Preparation and characterization of Fe/SiO2 core/shell nanocomposites
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2010���4��
�������ߣ�Ԭ���� �ռӻ� �ƹڽ� ̷���� �����
����ҳ�룺632 - 636
Key words��core-shell nanocomposites; iron; magnetic properties; nanoparticles
Abstract:
In a simple ethanol-water system, the magnetic ��-Fe nanoparticles (with an average diameter of 10-40 nm) were prepared by reduction of Fe2+ using potassium borohydride in the presence of surfactant. Then the shell was formed by hydrolysis- condensation polymerization of tetraethylorthosilicate (TEOS) on the surface of the Fe particles. The samples were characterized by XRD, TEM, SAED, TG-DSC and VSM. The results indicate that a thin film of silica is coated on the surface of Fe particles through a Si��O��Fe bond. The coated shell of silica can effectively protect the Fe cores from being oxidized.

YUAN Ming-liang(Ԭ����), TAO Jia-hua(�ռӻ�), YAN Guan-jie(�ƹڽ�),
TAN Mei-yi(̷����), QIU Guan-zhou(�����)
School of Mineral Processing and Bioengineering, Central South University, Changsha 410083, China
Received 9 March 2009; accepted 14 July 2009
Abstract: In a simple ethanol-water system, the magnetic ��-Fe nanoparticles (with an average diameter of 10-40 nm) were prepared by reduction of Fe2+ using potassium borohydride in the presence of surfactant. Then the shell was formed by hydrolysis- condensation polymerization of tetraethylorthosilicate (TEOS) on the surface of the Fe particles. The samples were characterized by XRD, TEM, SAED, TG-DSC and VSM. The results indicate that a thin film of silica is coated on the surface of Fe particles through a Si��O��Fe bond. The coated shell of silica can effectively protect the Fe cores from being oxidized.
Key words: core-shell nanocomposites; iron; magnetic properties; nanoparticles
1 Introduction
The properties of nanometer-sized magnetic iron particles have been investigated extensively by both experimental and theoretical analysis. On the one hand, these particles show promise for practical applications, such as catalysis, magnetic recording, magnetic fluids, and biomedical applications; on the other hand, their utility has been limited due to easy aggregation and uncontrolled oxidation[1]. Freshly formed iron nanoparticles often ignite spontaneously upon exposure to air or undergo a rapid exothermic reaction with oxygen under ambient aerobic condition[2]. One method for controlling the oxidation is to coat the particles, which means to create a core-shell structure. The shell could protect the nanoparticles from oxidation and reduce the dipole interaction. Furthermore, the coating is beneficial for nanomagnet, such as compatibility in biological system, functionality and high suspension stability under different solutions[3]. Generally, silica coating of oxides can be easily got, which can be combined by the silica with ��OH surface groups. However, silica deposition on pure metal particles is more complicated, because of the lack of ��OH groups on the metal surface[4]. Currently, the coating of magnetic metal particles without any additional organic molecules is still a challenge. There have been reported about the nanocoating techniques encapsulating iron nanoparticles. Fe nanoparticles have been coated with Au by a reverse micelle method[5]. Fe nanoparticles coated with SiO2 have been synthesized in aqueous solution[6]. Iron�Ciron oxide core�Cshell nanoparticles were also synthesized by laser pyrolysis followed by superficial oxidation[7]. Ni-based coating synthesized by rare earth was added into the Co-based coatings[8]. Though these nanocoatings can protect the metal nanoparticles against the oxidation, they still have some problems.
In this work, the preparation of ��-Fe nanoparticles by reduction of ferrous ion using KBH4 in simple ethanol-water system in the presence of surfactant has successfully performed. The synthesized ��-Fe nanoparticles is coated by SiO2 to prevent the iron from being oxidized. Silica coating helps dispersion of the synthesized nanoparticles. At the same time, silica coating helps dispersion of the synthesized ��-Fe nanoparticles.
2 Experimental
2.1 Materials
Tetraethylorthosilicate (TEOS), ammonia (25%, mass fraction), sodium dodecylsulfate (SDS, 99%), potassium borohydride (KBH4, 98%), chloride tetrahydrate (FeCl2?4H2O, 98%), nitrogen (99.5%) and absolute ethanol (95%) were all of analytic grade without further purifying.
2.2 Preparation of iron nanoparticles
Nanoscale zero-valence iron particles can be prepared in aqueous solutions via the reduction of ferric iron (Fe(��) or ferrous iron Fe(��)) with potassium borohydride[9-10], or via decomposition of iron pentacarbonyl (Fe(CO)5) in organic solution or in argon [11-14]. Zero-valence iron particles can also be prepared from hydrogen reduction of iron oxides. In this work, potassium borohydride was introduced to reduce ferrous iron (Fe2+) to prepare zero-valence iron particles (ZVI), according to the following reaction[15]:
![]()
The key advantage of this method is its simplicity. It can be safely done in most chemistry lab with simple chemical reagents. All procedures for synthesis and handling during this experiment were carried out under an atmosphere of N2 (99.5%). All solvents were degassed and saturated with N2 before using, based on the literatures[16-17]. Typically, some SDS was added into 100 mL ethanol-water solution containing 0.05 mol/L FeCl2 (here the volume ratio of ethanol to water was 10:1, the concentration of SDS was 0.04 mol/L) with vigorous stirring to form a mixture. Then 50 mL ethanol-water solution containing 0.25 mol/L KBH4 (here, the volume ratio of ethanol to water was 4:1) was added into the stirred mixture above. Then, the solution was stirred for 30 min at 45 �� and a black precipitate appeared. The precipitate was separated from the mixture with a magnet, and then it was washed for five times with absolute ethanol, finally it was dried in vacuum stove for 24 h at 60 ��.
2.3 Preparation of iron nanoparticles coated with silica
The above prepared iron seed solution was added into 100 mL ethanol-water solution (the finally volume ratio of ethanol to water was 10:1), with constant stirring, and then ammonia solution (25%) was injected to adjust pH of the solution at 8-9 and TEOS (molar ratio of TEOS to Fe2+ was 1:1) was added to the reaction mixture. The reaction was run for 4 h at room temperature with constant stirring, then aged for 24 h and washed with deionized water several times to remove dissociative polysiloxane, unreacted monomer and polysiloxane oligomers followed by absolute ethyl. The composite particles were then dried at 60 �� in oven for 6 h.
2.4 Characterization of iron nanoparticles
The X-ray diffraction patterns were obtained on a Bruker D8 Advance X-ray diffractometer with Cu K�� radiation (��=1.540 56 ?). The images of iron and SiO2-coated iron nanoparticles were recorded with a Philips EM 400T Transmission Electron Microscopy (TEM) (Philips Electronics Co, Eindhoven, Netherlands) operated at 100 kV. The thermal analysis experiment was carried out on a NETZSCH STA 449C Series thermo gravimetric analyzer (TGA) and a Perkin-Elmer differential scanning calorimeter (DSC) under the following operational conditions: with a 8 mg power sample heated from 20 to 550 �� under air atmosphere at a rate of 10 ��/min. Their magnetic properties at room temperature were investigated using a vibrating sample magnetometer (VSM).
3 Results and discussion
3.1 Structure of magnetic composite particles
TEOS would hydrolyze and happen in the presence of water, silicic acid was formed as one of the hydrolysates (Eq.(2)), and the purpose of adding ammonia was to promote the hydrolysis of TEOS. Then, silicic acid molecule would polymerize with other silicic acid or TEOS molecule (Eq.(3) or Eq.(4)); the product of this step was the monomer or oligomer of polysiloxane. Finally, the monomers or oligomers of polysiloxane along with silicic acid molecules had very high activities, and could be adsorbed on the surface of the iron particles rapidly, the polymerization of Eq.(5) occurred on the surface of the iron particles and the polysiloxane film thus covered around the particles tightly (Fig.1).
Si(OC2H5)4+4H2O��Si(OH)4+4C2H5OH (2)
Si(OH)4+ Si(OH)4��(HO)3Si-O-Si(OH)3+H2O (3)
Si(OH)4+(C2H5O)4Si��(HO)3Si��O��Si(OC2H5)3+C2H5OH (4)
![]()
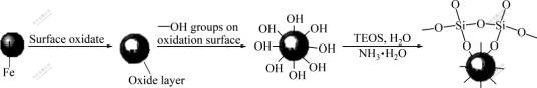
Fig.1 Mechanism of SiO2-coat on iron surface
3.2 Structure and antioxidation property of products
Fig.2 shows the X-ray diffraction patterns of the uncoated iron nanoparticles and the SiO2-coated iron nanoparticles, where 2�� is at the range of 10?-90?. Form Fig.2(a), it can be seen that there are five diffraction peaks at 11.82?, 35.36?, 62.47?, 38.08? and 44.60?, corresponding to ��-Fe reflection and Fe3O4 or Fe2O3 reflection, respectively, which show on the one hand that ��-Fe has indeed been synthesized, on the other hand that ��-Fe nanoparticles with highly surface activity can be oxidized. Fig.2(a) shows only two diffraction peaks of ferric oxide, it cannot be identified whether the oxidation product is Fe3O4, Fe2O3 or most likely a mixture of the two. The yellowish of the product suggests the presence of Fe2O3. The compact shell of Fe2O3 generated on the surface of ��-Fe particles hinders further oxidation of inner iron core. The background of XRD pattern in Fig.2(a) suggests that the crystallization of the products is far from completion, which was a result of low temperature synthesis.
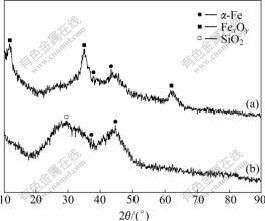
Fig.2 X-ray diffraction patterns of as-prepared samples: (a) Uncoated iron nanoparticles; (b) SiO2-coated iron nano- particles
By contrast, In Fig.2(b) we can see that there is no diffraction peak of any iron oxide, the only three diffraction peaks at 28.90?, 37.94? and 44.60? are observed. The diffraction peaks at 37.94? and 44.60? are clearly due to the existing of ��-Fe particles, while the peak at 28.90? may be attributed to silica coating, which shows not only that there are indeed ��-Fe in uncoated iron nanoparticles but also that SiO2-coated iron nanoparticles prevent the ��-Fe from oxidizing.
The TEM image was used to identify the morphology of uncoated and SiO2-coated iron nanoparticles with an average diameter of 10-40 nm. From Fig.3(b), we can see a very thin film of iron oxides with thickness of 2-4 nm on the surface of the iron powders, which may result in a slight inevitable surface oxidation during the process of cleaning and dryness, it agrees with the X-ray diffraction pattern of sample. The loose layer of iron oxides can not well protect the iron from further oxidation; however, it is essential for the successful coating in the absence of any surfactants. Generally, without any surfactants, the pure metallic iron was vitreophobe, which makes the interaction between silica and the metallic particles difficult. However, the thin layer of surface oxides changes the particle surface into vitreophilic, making the attachment of silica on iron much easier through the ��OH surface groups[4]. In Fig.3(c) the coating film was about 10nm. All the images also show that the most particles formed chain-like aggregation, which is due to the magneto static reaction and surface tension.

Fig.3 SAED pattern of uncoated iron nanoparticles (a), TEM pattern of uncoated iron nanoparticles (b) and TEM pattern of SiO2-coated iron nanoparticles (c)
In order to confirm the difference of anti-oxidation capability of the coated and uncoated samples, the TG-DSC curves were measured. Fig.4 shows that the mass change of the SiO2-coated iron nanoparticles is rather obvious from 20 to 550 ��. In the segment from 20 �� to 360 ��, the mass loss is 9.89%, associated with an endothermic peak at about 298 ��. This indicates a physical process, which vaporizes the absorbed gases. From 360 �� to 500 ��, most of SiO2-coated iron nanoparticles almost keep unchanged, this attributes to the protective SiO2 shell formed on the surface of the iron core. Fig.5 shows the TG-DSC curves of the uncoated iron nanoparticles, its mass loss and gaining stage are just the same with the SiO2-coated iron nanoparticles from 20 �� to 350 ��, and the mass loss rate is 8.85%. From 360 �� to 550 ��, TG-DSC curves exhibit an abrupt mass gain, associating with an exothermic peak at about 463 ��. This implied that the iron nanoparticles have been oxidized to iron oxides. The exothermal peaks at 463 �� is attributed to chemical reactions of oxidization, in which the iron core is oxidized to Fe2O3 and Fe3O4, which was confirmed with the X-ray diffraction patterns. So the two samples exhibited different resistance abilities to oxidation and the SiO2 shell can protect the iron nanoparticles from further oxidation.
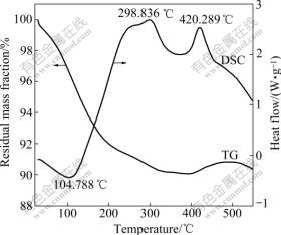
Fig.4 TG-DSC curves of SiO2-coated iron nanoparticles
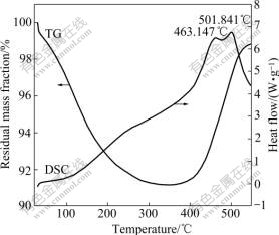
Fig.5 TG-DSC curves of uncoated iron nanoparticles
3.3 Magnetic properties
Magnetic properties of the two samples were investigated at room temperature using a VSM in an applied field -557.046 kA/m��H��557.046 kA/m. Fig.6 and Fig.7 show the magnetic hysteresis loops for the uncoated iron nanoparticles and SiO2-coated iron nanoparticles samples. Values of saturation magnetization (Ms), coercivity (Hc) are: 0.146 2 T, 20.769 858 kA/m for uncoated iron nanoparticles sample; 0.258 1 T, 20.929 014 kA/m for SiO2-coated iron nanoparticles sample. The magnetization discrepancy in magnetiza- tion value between bulk and our nanoparticles could be attributed to the small particle size effect. It is known that the saturation magnetization value of nanoparticles is smaller than that of corresponding bulk materials [18-22]. The magnetic iron nanoparticles coated some iron materials and SiO2 shell may serve as promising magnetic labels for highly efficient bioseparation/drug delivery and highly sensitive biodetection.
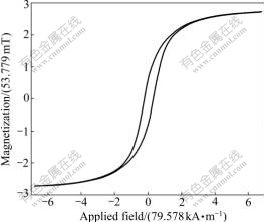
Fig.6 VSM curve of uncoated iron nanoparticles
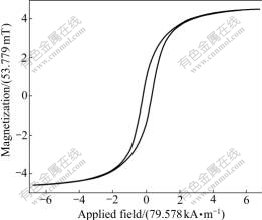
Fig.7 VSM curve of SiO2-coated iron nanoparticles
4 Conclusions
1) ��-Fe nanoparticles were synthesized by a simple ethanol-water system in the presence of surfactant and the average particle size was at the range of 10-40 nm. Shell silica was produced by the hydrolysis and condensation of tetraethyl orthosilicate (TEOS).
2) The analysis of XRD, TEM, SAED, TG-DSC and VSM indicates that the product has a core-shell structure which is combined through the bonding of Si��O��Fe.
3) Due to the large specific surface area of particles and high surface reactivity of the synthesized ��-Fe nanoparticles, the particles are found to be easily oxidized. The protective SiO2 shell effectively improves the thermal stability of Fe nanoparticles in air.
4) SiO2-coated iron nanoparticles are of great importance in applications, such as catalysis, magnetic recording, magnetic fluids, and biomedical applications.
References
[1] LIZ-MARZAN L M, MULVANEY P. The assembly of coated nanocrystals [J]. Phys Chem B, 2003, 107(30): 7312-7326.
[2] BOMATI-MIGUEL O, TARTAJ P, MORALES M P, BONVILLE P, GOLLA-SCHINDLER U, ZHAO X Q, VEINTEMILLAS- VERDAGUER S. Core-shell iron-iron oxide nanoparticles synthesized by laser-induced pyrolysis [J]. Small, 2006, 2(12): 1476- 1483.
[3] HARDIKAR V V, MATIJEVI?E. Coating of nanosize silver particles with silica [J]. Colloid Interface Sci, 2000, 221(12): 133-136.
[4] LIN J, ZHOU W L, KUMBHAR A, WIEMANN J, FANG J Y, CARPENTER E E, O'CONNOR C J. Coating Gd2O3: Eu phosphors with silica by solid-state reaction at room temperature [J]. Sun Powder Technol, 2004, 145(2): 149-153.
[5] LIN J, ZHOU W, KUMBHAR A, WIEMANM J, FANG J, CARPENTER E E. Gold-coated iron (Fe@Au) nanoparticles: Synthesis, characterization, and magnetic field-induced self- assembly solid [J]. State Chem. 2001, 159(1): 26-31.
[6] FERNANDEZ-PACHECO R, ARRUEBO M, MARQUINA C, IBARRA R, ARBIOL J, SANTAMARIA J. Highly magnetic silica-coated iron nanoparticles prepared by the arc-discharge method [J]. Nanotechnology, 2006, 17(5): 1188-1192.
[7] DUMITRACHE F, MORJAN I, ALEXANDRESCU R., CIUPINA V, PRODAN G, VOICU I, FLEACA C, ALBU L, SAVOIU M, SANDU I, POPOVICI E, SOARE I. Iron�Ciron oxide core�Cshell nanoparticles synthesized by laser pyrolysis followed by superficial oxidation [J]. Applied Surface Sci, 2005, 247(1/4): 25-31.
[8] ZHANG Z Y, LU X C, HAN B L, LUO J B. Rare earth effect on the microstructure and wear resistance of Ni-based coatings [J]. Mater Sci Eng A, 2007, 454/455: 194-202.
[9] ZHANG W X. Nanoscale iron particles for environmental remediation: An overview [J]. Nanoparticle Research, 2003, 5: 323-332.
[10] WANG C B, ZHANG W X. Synthesizing nanoscale iron particles for rapid and complete dechlorination of TCE and PCBs [J]. Environ Sci Technol, 1997, 31(7): 2154-2156.
[11] PENG S, WANG C, XIE J, SUN S H. Synthesis and stabilization of monodisperse Fe nanoparticles [J]. Am Chem Soc, 2006, 128(33): 10676-10677.
[12] KARLSSON M N A, DEPPERT K, WACASER B A, KARLSSON L S, MALM J O. Size-controlled nanoparticles by thermal cracking of iron pentacarbonyl [J]. Appl Phys A, 2005, 80(7): 1579-1583.
[13] CHOI C J, DONG X L, KIM B K. Microstructure and magnetic properties of Fe nanoparticles synthesized by chemical vapor condensation [J]. Mater Trans, 2001, 42(10): 2046-2049.
[14] ELIHN K, OTTEN F, BOMAN M, KRUIS F E, FISSAN H, CARLSSON J O. Nanoparticle formation by laser assisted photolysis of ferrocene [J]. Nanostruct Mater, 1999, 12(1): 79-82.
[15] LIEN H L, ZHANG W X. Dechlorination of chlorinated methanes in aqueous solution using nanoscale bimetallic particles [J]. Environ Eng, 1999, 125: 1042-1047.
[16] WANG C B, ZHANG W X. Synthesizing nanoscale iron particles for rapid and complete dechlorination of TCE and PCBS [J]. Environ Sci Technol, 1997, 31(7): 2154-2156.
[17] GLAVEE G N K J, SORENSEN C M, HADJIPANAYIS G C. Chemistry of borohydride reduction of iron(��) and iron(��) ions in aqueous and nonaqueous media: Formation of nanoscale Fe, FeB, and Fe2B powers [J]. Inorg Chem, 1995, 34: 28-35.
[18] SHAFI K V P M, KOLTYPIN Y, GEDANKEN A, PROZOROV R, BALOGH J. Sonochemical preparation of nanosized amorphous NiFe2O4 particles [J]. Phys Chem, 1997, 101: 6409-6414.
[19] MARTINEZ B, ROIG A, MOLINS E, GONZALEZ-CARRENO T, SEMA C J. Magnetic characterization of ��-Fe2O3 nanoparticles fabricated by aerosol pyrolysis [J]. Appl Phys, 1998, 83: 3256-3262.
[20] FELTIN N, PILENI M P. New technique for synthesizing iron ferrite magnetic nanosized particles [J]. Langmuir, 1997, 13(15): 3927- 3932.
[21] CEYLAN A, BAKER C C, HASANAIN S K, SHAH S I. Effect of particle size on the magnetic properties of core-shell structured nanoparticles [J]. Journal of Applied Physics, 2006, 100(3): 34301- 1-5.
[22] POZAS R, OCANA M, MORALES M P, SERNA C J. The influence of protective coatings on the magnetic properties of acicular iron nanoparticles [J]. Nanotechnology, 2006, 17(5): 1421-1427.
Corresponding author: YUAN Ming-liang; Tel: +86-731-88877209; E-mail: Chenke@mail.csu.edu.cn
DOI: 10.1016/S1003-6326(09)60190-4

