
Electrochemically switched ion exchange performances of capillary deposited nickel hexacyanoferrate thin films
HAO Xiao-gang(郝晓刚), GUO Jin-xia(郭金霞), LIU Shi-bin(刘世斌), SUN Yan-ping(孙彦平)
Department of Chemical Engineering, Taiyuan University of Technology, Taiyuan 030024, China
Received 17 August 2005; accepted 22 November 2005
Abstract: Thin films of capillary deposited nickel hexacyanoferrate(NiHCF) were investigated as electrochemically switched ion exchange(ESIX) materials. The films were generated on platinum and graphite substrates based on the ternary reagent diagram. In 1 mol/L KNO3 solution, cyclic voltammetry(CV) combined with energy-dispersive X-ray spectroscopy(EDS) was used to determine the influence of experimental conditions on the electroactivity of the NiHCF thin film on Pt substrates. The ion selectivity, ion-exchange capacity and the regenerability of NiHCF films on Pt and graphite substrates were investigated. The experiment results show that the NiHCF thin films from Ni2+-poor growth conditions have double peaks CV curves and contain relatively larger amount of potassium; while those from Ni2+-rich growth conditions are single peak CV curves and contain relatively smaller amount of potassium. It is demonstrated that the NiHCF thin films of capillary chemical deposition have good ESIX performances.
Key words: electrochemically switched ion exchange; nickel hexacyanoferrate; thin film; capillary chemical deposition
1 Introduction
Electrochemically switched ion exchange(ESIX) is an environmentally benign way to separate ions via reversible electrochemical modulation of the matrix charge density[1]. Ion loading and unloading can be easily controlled by modulating the redox states of ion exchange thin films prepared on conductive substrates, to perform the separation of ions from mixed solution and regeneration of the matrix. Since electrochemical rather than chemical potential (i.e. solution concentration) modulation is the main driving force for exchange, chemical regeneration of the ion-exchange matrix is not necessary[2]. The secondary waste created by chemical regenerants and associated rinse water is eliminated. So ESIX may replace the traditional ion exchange and have garnered intense recent interest[3-7].
Nickel hexacyanoferrate(NiHCF), an inorganic coordination compound with an open, zeolite-like structure, is an excellent candidate for ESIX separation of alkali cations because of its different affinity for alkali cations (Cs+>Rb+>K+>Na+>Li+)[8]. NiHCF can be made
as electroactive thin film by electrochemical methods: one is the anodic derivatization of a nickel surface[3, 4], another is cathodic deposition onto a conductive substrate[9,10]. Both methods consume lots of electrolyte and have to solve the recycle problem of depleted electrolyte; while the complex electrochemical system is also needed to perform the electrochemical reaction. So the cost is high and the operation is not convenient. Moreover, the traditional chemical deposition is simple but the good films are difficult to be deposited onto the conductive substrate[11-14].
In this study, we deposit NiHCF on the Pt and graphite substrates by capillary chemical deposition and get various structure and composition electroactive thin film samples according to the ternary reagent diagram. Cyclic voltammetry combined with the energy dispersive X-ray spectroscopy(EDS) is used to determine the preparation condition of NiHCF thin films with high electrochemical activity. This deposition method is simple and effective and can be used to screen the films with good ESIX properties (electroactivity, ion selectivity, stability and regeneration).
2 Experimental
2.1 Experiment instrument and reagents
All reagents were analytical grade and all solutions were prepared using Millipore water (18.2 MΩ?cm). All electrochemical experiments were performed using a VMP2 Potentiostat (Princeton USA) controlled with EC-Lab software. A three-electrode system was used with platinum sheet or graphite bar as the working electrode, a platinum wire as the counter electrode. All reported potentials are referenced to a saturated calomel electrode(SCE). EDS spectra were acquired using the American KEVEX SIGAMA unit.
2.2 Sample preparation
The platinum sheet electrode of 10 mm×10 mm surface area and 0.2 mm thickness was first mirror polished with different sandpapers. Prior to the capillary chemical deposition of a NiHCF film, the electrode surface was cleaned electrochemically in 1 mol/L H2SO4 by cyclic voltammetry from -275 to 1675 mV at 100 mV/s. The graphite rod electrode of 8 mm diameter was also polished by sandpapers, then rinsed with distilled water and dried in air.
Fig.1 shows a ternary diagram with varying volume fraction of three source reagents Fe3+(40 mmol/L K3Fe(CN)6), Ni2+(40 mmol/L NiSO4) and distilled H2O. The reagent percents labeled 1-13 were chosen to assess the range of materials produced with this combinatorial approach.
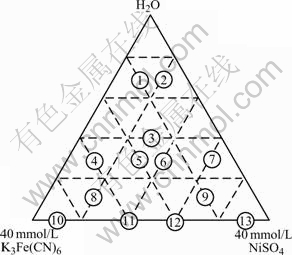
Fig.1 Ternary experimental design
Different NiHCF materials nucleated and grew on the surface of cleaned platinum and graphite electrodes by pipetting 4 μL of each reagent mixture into reagent well. The 7 mm×7 mm reagent wells were created using a PVC tape mask and the reagent mixing order was varied as follows:
Library A: Ni2+/H2O/Fe3+; Library B: Fe3+/H2O/Ni2+
The different combinations of Ni2+, Fe3+ and H2O were injected into the capillary aperture between the reagents well and a piece of hydrophobic PVC tape using a P2 pipetman (Gilson International), the chemical deposition of NiHCF was performed on the substrate in a room-temperature, water-saturated environment. The samples were left in the water-saturated environment for several hours. Then the NiHCF films were successfully prepared on substrates.
2.3 Electrochemistry
After film growth, each sample cycled 15 times in a 1 mol/L KNO3 solution from 100 to 1 000 mV at 25 mV/s. The sample was then rinsed with distilled H2O and cycled more 15 times in the same manner. After the cyclic voltammetry, the sample on platinum substrate was placed in oxidized (1 000 mV) state by holding the potential for 15 min. The sample was emersed from solution while still under potential control, then quickly rinsed, and dried in air. EDS spectra was acquired next.
Cation selectivity experiments were performed by electrochemical oxidation and reduction (cyclic volta- mmetry) of the NiHCF thin films on Pt and graphite substrates in various aqueous mixtures of Cs+ and K+ nitrate. After 25 cycles between 100 mV and 1 000 mV (at 25 mV/s) in a solution of interest, the electrode was quickly rinsed in deionized water to remove any electrolyte dragout and dried in air under ambient conditions. Seven different Cs+/K+ electrolyte mixtures were examined. These mixtures contained CsNO3 in the amounts 0, 10-5, 10-4, 10-3, 10-2, 0.1, and 1 mol/L, with KNO3 added to reach a total cation concentration of 1 mol/L.
3 Results and discussion
3.1 Influence of experimental conditions on NiHCF films
3.1.1 Electrochemical behavior of various NiHCF thin films
Fig.2 shows the plot of the final cyclic voltammo- gram(CV) for part films on Pt electrodes in 1 mol/L KNO3 solution. The positive currents correspond to the oxidation of NiHCF with K+ deintercalation from the matrix and negative currents denote the reduction with K+ intercalation. Fig.2 shows that the different preparation procedures for each film leads to quite different reversible charge densities and CV shapes. Some CVs from Ni2+-rich growth conditions display a predominant lower-voltage peak (0.48 V), such as A09, B12 in Fig.2(a); whereas other CVs from Ni2+-poor growth conditions show two comparable peaks (0.48 V and 0.6 V) separated by roughly 100 mV, such as A08, B10 in Fig.2(b).
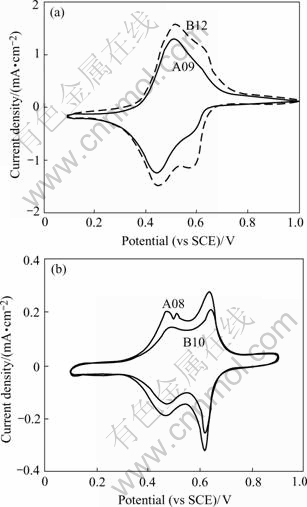
Fig.2 Cyclic voltammograms for NiHCF thin films on Pt cycled in 1 mol/L KNO3 at 25 mV/s
The relative electroactivity can be explored straightly by comparing the cyclic voltammogram area of each sample. The different processing conditions lead to different sample charge densities. The processing condition A12 creates films with the largest charge density (qA12), so we use it to normalize all the other conditions to get the relative charge density, xi=qi/qA12 for each sample i. The relative electroactivity of each sample and the reagent compositions that produce electroactive members in both libraries are shown schematically in Fig.3. Larger circles correspond to greater electroactivity, which indicate more films or thicker films produced in this sample. The ternary diagrams for the two libraries are plotted edge-to-edge to better show symmetry in the synthesis conditions. Fig.3 shows that high electroactivity samples in Library A are also high in Library B. The capacities of the samples in the middle of the diagram are quite high; whereas those of thin films from Fe3+-rich or Ni2+-rich growth conditions are usually low.
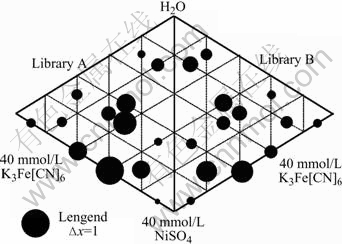
Fig.3 Electroactivity of NiHCF samples on ternary diagram
3.1.2 EDS spectroscopy and K+ contents of various NiHCF thin films
Energy dispersive X-ray(EDS) analysis is a fairly straightforward way to get at the relative cation content of different films. Fig.4 shows the oxidized-state EDS spectra for the same electroactive NiHCF samples shown in Fig.2. The peak at 3.3 keV corresponds to intercalated K ions while the peak at 6.4 keV is from Fe and 7.5 keV is from Ni. Each spectrum in Fig.4 is normalized to give identical integrated Fe peak intensities. Because the incident electrons easily penetrate through the entire NiHCF film and into the Pt substrate, a thin film analysis is possible (see details in [2, 6]). By normalizing with respect to Fe intensity (the films redox centers), the influence of film thickness is removed, so long as all the films are thin compared with the penetration depth of the electron beam. In this thin film limit, the K peak intensity is proportional to the amount of K intercalated into the matrix, so we can determine the K contents in the film by the ratio of K to Fe peak intensity (IK/IFe). Fig.4 shows that the NiHCF thin films from various preparation conditions have very different K intensity in oxidized state.
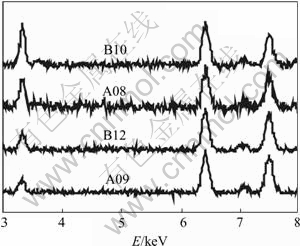
Fig.4 EDS spectra for parts of oxidized NiHCF samples
Eleven samples acquired the EDS spectra in Library A but only 10 samples in Library B. The ratios of IK/IFe in the EDS spectra for the 21 samples range from close to 0 (as A7) to greater than 1 (A11, B4, B10). These indicate the diversity of the samples with different composition and structure. Fig.5 shows the relationship between the K+ contents for each sample in both libraries and the reagent compositions that produce the film. The darker the circle, the higher the ratio of IK/IFe is; and the whiter the circle, the lower the ratio is. The IK/IFe ratio plot shows that the darker samples are from high iron areas and the whiter samples are from low iron areas. Thus, the NiHCF thin films from different film-growth areas or conditions will have different electrochemical behaviors and compositions or structures. The ternary diagrams for two libraries display good symmetry, indicating there is not too much dependence on mixing order.
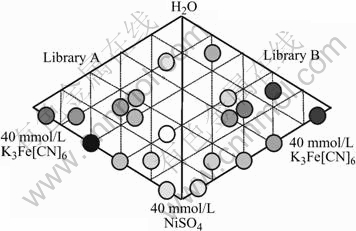
Fig.5 K+ contents of NiHCF samples on ternary diagram
The ideal ESIX thin film should have high electroactivity and low cation contents in oxidized state; therefore, NiHCF thin films from Ni2+-rich growth condition are suitable for separation of the alkali cations. In the following sections we will focus on sample 9 in Library B.
3.2 Ion selectivity of NiHCF thin film
Fig.6 shows the cyclic voltammograms for the NiHCF thin film on graphite substrate cycled in different electrolyte solutions with varied K+/Cs+ compositions. These curves illustrate the transition from predominantly K+ exchange to Cs+ exchange as the capillary deposited film is cycled in K+/Cs+ mixtures with increasing amount of Cs+. The redox potential of the reversible wave depends on the ionic radius of the exchanging cation in the film. Therefore, the shift of the redox potential toward more positive values in curves a through f qualitatively indicates an increasing amount of Cs+ loading and the higher potential of the cesium CV peak also indicates the selectivity of this species over K+[1, 2]. The film on Pt substrate has similar electrochemical characteristics.
In addition to a shift in redox potential, the reversible ion-exchange capacity is seen to decrease dramatically and the redox peak becomes flat as [Cs+/K+] in solution increases. Since the cyclic voltammetry in pure 1.0 mol/L KNO3 or 1.0 mol/L CsNO3 results in the exclusive exchange of K+ or Cs+, respectively, the CV peak in pure KNO3 solution is steep and that in pure CsNO3 solution is flat. The broad peak with low current indicates qualitatively the rate of loading and unloading of films. The broader the peak is, the slower the loading or unloading rate of ions is and the higher affinity for Cs+ the film has. These results are consistent with the NiHCF films prepared by anodic derivatization and cathodic deposition[1, 15].
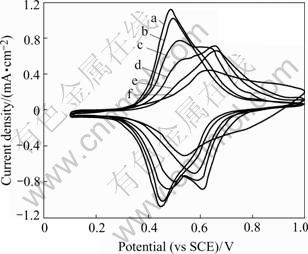
Fig.6 Cyclic voltammograms for NiHCF film on graphite substrate in a series of aqueous K+ and Cs+ nitrate mixtures (The total alkali content of ([Cs+] + [K+]) is 1 mol/L, the Cs+ concentrations in each solution were 10-5 mol/L(a), 10-4 mol/L(b), 10-3 mol/L(c), 10-2 mol/L, 10-1 mol/L and 1 mol/L(f).)
3.3 Cycle life of NiHCF thin film
The cycle life is an important property to assess ESIX films. The voltammetric cycles mimic redox switching in an ESIX process and are a measure of cycling stability. The CVs change of films on Pt and graphite are investigated in 1 mol/L KNO3 at 25 mV/s for 1000 times. Fig.7 clearly indicates decreases in cation loading capacity as a function of cycling. After 1 000 cycles, the Pt film demonstrates a loss of about 37.45% of its capacity and the graphite film lost only 24.5% of its capacity. But they still have good redox activity. Also note that two distinct peaks appear in the CVs of Pt substrate with the increasing of the cycle number. This may be caused by structural modifications in the NiHCF film with cycling. But the CVs of graphite substrate keep the same shape as the first cycle. So the film on graphite shows better stability and is suitable for the application of ESIX.
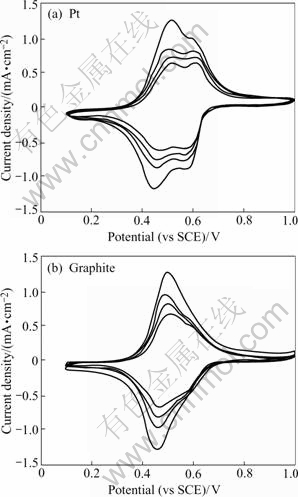
Fig.7 Cyclic voltammograms for repeated potential cycling of NiHCF films in 1 mol/L KNO3 (The cycling number is 1, 200, 500, and 1000 respectively from outside to inside.)
3.4 Electrochemical regeneration of NiHCF thin film
One advantage of ESIX technology is that the transition of oxidized and reduced states corresponding to unloading and loading of metal ions of NiHCF thin film is easily controlled by electrochemistry. Accordingly the metal ions are separated and thin films are regenerated. The film used in the solution containing Cs+ is called Cs+ form. The regeneration of the thin films on Pt and graphite substrates from the Cs-form to the K-form can be accomplished by cycling a Cs-loaded film in a K+-containing electrolyte. Fig.8 shows the voltammograms of regenerated films at the 25th cycle in 1 mol/L KNO3 solution. For comparison, the initial CVs of the films are also shown in this figure. After regeneration the CVs for Pt and graphite films have 77% and 80% as much ion-exchange capacity as initially CVs in K+-containing electrolyte respectively. The ion exchange capacity could be increased with the cyclic number and the regenerated NiHCF films on Pt and graphite still have good redox reversible activity.
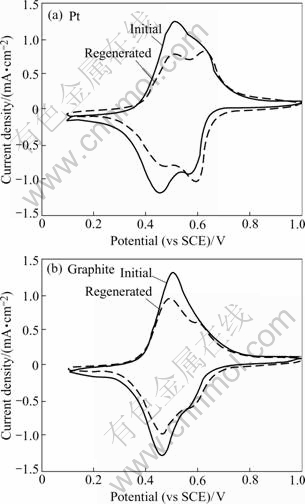
Fig.8 Cyclic voltammograms for matrix regeneration by potential cycling Cs-loaded film in 1 mol/L KNO3
4 Conclusions
1) Based on the ternary reagent diagram, the electroactivity NiHCF thin films are successfully prepared by capillary chemical deposition on platinum and graphite substrates. The ideal preparation for electroactivity NiHCF thin film is from Ni2+-rich growth condition according to cyclic voltammograme and EDS.
2) It has been demonstrated that the capillary deposited NiHCF thin films are suitable for ESIX processes. The NiHCF thin films have good Cs+ selectivity, stability and regenerability.
3) The ESIX performances of NiHCF film on graphite substrate are better than that on Pt substrate. It is important to the development of a practical and large-scale ESIX process.
Acknowledgments
The authors thank Prof. D. T. SCHWARTZ and Dr. W. A. STEEN (University of Washington) for discussions and beneficial suggestions.
References
[1] Rassat S D, Sukamto J H, ORTH M A, LILGA M A, HALLEN R T. Development of an electrically switched ion exchange process for selective ion separations [J]. Sep Puri Technol, 1999, 15: 207-222.
[2] Jeerage K M, Schwartz D T. Characterization of cathodically deposited nickel hexacyanoferrate for electrochemically switched ion exchange [J]. Sep Sci Technol, 2000, 35(15): 2375-2392.
[3] Lilga M A, Orth R J, Sukamto J P H, HAIGHT S M, SCHWARTZ D T. Metal ion separations using electrically switched exchange [J]. Sep Puri Technol, 1997, 11: 147-158.
[4] Lilga M A, Orth R J, Sukamto J P H, RASSAT S D, GENDERS J D, GOPAL R. Cesium separation using electrically switched ion exchange [J]. Sep Puri Technol, 2001, 24: 451-466.
[5] de Tacconi N R, Rajeshwar K. Metal hexacyanoferrate: electrosynthesis, in situ characterization and applications [J]. Chem Mater, 2003, 15: 3046-3062.
[6] Jeerage K M, Steen W A, Schwartz D T. Correlating nanoscale structure with ion intercalation in electrodeposited nickel hexacyanoferrate thin films [J]. Chem Mater, 2002, 14: 530-535.
[7] Reyes G J, Medina J A, Jeerage K M, STEEN W A, SCHWARTZ D T. High capacity SiO2-graphite composite electrodes with chemically incorporated metal MHCFs for electrochemically switched alkaline cation exchange [J]. J Electrochem Soc, 2004, 151(9): D87-92.
[8] Schneemeyer L F, Spengler S E, Murphy D W. Ion selectivity in nickel hexacyanoferrate films on electrode surfaces [J]. Inorg Chem, 1985, 24: 3044-3046.
[9] Steen W A, Jeerage K M, Schwartz D T. Raman spectroscopy of redox activity in cathodically electrodeposited nickel hexacyanoferrate thin films [J]. Appl Spectrosc, 2002, 56(8): 1021-1029.
[10] Jeerage K M, Steen W A, Schwartz D T. Charge- Density-Partitioning of Cs+ and K+ into nickel hexacyanoferrate matrixes [J]. Langmuir, 2002, 18: 3620-3625.
[11] Steen W A, Schwartz D T. A route to diverse combinatorial libraries of electroactive nickel hexacyanoferrate [J]. Chem Mater, 2003, 15: 2440-2453.
[12] Zamponi S, Berrettoni M, Kulesza P J, MIECZNIKOWSKI K, MALIK M A, MAKOWSKI O, MARASSI R. Influence of experimental conditions on electrochemical behavior of prussian blue type nickel hexacyanoferrate film [J]. Electrochim Acta, 2003, 48: 4261-4269.
[13] Loos-Neskovic C, Fedoroff M. Fixation mechanisms of cesium on nickel and zinc ferrocyanides [J]. Solvent Extr Ion Exch, 1989, 7: 131-158.
[14] Green-Pedersen H, Korshin G V. Separation of cesium from high ionic strength solutions using a cobalt hexacyanoferrate- modified graphite electrode [J]. Environ Sci Technol, 1999, 33: 2633-2637.
[15] Steen W A, Han S, Yu Q, GORDON R A, CROSS J O, STERN E A, SEIDLER G T, JEERAGE K M, SCHWARTZ D T. Structure of cathodically deposited nickel hexacyanoferrate thin films using XRD and EXAFS [J]. Langmuir, 2002, 18: 7714-7721.
Foundation item: Project(20006011) supported by the National Natural Science Foundation of China; Project(20021017) supported by the Natural Science Foundation of Shanxi Province; Project(2004-24) supported by the Scholar Council Foundation of Shanxi Province, China
Corresponding author: HAO Xiao-gang; Tel: +86-351-6018193; E-mail: xghao@tyut.edu.cn
(Edited by YUAN Sai-qian)