
Gas sensitivity of polyaniline/SnO2 hybrids to volatile organic compounds
GENG Li-na(������)
Department of Chemistry, Hebei Normal University, Shijiazhuang 050016, China
Received 10 August 2009; accepted 15 September 2009
Abstract: Polyaniline (PAn) was prepared by chemical oxidation polymerization and characterized by FT-IR. PAn/SnO2 materials with different mass fractions of PAn were prepared by mechanical mixing. Compared with single PAn and SnO2, the gas sensitivity of PAn/SnO2 materials to volatile organic compounds (VOCs, methanol, ethanol and acetone) was studied. The possible response mechanism of PAn/SnO2 materials to VOCs was also discussed. It is found that PAn/SnO2 materials have gas sensitivity to VOCs at 90 �� among the four operating temperatures (room temperature, 30, 60 and 90 ��), but PAn and SnO2 have no gas sensitivity at the above temperatures. The sensitivity of PAn/SnO2 materials shows linear increase with the increase of methanol concentration, but saturation with the increase of ethanol and acetone concentrations. PAn/SnO2 materials have high selectivity, fast response-recovery time and low operation temperature to VOCs, but pure PAn and SnO2 do not have.
Key words: gas sensitivity; polyaniline/SnO2; volatile organic compounds
1 Introduction
Environmental pollutions have greatly increased during the last few decades. VOCs (volatile organic compounds) can cause sick house syndrome, and be inflammable and explosive in plant and laboratory, so the detection of VOCs has become increasingly important. Many studies have focused on the development of the sensing materials, including inorganic and organic semi- conductors[1-3]. Though the inorganic semiconductors such as SnO2, Fe2O3 and ZnO have been used as gas leakage monitors, they must be worked at elevated temperature above 300 ��[4-5], which increases power consumption and reduce sensor life. Organic semiconductors are fit for operating at low temperature and have been applied in commercial devices, but the slow response time and insolubility are the most serious problem.
To complement the characteristics of pure inorganic and organic materials and explore the sensing materials with low operating temperature and good selectivity, organic-inorganic sensing hybrids have been developed[6-7]. Recently, JIANG et al[8-9] reported that PANI/TiO thin film to NH3 is superior to CO gas in response, reproducibility and stability, and studied the effect of polymerization temperature on the gas response of the PANI/TiO2 thin film gas sensor; HOSONO et al[10-11] synthesized PPy/MoO3 thin film and PPy/MoO3 pressed pellet, and studied the gas sensitivity to VOCs formaldehyde, ethanol, toluene, benzene, and so on. In addition, ARMES and MAEDS[12], and PARTCH et al[13] reported that these types of hybrid materials possess small grain size and high stability in air. But the research of PAn/SnO2 materials used for detecting VOCs has not been reported.
In the previous papers, we reported the primary gas sensitivity study of polypyrrole (PPy)/SnO2 and PPy/ZnO materials[14-15]. In this work, PAn was prepared by the similar polymerization method as polypyrrole and characterized by FT-IR. A series of PAn/SnO2 materials were prepared and measured for gas sensitivity to VOCs for the first time. The response mechanism of PAn/SnO2 materials was also presumed.
2 Experimental
2.1 Synthesis of H+ doped polyanline (PAn)
The reaction equation of aniline (An) with oxidant and HCl was[16-17]:

(0��y��1; x was a positive integer)
Aniline (An) monomer was distilled under reduced pressure before use.
Polyaniline (PAn) was synthesized by chemical polymerization at room temperature under nitrogen atmosphere[17]. An aqueous solution of ammonium persulfate (APS) was dropped into aniline solution in which the concentration of H+ was adjusted by HCl to 1 mol/L. The mole ratio of An to APS was 1?1. After APS was dropped, the mixed solution was stirred for 4 h. Then the precipitate was filtered, washed with 0.01 mol/L HCl and acetone three times respectively, followed by water wash to neutral. The product was dried in vacuum at 65 �� for 12 h.
PAn was characterized by Fourier transform infrared spectroscopy (FT-IR, Avatar 360 FT-IR spectrophotometer).
2.2 Preparation of PAn/SnO2 materials
A series of PAn/SnO2 materials were prepared by grinding PAn and SnO2 with different mass fractions and designated as PAn(1%)/SnO2, PAn(3%)/SnO2, PAn(5%)/SnO2, PAn(10%)/SnO2, PAn(20%)/SnO2, PAn(30%)/SnO2, PAn(40%)/SnO2 according to mass fractions of PAn.
SnO2 was purchased and the particle size was about 15 nm (calculated according to X-ray diffraction).
2.3 Fabrication and characterization of sensor elements
Gas-sensing properties of PAn, SnO2 and PAn/SnO2 materials to VOCs (methanol, ethanol and acetone) were measured at different operating temperatures: room temperature (RT), 30, 60 and 90 ��. The sensing samples were fabricated as thick-film sensors.
The test circuit (Fig.1) was described in our previous papers[14-15]. In Fig.1, Vc is a circuit voltage, Vout is a measured voltage, Vh is a heating voltage, and RL is the resistance of a loading resistor. The voltage (or resistance) across the sensor can be determined indirectly by measuring the Vout (or RL). The sensitivity (S) was defined as V(out)g/V(out)a (short as Vg/Va) (or RLg/RLa), where V(out)a (or RLa) is the initial voltage (or resistance) of the sensor and V(out)g (or RLg) is the voltage (or resistance) of the sensor when it was exposed to the testing gases. The response or recovery time is the time for the voltage (or resistance) change to reach 90% of the total change from V(out)g (or RLg) to V(out)a (or RLa) or vice versa. All experiments were carried out in a fixing humidity of 60%.
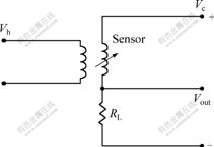
Fig.1 Electric circuit for gas sensing measurement
3 Results and discussion
3.1 FT-IR characterization of PAn
The FT-IR spectrum of H+ doped PAn using KBr pellets was recorded from 400 to 4 000 cm-1 (Fig.2). In the spectra of PAn, the specific absorption bands are observed at 1 557, 1 504, 1 300, 1 143, and 796 cm-1. They are close to those reported data[17-18]: 1 557 cm-1 is the stretching band of quinoid ring, 1 504 cm-1 is the stretching band of benzenoid ring, 1 300 cm-1 is the C��N stretching band of aryl amine (Ar-NH-Ar), 1 143 cm-1 is the vibration band of dopant anion, and 796 cm-1 is the para disubsticuted benzene ring.
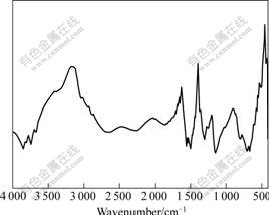
Fig.2 FT-IR spectrum of H+ doped PAn
3.2 Gas sensitivity measurement
Intrinsic PAn has no conductivity and gas sensitivity, but doped PAn has these properties depending on the preparation method and the fabrication method of sensors[17-18]. It is well known that the doped PAn is a p-type organic semiconductor, and has linear conjugate �� electron system in molecule, which can offer the transfer opportunity for current carrier (hole). PAn is suitable for working at low temperature, due to high temperature disadvantageous to the PAn conductivity. In this work, PAn was prepared by doping H+, and thick film sensors were fabricated to test. PAn has no gas sensitivity to VOCs at our operating temperatures (room temperature, 30, 60 and 90 ��), which may be related to the preparation method and the thick-film type sensor in our experiment.
SnO2 also has no gas sensitivity to VOCs when operating at the above operating temperatures, because SnO2 is an insulator and has no gas sensitivity at low temperature. It shows the properties of n-type semiconductor and gas sensitivity at elevated temperature.
PAn/SnO2 materials have no gas sensitivity to VOCs at RT, 30 �� or 60��, but PAn(3%)/SnO2, PAn(5%)/ SnO2, PAn(10%)/SnO2, PAn(20%)/SnO2 show gas sensitivity at 90 ��. Figs.3-5 show the response- recovery curves of PAn/SnO2 materials to methanol, ethanol and acetone, respectively. It can be seen that the response and recovery time is fast and the reversibility of PAn/SnO2 materials to VOCs is good. The response time of PAn/SnO2 materials to methanol, ethanol and acetone is 54-148, 10-32, 17-49 s, respectively, and the recovery time is 79-118, 47-109, 65-160 s, respectively. Figs.3-5 show that the resistance of PAn/SnO2 materials decreases when the materials are exposed to the electron-donating vapours (VOCs), which exhibit the properties of n-type semiconductors. The response mechanism of PAn/SnO2 materials may be similar to that of PPy/SnO2[19]. The electronic properties of the PAn/SnO2 materials appear to be governed by SnO2, due to PAn present at a low level in PAn/SnO2 materials. The gas sensitivity of PAn/SnO2 materials to VOCs when the materials are operated at 90 �� may be explained by the creation of positively charged depletion layer on the surface of the SnO2, which could be formed owing to inter-particle electron migration from SnO2 to PAn at the p-n heterojunctions. This would cause a lowering of the activation energy and enthalpy of physisorption for vapours with good electron-donating characteristics[19].
In order to explore the relationship of PAn/SnO2 materials�� sensitivity properties with the concentration of testing gases, a series concentration of gases were monitored. Fig.6 shows the sensitivity variation of PAn/SnO2 materials with increasing concentration of methanol, ethanol and acetone. It is clear that the sensitivity of PAn/SnO2 materials operated at 90 �� shows good dependence on methanol concentrations and exhibits an approximately linear sensitivity in the range of 0.05%-0.25%(volume fraction), even though they have different mass fractions of PAn. But the sensitivity of PAn/SnO2 materials shows saturation with ethanol and acetone at the concentration of 0.05%-0.25%. It may be due to that the hybrids achieve the highest sensitivity at
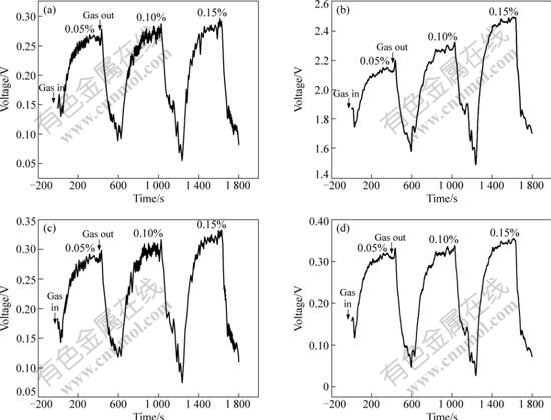
Fig.3 Response-recovery curves of PAn/SnO2 hybrids in methanol atmosphere: (a) PAn(3%)/SnO2, (b) PAn(5%)/SnO2; (c) PAn(10%)/SnO2; (d) PAn(20%)/SnO2
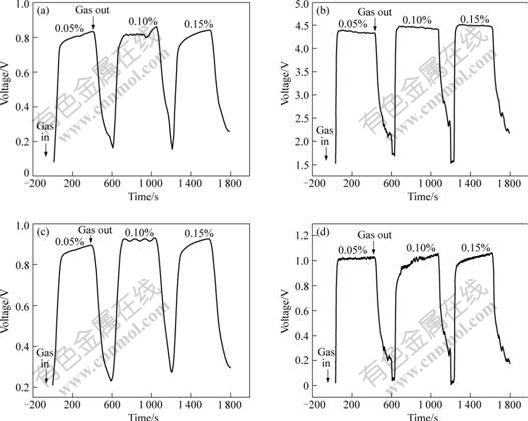
Fig.4 Response-recovery curves of PAn/SnO2 hybrids in ethanol atmosphere: (a) PAn(3%)/SnO2; (b) PAn(5%)/SnO2; (c) PAn(10%)/SnO2; (d) PAn(20%)/SnO2

Fig.5 Response-recovery curves of PAn/SnO2 hybrids in acetone atmosphere: (a) PAn(3%)/SnO2; (b) PAn(5%)/SnO2; (c) PAn(10%)/SnO2; (d) PAn(20%)/SnO2

Fig.6 Sensitivity of PAn/SnO2 materials vs different concentrations of VOCs: (a) Methanol; (b) Ethanol; (c) Acetone
0.05% ethanol or acetone, and the related experiment is being conducted.
Fig.7 shows the sensitivity of PAn/SnO2 materials with different mass fractions of PAN at 0.10% VOCs atmosphere respectively. We can see that with the increasing of PAn content, the sensitivity of PAn/SnO2 materials expresses the same tendency to different gases. It shows that PAn (1%)/SnO2 and PAn (40%)/SnO2 have low sensitivity to VOCs, and the sensitivities (S=RLg/RLa) of them are close to 1.0, but PAn (20%)/SnO2 has the highest sensitivity and can be used for practice.

Fig.7 Sensitivity of PAn/SnO2 materials at 0.10% VOCs atmosphere
4 Conclusions
1) PAn/SnO2 materials have gas sensitivity to VOCs at 90�� and can test VOCs at a wide concentration range, while PAn and SnO2 have no gas sensitivity in the experiment.
2) The response-recovery time of PAn/SnO2 materials to VOCs is fast. PAn (20%)/SnO2 has the highest sensitivity and is fit for practice.
3) The response mechanism of PAn/SnO2 materials to VOCs may be due to the existence of p-n heterojunction, but the true mechanism is under research.
References
[1] KUMAY V, SEN S, MUTHE K P, GAUR N K, GUPTA S K, YAKHMI J V. Copper doped SnO nanowires as highly sensitive H2S gas sensor [J]. Sens Actuators B, 2009, 138: 587-590.
[2] CHU De-wei, ZENG Yu-Ping, JIANG Dong-liang, MASUDA Y. In2O3�CSnO2 nano-toasts and nanorods: Precipitation preparation, formation, mechanism, and gas sensitive properties [J]. Sens Actuators B, 2009, 137: 630-636.
[3] LAITH A M, HENRY D T, WOJTEK W, RICHARD B K, KOUROSH K Z. Polypyrrole nanofiber surface acoustic wave gas sensors [J]. Sens Actuators B, 2008, 134: 826-831.
[4] SI Shu-feng, LI Chun-hui, WANG Xun, PENG Qing, LI Ya-dong. Fe2O3/ZnO core�Cshell nanorods for gas sensors [J]. Sens Actuators B, 2006, 119: 52-56.
[5] HSUEH T J, CHEN Y W, CHANG S J, WANG S F, HSU C L, LIN Y R, LIN T S, CHEN C C. ZnO nanowire-based CO sensors prepared on patterned ZnO:Ga/SiO2/Si templates [J]. Sens Actuators B, 2007, 125: 498-503.
[6] SURI K, ANNAPOORNI S, SARKAR A K, TANDON R P. Gas and humidity sensors based on iron oxide�Cpolypyrrole nanocomposites [J]. Sens Actuators B, 2002, 81: 277-282.
[7] NARDIS S, MONTI D, NATABLE C D, AMICO A D, SICILIANO P, FORLEO A, EPIFANI M, TAURINO A, RELLA R, PALLESSE R. Preparation and characterization of cobalt porphyrin modified tin dioxide films for sensor applications [J]. Sens Actuators B, 2004, 103: 339-343.
[8] TAI Hui-ling, JIANG Ya-dong, XIE Guang-zhong, YU Jun-sheng, CHEN Xuan. Fabrication and gas sensitivity of polyaniline-titanium dioxide nanocomposite thin film [J]. Sens Actuators B, 2007, 125: 644-650.
[9] TAI Hui-ling, JIANG Ya-dong, XIE Guang-zhong, YU Jun-sheng, CHEN Xuan, YING Zhi-hua. Influence of polymerization temperature on NH3 response of PANI/TiO2 thin film gas sensor [J]. Sens Actuators B, 2008, 129: 319-326.
[10] HOSONO K, MATSUBARA I, MURAYAMA N, WOOSUCK S, IZU N. Synthesis of polypyrrole/MoO3 hybrid thin film and their volatile organic compound gas-sensing properties [J]. Chem Mater, 2005, 17 (2): 349-354.
[11] MATSUBARA I, HOSONO K, MURAYAMA N, WOOSUCK S, IZU N. Synthesis and gas sensing properties of polypyrrole/MoO3- layered nanohybrids [J]. Bull Chem Soc Jpn, 2004, 77: 1231-1237.
[12] ARMES S, MAEDA S P. Preparation and characterization of polypyrrole-tin (IV) oxide nanocomposite collides [J]. Chem Mater, 1995, 7: 171-178.
[13] PARTCH R, GANGOLLI S G, MATIJEVIC E, CAL W, ARAJS S. Conducting polymer composites: (I). Surface induced polymerization of pyrrole on iron (III) and cerium (IV) oxide particles [J]. J Collid Interf Sci, 1991, 144 (1): 27-35.
[14] GENG Li-na, WANG Shu-rong, LI Peng, ZHAO Ying-qiang, ZHANG Shou-min, WU Shi-hua. Preparation and gas sensitivity study of polypyrrole/tin oxide hybrid material [J]. Chinese Jourmal of Inorganic Chemistry, 2005, 21(7): 977-981. (in Chinese)
[15] GENG Li-na, ZHAO Ying-qiang, HUANG Xue-liang, WANG Shu-rong, ZHANG Shou-min, HUANG Wei-ping, WU Shi-hua. The preparation and gas sensitivity study of polypyrrole/zinc oxide [J]. Synth Met, 2006, 156: 1078-1082.
[16] YANG Lan-sheng, XU Jin-mao, SHAN Zhong-qiang, CHEN Wei-xiang. The chemical synthesis of conducting polyaniline [J]. Chemical Industry and Engineering, 1994, 2 (11): 7-12. (in Chinese)
[17] WEI Chun-xiang. The preparation and characterization of polyaniline [D]. Nanjing: Nanjing University of Science and Technology, 2004: 3-4.
[18] MONKMAN A P, ADAMS P. Optical and electronic properties of strech-oriented solution-cast polyzniline films [J]. Synth Metals, 1991, 40: 87-96.
[19] BENJAMIN P J, PHILLIP E, RICHARD J E, COLIN L H, NORMAN M R. Novel composite organic-inorganic semiconductor sensors for the quantitative detection of target organic vapours [J]. J Mater Chem, 1996, 6 (3): 289-294.
(Edited by YANG You-ping)
Foundation item: Project (20070410866) supported by Postdoctoral Science Foundation of China; Project(L2006B18) supported by Doctoral Foundation of Hebei Normal University
Corresponding author: GENG Li-na; Tel: +86-311-86268311; E-mail: genglina0102@126.com