���̸�������������������������Һ�з�����
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2017���11��
�������ߣ����Ʒ� ������ ������ ����ΰ
����ҳ�룺2492 - 2502
�ؼ��ʣ����٣���������Һ�����̸������������
Key words��tungsten removal; molybdate solution; Fe-Mn binary oxide; adsorption
ժ Ҫ�����ǵ��١��ⲻͬ�ĵ���ѧ������Ϊ���о����̸���������(FMBO)����������(Fe(OH)3)�Ͷ�������(MnO2)��������������������Һ�з����١�������������̸���������(FMBO)���ʺϴ���������Һ�г��١����̸�����������һ���Ͽ�����pH��Χ��(6.9~11.3)���нϺõ������ԣ����Եõ���ȥ���ʳ���80%������ʧС��3%�����⣬���̸��������������3 mol/L���������������������ٵ�ȥ���ʾ���5������-����-������Ȼ���Ա��ֲ��䡣�������̸�������������������������Һ�г��پ������õ�Ӧ��DZ����
Abstract: Considering the different geochemical enrichment behaviors of W and Mo, Fe-Mn binary oxide (FMBO), ferric hydroxide (Fe(OH)3) and manganese dioxide (MnO2) were studied to separate W from molybdate solution, respectively. The experimental results demonstrated that Fe-Mn binary oxide (FMBO) was the most suitable adsorbent for the separation. Under a wide pH (6.9-11.3) region, more than 80%W removal efficiency and less than 3%Mo loss could be obtained. In addition, the Fe-Mn binary oxide adsorbent can be regenerated by treating with 3 mol/L NaOH, and the W adsorption efficiency was retained after five adsorption-desorption-regeneration cycles. All these indicate that the Fe-Mn binary oxides have the potential for the separation of W from molybdate solution.
Trans. Nonferrous Met. Soc. China 27(2017) 2492-2502
Yun-feng SONG1, Li-hua HE1,2, Xing-yu CHEN1, Zhong-wei ZHAO1
1. School of Metallurgy and Environment, Central South University, Changsha 410083, China;
2. Institute of Nuclear and New Energy Technology, Tsinghua University, Beijing 100084, China
Received 19 June 2016; accepted 24 May 2017
Abstract: Considering the different geochemical enrichment behaviors of W and Mo, Fe-Mn binary oxide (FMBO), ferric hydroxide (Fe(OH)3) and manganese dioxide (MnO2) were studied to separate W from molybdate solution, respectively. The experimental results demonstrated that Fe-Mn binary oxide (FMBO) was the most suitable adsorbent for the separation. Under a wide pH (6.9-11.3) region, more than 80%W removal efficiency and less than 3%Mo loss could be obtained. In addition, the Fe-Mn binary oxide adsorbent can be regenerated by treating with 3 mol/L NaOH, and the W adsorption efficiency was retained after five adsorption-desorption-regeneration cycles. All these indicate that the Fe-Mn binary oxides have the potential for the separation of W from molybdate solution.
Key words: tungsten removal; molybdate solution; Fe-Mn binary oxide; adsorption
1 Introduction
Tungsten content in molybdenum products needs to be strictly controlled, since the trace contaminate of W in molybdenum material would reduce its physical and mechanical properties [1]. However, with the continuous and excessive consumption of high quality molybdenum resources, molybdenum industry has to exploit and utilize the refractory Mo-W blend ores of which the amount of W is much higher. For instance, the mass ratio of W to Mo in Ni-Mo deposits can be as high as 1:10. Therefore, the efficient removal of W from molybdate solution is very crucial for the successful utilization of these resources.
The separation of W and Mo is a very difficult process due to their similar extraction behaviors in most cases [2]. There are a number of approaches available for the separation of molybdenum from tungstate solution, such as selectively precipitation [3,4] solvent extraction [5], ion exchange [6] and adsorption [7]. On the other hand, the studies of removing tungsten from molybdate solution were limited to only a few cases mainly on solvent extraction [8] and ion-exchange route [9,10]. Solvent extraction route is a costlier one [11], whereas the ion-exchange process has its own disadvantages such as complicated operation process and poor selectivity [8].
Recently, the adsorption methods aiming to remove a small quantity of certain metal ions by using hydrated oxides or hydroxides have been proved to be promising due to their low cost, environmental friendly, simple and effective operability [12-18]. Thus, it is reasonable to make an assumption that the method may have the potential for tungsten removal from molybdate solution as well, and the key of a successful separation of W and Mo is to find an efficient adsorbent.
Moreover, the geochemical behaviors of W and Mo have great referential significance concerning the separation of W and Mo. An analysis of the mineralogical occurrence of W and Mo showed that oxyphile element W can exist naturally with iron and manganese, forming compounds such as ferberite (FeWO4), huebnerite (MnWO4) and wolframite ((Fe,Mn)WO4) [19,20], while sulfophilic element Mo exists mainly as molybdenite (MoS2) in nature. Besides, researchers have found that the content of W increases markedly in limonite, pyrolusite and ocean manganese nodules, all of which contain abundant (hydr)oxides of iron and manganese [21,22].
The facts mentioned above imply that W might be removed from molybdate solution by (hydr)oxides of iron or manganese. However, when the removal of W from molybdate solution was carried out by iron and manganese compounds separately [23-29], all the methods have some drawbacks, either insufficient separation or high loss of Mo or impurities contamination due to in situ generated adsorbent. For example, SRIVASTAVA et al [11] used freshly ferric hydroxide to remove tungsten from molybdate, while ~12% loss of Mo was observed. Research by KASHIWABARA et al [30] also showed that in a single-sorbate solution��the adsorptions of W and Mo onto Fe (hydr)oxides were both more significant than onto Mn oxides. Therefore, it seems reasonable to assume that relatively high W removal efficiency and low Mo loss may be obtained at the same time by using Fe-Mn binary oxide to remove tungsten from molybdate solution.
The aim of this work is to determine a suitable adsorbent based on the iron oxide and manganese oxide for the removal of W from molybdate solution. In addition, the effects of different experimental parameters on the adsorption were also studied in order to develop an optimized technical approach.
2 Experimental
2.1 Materials and analytical methods
All reagents used were of analytical grade and all solutions were prepared with deionized water. The typical mixture solutions of 0.47 g/L WO3 and 50 g/L Mo were prepared by dissolving the sodium salts of both metals in deionized water. The solution pH value was measured by a Shanghai Jinqke Scientific Leici pH meter (PHS-25) with a Leici pH electrode (E-201-C). The concentrations of W and Mo were analyzed by the inductively coupled plasma-atomic emission spectrometer (ICP-AES, Thermo Electron Corporation, US). The adsorbent was characterized by X-ray diffraction (XRD, Rigaku, TTR III, Cu K�� radiation). The morphology of particles was observed in a JEOL JSM-6360LV scanning electron microscope (SEM) and the element was analyzed by X-ray electron dispersive spectroscopy (EDS).
2.2 Adsorbent preparation
In this work, three kinds of adsorbents (ferric hydroxide, manganese dioxide and Fe-Mn binary oxide) were prepared for the removal of W from molybdate solution. Ferric hydroxide was prepared by adding ferric chloride into the stirring caustic solution. The suspensions were filtrated and washed with deionized water, and then the freshly ferric hydroxide was obtained. Manganese dioxide was prepared by the reaction of potassium permanganate with manganese sulphate at pH value around 8. After the filtration, the obtained precipitate was washed with deionized water.
Fe-Mn binary oxide (FMBO) with a Fe to Mn molar ratio of 3:1 was prepared according to the following procedure. Firstly, potassium permanganate (KMnO4, 2.98 g) and sodium hydroxide were dissolved in a 200 mL of deionized water, and iron sulfate heptahydrate (FeSO4��7H2O, 15.81 g) was dissolved in another 200 mL of deionized water. Secondly, the FeSO4 solution was added into the KMnO4 solution under vigorous stirring, and NaOH was supplied to keep the mixture pH around 8. The formed suspension was continuously stirred for 1 h, washed repeatedly with deionized water, and then the nascent Fe-Mn binary oxide (FMBO) was obtained. Reaction equations can be written as follows:
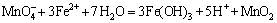 (1)
(1)
These three kinds of nascent adsorbents were dried at 50 ��C for 5 h. The dry materials were crushed and appeared in the form of fine powder.
2.3 Adsorption experiments
Adsorption experiments were carried out in 100 mL polypropylene bottle in a water-bath shaker with 200 r/min shaking rate at constant temperature. After reaching the desired reaction temperature, a specified amount of adsorbent was added into the solution. The initial pH value of 100 mL solution is around 9.3, in which the concentrations of WO3 and Mo are 0.47 g/L and 50 g/L, respectively. After a certain time, the liquor was filtered and the concentrations of W and Mo were analyzed by ICP. The adsorbent was desorbed using NaOH solution and then dried at 50 ��C for 5 h, thus, the adsorbent was regenerated.
The kinetic experiments were carried out at room temperature (25��1 ��C), and a defined amount of sodium tungstate or sodium molybdate salts was added to a 1000 mL glass vessel to obtain a 400 mL, 0.5 g/L WO3 or 0.5 g/L Mo solution. After that, 3 g of FMBO, ferric hydroxide, and manganese dioxide was added to the solutions to obtain a 7.5 g/L suspension, respectively. The suspension was mixed using a magnetic stirrer, and the pH was adjusted to 6.7 with the addition of 0.1 mol/L HCl and/or NaOH. 1 mL solution was taken from the suspension at the following intervals: 0.083, 0.167, 0.25, 0.333, 0.5, 0.667, 1, 2, 3, 4, 5, 7, 12, and 22 h of the reaction.
WO3 and Mo adsorption isotherms were determined at the pH of 6.7, (25��1) ��C. Initial WO3 or Mo concentrations were varied from 0.1 to 0.8 g/L. In each test, 0.4 g of the three adsorbent samples were added to the polypropylene bottle to obtain a 4 g/L suspension.
The molar distribution coefficients of tungsten and molybdenum were calculated as follows:
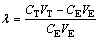 (2)
(2)
where �� is the molar distribution coefficient of metal; CT and CE are the initial and equilibrium metal concentration in solution; and VT and VE are the initial and final volumes of the solution. Separation factor  was then calculated as follows:
was then calculated as follows:
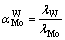 (3)
(3)
where ��Mo and ��W are the molar distribution coefficients of Mo and W, respectively.
3 Results and discussion
3.1 Selection of adsorbent
Three kinds of adsorbents (ferric hydroxide, manganese dioxide, and Fe-Mn binary oxide) were taken to study the adsorption effect on the removal of W from molybdate solution. As shown in Fig. 1(a), the W adsorption capability increased with the increase of Fe content in adsorbent, which is consistent with the literature result [30] that W shows larger affinity on Fe (hydr)oxides than on Mn oxides at higher equilibrium concentrations. However, the large number of surface active sites on nascent adsorbents also led to the increase of the molybdenum loss. When the adsorption rate of W reached 75%, the loss rate of molybdenum was nearly 10%.
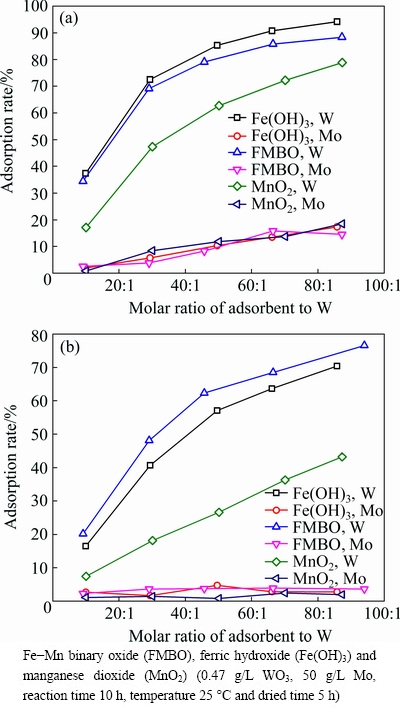
Fig. 1 Adsorption by three nascent (a) and dried (b) adsorbents
In order to minimize the loss of molybdenum, these nascent adsorbents were dried and their adsorption behaviors are shown in Fig. 1(b). It can be seen that the loss of molybdenum greatly reduced to 3%. Although the adsorption rate of W also dropped slightly, it was still acceptable.
Interestingly, the dried Fe-Mn binary oxide had a better adsorption result for W than dried iron oxides, which is consistent with the results of P, As, and Sb adsorption [31-33]. The XRD pattern of Fe-Mn binary adsorbent (Fig. 2) showed that no obvious crystalline peak was detected, indicating that both Fe and Mn oxides in the Fe-Mn binary composite exist mainly in amorphous form. According to Ref. [31], during the synthesis process of Fe-Mn binary adsorbent, this may be the formation of crystalline manganese oxide and iron(III) oxide was blocked by the co-existing of them.
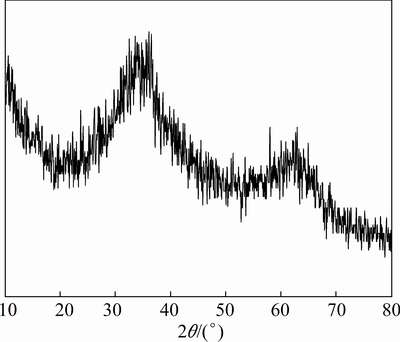
Fig. 2 XRD pattern of dried Fe-Mn binary oxide
The morphologies of dried Fe-Mn binary oxide and iron oxide were studied by scanning electron microscopy (SEM) (Figs. 3(a) and (b)). It can be seen that the surface of iron oxide was smooth, while the Fe-Mn binary oxide was constituted by many aggregated micro-particles, resulting in a rough surface and high activity. The EDS analysis (Fig. 3(c)) showed that Fe/Mn mole ratio on the surface was 3.04:1, which is very close to the value 3:1 of the bulk Fe/Mn mole ratio, revealing that Fe and Mn were evenly distributed on the surface. The chemical composition of Fe-Mn binary oxide was analyzed as 41.22% Fe, 13.48% Mn and 43% O (molecular formula is very close to Fe3MnO11H9 which is made up of three Fe(OH)3 molecules and one MnO2 molecule). In order to understand the adsorption mechanisms, the adsorption kinetics and isotherm experiments were done.

Fig. 3 SEM images of dried Fe(OH)3 (a) and FMBO (b), and EDS analysis result of FMBO (c)
3.1.1 Adsorption kinetics
Figure 4 highlighted the variation of the adsorbed W on these three adsorbents and Mo on FMBO with reaction time. It was observed that the adsorption process of W and Mo onto these adsorbents could be divided into two steps. In the first step, the adsorption rate was fast and almost 80% of the maximum adsorption capacity (Qmax) was achieved within the initial 5 h. This may be due to the fine particles of these three adsorbents powders. The smaller particle size was favorable for the diffusion of W and Mo molecules from bulk solution onto the active sites of the solid surface. In the following step, intraparticle diffusion dominated and the adsorption rate slowed down.
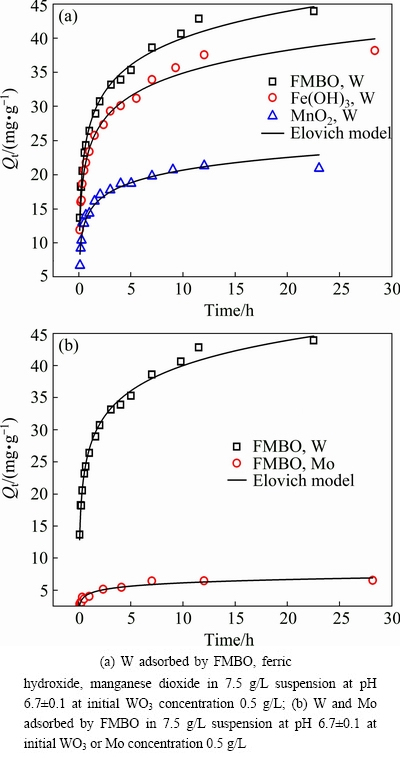
Fig. 4 Adsorption kinetics
In order to evaluate the kinetic mechanism that controls the adsorption process, the experimental data (Fig. 4) were analyzed using four different kinetic models: Pseudo-first-order equation, Pseudo-second- order, Parabolic diffusion and Elovich model [33]. The kinetic parameters obtained from the models are given in Table 1. It is found that the kinetic data of W adsorbed by these three adsorbents and Mo on FMBO fitted well with the Elovich model, indicating the occurrence of heterosphere diffusion reactions [33]. k values in the Elovich equation indicated the adsorption rate of adsorbate, and an increase of k would enhance the reaction rate [33,34]. It was observed that k values of W and Mo adsorption on these adsorbents were in the following order: FMBO,W (5.66 mg/(g��h)) > Fe(OH)3,W (4.80 mg/(g��h)) > MnO2,W (2.67 mg/(g��h)) > FMBO,Mo (0.74 mg/(g��h)), which indicated that FMBO had better selective adsorption performance for W over Mo compared with ferric hydroxide and manganese dioxide.
3.1.2 Adsorption isotherm
The adsorption capacities of these adsorbents for W and Mo were evaluated using the isotherms presented in Fig. 5. Both Freundlich and Langmuir models were employed to describe the adsorption isotherms, and the fitted constants are listed in Table 2. It is seen that the Freundlich model fitted the experimental data better than Langmuir model. The value of correlation coefficient (R2=0.99) confirmed the good representation of experimental data by this model. The Freundlich equation describes that the adsorbent has a heterogeneous surface with adsorption sites which have different energies of adsorption. The KF in the Freundlich equation is roughly an indicator of the adsorption capacity [32]. It was observed that the KF values of W and Mo adsorption on these adsorbents were in the same order: FMBO,W (3.64) > Fe(OH)3,W (2.82) > MnO2, W (2.01) > FMBO,Mo (0.51), which demonstrated that FMBO has a better adsorption selectivity for W than ferric hydroxide and manganese dioxide. The results of adsorption kinetics and isotherm experiments indicated that FMBO is the most potential adsorbent for the separation of W from molybdate solution. Thus, the dried Fe-Mn binary oxide (FMBO) was selected to remove W from molybdate solution.
3.2 Drying process of adsorbent
Table 1 Kinetic model correlation coefficient and parameters for adsorption of W on FMBO, ferric hydroxide, manganese dioxide, and Mo on FMBO at pH 6.7
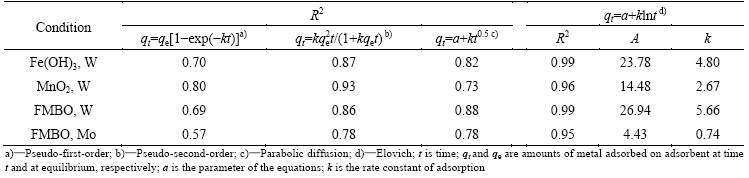
Table 2 Langmuir and Freundlich isotherm constants for adsorption of W on FMBO, ferric hydroxide, manganese dioxide, and Mo on FMBO at pH 6.7, (25��1) ��C
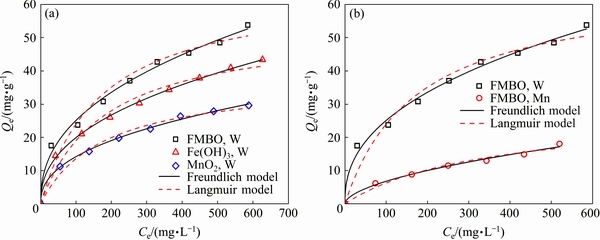
Fig. 5 Adsorption isotherms for W by FMBO, ferric hydroxide, manganese dioxide in 4 g/L suspension at pH 6.7��0.1, (25��1) ��C (a) and for W and Mo by FMBO in 4 g/L suspension at pH 6.7��0.1, (25��1) ��C (b)
It is observed from the previous section that the nascent Fe-Mn binary oxide and the dried Fe-Mn binary oxide have a great difference on the adsorption result, so it is necessary to study the drying process of adsorbent. The adsorptions for W and Mo both decreased along with the drying time. As shown in Fig. 6, the adsorption rates for W and Mo both dropped markedly in the first 5 h. The W adsorption rate dropped from 81% to 70%, while the loss of Mo also greatly reduced from 12% to 2%. Besides, the separation factor reached peak value 136, and it had almost no further impact on the adsorption of W and Mo with prolonging drying time. Therefore, the Fe-Mn binary oxide was dried for 5 h.
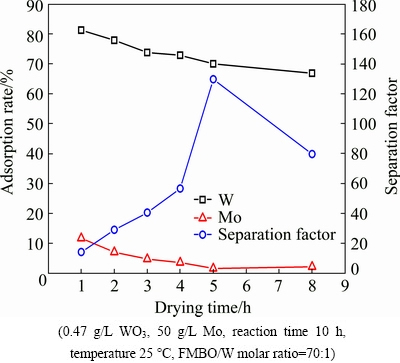
Fig. 6 Effect of dried time of Fe-Mn binary oxide (FMBO) on adsorption
3.3 Adsorption on Fe-Mn binary adsorbent
3.3.1 Effect of reaction time
The effect of time on the adsorption of W and Mo is shown in Fig. 7. The adsorption rate of W increased with reaction time, while that of Mo did not change basically.
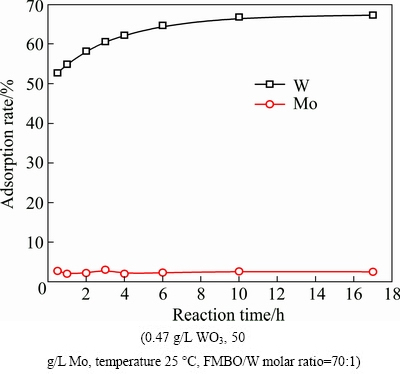
Fig. 7 Effect of reaction time on adsorption
When the reaction time was less than 4 h, the adsorption rate of W increased rapidly and then increased slightly. Moreover, the adsorption rate did not change after 10 h. Thus, 10 h was selected to study the effects of other factors on separation.
3.3.2 Effect of adsorbent dosage
Figure 8 presented the effect of the adsorbent (FMBO) dosage on the adsorption. The experiment data indicated that W was adsorbed prior to Mo. More specifically, when the FMBO/W molar ratio was below 60, the adsorption of W increased linearly with the FMBO dosage, while the loss of Mo basically remained unchanged. The adsorption curve of W bent when the molar ratio was greater than 60. When the ratio exceeds 80:1, the removal efficiency of W increased slowly, whereas the loss of Mo also increased. Considering that the increased dosage results in the cost of raw materials increased, FMBO/W ratio of 80:1 was found to be suitable.
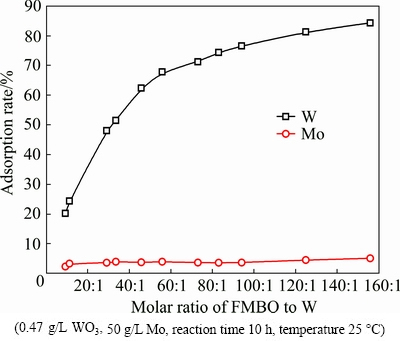
Fig. 8 Effect of FMBO dosage on adsorption
3.3.3 Effect of solution initial pH
Effect of initial solution pH from 3 to 13 on the removal of W was shown in Fig. 9. There existed a platform in the adsorption-pH curve. Fe-Mn binary oxide had a good adaptability for the solution pH. When the initial solution pH ranges within 6.9-11.3, the equilibrium pH was always around 9.7 (Fig. 9(b)) and the adsorption rate of W was basically the same (Fig. 9(a)).
This should be due to the protonation and deprotonation reactions of surface active group SOH with H+ and OH- [35-39]:
 (4)
(4)
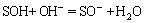 (5)
(5)
The adsorption of Mo increased with the decreasing pH, which displayed typical anion adsorption property. It was favorable to anion adsorption when pH decreased, because of the protonation reaction of surface active group (Eq. (4)), the adsorption of Mo increased. When the initial pH dropped to 7.0, the competition of Mo for the available active sorption sites increased due to the gradual formation of Mo polymeric ions, which caused a significant reduction of the adsorption of W [40]. Correspondingly, it was unfavorable to anion adsorption when pH increased, because of the deprotonation reaction of surface active group (Eq. (5)), thus, the adsorption of W and Mo decreased under high pH conditions. When the initial pH was greater than 10, the loss of Mo was below 1%. The results indicated that Fe-Mn binary had a good adaptability for the solution pH. The adsorbent can be added into weak alkaline solution (pH=6.9-11.3) directly without preadjusting the initial solution pH, the operation was greatly simplified.
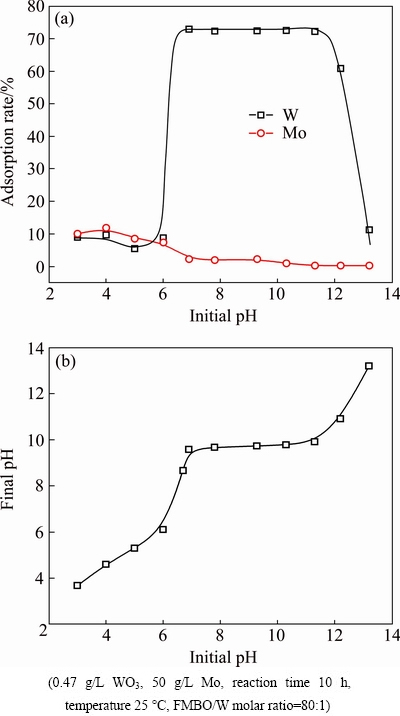
Fig. 9 Effect of initial solution pH on adsorption (a) and final pH (b)
3.3.4 Effect of temperature
The role of temperature on the removal of W was studied by varying reaction temperature at 15-80 ��C (Fig. 10).
Elevated temperature can not only promote the adsorption process but also the desorption process. However, the adsorbent has a greater affinity for W than Mo, leading to the increase of W adsorption and the slight decrease of Mo loss when reaction temperature increased. The adsorption rate of W increased from 64% to 76%, when temperature increased from 15 to 50 ��C. When the temperature was greater than 50 ��C, the removal efficiency of W reached a maximum gradually. Considering the energy consumption, 50 ��C was chosen as an optimal temperature. The adsorption rates of W and Mo were 76.17% and 2.15%, respectively, and the separation factor achieved to 145. After adsorption, the W concentration in filtrate dropped to 112 mg/L, and the mass ratio of Mo/WO3 reached 437:1.
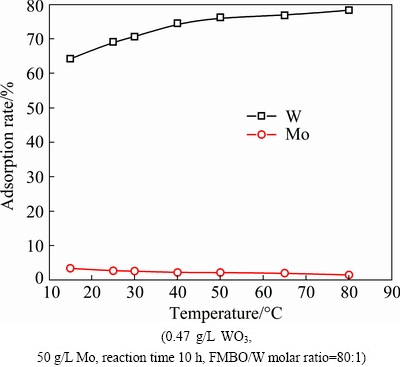
Fig. 10 Effect of temperature on adsorption
3.3.5 Effect of Mo/WO3 mass ratio
The effect of Mo/WO3 mass ratio on the W and Mo separation was studied by varying the W and Mo concentration under different adsorbent dosages.
In different Mo/WO3 mass ratio solutions (WO3 concentration was fixed at 0.47 g/L), the influence of Mo concentration and adsorbent dosage on the removal of W was investigated (Fig. 11(a)). It can be seen from Fig. 11(a) that the removal rate of W decreased with increasing Mo concentration. This can be explained that the competitive adsorption of Mo was intensified with the increase of Mo concentration. When the Mo concentration was 0.5 g/L (the FMBO/W molar ratio exceeded 70:1) and 5 g/L (the FMBO/W molar ratio was above 90:1), W in the filtrate was undetectable. When the Mo concentration was 50 g/L and the FMBO/W molar ratio was 100:1, the adsorption rates of W and Mo were 82.5% and 2.2%, respectively, the separation factor achieved to 210. After adsorption, the W concentration in filtrate dropped to 82 mg/L, and the mass ratio of Mo/WO3 reached 595.
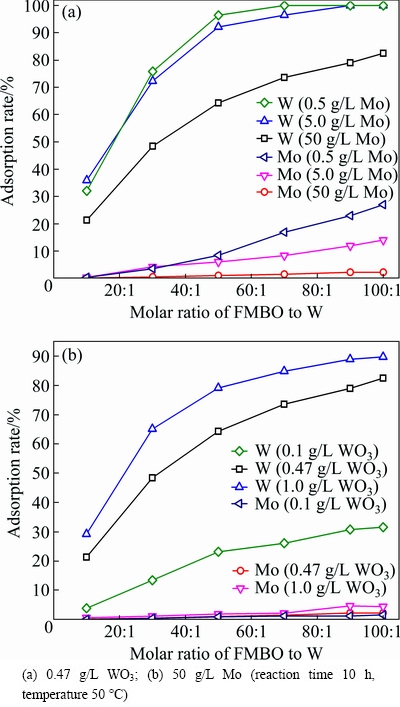
Fig. 11 Effect of Mo/WO3 mass ratio on adsorption
Under conditions of fixed Mo concentration (50 g/L) and different Mo/WO3 mass ratios, the separation results are shown in Fig. 11(b). It can be seen from Fig. 11(b) that the adsorption rate of W increased with adsorbent dosage, at the same FMBO/W molar ratio, the higher W concentration, the higher W removal efficiency. Obviously, this adsorption method has a better applicability in treating solution with a larger W/Mo molar ratio.
3.4 Desorption and recycle of adsorbent
The recoveries of W and Mo from spent adsorbent and the regeneration ability of adsorbent during several adsorption-desorption-regeneration cycles were studied.
In fact, hydrated metal oxides can be seen as weakly basic anion exchanger. Under the acid and weak alkaline conditions, anions would be exchanged with the surface hydroxyl group of hydrated oxides. The adsorption reaction would be reversed under strong alkaline conditions as Eq. (6), the adsorbed W and Mo anions would be recycled and the adsorbent was regenerated.
S-W(Mo)n-+OH- SOH+W(Mo)(n+1)- (6)
SOH+W(Mo)(n+1)- (6)
The adsorbent was desorbed under different concentrations of NaOH solution (0.1-5 mol/L) at room temperature. As shown in Fig. 12, the desorption rates of W and Mo both increased with desorption time and NaOH concentration. Under conditions of 3 mol/L NaOH solution and 10 h, the desorption rates of W and Mo reached 81% and 99% respectively. It had almost no further impact on the W and Mo desorption with the increase of NaOH concentration.
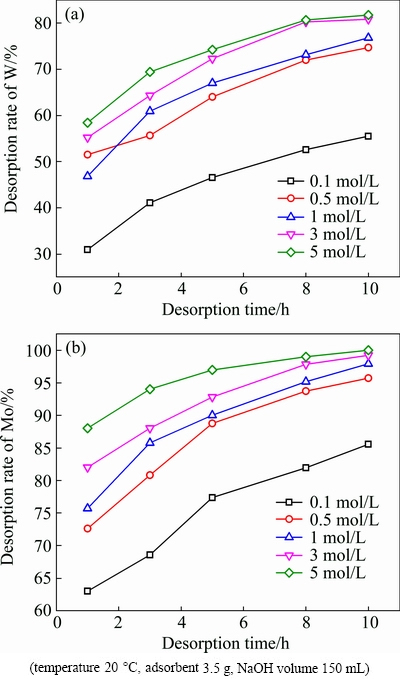
Fig. 12 Effect of NaOH concentration and desorption time on desorption of W (a) and Mo (b)
Adsorbents were desorbed for 10 h under different NaOH concentrations. They were dried at 50 ��C for 5 h and then recycled to remove W from molybdate solution. As shown in Fig. 13, the rates of W adsorption onto recycled adsorbent were all around 75% when the eluent alkali concentration was less than 3 mol/L. While it decreased to 64% at 5 mol/L NaOH condition. This indicated that too high alkali concentration may destroy the structure of adsorbent. Therefore, the adsorbent which desorbed under 3 mol/L NaOH solution for 10 h was selected to further study the regeneration ability.
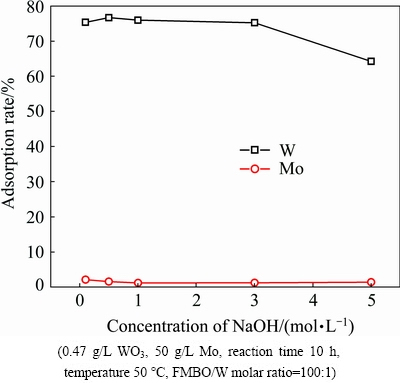
Fig. 13 Effect of eluent NaOH concentration on readsorption of W and Mo
The adsorbent was regenerated five times, and the adsorption efficiencies of W and Mo were shown in Fig. 14. The experimental data clarified that the adsorption rate of W slightly decreased after five consecutive cycles and it was greater than 71% and the loss of Mo was less than 2%. This indicated that the cycle performance of Fe-Mn binary oxide adsorbent was stable.
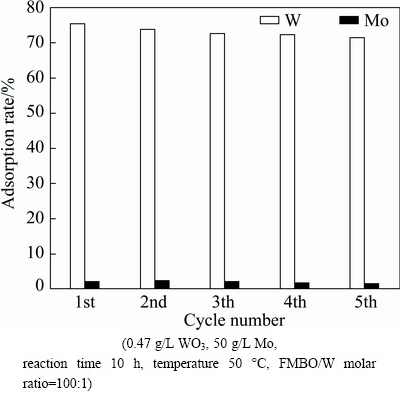
Fig. 14 Regeneration study with consecutive elution operation in five cycles with 3 mol/L NaOH
4 Conclusions
1) Three adsorbents of Fe-Mn binary oxide (FMBO), ferric hydroxide (Fe(OH)3) and manganese dioxide (MnO2) were investigated to remove W from molybdate solution. The Fe-Mn binary oxide (FMBO) was obtained by a simultaneous oxidation and co-precipitation process after being further dried for 5 h, and it was proved to be a more suitable adsorbent than others in the process of the W removal.
2) Fe-Mn binary oxides have a good adaptability for the solution pH. It can obtain satisfactory separation results without preadjusting the initial solution pH, which simplifies the operation and expands applicability of processing solution. The feed concentration experiments show that this adsorption method has a better applicability in treating solution with larger W/Mo molar ratios. The separation factor reached 210 at FMBO/W molar ratio of 100:1, 50 ��C for 10 h. At the same time, the adsorption rates of W and Mo were 82.5% and 2.2% respectively, and the Mo/WO3 mass ratio in purified filtrate reached 595:1.
3) The desorption rates of W and Mo reached 81% and 99%, respectively, when the adsorbent was desorbed after 10 h in 3 mol/L NaOH solution. The regeneration ability of the Fe-Mn binary oxide adsorbent was stable. All these demonstrated that Fe-Mn binary oxide could be one of the efficient and cost-effective potential materials for removing W from molybdate solution.
References
[1] ZHAN Zhi-hong. Analysis of effects on Mo wire quality and measures of technological improvement [J]. China Molybdenum Industry, 2006, 2: 28-31. (in Chinese)
[2] KEKESI T, TOROK T I, ISSHIKI M. Anion exchange of chromium, molybdenum and tungsten species of various oxidation states, providing the basis for separation and purification in HCl solutions [J]. Hydrometallurgy, 2005, 77(1): 81-88.
[3] LI Hong-gui, HUO Guang-sheng, SUN Pei-mei, ZHAO Zhong-wei, LI Yun-jiao, SU Peng-tuan, LIU Mao-sheng. Developing new reagent for selectively precipitation of molybdenum from tungstate solution [J]. Transactions of Nonferrous Metals Society of China, 2003, 13(1): 184-187.
[4] HUO Guang-sheng, ZHAO Zhong-wei, LI Hong-gui, SUN Pei-mei, LI Yun-jiao. Removing molybdenum from tungstate solution with metal sulfides [J]. The Chinese Journal of Nonferrous Metals, 2004, 14(2): 302-305. (in Chinese)
[5] ZHENG Qing-yuan, FAN Hui-hao. Separation of molybdenum from tungsten by di-2-ethylhexylphosphoric acid extractant [J]. Hydrometallurgy, 1986, 16(3): 263-270.
[6] XIAO Lian-sheng, ZHANG Qi-xiu, GONG Bo-fan, HUANG Shao-ying. Separation of molybdenum from tungstate solution by a combination of moving packed bed and fluid bed ion-exchange techniques [J]. International Journal of Refractory Metals and Hard Materials, 2001, 19(3): 145-148.
[7] ZHAO Zhong-wei, XIAO Lu-ping, SUN Fang, HUO Guang-sheng, LI Hong-gui. Study on removing mo from tungstate solution by activated carbon loaded with copper [J]. International Journal of Refractory Metals and Hard Materials, 2010, 28(4): 503-507.
[8] NING Peng-ge, CAO Hong-bin, ZHANG Yi. Selective extraction and deep removal of tungsten from sodium molybdate solution by primary amine N1923 [J]. Separation and Purification Technology, 2009, 70(1): 27-33.
[9] BLOKHIN A A, KALOSHIN J I, LYUBMAN H ��. Removing tungsten and purifying ammonia molybdate by catechol cation exchange resin [J]. Russian Journal of Applied Chemistry, 2005, 78: 425-427. (in Russian)
[10] XIAO Lian-sheng, GONG Bo-pan,WANG Xue-wen. A method for deep removal of trace tungsten from high concentration of molybdate solution: CN Patent 101264933A [P]. 2008-04-11.
[11] SRIVASTAVA R R, MITTAL N K, PADH B, REDDY B R. Removal of tungsten and other impurities from spent HDS catalyst leach liquor by an adsorption route [J]. Hydrometallurgy, 2012, 127: 77-83.
[12] WANG Shu-guang, GONG Wen-xin, LIU Xian-wei, YAO Ya-wei, GAO Bao-yu, YUE Qin-yan. Removal of lead (II) from aqueous solution by adsorption onto manganese oxide-coated carbon nanotubes [J]. Separation and Purification Technology, 2007, 58(1): 17-23.
[13] DEMIRKIRAN N. Copper adsorption by natural manganese dioxide [J]. Transactions of Nonferrous Metals Society of China, 2015, 25(2): 647-653.
[14] SONG Hai-lei, JIAO Fei-peng, JIANG Xin-yu, YU Jin-gang, CHEN Xiao-qing, DU Shao-long. Removal of vanadate anion by calcined Mg/Al-CO3 layered double hydroxide in aqueous solution [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(11): 3337-3345.
[15] WU Kun, LIU Ting, XUE Wen, WANG Xiao-chang. Arsenic (III) oxidation/adsorption behaviors on a new bimetal adsorbent of Mn-oxide-doped Al oxide [J]. Chemical Engineering Journal, 2012, 192: 343-349.
[16] SONMEZAY A,  N. Adsorption of lead and cadmium ions from aqueous solutions using manganoxide minerals [J]. Transactions of Nonferrous Metals Society of China, 2012, 22(12): 3131-3139.
N. Adsorption of lead and cadmium ions from aqueous solutions using manganoxide minerals [J]. Transactions of Nonferrous Metals Society of China, 2012, 22(12): 3131-3139.
[17] TAN Yan-ni, SONG Ni, LIU Yong, LUO Tao, DOU Yu-hai, ZHANG Qing, LIU Qian-nan, LUO Lan-lan. Synthesis of platy potassium magnesium titanate and its application in removal of copper ions from aqueous solution [J]. Transactions of Nonferrous Metals Society of China, 2015, 25(3): 981-990.
[18] XIAO Wei-hong, ZHANG Qing-mei, YOU Xiang-yu, LIU Zhan. Synthesis of the Mg-Al hydrotalcite and its adsorption properties for VO3- [J]. Nonferrous Metals Science and Engineering, 2015, 6(4): 37-40. (in Chinese)
[19] PAN Zhao-lu. Crystallography and mineralogy [M]. Beijing: Geological Publishing House, 1984, 2: 222-226. (in Chinese)
[20] VERMAAS F H S. South African scheelites and an X-ray method for determining members of the scheelite�Cpowellite series [J]. American Mineralogist, 1952, 37(9-10): 719-735.
[21] CHERESNOWSKY M J. Method for separating tungsten from molybednum: U.S. Patent 4 999 169 [P]. 1991-03-12.
[22] MA Dong-sheng. Progress in research on geochemistry of tungsten [J]. Geological Journal of China Universities, 2009, 15(1): 19-34. (in Chinese)
[23] HUANG Pu-xuan, FAN Wei. Research on the technology of separating tungsten and molybdenum with nascent state iron hydroxide [J]. Journal of Jilin Institute of Chemical Technology, 2001, 18(3): 18-20. (in Chinese)
[24] LU Ying, SUN Fang. Study of separation of tungsten and molybdenum from high W-containing molybdate acid sodium solution by Fe(OH)3 adsorption [J]. Rare Metals and Cemented Carbides, 2005, 33(3): 1-3. (in Chinese)
[25] ZHAO Zhong-wei, LI Jiang-tao, CAO Cai-fang, HUO Guang-sheng, ZHANG Gang, LI Hong-gui. Recovery and purification of molybdenum from Ni-Mo ore by direct air oxidation in alkaline solution [J]. Hydrometallurgy, 2010, 103(1): 68-73.
[26] SEMENOV M I, BLOKHINl A A, TAUSHKANOV V P. Application of hydrated polyvalent metal oxides for deep removal of tungsten from molybdenum-containing solutions [J]. Russian Journal of Applied Chemistry, 1984, 57(7): 1501-1506. (in Russian)
[27] CAO Cai-fang, ZHAO Zhong-wei, CHEN Xing-yu. Selective precipitation of tungsten from molybdate containing solution using divalent ions [J]. Hydrometallurgy, 2011, 110(1): 115-119.
[28] ZHAO Zhong-wei, CAO Cai-fang, CHEN Xing-yu. Separation of macro amounts of tungsten and molybdenum by precipitation with ferrous salt [J]. Transactions of Nonferrous Metals Society of China, 2011, 21(12): 2758-2763.
[29] ZHAO Zhong-wei, CAO Cai-fang, CHEN Xing-yu, HUO Guang-sheng. Separation of macro amounts of tungsten and molybdenum by selective precipitation [J]. Hydrometallurgy, 2011, 108(3): 229-232.
[30] KASHIWABARA T, TAKAHASHI Y, MARCUS M A, URUGA T, TANIDA H, TERADA Y, USUI A. Tungsten species in natural ferromanganese oxides related to its different behavior from molybdenum in oxic ocean [J]. Geochimica et Cosmochimica Acta, 2013, 106: 364-378.
[31] ZHANG Gao-sheng, QU Jiu-hui, LIU Hui-juan, LIU Rui-ping, WU Rong-cheng. Preparation and evaluation of a novel Fe-Mn binary oxide adsorbent for effective arsenite removal [J]. Water Research, 2007, 41(9): 1921-1928.
[32] ZHANG Gao-sheng, LIU Hui-juan, LIU Rui-ping, QU Jiu-hui. Removal of phosphate from water by a Fe-Mn binary oxide adsorbent [J]. Journal of Colloid and Interface Science, 2009, 335(2): 168-174.
[33] XU Wei, WANG Hong-jie, LIU Rui-ping, ZHAO Xu, QU Jiu-hui. The mechanism of antimony (III) removal and its reactions on the surfaces of Fe-Mn binary oxide [J]. Journal of Colloid and Interface Science, 2011, 363(1): 320-326.
[34] CHIEN S H, CLAYTON W R. Application of Elovich equation to the kinetics of phosphate release and sorption in soils [J]. Soil Science Society of America Journal, 1980, 44(2): 265-268.
[35] SU Ting-zhi, GUAN Xiao-hong, GU Guo-wei, WANG Jian-min. Adsorption characteristics of As (V), Se (IV), and V (V) onto activated alumina: Effects of pH, surface loading, and ionic strength [J]. Journal of Colloid and Interface Science, 2008, 326(2): 347-353.
[36] WANG Jian-min, WANG Tian, BURKEN J G, CHUSUEI C C, BAN Heng, LADWIG K, HUANG C P. Adsorption of arsenic (V) onto fly ash: A speciation-based approach [J]. Chemosphere, 2008, 72(3): 381-388.
[37] SU Ting-zhi, WANG Jian-min. Modeling batch leaching behavior of arsenic and selenium from bituminous coal fly ashes [J]. Chemosphere, 2011, 85(8): 1368-1374.
[38] GUAN Xiao-hong, SU Ting-zhi, WANG Jian-min. Quantifying effects of pH and surface loading on arsenic adsorption on nanoactive alumina using a speciation-based model [J]. Journal of Hazardous Materials, 2009, 166(1): 39-45.
[39] SU Ting-zhi, GUAN Xiao-hong, TANG Yu-lin, GU Guo-wei, WANG Jian-min. Predicting competitive adsorption behavior of major toxic anionic elements onto activated alumina: A speciation-based approach [J]. Journal of Hazardous Materials, 2010, 176(1-3): 466-472.
[40] ZHAO Zhong-wei, ZHANG Jia-liang, CHEN Xing-yu, LIU Xu-heng, LI Jiang-tao, ZHANG Wei-guang. Separation of tungsten and molybdenum using macroporous resin: Equilibrium adsorption for single and binary systems [J]. Hydrometallurgy, 2013, 140: 120-127.
���Ʒ�1��������1,2��������1������ΰ1
1. ���ϴ�ѧ ұ���뻷��ѧԺ����ɳ 410083��
2. �廪��ѧ ����������Դ�����о�Ժ������ 100084
ժ Ҫ�����ǵ��١��ⲻͬ�ĵ���ѧ������Ϊ���о����̸���������(FMBO)����������(Fe(OH)3)�Ͷ�������(MnO2)��������������������Һ�з����١�������������̸���������(FMBO)���ʺϴ���������Һ�г��١����̸�����������һ���Ͽ�����pH��Χ��(6.9~11.3)���нϺõ������ԣ����Եõ���ȥ���ʳ���80%������ʧС��3%�����⣬���̸��������������3 mol/L���������������������ٵ�ȥ���ʾ���5������-����-������Ȼ���Ա��ֲ��䡣�������̸�������������������������Һ�г��پ������õ�Ӧ��DZ����
�ؼ��ʣ����٣���������Һ�����̸������������
(Edited by Xiang-qun LI)
Foundation item: Projects (51334008, 51304243, 51604160) supported by the National Natural Science Foundation of China; Project (2016zzts037) supported by the Fundamental Research Funds for the Central Universities, China
Corresponding author: Li-hua HE; Tel: +86-731-88830476; E-mail: helihua@csu.edu.cn
DOI: 10.1016/S1003-6326(17)60273-5

