从工业鼓泡流化床锅炉底灰和粉煤灰中浸出重金属评价环境风险
来源期刊:中国有色金属学报(英文版)2016年第1期
论文作者:Risto POYKIO Mikko MAKELA Gary WATKINS Hannu NURMESNIEMI Olli DAHL
文章页码:256 - 264
关键词:底灰;粉煤灰;BCR;萃取;重金属;风险评价指数
Key words:bottom ash; fly ash; BCR; extraction; heavy metals; risk assessment code
摘 要:在鼓泡流化床锅炉(296 MW)中混合燃烧木渣和泥煤产生的底灰和粉煤灰中含有石英(SiO2)、微斜长石(KAlSi3O8)和钠长石(NaAlSi3O8)。X射线衍射不能区分从底灰和粉煤灰中释放的相关重金属。为了评估不同环境条件下底灰和粉煤灰中重金属的释放情况,根据BCR工艺将底灰和粉煤灰中的重金属依次萃取和分离成酸性可溶/可交换(CH3COOH)、可还原(NH2OH-HCl)和可氧化(H2O2/CH3COONH4)相,利用冰醋酸萃取组分和重金属总浓度计算从底灰和粉煤灰中浸出重金属的风险评价指数。浸出研究表明,从底灰和粉煤灰中萃取的不同组分中重金属量不同。从环境和利用前景来看,底灰和粉煤灰中的重金属具有不同水平环境污染风险,只有底灰中的砷呈现非常高的风险。底灰中高风险金属是镉,而粉煤灰中的是砷、镉和硒。
Abstract: The bottom ash and fly ash from the co-combustion of wood residues and peat at a bubbling fluidised bed boiler (296 MW) contained only quartz (SiO2), microcline (KAlSi3O8) and albite (NaAlSi3O8). Thus, X-ray powder diffraction (XRD) was not useful for clarifying the difference in the release of associated heavy metals from ash matrices. In order to assess the release of heavy metals from ashes under changing environmental conditions, they were sequentially extracted and fractionated by the BCR-procedure into acid soluble/exchangeable (CH3COOH), reducible (NH2OH-HCl) and oxidizable (H2O2/CH3COONH4) phases. The CH3COOH extractable fraction in conjunction with the total heavy metals concentrations were used to calculate the risk assessment code values for heavy metals leaching from the ash matrix. The leaching studies indicate that the heavy metals in the bottom ash and fly ash are bound to different fractions with different strengths. From the environmental and utilization perspectives, heavy metals in ashes posed different levels of environmental contamination risk. Only As in the bottom ash posed a very high risk. High risk metals were Cd in the bottom ash as well as As, Cd and Se in the fly ash.
Risto  1, Mikko
1, Mikko  2,3, Gary WATKINS4, Hannu NURMESNIEMI5, Olli DAHL4
2,3, Gary WATKINS4, Hannu NURMESNIEMI5, Olli DAHL4
1. Department of Environmental Protection, City of Kemi, Valtakatu 26, FI- 94100 Kemi, Finland;
2. Division of Biomass Technology and Chemistry, Department of Forest Biomaterials and Technology,
Swedish University of Agricultural Sciences, P.O. Box SLU, SE-90183 Umea, Sweden;
3. Department of Environmental Science and Technology, Tokyo Institute of Technology,
4259 Nagatsuta-cho, Midori-ku, Yokohama 226-8502, Japan;
4. Department of Forest Products Technology, School of Chemical Technology,
Aalto University, P.O. Box 16400, FI-00076 Aalto, Finland;
5. Department of Environmental Protection, Stora Enso Oyj, Veitsiluoto Mill, FI-94800 Kemi, Finland
Received 10 February 2015; accepted 12 October 2015
Abstract: The bottom ash and fly ash from the co-combustion of wood residues and peat at a bubbling fluidised bed boiler (296 MW) contained only quartz (SiO2), microcline (KAlSi3O8) and albite (NaAlSi3O8). Thus, X-ray powder diffraction (XRD) was not useful for clarifying the difference in the release of associated heavy metals from ash matrices. In order to assess the release of heavy metals from ashes under changing environmental conditions, they were sequentially extracted and fractionated by the BCR-procedure into acid soluble/exchangeable (CH3COOH), reducible (NH2OH-HCl) and oxidizable (H2O2/CH3COONH4) phases. The CH3COOH extractable fraction in conjunction with the total heavy metals concentrations were used to calculate the risk assessment code values for heavy metals leaching from the ash matrix. The leaching studies indicate that the heavy metals in the bottom ash and fly ash are bound to different fractions with different strengths. From the environmental and utilization perspectives, heavy metals in ashes posed different levels of environmental contamination risk. Only As in the bottom ash posed a very high risk. High risk metals were Cd in the bottom ash as well as As, Cd and Se in the fly ash.
Key words: bottom ash; fly ash; BCR; extraction; heavy metals; risk assessment code
1 Introduction
Energy generation from biomass fuels such as clean forest residues (i.e., bark, woodchip and sawdust) and peat is a sustainable and environmentally friendly alternative to the use of fossil fuels such as coal or heavy fuel oil, since they are domestic and usually local fuels [1]. Fly ash and bottom ash arise as a result of organic residue combustion for energy recovery. Before the utilization or landfill disposal of ash, its total and extractable heavy metal concentrations have to be known [2]. Although the determination of total concentration of heavy metals in wastes is undoubtedly important to know, the total concentration of heavy metals does not give any information about the solid-phase partitioning and potential bioavailability or mobility of metals in waste [3,4], because these properties are basically dependent on the different chemical forms of binding between the metals and solid phases of the sample [4]. Availability, mobility and the potential risk of contaminants are strongly affected by the manner of appearance of elements, the so-called speciation. Operational fractionation methods like sequential extraction have been applied for a long time to determine the solid phase speciation of heavy metals in industrial samples and today are the most frequently applied analytical methods for studying the solid phase speciation of environmentally relevant heavy metals [5].
The three-step sequential extraction scheme developed by the European Commission’s Community Bureau of Reference (BCR), which includes acetic acid (CH3COOH), hydroxylamine hydrochloride (NH2OH- HCl), and hydrogen peroxide (H2O2) digestion following the ammonium acetate (CH3COONH4) extractable fractions, has been demonstrated to be a successful method for determining the speciation of heavy metals in various environmental samples [6,7]. This extraction scheme distinguishes the metals correspondingly into the acid soluble/exchangeable, reducible and oxidizable fractions. The basic assumption of sequential extraction is that the reagents used are able to dissolve one mineral phase selectively without solubilising the others [8]. This extraction scheme is validated by inter-laboratory comparison experiments and certified reference materials [9]. In view of this, the above mentioned three-step sequential extraction procedure, which is also known as BCR-extraction, is commonly applied in environmental studies for assessing the potential bioavailability of heavy metals in various environmental samples, e.g., in wastes [7].
The objective of this study was to assess the distribution, mobility and potential ecological risks of heavy metals in bottom ash and fly ash fractions originating from the co-combustion of wood residues and peat at an industrial-scale (296 MW) bubbling fluidised bed (BFB) boiler. To address this objective, a three-stage BCR-sequential extraction procedure was used in combination with the risk assessment code (RAC). The RAC considers the different binding strengths of metals in various BCR fractions. It assesses the availability of metals in solution by applying a scale to the percentage of metals in the acetic acid (CH3COOH) extractable fraction [10,11]. This kind of environmental risk assessment, which is based on the use of RAC, has not yet been published for Finnish bottom ash and fly ash fractions, hence the motivation for this study.
2 Experimental
2.1 Collection and preparation of ashes for chemical analyses
The bottom ash and fly ash investigated in this study originated from the co-combustion of wood residues and peat at an industrial-scale (296 MW) power plant which uses a bubbling fluidised bed boiler for energy production. During the sampling period, when bottom ash was sampled from the outlet of BFB boiler and fly ash from the boiler’s electrostatic precipitator (ESP), approximately 61% of the energy produced by the BFB boiler originated from the incineration of clean forest residues (i.e., bark and woodchips), and 39% of it from the incineration of commercial peat, which was of domestic origin. The bark and woodchips were mostly derived from pine (Pinus sylvestris) trees, and the proportion of spruce (Picea abies) was only a few percent. Sampling of the bottom ash and fly ash fractions was carried out over a period of 4 d, with individual daily samples of 1 kg. The sampling period represented normal process operating conditions for the power plant, for instance in terms of O2 content and temperature. The incineration temperature in the BFB boiler is about 850 °C, while in the electrostatic precipitator it is about 145 °C. After sampling, the samples were stored in plastic bags in a refrigerator (4 °C). A coning and quartering method [12] was repeatedly applied to reducing the ash samples to a size suitable for conducting laboratory analyses. All chemical analyses were carried out at a sieved (<2 mm) sample.
2.2 Determination of relevant physical and chemical properties of ashes
To determine the mineralogical composition of the ashes, X-ray diffractograms of powdered samples were obtained with a Siemens D 5000 diffractometer (Siemens AG, Karlsruhe, Germany) using Cu Kα radiation. The scan was run from 2° to 80° (2θ scale), with increments of 0.02° and a counting time of 1.0 s per step. The operating conditions were 40 kV and 40 mA. Peak identification was carried out with the DIFFRACplus BASIC Evaluation Package PDFMaint 12 (Bruker axs, Germany) and the software package ICDD PDF-2 Release 2006 (Pennsylvania, USA).
The pH of the ashes was determined using a pH/EC analyser equipped with a Thermo Orion Sure Flow pH electrode (Turnhout, Belgium). The determination of pH was carried out according to European standard SFS-EN 12880 at a solid-to-liquid (i.e., ultrapure H2O) ratio of 1:5. The moisture content of the ashes was determined according to European standard SFS-EN 12048 [14]. Determination of the dry matter content of the ashes was carried out according to European standard SFS-EN 12880, in which a sample was dried overnight to a constant mass in an oven at 105 °C. A comprehensive review of the standards, analytical methods and instrumentation is presented in our previous paper [13].
2.3 Determination of total nutrient and heavy metal concentrations in ashes
To determine the total nutrient (P, Ca, Na, K, Mg, Zn and Cu) and heavy metal concentrations in the ashes, the dried sample was digested with a mixture of HCl (3 mL) and HNO3 (9 mL) in a CEM Mars 5 microprocessor-controlled microwave oven with a CEM HP 500 Teflon vessels (CEM Corp., Matthews, USA) using USEPA method 3051A [14]. The cooled solutions were transferred to 100 mL volumetric flasks and the solutions diluted to volume with ultrapure water. The ultrapure water was generated by an Elgastat Prima reverse osmosis and Elgastat Maxima ion exchange water purification system (Elga Ltd; Bucks, England). All reagents and acids were superpure or of pro-analysis quality. Apart from Hg, the total nutrient (P, Ca, Na, K, Mg, and Zn) and heavy metal concentrations in ashes were determined with ICP-OES (Thermo Fisher Scientific iCAP6500 Duo, UK). The concentration of Hg in the ashes was determined with a Perkin Elmer Aanalyst 700 cold-vapour AAS equipped with a Perkin Elmer FIAS 400 and AS 90 plus auto-sampler. A more comprehensive review of the standards, analytical method and instrumentation was provided in our previous paper [13].
2.4 Determination of solid phase speciation of heavy metals in ashes
The solid phase speciations of heavy metals in the bottom ash and fly ash fractions were carried out according to three-step sequential extraction BCR scheme, which is fully described in Refs. [7,15]. Briefly, the extraction was performed by sequentially extraction of 0.5 g of the bottom ash and fly ash in a 50 mL centrifuge tube at room temperature using a mechanical shaker with 20 mL 0.11 mol/L acetic acid (CH3COOH) (16 h shaking), 20 mL 0.1 mol/L hydroxylamine hydrochloride (NH2OH-HCl) in nitric acid (HNO3) medium (16 h shaking), a two-fold 5 mL 9.8 mol/L hydrogen peroxide (H2O2) (pH 2-3) digestion at 85 °C and subsequently extraction with 25 mL 1 mol/L ammonium acetate (CH3COONH4) (16 h shaking). In order to minimize possible chemical and/or microbiological changes in the ashes, the extraction was carried out using a sample as such, instead of a dried sample, since according to KOSSON et al [16], it is preferable to avoid sample drying before extraction.
After shaking, the suspension was each time centrifuged at 3000 r/min and the supernatant solution was decanted, filtered through a 0.45 μm membrane filter (47 mm diameter; Schleicher & Schuell, Dassel, Germany) and put aside for analysis. After each of the first two steps, the ash residue was washed each time with 10 mL ultrapure H2O (15 min). After centrifuging, the washing solution was carefully decanted and discarded. In order to avoid loss between the extraction stages, the filters and adhering ash particles from the previous extraction stage were also included in the next stage. After the addition of 200 μL of 65% HNO3 to the supernatant phase, it was stored under dark and cool (4 °C) conditions in the laboratory. The heavy metal concentrations in the extracts were determined with ICP-OES (Thermo Fisher Scientific iCAP6500 Duo, UK).
2.5 Quality control of BCR extraction procedure
The accuracy of the BCR extraction procedure consisted of calibration standards, standard solutions analyzed as an unknown (quality control solutions), method blanks and a certified reference material of lake sediment BCR-701, which is developed especially for BCR extraction by the Institute for Reference Materials and Measurements of the European Commission (Geel, Belgium). The BCR extraction procedure and methods for heavy metals determination in the extracts of the reference material were the same as for the ash fractions.
3 Results and discussion
3.1 Relevant physical and chemical properties of ashes
According to the results in Table 1, the pH values of bottom ash and fly ash were 12.7 and 12.8, respectively. The high alkaline pH value of the ashes agrees with the findings of PAN and EBERHARDT [17], who reported that the high pH value is consistent with the high concentrations of alkaline (Na, K) and alkaline earth metals (Mg, Ca, Ba). According to van HERCK and VANDECASTEELE [18], the high pH indicates the occurrence of basic metal salts, oxides, hydroxides, and/or carbonates in the ash. However, the X-ray diffraction powder (XRD) data of crystalline phases in the ashes do not support the findings of van HERCK and VANDECASTEELE [18], because they contained only silicate minerals such as silicon oxide (quartz; SiO2), potassium aluminium silicate (microcline; KAlSi3O8), and sodium aluminium silica (albite; NaAlSi3O8). This is because the X-ray spectrometer is normally operated with a limit of detection of 1%-2% and is not able to identify non-crystallised phases. The higher amount of quartz in the bottom ash fraction compared with that of the fly ash fraction was to be expected because the bed material of BFB-boiler consists of silica sand. The silicate minerals in these ash fractions may also be partly due to sand and soil particle contamination of forest residues during harvesting, transportation and handling [19]. In addition, it may partly derive from the decomposition of plant tissue-derived Si-based minerals during incineration, such as phytolith (SiO2 × nH2O), which is often a structural component of plant tissues, deposited between and within plant cells [20].
Table 1 Relevant physical and chemical properties (n=1), total (USEPA 3051A) nutrient concentrations (n=3) and crystalline mineral phases in bottom ash and fly ash fractions (n means number of replicates)
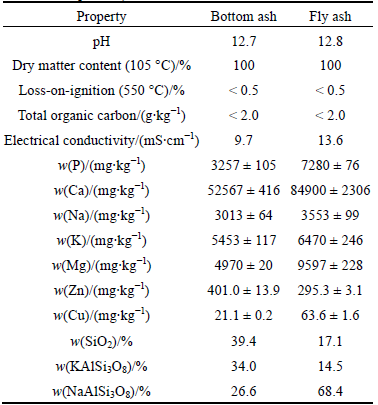
According to Ref. [21], quartz and microcline are typical mineral phases in finnish ashes originating from the co-combustion of wood and peat, although oxide and sulphate are also observed. The high (100%) dry matter content of the ash fractions is a disadvantage if these residues are to be physically handled or transported to do their likely dusting characteristics. However, it is possible to avoid dust problems if ashes are watered before handling. The very low total organic carbon (<2.0 g/kg) and loss-on-ignition (<0.5%) values in the ash fractions indicate the efficient combustion of organic matter in our BFB-boiler. Additionally, as indicated by the electrical conductivity of 13.6 mS/cm, the extraction solution of fly ash had a higher ionic strength than that of the bottom ash (9.7 mS/cm).
3.2 Total nutrient and heavy metal concentrations in ashes
The total nutrient (P, Ca, Na, K, Mg, Zn and Cu) and heavy metal concentrations in the bottom ash and fly ash fractions expressed on a dry mass and means of triplicate samples are presented in Tables 1 and 2, respectively. Except for Zn which was 295.3 mg/kg in the fly ash and 401.0 mg/kg in the bottom ash, the other nutrient concentrations in the fly ash were between 1.2 (Na and K) and 3.0 (Cu) times higher than those in the bottom ash (Table 1). Although the Zn concentration in the fly ash is generally elevated compared with that in the bottom ash, we have observed the opposite phenomenon also in our previous study [13], in which wood residues and peat were also co-incinerated in a BFB-boiler. This phenomenon is probably partly due to the semi-volatile property of Zn [17]. If we disregard those heavy metals where the concentrations were below the detection limits, the heavy metals concentrations in the fly ash were between circa 1.5 (Mn) and 5.9 (Pb) times higher than those in the bottom ash (Table 2). The high enrichment of Pb in the fly ash is due to the fact that it is volatile and forms gaseous species even at relatively low temperatures ranging from 550 to 725 °C [17], which are much lower than that of the BFB-boiler (850 °C) investigated in this study. According to BOGUSH et al [22], the presence of elements in specific species and their concentrations in the fly ash definitely depends on the type of combusted material, its content of chloride, sulphur and water, the oxidation level, incineration time and temperature, as well as the boiling point of the different phases. Since this phenomenon is well known and reported elsewhere [20-23], we do not focus on it in the present work.
3.3 Solid phase speciation of heavy metals in ash fractions
Metal speciation and solubility affect the mobility and bioavailability of metals significantly [24]. Metal speciation using a sequential extraction procedure is applied to estimating the potential release of metals under changing environmental conditions [25]. Table 2 presents the chemical speciation of heavy metals in the bottom ash and fly ash fractions into acid soluble/exchangeable (step 1: CH3COOH), reducible (step 2: NH2OH-HCl in nitric acid medium) and oxidizable (step 3: H2O2/CH3COONH4) phases.However, Hg is not fractionated, because of the very low total Hg concentrations in both ash fractions.
The extraction step 1, in which acetic acid (CH3COOH) is used as extractant, releases relatively weakly adsorbed heavy metals from the solid surface of ash particles that can be released by ion-exchangeable processes [4,26].Metals in this fraction are easily mobile and are assumed to be highly available. According to KOUASSI et al [11], this phase may pose the main risk for the environment because metals in this fraction are easily leached in neutral or slightly acidic conditions. If we compare the extractable heavy metal concentration into the acid soluble/exchangeable (step 1) phase (Table 2), we notice that, the highest individual extractable concentrations were observed for Zn (107 mg/kg), Mn (88.3 mg/kg), Al (25.5 mg/kg) and Ba (16.8 mg/kg) in the bottom ash, whereas in the fly ash the highest concentrations were as follows: Mn(239 mg/kg), Zn (32.3 mg/kg) and Al (19.7 mg/kg).
The extraction step 2, in which a reducing agent of hydroxylamine hydrochloride (NH2OH-HCl) is used as extractant, releases heavy metals from the matrix, which are bound to easily reducible phases, e.g., Fe and Mn oxides [5,27]. This fraction is moderately mobile. The metals in this fraction are sensitive to redox potential changes and represent a fraction which can be solubilised under reducible conditions [4,28] or caused by oxygen deficiency [29].
If we compare the extractable heavy metal concentration into the reducible (step 2) phase, we notice that, the highest individual extractable concentrations were observed both in the bottom ash and fly ash fractions for the same heavy metals as follows: Al (8170 and 7970 mg/kg), Fe (5430 and 3980 mg/kg), Mn (660 and 495 mg/kg), and Ba (270 and 480 mg/kg).
Table 2 Total (USEPA 3051A, n=3) and extractable (n=1) heavy metal concentrations in bottom ash and fly ash fractions among fractions CH3COOH (step 1), NH2OH-HCl in nitric acid medium (step 2) and H2O2/CH3COONH4 (step 3)
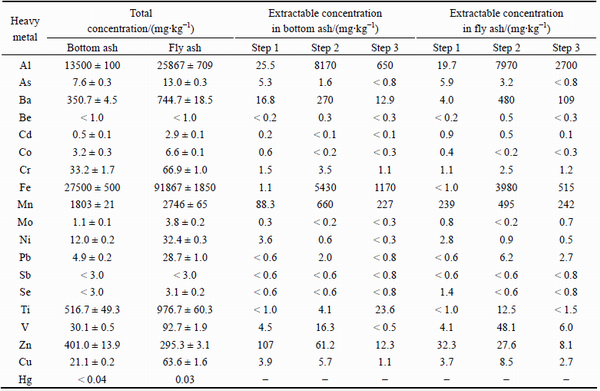
The extraction step 3, in which the sample is first digested with hydrogen peroxide (H2O2) and after that extracted with ammonium acetate (CH3COONH4), releases those heavy metals from the matrix, which are complexed by organics and/or associated with sulphides [27]. The metals in this phase would be released if conditions become oxidizing [4]. According to KARAKASEVA et al [28], this fraction is temporarily inaccessible and it can be solubilized by chemical oxidation. However, according to SMICHOWSKI et al [30], the organic fraction released in this extraction step is not considered to be very mobile and available. If we compare the extractable heavy metal concentration into oxidizable (step 3) phase, we notice that, the highest individual extractable concentrations were observed for different heavy metals in the bottom ash and fly ash fractions. In the bottom ash, they were as follows: Fe (1170 mg/kg), Al (650 mg/kg), Mn (227 mg/kg) and Ti (23.6 mg/kg), whereas in the fly ash they were as follows: Al (2700 mg/kg), Fe (515 mg/kg), Mn (242 mg/kg) and Ba (109 mg/kg).
3.4 Risk assessment of heavy metals leaching from ash fractions
The mobility and stability of heavy metals in waste depend on binding forms in different phases. It is evident from the results of the BCR-extraction study that the heavy metals in the bottom ash and fly ash fractions investigated in this study are bound to different solid phases with different strengths. Risk assessment code (RAC), is a method which can be used for evaluating the potential environmental risk of heavy metals leaching from the sample matrix [10,31]. It classifies the risk levels based on the chemical speciation of heavy metals using the BCR-sequential extraction procedure. The RAC determines the availability of heavy metals in the ash residue by applying a scale to the percentage of heavy metals in the exchangeable/acid-extractable phase (step 1: CH3COOH extraction), because this extraction phase is considered to be the most unstable and reactive and has therefore greater potential for adverse effects on the environment compared to the other phases [10,31]. According to RAC classification [10,31], for any metal, when the ratio of the heavy metal concentration in exchangeable/acid-extractable phase (step 1: CH3COOH extraction) is less than 1% compared with the total metal concentration of this metal, the heavy metal has no adverse impact on the environment. If the ratio is between 1% and 10%, a low risk is defined, while medium risk is 11%-30%, and 31%-50% indicates a high risk. A higher ratio than 50% of the metal indicates that the hazard is very high and the heavy metal is very easily to release from the sample matrix and poses a very high risk to the environment [10,31].
If we disregard those heavy metals where concentrations were below the detection limits, according to results in Table 3, only As in the bottom ash poses a very high environmental risk, whereas Cd both in the bottom ash and fly ash poses a high risk, as well as As and Se in the fly ash. Medium risk is caused by Mo and Zn in the fly ash and also by Co, Mo, Ni, V, Zn and Cu in the bottom ash. In the bottom ash, heavy metals such as Ba, Cr and Mn as well as Co, Cr, Mn, Ni, V and Cu in the fly ash exhibited a low risk. Although the total concentrations of Al (13500 mg/kg) and Fe (27500 mg/kg) in the bottom ash, as well as Al (25867 mg/kg) and Ba (744.7 mg/kg) in the fly ash are very high, they pose no environmental risk, because their leachabilities into the acid soluble/exchangeable (CH3COOH) phase are low enough not to cause higher risk. Our results agree with the findings of PAN et al [31] that the heavy metal in ash which has a low total concentration may pose a very high or high environmental risk if it is loosely bound into the ash matrix and easily extractable using CH3COOH.
Table 3 Risk assessment category and classification of risk assessment code (RAC) values [10,31] as well as environmental risk of heavy metals leaching from bottom ash and fly ash fractions investigated in this study
![说明: C:\Users\Administrator\AppData\Roaming\Tencent\Users\943171351\QQ\WinTemp\RichOle\$Q{L7EK@Q$_CMU1Z])QGZSP.png](/web/fileinfo/upload/magazine/12511/310843/image006.jpg)
3.5 Quality control results of BCR-701 certified reference material
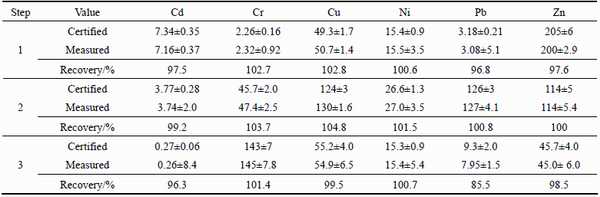
The quality control of the BCR-extraction procedure was evaluated using the certified reference material BCR-701 (lake sediment). Validation of the three-step sequential extraction procedure used in this study was carried out using this reference material, because this reference material is certified in an inter-certification campaign for extractable Cd, Cr, Cu, Ni, Pb and Zn in the three steps of the BCR scheme [32]. The extractable concentrations and recoveries in each extraction step of the standard reference material BCR-701 are presented in Table 4. The accuracy of concentrations in each extraction step was assessed by the five replicate analyses.
Except for Pb in the third extraction step (H2O2/CH3COONH4), our extractable concentrations (mean ± standard deviation) of other heavy metals in the reference material are generally in good agreement with the certified values and are considered satisfactory. In the first extraction step (CH3COOH), the recoveries of metals were between 96.8% (Pb) and 102.7% (Cu). Furthermore, in the second extraction step (NH2OH- HCl), the recoveries were between 99.2% (Cd) and 104.8% (Cu), and in the third extraction step (H2O2/CH3COONH4) they were between 85.5% (Pb) and 101.4% (Cr). This suggests that the extraction procedure and the instrumental analysis of the extraction were accurate.
Except for Pb in the third extraction step, our recovery values for the reference material BCR-701 agree well with the findings of OZCAN and ALTUNDAG [33], who reported values between 92.5% and 103.4% for heavy metals in this reference material, as well as those of SUNGUR et al [34], who observed values between 92.6% and 104.0%. However, our relatively low recovery value of 85.5% for Pb in the third extraction step is consistent with the value of 88.2% observed by SILVA et al [35] and with the value of 84.9% observed both by LACAL et al [36] and by CAPPUYNS et al [37]. The exact reasons for low Pb in the third extraction step is not possible to know.According to CHI [38], the low recovery value for Pb could be explained for example by the possible matrix effects, spectroscopic interference during analysis and incorrect concentration or volume of reagent added, as well as the washing of samples with water between extraction steps.
Table 4 Certified and measured (n=5) values of heavy metals in reference material BCR-701 (lake sediment) between fractions CH3COOH (step 1), NH2OH-HCl in nitric acid medium (step 2) and H2O2/CH3COONH4 (step 3) (All concentrations are expressed as mg/kg on a dry mass basis)
4 Conclusions
1) The bottom ash and fly ash from the co-combustion of wood residues (i.e. bark, woodchip and sawdust) and peat at a large-sized (296 MW) bubbling fluidized bed (BFB) boiler were investigated to determine the characteristics, leaching behaviour and risks of heavy metals to biota. The X-ray powder diffraction was not able to clarify the difference in the mineral composition of the ashes because both ashes contained only quartz (SiO2), microcline (KAlSi3O8) and albite (NaAlSi3O8). Therefore, for clarifying the difference in the release of associated heavy metals from ash matrices, the BCR sequential extraction procedure, in which heavy metals were partitioned between acid soluble/exchangeable (CH3COOH), reducible (NH2OH-HCl) and oxidizable (H2O2/CH3COONH4) phases, was applied.
2) The leaching studies indicate that the heavy metals in the bottom ash and fly ash are bound to different fractions with different strengths. From the environmental and utilization perspectives, heavy metals in ashes pose different levels of environmental contamination risk. According to the risk assessment codes, which were applied to assessing the environmental risk of the leaching of each heavy metal from the ashes, only two metals in the bottom ash (Al and Fe) and fly ash (Al and Ba) showed no risk for the environment if they leach out from the ash matrix. Low risk is caused by three metals (Ba, Cr and Mn) in the bottom ash and six metals (Co, Cr, Mn, Ni, V and Cu) in the fly ash. Medium risk metals were Co, Mo, Ni, V, Zn and Cu in the bottom ash and Mo and Zn in the fly ash. Cadmium (Cd) in the bottom ash poses a high environmental risk, whereas in the fly ash they are As, Cd and Se. Only As in the bottom ash posed a very high risk.
3) The results of this study show that the three-step BCR sequential extraction procedure together with a risk assessment codes can be used as valuable tools to provide information about the differentiation of the relative bonding strength of the metals in ash matrix to various solid phases and the potential reactivity of the metals under different physic-chemical and environmental conditions. This kind of information can be utilized for example in environmental permit applications and in the decision making process of environmental authorities because in Finland and elsewhere in the EU, the utilization of ash needs an environmental permit.
Acknowledgement
The authors wish to thank the technical staff of Ahma YMPARISTO OY, who kindly conducted all the chemical analyses. Thanks also belong to the technical staff at the Institute of Electron Optics of University of Oulu, for the XRD data.
References
[1] SOKKA L, PAKARINEN S, MELANEN M. Industrial symbiosis contributing to more sustainable energy use―An example from the forest industry in Kymenlaakso, Finland [J]. Journal of Cleaner Production, 2011, 19(4): 285-293.
[2] KILPIMAA S, KUOKKANEN T, LASSI U. Characterization and utilization potential of wood ash from combustion process and carbon residue from gasification process [J]. Bioresources, 2013, 8(1): 1011-1027.
[3]  Study of application of BCR sequential extraction procedure for fractionation of heavy metal content of soils, sediments, and gravitation dusts [J]. Toxicological & Environmental Chemistry, 2010, 92(3): 429-441.
Study of application of BCR sequential extraction procedure for fractionation of heavy metal content of soils, sediments, and gravitation dusts [J]. Toxicological & Environmental Chemistry, 2010, 92(3): 429-441.
[4] da SILVA L, COELHO L M, ROSOLEN V, COELHO N M. Metal speciation in surface sediments of the Uberabinha river in Uberlandia, MG state, Brazil [J]. Journal of Brazilian Chemical Society, 2011, 22(11): 2094-2100.
[5] van HERREWEGHE S, SWENNEN R, VANDECASTEELE C, CAPPUYNS V. Solid phase speciation of arsenic by sequential extraction in standard reference materials and industrially contaminated soil samples [J]. Environmental Pollution, 2003, 122(3): 323-342.
[6] MUKHTAR A, LIMBECK A. Recent developments in assessment of bio-accessible trace metal fractions in airborne particulate matter: A review [J]. Analytica Chimica Acta, 2013, 774: 11-25.
[7] FILGUEIRAS A, LAVILLA I, BENDICHO C. Chemical sequential extraction for metal partitioning in environmental solid samples [J]. Journal of Environmental Monitoring, 2002, 4(6): 823-857.
[8] ESTIFANOS S. Heavy metal pollution assessment by partial geochemical extraction technique [J]. Momona Ethiopian Journal of Science, 2013, 5(1): 71-88.
[9] URE A M, QUEVAUVILLER Ph, MUNTAU H, GRIEPINK B. Speciation of heavy metals in soils and sediments. An account of the improvement and harmonization of extraction techniques undertaken under the auspices of the BCR of the commission of the European communities [J]. International Journal of Environmental analytical Chemistry, 1993, 51(1-4): 135-151.
[10] ZHOU Y, NING X, LIAO X, LIN M, LIU J, WANG J. Characterization and environmental risk assessment of heavy metals found in fly ashes from waste filter bags obtained from a Chinese steel plant [J]. Ecotoxicology and Environmental Safety, 2013, 95: 130-136.
[11] KOUASSI N L B, YAO K M, TROKOUREY A, SORO M B. Preliminary assessment of cadmium mobility in surface sediments of a tropical estuary [J]. Bulletin of the Chemical Society of Ethiopia, 2014, 28(2), 245-254.
[12] GERLACH R, DOBB D, RAAB G, NOCERINO J. Gy sampling theory in environmental studies 1. Assessing soil splitting protocols [J]. Journal of Chemometrics, 2002, 16(7): 321-328.
[13] NURMESNIEMI H, M,
M,  R, MANSKINEN K, DAHL O. Comparison of the forest fertilizer properties of ash fractions from two power plants of pulp and paper mills incinerating biomass-based fuels [J]. Fuel Processing Technology, 2012, 104: 1-6.
R, MANSKINEN K, DAHL O. Comparison of the forest fertilizer properties of ash fractions from two power plants of pulp and paper mills incinerating biomass-based fuels [J]. Fuel Processing Technology, 2012, 104: 1-6.
[14] CHEN M, MA L Q. Comparison of four USEPA digestion methods for trace metal analysis using certified and Florida soils [J]. Journal of Environmental Quality, 1998, 27(6): 1294-1300.
[15] MANSKINEN K,  R, NURMESNIEMI H. Comparison of the total and fractionated heavy metal and sulphur concentrations in bottom ash and fly ash from a large-sized (120 MW) power plant of a fluting board mill [J]. Chemija, 2011, 22(1): 46-55.
R, NURMESNIEMI H. Comparison of the total and fractionated heavy metal and sulphur concentrations in bottom ash and fly ash from a large-sized (120 MW) power plant of a fluting board mill [J]. Chemija, 2011, 22(1): 46-55.
[16] KOSSON D S, van der SLOOT H A, SANCHEZ F, CARRABRANTS A C. An integrated framework for evaluating leaching in waste management and utilization of secondary materials [J]. Environmental Engineering Science, 2002, 19(3): 159-204.
[17] PAN H, EBERHARDT T L. Characterization of fly ash from the gasification of wood and assessment for its application as a soil amendment [J]. Bioresources, 2011, 6(4): 3987-4004.
[18] van HERCK P, VANDECASTEELE C. Evaluation of the use of a sequential extraction procedure for the characterization and treatment of metal containing solid waste [J]. Waste Management, 2001, 21(8): 685-694.
[19] STEENARI B M, LINDQVIST O. Fly ash characteristics in co-combustion of wood with coal, oil or peat [J]. Fuel, 1999, 78(4): 479-488.
[20] HUMPHREYS G, RAVEN D, FIELD H. Wood-ash stone in Angophora costata (Gaertn.) J. Britt. following Sydney bushfires [J]. Australian Forestry, 2004, 67(1): 39-43.
[21] MANSKINEN K. Utilisation aspects of ashes and green liquor dregs from an integrated semichemical pulp and board mill [D]. Unigrafia Oy, Helsinki, Finland: Department of Forest Products Technology, Aalto University, 2013.
[22] BOGUSH A, STEGEMANN J A, WOOD I, ROY A. Element composition and mineralogical characterisation of air pollution control residue from UK energy-from-waste facilities [J]. Waste Management, 2015, 36: 119-129.
[23] STEENARI B M, LINDQVIST O. Stabilisation of biofuel ashes for recycling to forest soil [J]. Biomass and Bioenergy, 1997, 13(1-2): 39-50.
[24] WANG Shao-feng, JIA Yong-feng, WANG Shu-ying, WANG Xin, WANG He, ZHAO Zhi-xin, LIU Bing-zhu. Fractionation of heavy metals in shallow marine sediments from Jinzhou Bay, China [J]. Journal of Environmental Sciences, 2010, 22(1): 23-31.
[25] RYAN P C, HILLIER S, WALL A J. Stepwise effects of the BCR sequential chemical extraction procedure on dissolution and metal release from common ferromagnesian clay minerals: A combined solution chemistry and X-ray powder diffraction study [J]. Science of the Total Environment, 2008, 407(1): 603-614.
[26] YANG J, CAO L L, WANG J, LIU C L, HUANG C G, CAI W X, FANG H, PENG X. Speciation of metals and assessment of contamination in surface sediments from Daya Bay, South China Sea [J]. Sustainability, 2014, 6(12): 9096-9113.
[27] HELTAI G, Various fractionation procedures in study of heavy metals mobility in the environment [J]. Ecological Chemistry and Engineering S, 2011, 18(1): 55-65.
Various fractionation procedures in study of heavy metals mobility in the environment [J]. Ecological Chemistry and Engineering S, 2011, 18(1): 55-65.
[28] KARAKASEVA E, BOEV B, ZAJKOVA V. Total and extractable forms of Cu, Zn, Ni, Cr, Pb and Fe in vineyard soil (Valandovo Valley, Macedonia) determined by a sequential extraction procedure [J]. Macedonian Journal of Chemistry and Chemical Engineering, 2012, 31(2): 271-283.
[29] WIELGOSINSKI G, WASIAK D, ZAWADZKA A. The use of sequential extraction for assessing environmental risks of waste incineration bottom ash [J]. Ecological Chemistry and Engineering S, 2014, 21(3): 413-423.
[30] SMICHOWSKI P, POLLA G, GOMEZ D. Metal fractionation of atmospheric aerosols via sequential chemical extraction: A review [J]. Analytical and Bioanalytical Chemistry, 2005, 381(2): 302-316.
[31] PAN Y, WU Z M, ZHOU J Z, ZHAO J, RUAN X X, LIU J Y, QIAN G R. Chemical characteristics and risk assessment of typical municipal solid waste incineration (MSWI) fly ash in China [J]. Journal of Hazardous Materials, 2013, 261: 269-276.
[32] FOSTNER U. Traceability of sediment analysis [J]. Trends in Analytical Chemistry, 2004, 23(3): 217-236.
[33] OZCAN N, ALTUNDAG H. Speciation of heavy metals in street dust samples from Sakarya I. Organized industrial district using the BCR sequential extraction procedure by ICP-OES [J]. Bulletin of the Chemical Society of Ethiopia, 2013, 27(2): 205-212.
[34] SUNGUR A, SOYLAK M, OZCAN H. Investigation of heavy metal mobility and availability by the BCR sequential extraction procedure: Relationship between soil properties and heavy metals availability [J]. Chemical Speciation & Bioavailability, 2014, 26(4): 219-230.
[35] da SILVA G S, do NASCIMENTO A S, de SOUSA E R, MARQUES E P, MARQUES A L B, CORREA L B, SILVA G S. Distribution and fractionation of metals in mangrove sediment from the Tibiri River estuary on Maranhao Island [J]. Revista Virtual de Quimica, 2014, 6(2): 323-336.
[36] LACAL J, da SILVA M P, GARCIA R, SEVILLA M T, PROCOPIO J R, HERNANDEZ L. Study of fractionation and potential mobility of metal in sludge from pyrite mining and affected river sediments: Changes in mobility over time and use of artificial ageing as a tool in environmental impact assessment [J]. Environmental Pollution, 2003, 124(2): 291-305.
[37] CAPPUYNS V, SWENNEN R, NICLAES M. Application of the BCR sequential extraction scheme to dredged pond sediments contaminated by Pb-Zn mining: A combined geochemical and mineralogical approach [J]. Journal Geochemical Exploration, 2007, 93(2): 78-90.
[38] CHI C Y. An investigation of the bioavailability of heavy metals in tailing run-off sediment at Devon Great Consols [J]. The Plymouth Student Scientist, 2013, 6(2): 211-309.
Risto  1, Mikko
1, Mikko  2,3, Gary WATKINS4, Hannu NURMESNIEMI5, Olli DAHL4
2,3, Gary WATKINS4, Hannu NURMESNIEMI5, Olli DAHL4
1. Department of Environmental Protection, City of Kemi, Valtakatu 26, FI- 94100 Kemi, Finland;
2. Division of Biomass Technology and Chemistry, Department of Forest Biomaterials and Technology,
Swedish University of Agricultural Sciences, P.O. Box SLU, SE-90183 Umea, Sweden;
3. Department of Environmental Science and Technology, Tokyo Institute of Technology,
4259 Nagatsuta-cho, Midori-ku, Yokohama 226-8502, Japan;
4. Department of Forest Products Technology, School of Chemical Technology,
Aalto University, P.O. Box 16400, FI-00076 Aalto, Finland;
5. Department of Environmental Protection, Stora Enso Oyj, Veitsiluoto Mill, FI-94800 Kemi, Finland
摘 要:在鼓泡流化床锅炉(296 MW)中混合燃烧木渣和泥煤产生的底灰和粉煤灰中含有石英(SiO2)、微斜长石(KAlSi3O8)和钠长石(NaAlSi3O8)。X射线衍射不能区分从底灰和粉煤灰中释放的相关重金属。为了评估不同环境条件下底灰和粉煤灰中重金属的释放情况,根据BCR工艺将底灰和粉煤灰中的重金属依次萃取和分离成酸性可溶/可交换(CH3COOH)、可还原(NH2OH-HCl)和可氧化(H2O2/CH3COONH4)相,利用冰醋酸萃取组分和重金属总浓度计算从底灰和粉煤灰中浸出重金属的风险评价指数。浸出研究表明,从底灰和粉煤灰中萃取的不同组分中重金属量不同。从环境和利用前景来看,底灰和粉煤灰中的重金属具有不同水平环境污染风险,只有底灰中的砷呈现非常高的风险。底灰中高风险金属是镉,而粉煤灰中的是砷、镉和硒。
关键词:底灰;粉煤灰;BCR;萃取;重金属;风险评价指数
(Edited by Xiang-qun LI)
Corresponding author: Risto  ; Tel: +358-16-259673; Fax: +358-16-259481; E-mail: risto.poykio@kemi.fi
; Tel: +358-16-259673; Fax: +358-16-259481; E-mail: risto.poykio@kemi.fi
DOI: 10.1016/S1003-6326(16)64112-2

