oxidation of iron ion.
Figure 9(d) shows the XPS analysis of S 2p for spent Bayer red mud sample. The S 2p peaks can be split into sub-peaks at 168.6 eV and 167.5 eV, which are ascribed to SO42�C and SO32�C [30].
3.4.4 SEM analysis
The SEM images of raw Bayer red mud, spent Bayer red mud with O2 and the spent Bayer red mud without O2 are shown in Figure 10. As observed in Figure 10(a), the raw red mud is mainly composed of dispersed and uniform particles with the size ranging from 2 to 14 ��m in diameter. Moreover, the surface of flocculent particles is rough, which is owing to the existing different minerals in the Bayer red mud [31]. Figures 10(c)�C(f) show the morphologic change after the desulfurization with O2 and in the absence of O2. The microcosmic morphology exhibits a significant difference. Schistose and layered particles are formed in the spent Bayer red mud with O2, the size of which is ranging from 2 to 16 ��m in diameter and the surface has increased. The possible reason is the new-formed material during the introduction of SO2 to the red mud with the condition of O2. For the spent red mud in the absence of O2, its structure is relatively loose compared with the raw red mud, flower-like and spherical particles with the size ranging from 1 to 6 ��m in diameter are the main form, which might attribute to SO2. SO2 makes block components disappear and then the red mud particles become smaller.
3.4.5 BET analysis
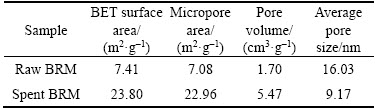
Figure 11 shows the N2 adsorption�Cdesorption isotherm of the raw and the spent red mud. Results indicate the isotherms all belong to the IV type adsorption isotherm based on the hysteresis loop, which is in accordance with the previous findings [8, 32]. Table 6 shows that the BET specific surface area increased from 7.41 m2/g for the raw Bayer red mud to 23.80 m2/g for the spent Bayer red mud, which is due to the involvement of the solid component of the red mud in the desulfurization process. The result is highly coincident with the results of SEM.

Figure 10 SEM spectra of raw red mud (a, b), spent red mud with O2 (c, d), and without O2 (e, f)
3.5 Reaction mechanism of flue gas desulfurization
1) Based on the above experiment data and results of characterization, the reaction mechanism of flue gas desulfurization by using Bayer red mud can be described as follows: 1) The gaseous SO2 is transferred from the flue gas to the Bayer red mud slurry to form the HSO3�C and SO3�C. The reaction can be described by Eq. (4) [33].
SO2+H2O H++HSO3�C
H++HSO3�C 2H++SO32�C (4)
2H++SO32�C (4)
2) The alkaline components consist of soluble alkali including NaOH and NaCO3, and insoluble alkali such as sodium aluminosilicate hydrate [17]. The soluble alkali can react with SO2; Besides, insoluble alkali can react with SO32�C to form CaSO3, Na2SO3 and Al2(SO4)3, thereafter CaSO3 and Na2SO3 can be oxidized by the O2 from the flue gas. Corresponding reactions can be expressed by the following Eqs. (5)�C(11) [17, 34].
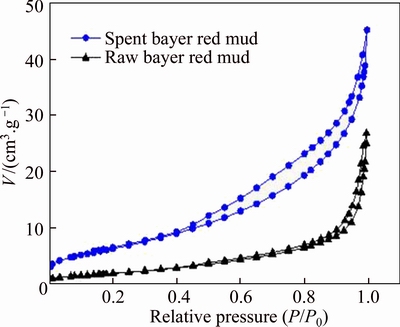
Figure 11 N2 adsorption�Cdesorption isotherms of red mud
Table 6 BET analysis of raw and spent Bayer red mud
2NaAlO2+SO2+3H2O=Na2SO3+2Al(OH)3 (5)
4CaO+4SO2=3CaSO3+CaS (6)
Na2O+SO2=Na2SO3 (7)
4Na2O+4SO2=3Na2SO3+Na2S (8)
Al2O3+4.5SO2=Al2(SO4)3+1.5S (9)
CaSO3+0.5O2+2H2O=CaSO4��2H2O (10)
Na2SO3+0.5O2+2H2O=Na2SO4��2H2O (11)
3) After the consumption of alkali, the pH of the slurry reduces and Fe2+ is leached from the red mud, then the dissolved Fe2+ promotes the absorption of SO2 by liquid-phase catalytic oxidation of metal ions, and Fe3+ plays a vital role in the oxidation property of the slurry. Reactions can be described by Eqs. (12) and (13) [35].
2Fe2++SO2+O2=2Fe3++SO42�C (12)
2Fe3++SO2+2H2O=2Fe2++SO42�C+4H+ (13)
4 Conclusions
In this work, absorbents were prepared using Bayer red mud and water. These absorbents were applied to removing SO2 from flue gas. The results indicate that L/S was the prominent factor, followed by the inlet SO2 concentration and the temperature was the least among them. The optimum desulfurization condition was as follows: liquid�C solid ratio, the temperature and the concentration of SO2 were 20:1, 25 ��C and 1000 mg/m3, respectively, under the gas flow of 1.5 L/min. The desulfurization efficiency significantly increased with the increase of O2 concentration ranging from 0 to 7%, and was not significantly influenced when O2 concentration was more than 7%. The accumulation of SO42�C inhibited the desulfurization efficiency. The contributions of alkali and metal ions liquid catalytic oxidation have played a vital role in flue gas desulfurization, which accounts for 98.61%. These results indicate that the Bayer red mud has great application potential for flue gas desulfurization.
References
[1] RAGHUNATH C V, MONDAL M K. Experimental scale multi component absorption of SO2 and NO by NH3/NaClO scrubbing [J]. Chemical Engineering Journal, 2017, 314: 537�C547. DOI: 10.1016/j.cej.2016.12.011.
[2] YU Qing-chun, DENG Yong, WANG Fei, FENG Yue-bin, YANG Bin, XU Bao-qiang, LIU Da-chun. Comparison of desulfurization kinetics of copper oxide sorbent [J]. Journal of Central South University, 2015, 22: 2902�C2908. DOI: 10.1007/s11771-015-2824-z.
[3] WU Xue-cheng, WU Kai, ZHANG Yong-xin, HONG Qiao-qiao, ZHENG Cheng-hang, GAO Xiang, CEN Ke-fa. Comparative life cycle assessment and economic analysis of typical flue-gas cleaning processes of coal-fired power plants in China [J]. Journal of Cleaner Production, 2017, 142: 3236�C3242. DOI: 10.1016/j.jclepro.2016.10.146.
[4] LIU Wan-chao, CHEN Xiang-qing, LI Wang-xing, YU Yan-fen, YAN Kun. Environmental assessment, management and utilization of red mud in China [J]. Journal of Cleaner Production, 2014, 84: 606�C610. DOI: 10.1016/j.jclepro.2014. 06.080.
[5] XUE Sheng-guo, WU Yu-jun, LI Yi-wei, KONG Xiang-feng, ZHU Feng, WILLIAM Hartley, LI Xiao-fei, YE Yu-zhen. Industrial wastes applications for alkalinity regulation in bauxite residue: A comprehensive review [J]. Journal of Central South University, 2019, 26(2): 268�C288.
[6] ZHU Feng, HOU Jing-tao, XUE Sheng-guo, WU Chuan, WANG Qiong-li, HARTLEY W. Vermicompost and gypsum amendments improve aggregate formation in bauxite residue [J]. Land Degradation and Development, 2017, 28(7): 2109�C2120. DOI: 10.1002/ldr.2737.
[7] YANG Wei, HUSSAIN A, ZHANG Jun, LIU Yang-xian. Removal of elemental mercury from flue gas using red mud impregnated by KBr and KI reagent [J]. Chemical Engineering Journal, 2018, 341: 483�C494. DOI: 10.1016/ j.cej.2018.02.023.
[8] WANG Meng, HU Hui-ping, LIU Jin-wei, CHEN Qi-yuan. Negative effect of dissolved organic compounds on settling behavior of synthetic monominerals in red mud [J]. Journal of Central South University, 2016, 23(7): 1591�C1602. DOI: 10.1016/S1003-6326(17)60049-9.
[9] LIAO Jia-xin, JIANG Jun, XUE Sheng-guo, CHENG Qing-yu, WU Hao, RAJENDRAN M, HARTLEY W, HUANG Long-bin. A novel acid-producing fungus isolated from bauxite residue: the potential to reduce the alkalinity [J]. Geomicrobiology Journal, 2018, 35(10): 840�C847. DOI: 10.1080/01490451.2018.1479807.
[10] KONG Xiang-feng, JIANG Xing-xing, XUE Sheng-guo, HUANG Ling, HARTLEY W, WU Chuan, LI Xiao-fei. Migration and distribution of saline ions in bauxite residue during water leaching [J]. Transactions of Nonferrous Metals Society of China, 2018, 28(3): 534�C541. DOI: 10.1016/ S1003-6326(18)64686-2.
[11] KONG Xiang-feng, TIAN Tao, XUE Sheng-guo, HARTLEY W, HUANG Long-bin, WU Chuan, LI Chu-xuan. Development of alkaline electrochemical characteristics demonstrates soil formation in bauxite residue undergoing natural rehabilitation [J]. Land Degradation and Development, 2018, 29(1): 58�C67. DOI: 10.1002/ldr.2836.
[12] XUE Sheng-guo, ZHU Feng, KONG Xiang-feng, WU Chuan, HUANG Ling, HUANG Nan, HARTLEY W. A review of the characterization and revegetation of bauxite residues (Red mud) [J]. Environmental Science and Pollution Research, 2016, 23(2): 1120�C1132. DOI: 10.1007/s11356- 015-4558-8.
[13] XUE Sheng-guo, YE Yu-zhen, ZHU Feng, WANG Qiong-li, JIANG Jun, HARTLEY W. Changes in distribution and microstructure of bauxite residue aggregates following amendments addition [J]. Journal of Environmental Sciences, 2019, 78: 276�C286. DOI: 10.1016/j.jes.2018.10.010.
[14] XUE Sheng-guo, LI Meng, JIANG Jun, MILLAR G J, LI Chu-xuan, KONG Xiang-feng. Phosphogypsum stabilization of bauxite residue: Conversion of its alkaline characteristics [J]. Journal of Environmental Sciences, 2019, 77: 1�C10. DOI: 10.1016/j.jes.2018.05.016.
[15] ZHU Feng, ZHOU Jia-yi, XUE Sheng-guo, HARTLEY W, WU Chuan, GUO Ying. Aging of bauxite residue in association of regeneration: A comparison of methods to determine aggregate stability & erosion resistance [J]. Ecological Engineering, 2016, 92: 47�C54. DOI: 10.1016/ j.ecoleng.2016.03.025.
[16] ZHU Feng, CHENG Qing-yu, XUE Sheng-guo, LI Chu-xuan, HARTLEY W, WU Chuan, TIAN Tao. Influence of natural regeneration on fractal features of residue microaggregates in bauxite residue disposal areas [J]. Land Degradation and Development, 2018, 29(1): 138�C149. DOI: 10.1002/ ldr.2848.
[17] ZHU Feng, LIAO Jia-xin, XUE Sheng-guo, HARTLEY W, ZOU Qi, WU Hao. Evaluation of aggregate microstructures following natural regeneration in bauxite residue as characterized by synchrotron-based X-ray micro-computed tomography [J]. Science of the Total Environment, 2016, 573: 155�C163. DOI: 10.1016/j.scitotenv.2016.08.108.
[18] HAMID S, BAE S, LEE W. Novel bimetallic catalyst supported by red mud for enhanced nitrate reduction [J]. Chemical Engineering Journal, 2018, 348: 877�C887. DOI: 10.1016/j.cej.2018.05.016.
[19] XUE Sheng-guo, KONG Xiang-feng, ZHU Feng, HARTLEY W, LI Xiao-fei, LI Yi-wei. Proposal for management and alkalinity transformation of bauxite residue in China [J]. Environmental Science and Pollution Research, 2016, 23(13): 12822�C34. DOI: 10.1007/s11356-016-6478-7.
[20] ELISABETTA F, ANTONIO L, MURA G. Sulfur dioxide absorption in a bubbling reactor with suspensions of Bayer red mud [J]. Journal of Industrial and Engineering Chemistry, 2007, 46: 6770�C6776. DOI: 10.1021/ie0616904.
[21] ZHOU Yang, LI Cai-ting, FAN Chun-zhen, FU Meng-fan, TAO Li, YU Min-ge, ZHANG Meng-ying. Wet removal of sulfur dioxide and nitrogen oxides from simulated flue gas by Ca(ClO)2 solution [J]. Environmental Progress & Sustainable Energy, 2015, 34(6): 1586�C1595. DOI: 10.1002/ ep.12153.
[22] LIU Hong-ying, HUANG Tian, JIANG Xia, JIANG Wen-ju. Preparation and desulfurization performance of pyrolusite modified activated coke [J]. Environmental Progress & Sustainable Energy, 2016, 35(6): 1679�C1686. DOI: 10.1002/ ep.12416.
[23] KARAGOZ O, COPUR M, KOCAKERIM M M. Kinetic analysis of retention of SO2 using waste ulexite ore in an aqueous medium [J]. Journal of Hazardous Materials, 2018, 353: 214�C226. DOI: 10.1016/j.jhazmat.2018.04.006.
[24] KONG Ling-nan, ZOU Si-jie, MEI Jian, GENG Yang, ZHAO Hui, YANG Shi-jian. Outstanding resistance of H2S-Modified Cu/TiO2 to SO2 for capturing gaseous Hg(0) from nonferrous metal smelting flue gas: Performance and reaction mechanism [J]. Environmental Science & Technology, 2018, 52(17): 10003�C10010. DOI: 10.1021/acs. est.8b03484.
[25] GIL A F, SALGADO L, GALICIA L, GONZ L I. Predominance-zone diagrams of Fe(III) and Fe(II) sulfate complexes in acidic media. Voltammetric and spectrophotometric studies [J]. Talanta, 1995, 42(3): 407�C414. DOI: 10.1016/0039-9140(95)01424-A.
[26] ROKNI E, LEVENDIS Y A. Utilization of a high-alkali lignite coal ash for SO2 capture in power generation [J]. Journal of Energy Engineering, 2017, 143(4): 04016067. DOI: 10.1061/(ASCE)EY.1943-7897.0000423.
[27] PANG Hao, LI Xue-ping, JIN Su-jing, WEI Peng, LI Hui-ping. Analysis of alumina red mud wet flue gas desulfurization (FGD) technology [J]. Advanced Materials Research, 2013, 634�C638: 198�C203. DOI: 10.4028/www. scientific.net/AMR.634-638.198.
[28] ZHAO Ling-kui, LI Cai-ting, ZHANG Jie, ZHANG Xu-nan, ZHAN Fu-man, MA Jin-feng, XIE Yin-e, ZENG Guang-ming. Promotional effect of CeO2 modified support on V2O5�CWO3/TiO2 catalyst for elemental mercury oxidation in simulated coal-fired flue gas [J]. Fuel, 2015, 153: 361�C369. DOI: 10.1016/j.fuel.2015.03.001.
[29] YAMASHITA T, HAYES P. Analysis of XPS spectra of Fe2+ and Fe3+ ions in oxide materials [J]. Applied Surface Science, 2008, 254(8): 2441�C2449. DOI: 10.1016/j.apsusc.2007. 09.063.
[30] HAO Run-long, ZHANG Yao-yu, WANG Zhao-yue, LI Yuan-peng, YUAN Bo, MAO Xing-zhou, ZHAO Yi. An advanced wet method for simultaneous removal of SO2 and NO from coal-fired flue gas by utilizing a complex absorbent [J]. Chemical Engineering Journal, 2017, 307: 562�C571. DOI: 10.1016/j.cej.2016.08.103.
[31] DEIHIMI N, IRANNAJAD M, REZAI B. Characterization studies of red mud modification processes as adsorbent for enhancing ferricyanide removal [J]. Journal of Environmental Management, 2018, 206: 266�C275. DOI: 10.1016/j.jenvman.2017.10.037.
[32] MASTALERZ M, HE Li-lin, MELNICHENKO Y B, RUPP J A. Porosity of coal and shale: insights from gas adsorption and SANS/USANS techniques [J]. Energy & Fuels, 2012, 26(8): 5109�C5120. DOI: 10.1021/ef300735t.
[33] CHEN Min, DENG Xian-he, HE Fei-qiang. Removal of SO2 from flue gas using basic aluminum sulfate solution with the byproduct oxidation inhibition by ethylene glycol [J]. Energy & Fuels, 2016, 30: 1183�C1191. DOI: 10.1021/acs. energyfuels.5b02411.
[34] BAHRABADI-JOVEIN I, SEDDIGHI S, BASHTANI J. Sulfur dioxide removal using hydrogen peroxide in sodium- and calcium-based absorbers [J]. Energy & Fuels, 2017, 31(12): 14007�C14017. DOI: 10.1021/acs.energyfuels. 7b02722.
[35] SUN Pei-shi, NING Ping, SONG Wen-biao. Liquid-phase catalytic oxidation of smelting-gases containing SO2 in low concentration [J]. Journal of Cleaner Production, 1998, 6: 323�C327. DOI: 10.1016/s0959-6526(98)00020-1.
(Edited by YANG Hua)
���ĵ���
�ݶ�������������������Ӱ�����������
ժҪ�������о����ɰݶ���������ˮ�Ʊ����ռ���Ӧ�������������о��˹�Һ�ȡ������¶ȡ����SO2Ũ�ȡ�O2������SO42�C�Ͱݶ�������������ֶ����������Ӱ�죬��������������Ļ��ơ����������Щ�����й�Һ��������Ҫ�����أ���������SO2Ũ�ȣ��¶ȵ�Ӱ����С������������Ϊ1.5 L/minʱ����ѵķ�Ӧ����Ϊ��Һ�̱�20:1�������¶�25 ��C�����SO2Ũ��1000 mg/m3����O2��������7%��O2����������Ч�ʵ�Ӱ���С��SO42�C���ۻ�����������Ч�ʡ���Һ���ա�Һ���������ռ������������ܹ���98.61%��
�ؼ��ʣ��ݶ������ࣻ��������ʪʽ��������������
Foundation item: Project(2017YFC0210500) supported by the National Key Technology R&D Program of China; Project(2017ACA092) supported by the Major Projects of Technical Innovation in Hubei Province, China
Received date: 2018-10-25; Accepted date: 2018-12-12
Corresponding author: LI Bin, PhD, Associate Professor; Tel: +86-13529198525; E-mail: libin@kmust.edu.cn; ORCID: 0000-0002- 3588-1296

