
Establishment of hydrogen measurement system
for magnesium alloy melt
XU Si-xiang(������), WU Shu-sen(����ɭ), MAO You-wu(ë����), AN Ping(�� Ƽ)
State Key Laboratory of Material Processing and Die & Mould Technology,
Huazhong University of Science and Technology, Wuhan 430074, China
Received 28 July 2006; accepted 15 September 2006
Abstract: The equations of hydrogen solubility in pure magnesium and its alloy were deduced based on thermodynamic analysis: for pure magnesium, lgc(H)=0.5lg p(H2)-1 332/T+0.568; for AZ91 alloy, lgc(H)=0.5lg p(H2)-1 332/T+0.483. Based on the above equations, a rapid and reliable measurement system for hydrogen content in magnesium melt was set up with CPU controller and electric circuit. With this instrument, measurement experiments were carried out to determine hydrogen content in AZ91 melt. The results show that the actual hydrogen level of AZ91 melt under gas protection varies from 0.06 mL/g to 0.14 mL/g at the temperature range from 650 �� to 750 ��, and hydrogen content lineally increases with the increase of temperature.
Key words: magnesium alloy melt; hydrogen content; thermodynamic equation; measurement
1 Introduction
The most notable gas is hydrogen, which is a common gaseous impurity in magnesium alloy melt[1]. Microporosity from hydrogen redistribution during solidification in the magnesium castings can reduce mechanical properties[2-4], such as ultimate strength, yield strength, ductility and fatigue resistance. This problem can only be dealt with and solved if hydrogen content in the liquid metal can be measured reliably and preferably on-line. There are at least five different methods for determination of hydrogen in magnesium[5-9]. These methods are based on two main principles. One is direct measurement of the gas that can be extracted from a restricted sample and the other is measurement of the hydrogen partial pressure above the melt. The former requires very careful handling and preparation of the samples and the extraction takes about 1-2 h; the later strongly depends on the diffusion-rate of hydrogen in the melt and it is also time and labor consuming. These methods are mainly used in experiment in laboratory as well. Yet no commercial instrument is available even though several attempts have been made. The main aim of this paper is to develop a reliable and rapid technique for analysis of hydrogen content in magnesium melt. The relation between temperature and hydrogen content of magnesium alloy melt under gas protection is investigated by using a self-developed instrument.
2 Hydrogen solubility in magnesium melt
2.1 Hydrogen solubility in pure magnesium melt
The original hydrogen in molten magnesium comes from the moisture of dissolvent, moisture adsorbed on metal surface and metal corrosion. The mechanism of gas absorption is as follows:
Mg(l)+H2O(g)=MgO(s)+H2(g)
1/2H2(g)=[H] (1)
where [H] denotes hydrogen solute in magnesium melt. The solute is so dilute that Sievert��s law applies. According to Sievert��s law, hydrogen solubility can be expressed as
lgc(H)=0.5lgp(H2)-A/T+B (2)
References[10-11] summarized the experimental hydrogen solubility data obtained by several investigators[12-17]. The correlation of the temperature and hydrogen content at 1.0��105 Pa in magnesium is fitted with the least squares method, as shown in Fig.1. The values of A and B can be obtained based on the fitted curve of Fig.1(limited to the liquid phase stage):
A=1332; B=0.568 (3)
That is, mathematic equation of hydrogen content in magnesium melt can be expressed as
lg c(H)=0.5lg p(H2) -1 332/T��0.568 (4)

Fig. 1 Hydrogen solubility in pure magnesium[10-11]
2.2 Hydrogen solubility in AZ91 magnesium alloy melt
Activity coefficient is introduced in the expression involving in magnesium alloy, therefore, Eqn.(1) becomes
lg c(H)=-lg fH+0.5lg p(H2)-A/T+B (5)
In pure magnesium, the activity coefficient fH may be given as unity, i.e. fH=1. While in magnesium alloys, fH changes with the components and contents of alloying elements. For example, for AZ91, the correlation of fH
and the interaction coefficient 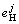 is given by
is given by
lg f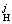 =
=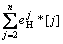 (6)
(6)
where j denotes the effect of the alloying elements. Al and Zn are the major alloying elements in AZ91, which have contents of 9% and 1% (mass fraction), respectively. Ref.[18] presented the interaction coefficients: 0.0087 for Al; 0.006 4 for Zn. Thus the activity coefficient, fH, can be calculated out, and accordingly the solubility of hydrogen in AZ91 magnesium alloy melt is expressed as
lg c(H)=0.5lg p(H2)-1 332/T+0.483 (7)
The thermodynamic equations of hydrogen solubility in magnesium melt are sound. Ref.[19] verified them.
3 Principles of measurement method of hydrogen content in magnesium melt
Based on the above thermodynamic equation, a rapid measurement apparatus was developed and its schematic diagram is shown in Fig. 2. It consists of three parts: a sample chamber including crucible, heater block and sealing unit; a vacuum system including vacuum pump, electromagnetic valves and pipeline; and a data processing system including high sensitive pressure transmitter, thermocouples and CPU circuit.
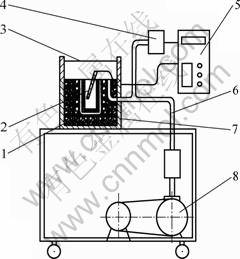
Fig.2 Schematic diagram of hydrogen content measurement instrument: 1 Crucible; 2 Thermocouple; 3 Cover and glass; 4 Pressure transmitter; 5 CPU circuit system; 6 Vacuum pipeline; 7 Resistance furnace; 8 Vacuum pump
It can be seen from Eqn.(5) that hydrogen content can be determined by measuring the temperature and pressure above the melt at the same time when the first bubble immerges. In this study, the hydrogen pressure in the bubble in the melt is equal to the pressure in the space above the melt. The system diagram is shown in Fig.3.
The temperature is collected and converted to an electrical signal in thermocouple, then by temperature module the electrical signal is converted back to the actual temperature and data signal and enters into the CPU controller. In the same way the pressure is transported into the CPU controller. CPU system deals with these data and calculates out hydrogen content in

Fig.3 Schematic diagram of hydrogen measurement system in magnesium melt
magnesium melt. Then the results are displayed in text displayer and printed out with a microprinter.
The diagram of structural program is shown in Fig.4.

Fig.4 Structural program diagram of hydrogen measurement system
4 Measurements of hydrogen level in Mg alloy melt
With the self-developed instrument, the correlation between the temperature and hydrogen content in Mg alloy molten under gas protection was investigated. The chemical compositions of AZ91 magnesium alloy are: 9.03% Al, 0.64% Zn, 0.33% Mn, 0.031% Si, 0.004 9% Cu, 0.001 1% Fe, 0.000 3% Ni, 0.001 4% Be, balance Mg. Tools and raw materials used in the experiment were preheated to 200�� in the stove before experiment in order to eliminate the water. About 10 kg raw materials of AZ91 alloy were melted under protection of Ar��HFC-134a mixture gases in an electrical resistance crucible furnace. Hydrogen contents were measured at the temperatures of 650, 675, 700, 725 and 750 �� respectively. Hydrogen content at every temperature was measured three times. The technological flow chart of hydrogen measurement is shown in Fig.5. During the measurement, about 100 g Mg alloy melt was taken from the melting furnace, then put into the small crucible of the instrument, as shown in Fig.2. After the cover was closed, the vacuum pump was started to extract air in the system. The measurement of temperature and pressure was done when the hydrogen bubble immerged. Then the hydrogen content was calculated out by the CPU and displayed.

Fig.5 Technological flow chart of measurement
The measured results are fitted by least squares method; and the correlation curve between temperature and hydrogen content is obtained, as shown in Fig.6.

Fig.6 Correlation of temperature and hydrogen content in AZ91 alloy melt
It is known from Fig.6 that the actual hydrogen level of AZ91 melt increases proportionally with the increase of temperature. The hydrogen level in AZ91 melt is about 0.06 mL/g at 650 ��, about 0.1 mL/g at 700 ��, and about 0.14 mL/g at 750 �� respectively. The data are fitted by the least squares method and Eqn.(6) is obtained:
y=-46.499 68+0.081 15t (8)
where t denotes temperature in ��, y is hydrogen content in Mg melt in 10-2mL/g. The correlation coefficient, R, is 0.99622.
BAKKE et al[20] set forth the five steps of hydrogen absorption of magnesium melt from the atmosphere: 1) Water vapor diffuses through the gas boundary layer at the melt surface. 2) Water molecules are adsorbed at the surface. 3) Adsorbed water molecules react with magnesium, forming gaseous hydrogen: Mg+H2O=MgO+H2. 4) The gaseous hydrogen dissociates at the surface, forming atomic hydrogen according to H2=2H. 5) Hydrogen atoms are transported through the melt surface layer by diffusion and convection. It is seen that with the increase of temperature, the reactions of the step 3) and step 4) are aggravated so that the hydrogen content becomes greater. At the same time, the diffusion rate is faster, thus, hydrogen easily transfers into the melt and hydrogen content increases.
5 Conclusions
1) The equations of hydrogen solubility in pure magnesium and AZ91 alloy are modified based on thermodynamic analysis:
For pure magnesium: lg c(H)=0.5lg p(H2)-1 332/ T+0.568;
For AZ91 alloy, lg c(H)=0.5lg p(H2)-1 332/T+ 0.483.
2) A fast measurement system for hydrogen content of magnesium melt is developed with a CPU controller, temperature and pressure measurement parts. This unit can be used as commercial analyzing equipment in the future.
3) Hydrogen content increases proportionally with the increase of temperature in AZ91 magnesium alloy melt, which is melted under protection of Ar+HFC-134a mixture gases.
References
[1] ENGH T A. Principles of Metal Refining[M]. Oxford University Press, 1992. 302.
[2] YUAN Guang-yin, LIU Man-ping, DING Wen-jiang, INOUE A. Microstructure and mechanical properties of Mg-Zn-Si-based alloys[J]. Mater Sci Eng A, 2003, 357(1/2): 314-320.
[3] DU Wen-wen, SUN Yang-shan, MIN Xue-gang, XUE Feng, ZHU Min, WU Deng-yun. Microstructure and mechanical properties of Mg-Al based alloy with calcium and rare earth additions[J]. Mater Sci Eng A, 2003, 356(1/2): 1-7.
[4] YUAN Guang-yin, SUN Yang-shan, DING Wen-jiang. Effects of bismuth and antimony additions on the microstructure and mechanical properties of AZ91 magnesium alloy[J]. Mater Sci Eng A, 2001, 308(1/2): 38-44.
[5] SIMENSEN C J. Gas-chromatic determination of hydrogen in magnesium by means of the capsule method[J]. Z Anal Chem, 1978, 302: 207-217.
[6] COE F R, JENKINS N, PARKER D H. Encapsulation as a technique for the measurement of hydrogen in volatile metals[A]. Nottingham S.A.C. Conference[C]. Nottingham, 1966: 274-279.
[7] WATANABE T, HUANG Y C, KOMATSU R. Solubility of hydrogen in magnesium[J]. Light Metals, 1975: 76-81.
[8] IMABAYASHI M. On a method for measuring gas content of light alloys, containing volatile constituents[J]. Light Metals Keikinzoku, 1965: 167-174.
[9] BAKKE P. Measurement and removal of inclusions and hydrogen in magnesium[D]. Norwegian Institute of Technology, 1992.
[10] SHEAROUSE J D ��MIKUCKI B A. The origin of microporosity in magnesium alloy AZ91[J]. J Mater Manufacturing, 1994, 103: 542-552.
[11] ZENG K, KLASSEN T, OELERICH W, BORMANN R. Critical assessment and thermodynamic modeling of the Mg-H system[A]. Inter J Hydrogen Energy, 1999, 24: 989-1004.
[12] KOENEMAN J, METCALFE A G. The solubility of hydrogen in magnesium[J]. Trans ASM, 1959, 51: 1072-1082.
[13] HUANG Y C, WATANABE T, KOMATSU R. Hydrogen in magnesium and its alloy[A]. 4th Intl Conf Vacuum Metallurgy[C]. Tokyo, 1973: 176-179.
[14] SHAPOVALOV V I, SERDYUK N P, SEMIK A P. Magnesium-hydrogen and aluminum-hydrogen phase diagrams[J]. Dep Akad Nauk Ukr RSR.Ser A Fiz-Mat Tekh Nauki, 1981, 6: 99-101.
[15] CHENEGA D F, GOTV V, PRIS C. Diffusion and solubility of hydrogen in liquid magnesium[J]. Betsn Kiev Politexn Inta Kiev, 1979, 17: 56-58.
[16] POPVIC Z D, PIERCY G R. Measurement of the solubility of hydrogen in solid magnesium[J]. Metall Trans A, 1975, 6A: 1915-1917.
[17] SHAPOVALOV V I, SEMIK A P, TIMCHENKO A G. On the solubility of hydrogen in liquid magnesium[J]. Metallurgy, 1993, 3: 25-28.
[18] OVRELID E, ENGH T A, OYMO D. Hydrogen measurement in pure and alloyed magnesium[J]. Light Metals, 1994: 771-778.
[19] XU Si-xiang, WU Shu-sen, MAO You-wu, AN Ping, GAO Pei-qing. Variation of hydrogen level in magnesium alloy melt[J]. China Foundry, 2006: 4.
[20] BAKKE P, LAURITZEN J L, ENGH T A, OYMO D. Hydrogen in magnesium absorption, removal and measurement[J]. Light Metals, 1991: 1015-1023.
(Edited by YANG Bing)
Corresponding author: WU Shu-sen; Tel: +86-27-87556262; E-mail: ssw636@hotmail.com