
Synthesis of Y2O3:Eu3+ phosphors by surface diffusion and their photoluminescence properties
ZHANG Ming(张 明), LI Xin-hai(李新海), WANG Zhi-xing(王志兴), HU Qi-yang(胡启阳), GUO Hua-jun(郭华军)
School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China
Received 21 October 2008; accepted 13 March 2009
Abstract: Y2O3:Eu3+ phosphors were synthesized by the surface diffusion method (SDM). X-ray diffractometry (XRD), scanning electron microscopy (SEM), and energy dispersive spectrometry (EDS) were used to characterize the structure, morphology and component of Y2O3:Eu3+ phosphors. The photoluminescent (PL) properties were also investigated. The results reveal that the PL intensity of Y2O3:Eu3+ phosphors prepared by the surface diffusion method (SDM) is much higher than that prepared by homogeneous co-precipitation. The luminescence efficiency of the sample (Y0.997, Eu0.003)2O3 prepared by the SDM is almost 1.9 times that by homogeneous co-precipitation. The concentration of Eu3+ in the phosphor Y2O3:Eu3+ prepared by the surface diffusion can be reduced greatly owing to the activator, Eu3+ ions, distributing mainly in the outer layer of the phosphors where the photon generation process occurs.
Key words: phosphor; rare earth; surface diffusion method; photoluminescence properties
1 Introduction
Rare earth luminescent materials have considerable application in devices involving fluorescent lighting devices and display devices, such as tricolor lamps, cathode ray tubes (CRTs), liquid crystal displays (LCDs), field emission displays (FEDs), and plasma display panels (PDPs)[1-3]. Among them, europium-doped yttrium oxide (Y2O3:Eu3+) fulfills all the requirements for a good red-emitting phosphor because of its excellent luminescence efficiency, color purity and stability. It can be easily excited by 254 nm from mercury radiation and its sharp emission is located at 613 nm and all other emission lines are weak[4].
The conventional preparation method of Y2O3:Eu3+ phosphor is based on solid state reaction, in which all materials are mixed by grinding mill and then undergo a high temperature reaction[5]. It has been reported that the optical properties of luminescent materials can be greatly affected by the particle size and morphology. This motivated the development of other alternative preparation techniques[6-10], such as combustion synthesis, hydrothermal process, microwave irradiation method, co-precipitation method, spray pyrolysis and sol-gel route. Recently, it has been studied that the penetration depth of electrons increases exponentially with increasing the accelerating voltage. When the accelerating voltage was less than 1 kV, the penetration depth was less than several nanometers so that most of electrons were consumed in the surface layer[11-12]. As a result, only a thin outer layer of the micron-sized commercial phosphors took part in the photon generation process. With nanoparticles, the penetration by low-voltage electrons was sufficient for efficient material utilization[13]. However, the activators in the center of the phosphor prepared by these methods do not take part in the photon generation process because the energy of 254 nm radiation is too low to reach several microns. At the same time, the rare earth oxides are very expensive, limiting their application to a great extent. It has been found that coating relatively inexpensive cores with Y2O3:Eu3+ shell will reduce the amount of expensive rare earth phosphors used[14-15]. However, there is a problem that the intensity of these core-shell particles is lower than that of bulk Y2O3:Eu3+.
Our strategy to solve these problems has focused on materials with higher luminescent intensity and lower cost. In this work, we developed a simple and economical way to embed the activator Eu3+ into the outer surface layer of the bulk Y2O3; and surface diffusion method (SDM), in order to save the cost of the phosphors, was used.
2 Experimental
2.1 Materials
Yttrium oxide (Y2O3, 99.99%) and europium oxide (Eu2O3, 99.999%) were obtained from Hunan Research Institute of Rare Earth (Changsha, China). NH4HCO3 and HNO3 were of analytical grade and used without further purification.
2.2 Sample preparation
Y2O3:Eu phosphors were prepared by surface diffusion and homogeneous co-precipitation. Surface diffusion method was follows. Stoichiometric amounts of Eu2O3 was firstly dissolved in nitric acid and evaporated to dryness, then dissolved in distilled water to form 0.02 mol/L europium nitrate solution. NH4HCO3 was dissolved in distilled water to form 0.07 mol/L solution. Secondly, 0.02 mol Y2O3 was ultrasonically dispersed in 200 mL distilled water for 30 min, then Eu(NO3)3 and NH4HCO3 solutions were slowly dripped into a continuously agitated Y2O3 suspension solution, simultaneously. The temperature of the solution was stabilized at 50 ℃ in a water bath. Soon, the precipitation was achieved on the Y2O3 surface. The precursor precipitate was filtered, washed with distilled water several times and dried at 80 ℃ for 24 h. After that, the precursor was ground and heated in air at 1 200 ℃ for 3 h. The resulting calcined Y2O3:Eu3+ samples were designated as SDM-n, where n stands for the concentration of Eu3+. For example, SDM-3 means that the concentration of Eu3+ is 0.3% (molar fraction).
For comparison, Y2O3:Eu3+ was prepared by traditional homogeneous co-precipitation method. The samples were designated as HCP-n, where n stands for the concentration of Eu3+. For example, HCP-3 means that the concentration of Eu3+ is 0.3% (molar fraction).
2.3 Characterization
X-ray diffraction (XRD) measurements of powder samples were carried out on a Rigaku diffractometer equipped with Cu Kα at 40 kV and 300 mA. Scanning electron microscope (SEM) and energy dispersive spectrometer (EDS) were used to characterize the morphology and the component of particles by a Hitachi S530 spectrometer. The excitation and emission spectra of the samples were taken on a Hitachi F-4500 fluorescence spectrophotometer using a Xe lamp as the excitation source. All spectra were measured at room temperature.
3 Results and discussion
3.1 Luminescence properties
The excitation and emission spectra of (Y0.997, Eu0.003)2O3, synthesized by homogeneous co- precipitation (HCP-3) and surface diffusion (SDM-3) are shown in Fig.1. The excitation spectrum (Fig.1(a)) consists of a broad band with the maximum at 242 nm due to the charge transfer (CT) band, which relates to the electronic transition from the 2p orbital of O2- to the 4f orbital of Eu3+. In Fig.1(b), the emission spectra of the two samples are composed of 5D0→7FJ(J=0, 1, 2, 3, 4) emission line of Eu3+, dominated by the hypersensitive red emission 5D0→7F2 (forced) electric dipole transition of Eu3+ at 609 nm. All the other magnetic dipole transitions originating from the 5D0 state to 7DJ (J=0, 1, 3, 4) level could also be observed, as labeled in Fig.1. Fig.1 demonstrates that the luminescent intensities of the two samples change greatly. The luminescent intensity in the SDM-3 increases remarkably over that in the HCP-3, for either the CT or the f-f inner-shell excitations. The consistency of the phenomenon was verified with different sets of such phosphor prepared for different times. To have a quantitative idea of the enhancement, the integrated luminescence efficiency in the wave length region of 550-640 nm, in each case, was determined by calculating the area under the curve. The ratio of the integrated luminescence efficiency of the two samples (SDM-3 to HCP-3, 27947.5?14725.4=1.9) shows that the luminescence efficiency of the SDM-3 sample is almost 1.9 times that of the HCP-3 sample.
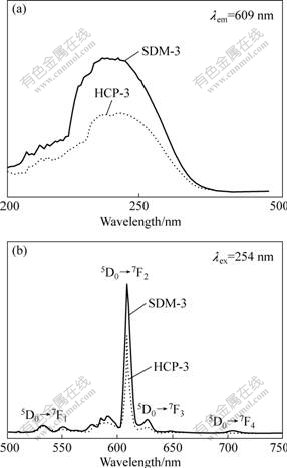
Fig.1 Photoluminescence excitation (a) and emission (b) spectra of SDM-3 and HCP-3
To investigate the PL behaviors of Y2O3:Eu3+, the electron penetration depth into the phosphors should be considered. According to Feldman’s equation, the penetration depth of an electron will increase exponentially with increasing excitation energies[16]. The penetration depth of an electron is given as follows:
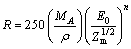 (1)
(1)
where R is the penetration depth of an electron; MA is molecular mass; ρ is bulk density; Zm is mean atomic number; n=1.2/(1-0.29lgZm); and E0 is excitation energy. JUNG et al[12] suggested that the penetration depth of a electron in (Y, Zn)2O3:Eu3+ phosphor increases exponentially with increasing the accelerating voltage. For Y2O2S:Tb, Zn2SiO4:Mn and La2O2S:Tb phosphors, GUTALER[17] calculated the penetration depth to be 5 nm at a screen potential of 300 V.
According the following equation, the energy of a photon of 254 nm UV is 4.88 eV:
E=hc/λ (2)
where h is Planck’s constant; c is the velocity of light. The penetration depth of the photon is within several nanometers. It can be deduced that only the Eu3+ activator ions in the outer layer of micro-sized Y2O3:Eu3+ take part in the photon generation process, and most of the Eu3+ ions in the inner layer do not. According to the phenomenon that the PL intensity of SDM-3 increases greatly over that of HCP-3, we can deduce that the concentration of the Eu3+ ions in the outer layer of SDM-3 is more than that of HCP-3 because the PL intensity greatly depends on the concentration of activator ions.
3.2 Electron microscopic studies
Fig.2 shows the scanning electron micrographs of the Y2O3 and SDM-3. Obviously, the surface of Y2O3 is smooth and clear. After being coated, the surface of SDM-3 is covered with a discontinuous layer.

Fig.2 SEM photographs of Y2O3 (a) and SDM-3 prepared by surface diffusion method (b)
X-ray energy dispersive spectrum performed on the particles in Fig.3 suggests the existence of Y and O, as well as Eu. The Eu element was not detected clearly due to its low concentration.
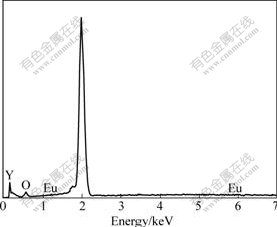
Fig.3 EDS spectrum of SDM-3 prepared by surface diffusion method
3.3 X-ray diffraction patterns
In order to assess the crystal lattice of each phosphor, XRD was used. Fig.4 and Table 1 show respectively the XRD patterns and the crystal cell parameters of the (Y1-xEux)2O3 synthesized by homogeneous co-precipitation (HCP) and surface diffusion method (SDM). Both samples have pure cubic Y2O3 phase with high crystallinity.
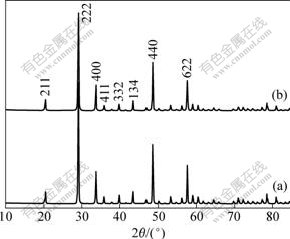
Fig.4 XRD patterns of SDM-3 (a) and HCP-3 (b)
Table 1 Cell parameters of (Y1-xEux)2O3
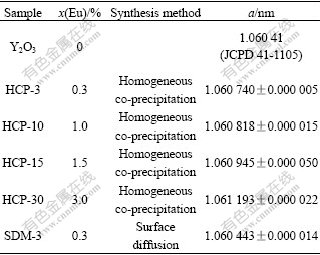
As shown in Table 1, with increase in the concentration of Eu3+ from 0 to 3%, the lattice constant of the phosphors increases. Since the ionic radius of europium ion (0.095 nm) is larger than that of yttrium (0.09 nm), the substitution of Eu in cation sites takes place. However, the lattice constant of the sample SDM-3 has a special phenomenon. It is somewhat smaller than that of HCP-3, which has the same initial concentration of Eu3+ ion, and is close to that of Y2O3, in which the concentration of Eu3+ ion is 0. This indicates that the concentration of Eu3+ ion in the inner parts of the particles is small.
4 Conclusions
1) Cubic crystalline Y2O3:Eu3+ was successfully prepared using a novel approach, surface diffusion method. The results of the XRD, SEM and EDS investigation show that Eu3+ ions exist in the outer layer of the phosphors.
2) Compared with the traditional homogeneous co-precipitation method, the phosphors prepared by surface diffusion method show excellent PL properties. In the wave length region of 550-640 nm, the luminescence efficiency of the SDM-3 sample is almost 1.9 times that of the HCP-3 sample.
3) The concentration of Eu3+ in the phosphor Y2O3:Eu3+ prepared by the surface diffusion method can be reduced greatly owing to the activator, Eu3+ ions, distributing mainly in the outer layer of the phosphors where the photon generation process occurs. It is an efficient way to save the cost of the phosphors while the luminescent properties are retained.
References
[1] JACK S, ROBERT W, ANTHONY L, TERRY G I, GEORGE R F. Low-voltage cathodoluminescent red emitting phosphors for field emission displays [J]. Journal of Luminescence, 2007, 122/123: 562-566.
[2] NGUYEN V, TRAN K A, GYYU-CHUI Y, STREK W. Photoluminescence and cathodoluminescence properties of Y2O3:Eu nanophosphors prepared by combustion synthesis [J]. Journal of Luminescence, 2007, 122/123: 776-779.
[3] ZHANG Ming, LI Xin-hai, HU Qi-yang, WANG Zhi-xing, GUO Hua-jun. Preparation methods of novel structural Y2O3:Eu3+ [J]. Rare Metal Materials and Engineering, 2008, 37(1): 2065-2068. (in Chinese)
[4] BLASSE G, GRABMETER B C. Luminescent materials [M]. Berlin: Springer-Verlag, 1994: 116-117.
[5] DEBNATH R, NAYAK A, GHOSH A. On the enhancement of luminescence efficiency of Y2O3:Eu3+ red phosphor by incorporating (Al3+, B3+) in the host lattice [J]. Chemical Physics Letters, 2007, 444: 324-327.
[6] TAKEDA T, KOSHIBA D, KIKKAWA S. Gel combustion synthesis of fine crystalline (Y0.95Eu0.05)2O3 phosphor in presence of lithium flex [J]. Journal of Alloys and Compounds, 2006, 408/412: 879-882.
[7] YAO G, SU L B, XU X D, XU J. Eu:Y2O3 nano-phosphor prepared by novel energy-saving solution combustion method [J]. Journal of Alloys and Compounds, 2008, 462: 381-385.
[8] HUANG Yan, YE Hong-qi, ZHUANG Wei-dong, HU Yun-sheng, ZHAO Chun-lei, LI Cui, GUO Song-xia. Preparation of Y2O3:Eu3+ phosphor by molten salt assisted method [J]. Transactions of Nonferrous Metals Society of China, 2007, 17(3): 644-648.
[9] SI Wei, JIANG Da, GAO Hong, ZHAI Yu-chun. Preparation and photoluminescence of nanocrystal Eu0.12Y1.88-xMxO3-d (M-Mg, Ca) [J]. Transactions of Nonferrous Metals Society of China, 2008, 18(5): 1879-1884.
[10] JOFFIN N, CAILLIER B, GARCIA A, GUILLOT P, GALY J, FERNANDES A, MAURICOT R, DEXPERT-GHYS J. Phosphor powders elaborated by spray-pyrolysis: Characterizations and possible applications [J]. Optical Materials, 2006, 28: 597-601.
[11] SHIN S H, KANG J H, JEON D Y, CHOI S H, LEE S H, YOU Y C, ZANG D S. Cathodoluminescence change of Y2O3:Eu phosphors by incorporation of Zn ions [J]. Solid State Communications, 2005, 135: 30-33.
[12] JUNG K Y, HAN K H, KO Y S. Cathodoluminescence characteristics of particles and film of (Y,Zn)2O3:Eu phosphor prepared by spray pyrolysis [J]. Journal of Luminescence, 2007, 127: 391-396.
[13] MCKITTRICK J, SHEA L E, BACALSKI C F, BOSZE E J. The influence of processing parameters on luminescent oxides produced by combustion synthesis [J]. Displays, 1999, 19: 169-172.
[14] FENG H J, CHEN Y, TANG F Q, REN J. Synthesis and characterization of monodispersed SiO2/Y2O3:Eu3+ core-shell submicrospheres [J]. Materials Letters, 2006, 60: 737-740.
[15] WANG H, LIN C K, LIU X M, LIN J. Monodisperse spherical core-shell-structured phosphors obtained by functionalization of silica spheres with Y2O3:Eu3+ layers for field emission displays [J]. Applied Physics Letters, 2005, 87: 181907.
[16] FELDMAN C. Range of 1-10 keV electrons in solids[J]. Phys Rev, 1960, 117: 455-459.
[17] GUTALER D E. The efficiency of Y2O2S:Tb under low energy electron bombardment [J]. J Electrochem Soc, 1979, 126: 571-573.
Foundation item: Project(2007CB613607) supported by the National Basic Research Program of China
Corresponding author: LI Xin-hai; Tel: +86-731-88836633; E-mail: xhli@mail.csu.edu.cn
DOI: 10.1016/S1003-6326(09)60106-0
(Edited by YANG Hua)