文章编号:1004-0609(2015)-07-2006-09
3-己基-4-氨基-1,2,4-三唑-5-硫酮在黄铜矿表面的吸附动力学与热力学
曲肖彦,刘广义,刘 胜,钟 宏,肖静晶
(中南大学 化学化工学院,长沙 410083)
摘 要:合成了一种新型的三唑硫酮类表面活性剂―3-己基-4-氨基-1,2,4-三唑-5-硫酮(HATT),通过红外光谱和核磁共振氢谱及磁谱对其结构进行了表征,研究其在黄铜矿表面的吸附动力学和热力学。结果表明:黄铜矿吸附HATT优选的pH值为4~8,吸附量随着温度的升高而增大;吸附过程符合准二级动力学方程,吸附活化能为13.06 kJ/mol;HATT在黄铜矿表面的吸附等温线符合Langmuir模型,吸附焓变ΔH为71.52 kJ/mol,熵变ΔS为348.7 J/(mol・K),吸附自由能变ΔG为-28.95 kJ/mol(298K)。HATT可能以单分子层化学吸附于黄铜矿表面,吸附为自发的吸热过程。FTIR光谱分析进一步证实HATT以化学方式吸附在黄铜矿的表面。浮选试验结果也表明,HATT是黄铜矿的优良捕收剂。
关键词:3-己基-4-氨基-1,2,4-三唑-5-硫酮;黄铜矿;热力学;动力学; 吸附
中图分类号:TQ 311;O64 文献标志码:A
Adsorption kinetics and thermodynamics of 3-hexyl-4-amino-1,2,4-triazole-5-thione on surface of chalcopyrite
QU Xiao-yan, LIU Guang-yi, LIU Sheng, ZHONG Hong, XIAO Jing-jing
(School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China)
Abstract: A novel surfactant, 3-hexyl-4-amino-1,2,4-triazole-5-thione (HATT) was synthesized and characterized by infrared spectroscopy, 1HNMR and 13CNMR. And its adsorption kinetics and thermodynamics on the chalcopyrite surface were evaluated. The results indicate that the preferable pH values for HATT adsorption on chalcopyrite surface are 4-8 and the adsorption amount increases with increasing temperature. The adsorption process conforms to the pseudo-second-order kinetics equation and the adsorption activation energy is 13.06 kJ/mol. The adsorption isotherm agrees well with Langmuir model and the thermodynamics parameters ΔH, ΔS and ΔG are 71.52 kJ/mol, 348.7 J/(mol・K) and -28.95 kJ/mol (298K), respectively. The adsorption of HATT on the chalcopyrite surface is a spontaneous endothermic monolayer chemisorption. The FTIR spectra further verify the chemisorption process. The flotation results also show that HATT is a superior collector for chalcopyrite.
Key words: 3-hexyl-4-amino-1,2,4-triazole-5-thione; chalcopyrite; thermodynamics; kinetics; adsorption
三唑硫酮类化合物具有良好的杀菌、抑菌、调节植物生长和螯合金属等性质[1-3]。在生物医药领域[3-8],该类化合物具有消炎、镇静、抗焦虑、抗微生物、抗真菌等多种生物活性;在农业领域[9-10],可用于开发高效低毒的除草剂、杀虫剂和植物生长调节剂;在金属防腐领域[11-13],其用作金属缓蚀剂。
三唑硫酮类化合物中的硫原子和氮原子具有孤对电子,可作为电子给予体与金属离子的空轨道形成配位键[14-15],且这类杂环化合物中的杂环氮原子和环外硫原子能通过共轭作用离域它们的价电子,增强其对过渡金属的络合选择性。KAZYMOVA等[16]研究了3-甲基-4-氨基-1,2,4-三唑-5-硫酮(EATT)与Ni(Ⅱ)的络合反应,发现Ni能与EATT的S原子和环外N原子成键。YI等[17]报道3,5-二甲基-1,2,4-三唑-镉金属配位化合物的单晶结构。结果表明:镉原子分别与三唑环上的两个氮原子形成六元环状对称结构。SHIRINKAM等[18]考察4-(2,4-二氯氨基苯)-5-甲基-1,2,4-三唑-3-硫酮(LH)与硝酸银和三苯基膦三元络合物的单晶结构,表明配合物中银离子通过与LH中的一个S原子及三苯基膦中的3个P原子成键形成一个四面体络合物。OUICI等[19]通过动电位极化及电化学阻抗法研究发现5-苯基-1,2,4-三唑-5-硫酮在1 mol/L的酸性介质中对C38钢具有良好的缓蚀效果。王鹏[20]报道了3-十一烷基-4-氨基-5-巯基-1,2,4-三唑通过其π(C=N)轨道和S原子的p轨道提供电子与铜的d轨道成键而吸附在铜币表面,减少了铜币的腐蚀。
虽然三唑硫酮类杂环化合物是过渡金属的优良螯合剂和防腐剂,但目前鲜有其用作浮选捕收剂从金属矿石中回收有价金属矿物的报道。因此,开展对此类物质的吸附及浮选性能研究,研究其吸附机理,有望开发出一类新型的浮选捕收剂。
1 实验
1.1 试剂与仪器
二氨基硫脲(TCH)和3-乙乙基-4-氨基-1,2,4-三唑-5-硫酮(HATT)由本课题组实验室制备,其他化学药品购自商业公司,其纯度为化学纯及以上。黄铜矿源自墨西哥,购自美国WARD’S 自然科学有限公司,为手捡分选样品。试验、分析仪器主要有:SHA-C型水浴恒温振荡器(常州澳华仪器有限公司生产,中国)、UV-1750型紫外可见分光光度计(岛津公司生产,日本)、pHS-3C型pH计(上海精密科学仪器有限公司镭磁仪器厂生产,中国)、Nicolet FTIR-740 型傅里叶变换红外光谱仪(尼高力公司生产,美国);AVANCE III500M型核磁共振波谱仪(布鲁克公司生产,瑞士)。实验用水为一次蒸馏水。
1.2 HATT合成与表征
准确称量2.125 g二氨基硫脲(0.02 mol)于50 mL三口瓶中,向其中加入8.5 mL正庚酸(0.06 mol)(n(TCH):n(n-C6H13COOH)=1:3),油浴中加热回流(回流温度为145~150 ℃)2 h,室温冷却析出白色晶体后抽滤,蒸馏水洗,用乙醇-水重结晶得到白色针状晶体,合成的化学反应方程如式(1)所示:
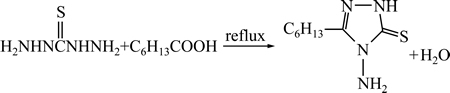 (1)
(1)
目标产品为白色针状晶体,产率80.5%,熔程为111~113 ℃,与文献[2]的值一致。采用KBr压片法对产品进行红外光谱测定,在波长3157、3283和3043 cm-1处为N―H的伸缩振动,2764 cm-1为S―H的伸缩振动,1622 cm-1为C=N 伸缩振动,1562、1485、1395、1296和1117 cm-1处为C(=S)―NH的复合伸缩振动。在测定频率为500 MHz ,溶剂为DMSO的条件下对产品进行核磁氢谱(1H NMR)和碳谱(13C NMR)分析。化学位移0.866×10-6 处的四重峰归属为CH3上的3个氢,1.279×10-6处的多重峰归属为3个化学环境相同的CH2上的6个氢,1.63×10-6处的四重峰归属为CH2上的两个氢,2.614×10-6处的多重峰归属为CH2上的两个氢,5.523×10-6处的单峰归属于NH2上的两个氢,13.435×10-6处的单峰归属为NH或者SH上的氢。在位移为166.154×10-6、152.622×10-6、31.303×10-6、28.552×10-6、25.881×10-6、 24.638×10-6、22.422×10-6和14.364×10-6处检测到碳的出现,与目标产品碳的种类和位移吻合。
1.3 矿样准备
黄铜矿经过手碎、手选和玛瑙研钵研磨,取37~74 μm的矿样用于吸附和浮选试验,黄铜矿比表面积为0.165 m2/g(BET法),其XRD谱如图1所示,XRF分析结果见表1。结果表明,该黄铜矿矿样中黄铜矿含量大于91%,蓝晶石(Al2O[SiO4])含量大于7%,黄铁矿含量小于1%。实验前用超声波洗涤黄铜矿矿样,每次洗涤5 min,静置1 min后倒掉上层清液,重复洗涤3次,抽滤,自然晾干。
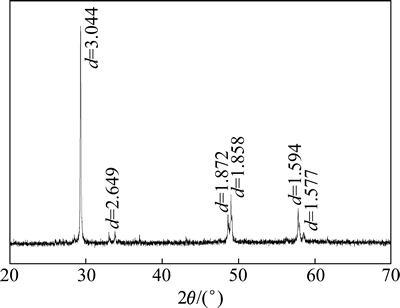
图1 黄铜矿XRD谱
Fig. 1 XRD pattern of chalcopyrite
1.4 HATT溶液标准曲线
准确称取0.1002 g HATT,溶解于500 mL蒸馏水中得到1 mmol/L HATT溶液,稀释母液得到一定浓度梯度的HATT溶液,在特征波长247 nm处,用紫外分光光度计测定其吸光度(A),然后以吸光度为纵坐标,浓度(X)为横坐标,得线性回归方程:A=0.009 65+ 15 857.408X,R2=0.999 9。
表1 黄铜矿XRF分析结果
Table 1 Element content of chalcopyrite by XRF (mass fraction, %)

1.5 吸附试验
移取50 mL一定初始浓度的HATT溶液于150 mL锥形瓶中,加入0.5 g黄铜矿,根据需要调节溶液pH或温度,置于恒温水浴振荡器中,转速为200 r/min,不同温度(288、299、308 K)下震荡一段时间,过滤,取上层清液测定其吸光度。
根据所得数据按照式(2)计算出任意时刻吸附量Qt:
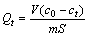 (2)
(2)
式中:c0为HATT溶液初始浓度,mol/L;ct为任意时刻HATT溶液浓度,mol/L;V为溶液体积,L;m为黄铜矿的加入量,g;Qt为任意时刻黄铜矿的吸附量,mol/m2;S为黄铜矿的比表面积,m2/g。
1.6 红外光谱
将浓度均为1 mmol/L的HATT溶液Cu2+/Cu+(由抗坏血酸还原铜离子生成)溶液按体积比2.5:1混合,产生黄色/灰绿色沉淀;取150 mL浓度为1 mmol/L的HATT溶液于250 mL锥形瓶中,加入0.5 g粒度<37 μm的黄铜矿,室温下在恒温水浴振荡器中震荡20 h。将药剂与金属离子或矿物作用后的固体经过滤、蒸馏水洗、真空干燥后进行红外检测。
红外光谱在Nicolet FTIR-740 型傅里叶变换红外光谱仪上采用溴化钾压片法进行红外测试,波数范围为4000~500 cm-1。
1.7 单矿物浮选实验
实验采用单泡管,每次取已处理好的单矿物2 g于烧杯中,并向其中加入已调好浓度的224 mL HATT溶液,搅拌1 min。用HCl或KOH调节矿浆pH值,再搅拌3 min后,加入1 mL甲基异丁基甲醇(MIBC)使其在浮选时浓度为15 mg/L,继续搅拌1 min后,静置30 s将上清液加入单泡管中,通入N2,流速控制在200 mL/min,加入矿物,浮选3 min,收集泡沫产品和槽内产品,经干燥、烘干、称取质量后计算回收率。浮选回收率ε按式(3)计算。
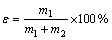 (3)
(3)
式中:m1为泡沫产品的质量,g;m2为槽内产品的质量,g。
2 结果与讨论
2.1 时间对吸附量的影响
在HATT初始浓度为0.1 mmol/L、不同温度(288 K、298 K、308 K)下黄铜矿对HATT的吸附量随时间变化的关系见图2。由图2可看出,同一温度下,黄铜矿对HATT的吸附量随吸附时间的增加而增大,在吸附开始的0~15 h内,黄铜矿对HATT的吸附速率最大,吸附量增长最快;超过15 h之后,随着时间的增加,吸附量增加变缓;18 h时,吸附量基本达到饱和。同一吸附时间下,温度越高,吸附量越大,说明此吸附过程为吸热过程。

图2 黄铜矿对HATT的吸附量随时间变化的关系曲线
Fig. 2 Adsorption amount of HATT on surface of chalcopyrite as function of time
2.2 pH对吸附量的影响
在吸附时间为18 h、温度为298 K时,HATT在黄铜矿表面的吸附量与pH值的关系见图3。由图3可看出,随着pH的增加,黄铜矿对HATT的吸附量先增加后减少,pH约为7.3时达到最大。在试验pH 2~12的范围内,HATT均能较好吸附在黄铜矿表面,但pH 4~8为其优选的吸附pH范围。
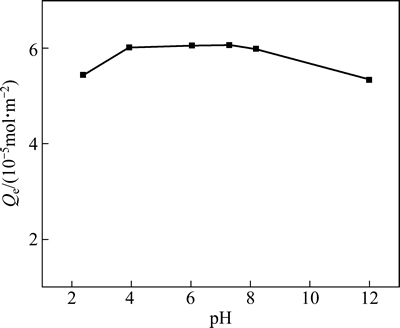
图3 HATT在黄铜矿表面的吸附与pH值的关系
Fig. 3 Relationship between adsorption of HATT on chalcopyrite surface and pH value
2.3 等温吸附
在吸附时间为18 h、pH 7.3时,不同温度(298 K、318 K、328 K)下黄铜矿对HATT的平衡吸附量(Qe)与HATT的平衡浓度(ce)的关系见图4。
由图4可知,同一温度下,黄铜矿对HATT的平衡吸附量随着HATT平衡浓度的增加而增加;在相同的平衡浓度条件下,随着温度的升高,黄铜矿对HATT的平衡吸附量也升高,表明该吸附过程为吸热过程。
分别采用Langmuir[21](见式(4))和Freundlich[22](见式(5))等温吸附方程对图4中实验数据进行线性拟合,并对两者进行了比较,其结果见图5、表2以及图6、表3。
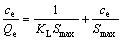 (4)
(4)
 (5)
(5)
式中:KL为Langmuir常数;Smax为单位面积最大吸附容量,mol/m2;Kf和n为Freundlich参数。
由图5、表2以及图6、表3的拟合结果可知,在实验范围内,黄铜矿对HATT的吸附既符合Langmuir等温吸附方程,也符合Freundlich等温吸附方程(R2>0.90),但Langmuir等温吸附方程拟合的线性相关性更佳(R2>0.98),说明HATT在黄铜矿表面的吸附以单层化学吸附为主。
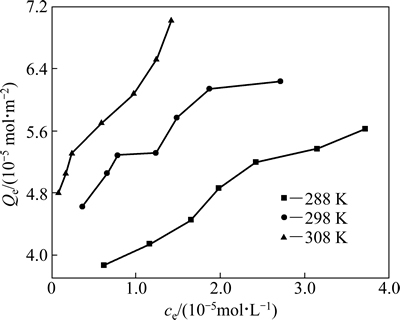
图4 黄铜矿对HATT的吸附等温线
Fig. 4 Adsorption isotherm of HATT on chalcopyrite surface
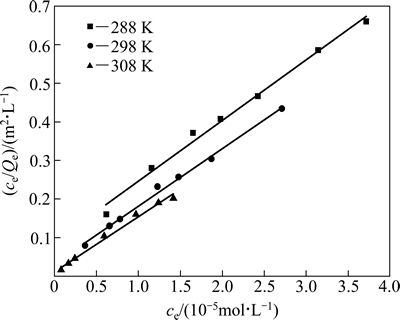
图5 图4中数据的Langmuir拟合曲线
Fig. 5 Langmuir fitting curves of data in Fig. 4
表2 Langmuir等温方程拟合结果
Table 2 Fitting results of Langmuir isotherm equation
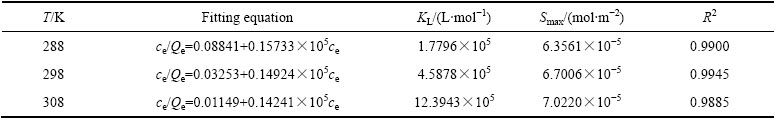
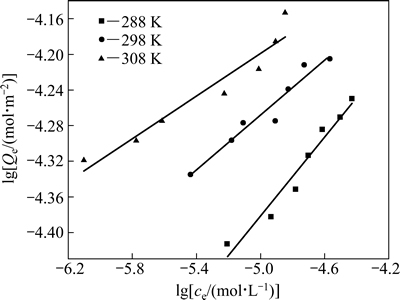
图6 图4中数据的Freundlich拟合曲线
Fig. 6 Freundlich fitting curves of data in Fig. 4
表3 Freundlich等温方程拟合结果
Table 3 Fitting results of Freundlich isotherm equation

2.4 吸附热力学
吸附自由能变(ΔG)、焓变(ΔH)及熵变(ΔS)是测定吸附过程热量变化及自发性的重要热力学参数,能够反映出温度对HATT在黄铜矿表面吸附行为的影响。上述热力学参数可以通过式(6)和(7)进行计算[23]:
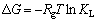 (6)
(6)
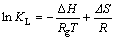 (7)
(7)
式中:Rg是摩尔气体常数,8.314 J/(mol・K);T为绝对温度,K;KL为Langmuir常数。
ΔH和ΔS可通过lnKL对1/T作图所得斜率和截距求得,其结果见表4和图7。表4和图7的结果表明,在288、298和308 K下,ΔG均为负值,表明这3种温度下的吸附过程均为自发过程。ΔH为正值,表明HATT在黄铜矿表面的吸附过程为吸热过程,高温有利于吸附,这与等温吸附的实验结果一致。ΔS为正值表明吸附过程为熵驱动过程而非焓驱动过程[24]。从微观上讲,对固液反应,根据吸附交换理论,HATT从水溶液迁移到固液界面并发生化学吸附,有较强的成键作用,HATT由无序趋向有序,此过程通常放热且熵减小。然而,黄铜矿表面吸附的水分子同时也会解吸到液相中,该过程需消耗能量且熵增大;且HATT分子中含杂环氮原子及胺基,在溶液中易与水分子形成氢键和范德华力,HATT化学吸附于黄铜矿表面后必将削弱与水分子间的作用。因而,HATT在黄铜矿表面的吸附过程所消耗的能量可能大于释放的能量,熵增大于熵减,焓和熵都增加[25]。
表4 HATT在黄铜矿表面吸附的热力学参数
Table 4 Thermodynamics parameters of HATT adsorbing on chalcopyrite surface

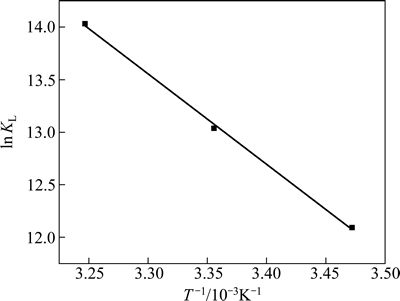
图7 lnKL与T-1的关系
Fig. 7 Relationship between lnKL and T-1
2.5 吸附动力学
为了研究不同温度下HATT在黄铜矿表面的吸附过程,分别采用准一级反应动力学方程[26](式(8))和准二级反应动力学方程[27](式(9))对动力学数据(实验数据见图2)进行拟合,并对两者进行了比较。
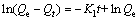 (8)
(8)
 (9)
(9)
式中:K1为准一级动力学吸附速率常数,h-1;K2为准二级动力学吸附速率常数,m-2/(mol・h)。
准一级动力学和准二级动力学拟合曲线分别见图8和9,K1可通过拟合曲线的斜率求得, K2可通过拟合曲线的斜率和截距求得,其结果见表5。
由表5的拟合结果可知,在实验范围内,准二级动力学方程拟合的线性相关性更佳(R2>0.99),且平衡吸附量与实测值接近,表明吸附过程符合准二级动力学方程。Qe随着温度的升高而增大,表明吸附过程为吸热过程,这与热力学分析的结论相符。
表5 HATT在黄铜矿表面吸附动力学参数
Table 5 Kinetics parameters of HATT adsorbing on chalcopyrite surface
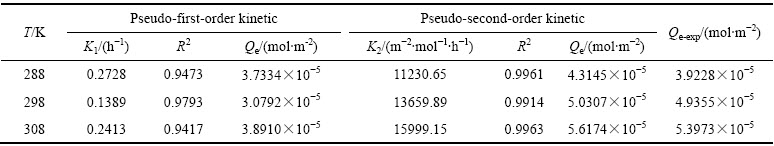
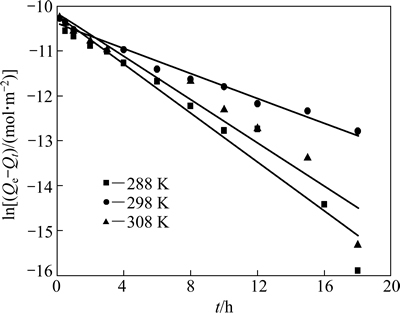
图8 准一级动力学拟合曲线
Fig. 8 Pseudo-first-order kinetic fitting curves
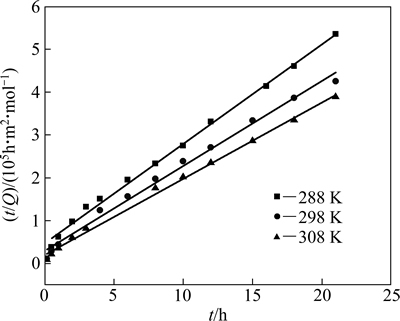
图9 准二级动力学拟合曲线
Fig. 9 Pseudo-second-order kinetic fitting curves
根据准二级动力学方程的吸附速率常数K2采用Arrhenius公式来计算活化能Ea,如式(10)所示:
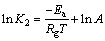 (10)
(10)
式中:Ea为吸附活化能,J・mol-1;K2为准二级动力学方程吸附速率常数,m-2・(mol-1・h-1);A为常数。
由表5中不同温度下的K2值,作lnK2-T-1图,其结果见图10。
由图10可知,lnK2与1/T有很好的线性关系(R2= 0.996 5),线性拟合方程为
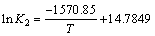 (11)
(11)
通过方程的斜率可计算出吸附活化能为13.06 kJ/mol。
2.6 红外光谱分析
HATT、HATT与Cu2+或Cu+作用产物的红外光谱见图11,黄铜矿与HATT作用前后的红外光谱见图12。
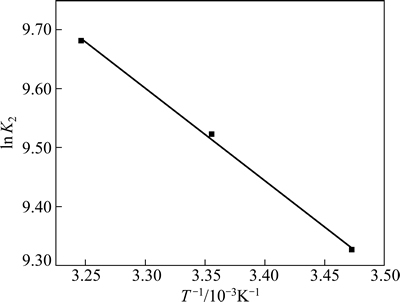
图10 lnK2与T-1的关系
Fig. 10 Relationship between lnK2 and T-1
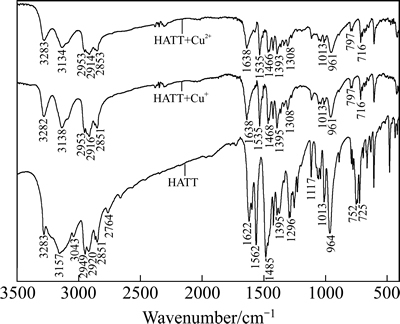
图11 HATT与Cu2+或Cu+作用前后的红外光谱图
Fig. 11 IR spectra of HATT before and after its response to Cu2+ or Cu+
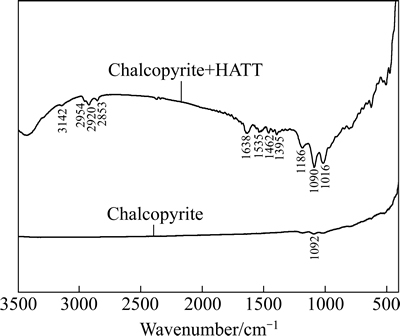
图12 黄铜矿与HATT作用前后的红外光谱图
Fig. 12 FTIR spectra of chalcopyrite before and after HATT treatment
图11表明,HATT与Cu2+或Cu+作用后,其3157 cm-1附近的N―H伸缩振动有所减弱,3043 cm-1处的N―H红外吸附峰基本消失;与此同时,2764 cm-1处的S―H伸缩振动也在HATT与Cu2+或Cu+的络合物中消失。与HATT相比,HATT与Cu2+或Cu+络合物中红外特征吸收峰强度或位移发生了明显变化的还有:1622 cm-1处的C=N振动向高频处移动了16cm-1,出现在1638 cm-1左右(HATT与Cu2+或Cu+络合物中);1562 cm-1处的C(=S)―N伸缩振动[28]向低频处移动了27 cm-1,在HATT与Cu2+或Cu+络合物中出现在1535 cm-1处;同时,1485 cm-1处的C(=S)―N也向低频处移动了17~19 cm-1;964 cm-1处的C=S峰强明显减弱,且稍微移向低频(HATT与Cu2+络合物为964 cm-1、与Cu+络合物为959 cm-1)。图11中这些特征官能团的红外吸收峰变化说明,在HATT与Cu2+或Cu+的络合物中,HATT可能通过其硫代羰基硫原子与铜原子成键,使得硫代酰胺基C(=S)―N中电子云密度有所降低,其红外吸收峰稍微移向低频,并释放出S―H(C(―SH)=N=和C(=S)―NH―互为共振结构)中的氢离子进入溶液;与此同时,C=N键中电子密度增大,其红外吸收峰稍微移向高频[29-30]。N―H伸缩振动的红外吸附峰有所减弱也表明,HATT可能还通过其环外氮原子与铜原子成键,并释放出N―H键中的一个氢离子进入溶液。
图12表明,HATT处理后,HATT与Cu2+或Cu+络合物的红外特征吸收峰出现在黄铜矿表面,这些吸收带包括2954、2920和2853 cm-1处的C―H伸缩振动,1638 cm-1处的C=N伸缩振动吸收峰,1535和1462 cm-1处的硫代酰胺基C(=S)―N。这充分说明,HATT通过与黄铜矿表面的铜/亚铜离子发生化学反应而吸附在其表面,可能其硫代羰基硫原子和环外氮原子都参加了与铜原子的成键作用。红外光谱进一步证实HATT以化学方式吸附在黄铜矿表面,这与吸附热力学的结论一致。
2.7 HATT对单矿物浮选行为的影响
在HATT浓度为5×10-5 mol/L时,矿浆pH值对黄铜矿浮选回收率的影响见图13。
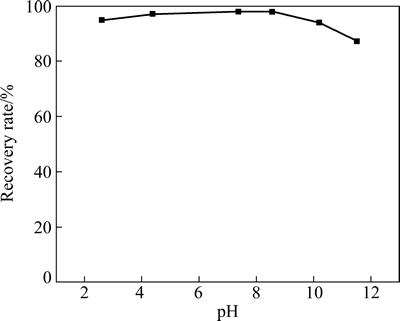
图13 pH对黄铜矿浮选回收率的影响
Fig. 13 Effect of pH values on flotation recovery of chalcopyrite
由图13可知,随着pH的增加,黄铜矿的回收率先增加后减小,在整个实验pH范围内,HATT对黄铜矿的浮选捕收效果较好,pH=7.3时,HATT对黄铜矿回收率的达到97.89%;pH 4~9为其优选的浮选pH范围,这与吸附实验结果一致。
在矿浆pH值为7.3时,HATT初始浓度对黄铜矿浮选回收率的影响见图14。
图14表明,黄铜矿的浮选回收率随着HATT初始浓度的增加而增大,当HATT浓度为2×10-5 mol/L时,黄铜矿的回收率达到96.07%,之后,随着HATT浓度的增加,回收率增加不明显。
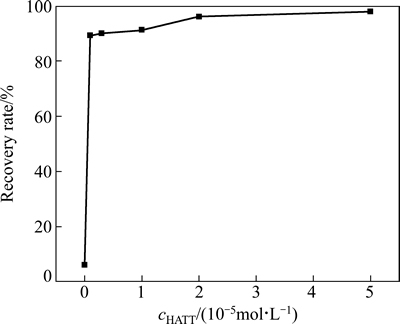
图14 HATT初始浓度和单矿物浮选回收率的关系
Fig. 14 Relationship between HATT initial concentration and flotation recovery rate of chalcopyrite
3 结论
1) 研究一种新型的三唑硫酮类表面活性剂HATT在黄铜矿表面的吸附动力学和热力学。结果表明:黄铜矿吸附HATT优选的pH值为4~8,吸附量随着温度的升高而增大。
2) 吸附动力学分析表明:吸附过程符合准二级动力学方程,吸附活化能为13.06 kJ/mol。吸附热力学分析表明:HATT在黄铜矿表面的吸附可能为单分子层化学吸附,吸附等温线符合Langmuir模型,为自发的吸热过程。
3) FTIR光谱分析证实HATT以化学方式吸附在黄铜矿的表面,可能通过硫代羰基硫原子和环外氮原子与黄铜矿表面的铜原子作用。浮选试验结果表明:HATT是黄铜矿的优良捕收剂。
REFERENCES
[1] 陆文婷, 孙晓红, 刘源发,陈 邦, 勒如意. 烷基三唑硫酮席夫碱的合成和生物活性研究[J]. 化学工程, 2012, 40(1): 5-8.
LU Wen-ting, SUN Xiao-hong, LIU Yuan-fa, CHEN Bang, LE Ru-yi. Synthesis and biological activities of 5-alkyl-4- amino-1,2,4-triazole-3-thione Schiff bases[J]. Chemical Engineering, 2012, 40(1): 5-8.
[2] SMI IUS R, BURBULIENE M M, JAKUBKIENE V, UDRENAITE E, VAINILAVI
IUS R, BURBULIENE M M, JAKUBKIENE V, UDRENAITE E, VAINILAVI IUS P. Convenient way to 5-substituted 4-amino-2, 3-dihydro-4H-1, 2, 4-triazole- 3-thiones[J]. Journal of Heterocyclic Chemistry, 2007, 44(2): 279-284.
IUS P. Convenient way to 5-substituted 4-amino-2, 3-dihydro-4H-1, 2, 4-triazole- 3-thiones[J]. Journal of Heterocyclic Chemistry, 2007, 44(2): 279-284.
[3] BENTISS F, BOUANIS M, MERNARI B, TRAISNEL M, VEZIN H, LAGRENEE M. Understanding the adsorption of 4H-1,2,4-triazole derivatives on mild steel surface in molar hydrochloric acid[J]. Applied Surface Science, 2007, 253: 3696-3704.
[4] PLECH T, KAPRO B, LUSZCZKI J J, WUJEC M, PANETH A, SIWEK A, KOLACZKOWSKI M, ZOLNIEREK M, NOWAK G. Studies on the anticonvulsant activity of 4-alkyl-1,2,4-triazole-3-thiones and their effect on GABAergic system[J]. European Journal Medicinal Chemistry, 2014, 86: 690-699.
B, LUSZCZKI J J, WUJEC M, PANETH A, SIWEK A, KOLACZKOWSKI M, ZOLNIEREK M, NOWAK G. Studies on the anticonvulsant activity of 4-alkyl-1,2,4-triazole-3-thiones and their effect on GABAergic system[J]. European Journal Medicinal Chemistry, 2014, 86: 690-699.
[5] COSTEA L V, BERCEAN V, BADEA V, CHIRIAC A. Substituent effects on the anodic oxidation of some 1H- 3-methyl-4-ethoxycarbonyl-5-substituted-benzylidenehydrazino-pyrazoles Ⅱ[J]. Annals of West University of Timisoara-Series of Chemistry, 2006, 15(2): 225-230.
[6] KHAN M H, HAMEED S, YASIN K A, AKHTAR T, KHAN K M. Design, synthesis,and urease inhibition studies of a series of 4-amino-5-aryl-3H-1,2,4-triazole-3-thiones[J]. Monatsh Chem, 2010, 10: 479-484.
[7] SAHOO P K, SHARMA R, PATTANAYAK P. Synthesis and evaluation of 4-amino-5-phenyl-4H-[1,2,4]- triazole-3-thiol derivatives as antimicrobial agents[J]. Medicinal Chemistry Research, 2010, 19: 127-135.
[8]  . Recent advances bioactive 1,2,4-triazole-3-thiones[J]. European Journal of Medicinal Chemistry, 2014, http://dx.doi.org/10.1016/j.ejmech. 2014. 11.033.
. Recent advances bioactive 1,2,4-triazole-3-thiones[J]. European Journal of Medicinal Chemistry, 2014, http://dx.doi.org/10.1016/j.ejmech. 2014. 11.033.
[9] 张静静, 姜鸿飞, 张金波, 迟会伟, 李志念, 刘长令. 三唑硫酮类化合物的合成与生物活性[J]. 农药, 2012, 51(4): 243-245.
ZHANG Jing-jing, JIANG Hong-fei, ZAHNG Jin-bo, CHI Hui-wei, LI Zhi-nian, LIU Chang-ling. Synthesis and research of biological activity of triazole thiones[J]. Agrochemicals, 2012, 51(4): 243-245.
[10]  , TABANCA N, ALI A, KHAN S I, KHAN I A, WEDGE D E. Synthesis and biological activity of substituted urea and thiourea derivatives containing 1,2,4-triazole moieties[J]. Molecules, 2013, 18: 3562-3576.
, TABANCA N, ALI A, KHAN S I, KHAN I A, WEDGE D E. Synthesis and biological activity of substituted urea and thiourea derivatives containing 1,2,4-triazole moieties[J]. Molecules, 2013, 18: 3562-3576.
[11] BENTISS F, TRAISNEL M, VEZIN H, LAGRENEE M. Linear resistance model of the inhibition mechanism of steel in HCl by triazole and oxadiazole derivatives: structure-activity correlations[J]. Corrosion Science, 2002, 45(2): 371-380.
[12] MEHDI B E, MERNARI B, TRAISNEL M, BENTISS F, LAGRENEE M. Synthesis and comparative study of the inhibitive effect of some new triazole derivatives towards corrosion of mild steel in hydrochloric acid solution[J]. Materials Chemistry and Physics, 2003, 15: 489-496.
[13] KUMAR M S, KUMAR S L A, SREEKANTH A. Anticorrosion potential of 4-amino-3-methyl-1,2,4-triazole-5-thione derivatives (SAMTT and DBAMTT) on mild steel in hydrochloric acid solution[J]. Industrial & Engineering Chemistry Research, 2012, 51(15): 5408-5418.
[14] ELDAKAR N, NOBE K E N. Electrochemical and corrosion behavior of iron in presence of substituted benzotriazoles[J]. Corrosion, 1977, 33(4): 128-130.
[15] LIU C Y, LU G H, JIANG L Y, JIANG L P. Study on the electrochemical behavior of dopamine and uric acid at a 2-amino-5-mercapto-[1,3,4] triazole self-assembled monolayers electrode[J]. Electroanalysis, 2006, 18(3): 291-297.
[16] KAZYMOVA M A, SHUMILOVA T A, MIKHAILOV O V, SOLOVIEVA S E. Complexing reactions in the Ni(II)-5- methyl-4-amino-3-thiooxo-1,2,4-triazapentene-1-methanal and Ni(II)-5-methyl-4-amino-3-thiooxo-1,2,4-triazapentene-1-propanone triple systems[J]. Journal of Coordination Chemistry, 2009, 62(17): 2792-2795.
[17] YI L, DING B, ZHAO B, CHENG P, LIAO D Z, YAN S P, JIANG Z H. Novel triazole-bridged cadmium coordination polymers varying from zero-to three-dimensionality[J]. Inorganic Chemistry, 2004, 43: 33-43.
[18] SHIRINKAM B, TABATABAEE M, KUKOVEC B M, OLIVER C L, GHASSEMZADEH M. Preparation, spectroscopic characterization, and crystal structure of a mixed-ligand silver (I) complex with 1,2,4-triazole-based Schiff base and triphenylphosphine[J]. Monatsh Chem, 2014, 145(11): 1753-1757.
[19] OUICI H B, BELKHOUDA M, BENALI O, SALGHI R, BAMMOU L, ZARROUK A, HAMMOUTI B. Adsorption and inhibition effect of 5-phenyl-1,2,4-triazole-3-thione on C38 steel corrosion in 1M HCl[J]. doi: 10.1007/s11164-014-1556-2, Research on Chemical Intermediates, 2014.
[20] 王 鹏. 铜质币腐蚀变色机理及抗变色工艺研究[D]. 大连: 大连理工大学, 2009.
WANG Peng. Tarnishing mechanism and anti-tarnishing techniques of Copper and brass coins[D]. Dalian: Dalian University of Technology, 2009.
[21] LANGMIUR I. The adsorption of gases on plane surfaces of glass, mica and platinum[J]. Journal of the American Chemistry Society, 1918, 40(9): 1361-1403.
[22] FREUNDLICH H M F. Over the adsorption in solution[J]. Zeitschriftfür Physikalische Chemie, 1906, 57: 385-471.
[23] AL-GHOUTI M, KHRAISHEH M A M, AHMAD M N M, ALLEN S. Thermodynamic behavior and the effect of temperature on the removal of dyes form aqueous solution using modified diatomite: A kinetic study[J]. Journal of Colloid Interface Science, 2005, 287(1): 6-13.
[24] YANG J B, YU M Q, QIU T. Adsorption thermodynamics and kinetics of Cr(Ⅵ) on KIP210 resin[J]. Journal of Industrial and Engineering Chemistry, 2014, 20(2): 480-486.
[25] ZHAO Z W, XU X Y, CHEN X Y, HUO G S, CHEN A L, LIU XH, XU H. Thermodynamics and kinetics of adsorption of molybdenum blue with D301 ion exchange resin[J]. Transactions of Nonferrous Metals Society of China, 2012, 22(3): 686-693.
[26]  . Adsorption of lead and cadmium ions from aqueous solutions using manganoxide minerals[J]. Transactions of Nonferrous Metals Society of China, 2012, 22(5): 3131-3139.
. Adsorption of lead and cadmium ions from aqueous solutions using manganoxide minerals[J]. Transactions of Nonferrous Metals Society of China, 2012, 22(5): 3131-3139.
[27] LIU Guang-yi, XIAO Jing-jing, REN Heng, ZHONG Hong. Adsorption thermodynamics and kinetics of N, N′-diisopropoxypropyl-N″, N″′-oxydiethylenedicarbonyl bis (thiourea) on chalcopyrite surfaces[J]. Journal of Industrial and Engineering Chemistry, 2015, 21: 1306-1313.
[28] 刘广义, 钟 宏, 戴塔根, 夏柳荫. 中碱度条件下乙氧羰基硫脲浮选分离铜硫[J]. 中国有色金属学报, 2009, 19(2): 389-396.
LIU Guang-yi, ZHONG Hong, DAI Ta-gen, XAI Liu-yin. Flotation separation of Cu/Fe sulfide minerals by ethoxycarbonyl thiourea under middle alkaline conditions[J]. Transactions of Nonferrous Metals Society of China, 2009, 19(2): 389-396.
[29] 刘广义, 任 恒, 詹金华, 钟 宏. 3, 3'-二乙基-1,1'-一缩二乙二醇二羰基双硫脲的合成、表征与性能[J]. 中国有色金属学报, 2013, 23(1): 290-296.
LIU Guang-yi, REN Heng, ZHAN Jin-hua, ZHONG Hong. Synthesis, characterization and properties of 3, 3’-diethyl-1,1’-oxydiethylenedicarbonyl bis(thiourea)[J]. The Chinese Journal of Nonferrous Metals, 2013, 23(1): 290-296.
[30] 狄 宁, 肖静晶, 刘广义, 黄志强, 曹占芳, 王 帅. N-丁氧丙基-N′-乙氧羰基硫脲对硫化矿物的浮选行为与吸附机理[J]. 中国有色金属学报, 2014, 24(2): 561-568.
DI Ning, XIAO Jing-jing, LIU Guang-yi, HUANG Zhi-qiang, CAO Zhan-fang, WANG Shuai. Adsorption mechanism and flotation behaviors of N-butoxypropyl-N’-ethoxycarbonyl thiourea with sulfide minerals[J]. The Chinese Journal of Nonferrous Metals, 2014, 24(2): 561-568.
(编辑 李艳红)
基金项目:国家高技术研究发展计划资助项目(2013AA064101);国家基础研究发展计划资助项目(2014CB643403);国家自然科学基金资助项目(51474253)
收稿日期:2014-12-09;修订日期:2015-03-28
通信作者:刘广义,教授,博士;电话:0731-88830654;E-mail:guangyiliu@csu.edu.cn