Preliminary bioleaching of heavy metals from contaminated soil employing indigenous Penicillium Chrysogenum strain F1
��Դ�ڿ������ϴ�ѧѧ��(Ӣ�İ�)2012���7��
�������ߣ����»� ����Ԫ ��־�� ʯ�� ͯ��ϼ ������
����ҳ�룺1973 - 1979
Key words��heavy metal; bioleaching; Penicillium Chrysogenum; organic acid
Abstract: Bioleaching is an environment-friendly and economical technique to remove heavy metals from contaminated soil. The objective of this work is to find out an indigenous strain to remedy soil contaminated by Zn, Pb, Cu and Cd. A strain which was selected from the soil of a local smelting industry was found to be able to produce many organic acids and degrade pH value of the liquid medium. The fungus strain is identified as Penicillium Chrysogenum (P. Chrysogenum) by sequencing 18srDNA and ITS. Bioleaching condition using P. Chrysogenum is optimized. Glucose is the best carbon source for P. Chrysogenum and inorganic nitrogen is better than organic nitrogen. In addition, neutral solution and room temperature are fit for P. Chrysogenum to bioleach. In the one-step bioleaching, the bioleaching ratios are 39.95% for Zn, 9.4% for Pb, 34.89% for Cu and 49.59% for Cd, which are 53.89% for Zn, 14.44% for Pb, 55.53% for Cu and 62.81% for Cd in the two-step bioleaching. The efficiency of two-step bioleaching is better than the one-step bioleaching. P. Chrysogenum is effective in removing heavy metals from the contaminated soil.
J. Cent. South Univ. (2012) 19: 1973-1979
DOI: 10.1007/s11771-012-1234-8![]()
DENG Xin-hui(���»�)1, 2, CHAI Li-yuan(����Ԫ)1, YANG Zhi-hui(��־��)1, SHI Yan(ʯ��)1,
TONG Hai-xia(ͯ��ϼ)1, WANG Zhen-xin(������)3
1. College of Metallurgical Science and Engineering, Central South University, Changsha 410083, China;
2. College of Packing and Material Engineering, Hunan Industry University, Zhuzhou 412007, China;
3. South China Institute of Environmental Sciences,Ministry of Environmental Protection, Guangzhou 510655, China;
? Central South University Press and Springer-Verlag Berlin Heidelberg 2012
Abstract: Bioleaching is an environment-friendly and economical technique to remove heavy metals from contaminated soil. The objective of this work is to find out an indigenous strain to remedy soil contaminated by Zn, Pb, Cu and Cd. A strain which was selected from the soil of a local smelting industry was found to be able to produce many organic acids and degrade pH value of the liquid medium. The fungus strain is identified as Penicillium Chrysogenum (P. Chrysogenum) by sequencing 18srDNA and ITS. Bioleaching condition using P. Chrysogenum is optimized. Glucose is the best carbon source for P. Chrysogenum and inorganic nitrogen is better than organic nitrogen. In addition, neutral solution and room temperature are fit for P. Chrysogenum to bioleach. In the one-step bioleaching, the bioleaching ratios are 39.95% for Zn, 9.4% for Pb, 34.89% for Cu and 49.59% for Cd, which are 53.89% for Zn, 14.44% for Pb, 55.53% for Cu and 62.81% for Cd in the two-step bioleaching. The efficiency of two-step bioleaching is better than the one-step bioleaching. P. Chrysogenum is effective in removing heavy metals from the contaminated soil.
Key words: heavy metal; bioleaching; Penicillium Chrysogenum; organic acid
1 Introduction
Soil contamination has become a serious problem with the economy and industry development. Heavy metal soil contamination is more serious than any other soil contamination.
Smelting and beneficiation are the main ways of soil heavy metal contamination. Smelting slags increase with the development of smelting industry and significant quantities of metals are embodied in them (e.g. Al, Zn, Pb and Cd). Thus, soil heavy metal contamination under the smelting slags becomes more serious.
Generally speaking, there are two principles to remedy the contaminated soil: 1) The heavy metal is retained in the soil, but the migration or bioavailability of heavy metal is degraded by changing the forms of the heavy metal. Some chemical reagents are often added to the soil so that the chemical forms of heavy metal in the soil are changed. In addition, some bacteria are able to degrade the toxicity of heavy metal through redox reaction. For example, Cr6+ is able to be reduced to Cr3+ by Pannonibacter sp., Bacillus sphaericus, Achromobacter sp., Desulfovibrio vulgaris, Enterobacter cloacae, Pseudominas aeruginosa [1-3], and As3+ can be oxided to As5+ by Aeidovorax sp., and Bosea sp. [4-5]; 2) The heavy metal is leached from the contaminated soil and its content is decreased. Some chemical reagents including inorganic acids, organic acids and organic complexants are used, such as H2SO4, HNO3, HCl, citric acid, oxalic acid, malic acid and EDTA [6]. Heavy metal is usually able to be solubilized or extracted from the contaminated soil by these reagents. Moreover, heavy metal in the contaminated soil is leached by the metabolites produced by some fungi. For example, Aspergillus genus and Penicillium genus are often used to bioleach the contaminated soil [7-8].
In accordance with the above two principles, some methods are searched out, such as soil removed engineering, chemical process, physico-chemical process and biological process. Mixing of soil is a time, manpower and material resource consuming method, thus it is only suitable for the contaminated soil in a small area [9]. There is no bad aftereffect when using method of physico-chemical process (electrothermal or electrokinetic). But electricity is more precious at present and the scope of application of this method is small. Chemical process is not a permanent remediation because the precipitates may be solubilized with the change of external environment and a secondary contamination may be caused at any time with the change of external environment.
Biological process is often used to remedy contaminated soil [10-11]. Especially, microremediation is more popular than long-time plant remediation and less-efficient animal remediation. Microorganism is usually used to degrade the toxicity of heavy metal through redox or oxidation, also through bioleaching to decrease heavy metal content in the soil. At present, more and more concerns have been drawn on bioleaching for its economic, ecological and friendly advantages. Some filamentous fungi belonging to Aspergillus genus and Penicillium genus were used to solublize metallic elements and remove heavy metal in the soil or tailings [6, 12]. But the bioleaching efficiency is needed to be raised.
The aims of this work are: 1) to find out an indigenous strain capable of producing organic acids from the contaminated soil, 2) to optimize the conditions for producing organic acids, 3) to select the best conditions of bioleaching, and 4) to use it to remove heavy metal from the contaminated soil.
2 Materials and method
2.1 Sample collection and chemical analysis
The contaminated soil used in this work was collected from the top soil (0-20 cm) of the sites under a slag heap at the Smelting Industry, which was built in 1956 and located in Zhuzhou city, Hunan Province, central south of China. The Smelting Industry produces metals such as Pb, Zn, Cu, Ag and Au. The area is well known in China for its notorious heavy metal contamination. The soil sample was air dried and sieved through a 2 mm sieve to break soil clumps and to remove sundries, and then was mechanically mixed to ensure homogeneity and stored in a plastic container for subsequent experiments. The total heavy metal content in the soil was determined by an electric heating plate-acid digestion method (HCl+HNO3+HClO4+HF) [13-14]. The digested colloid was dissolved by dilute nitric acid and then transferred to 100 mL volumetric flask, diluted 10 times and analyzed for the heavy metal content using a Perkin-Optima 3 000 V inductively coupled plasma- optical emission spectrometer (ICP-OES). The pH value of the soil was determined according to the method of potentiometric determination of solution pH value using a pH-meter (pHS-3B), in which the ratio of soil to water was 1:2.5 [15]. All experiments were run in triplicate. The physico-chemical characteristics of contaminated soil are presented in Table 1. The soil was highly contaminated by kinds of heavy metals, and the national standard is largely outnumbered by the concentration of heavy metals in the soil and a significant hazard for human health and the environment is posed.
2.2 Fungal isolating, purification and acclimation
For some indigenous strains isolated to be leached soils, 0.1 g soil was added to 9.9 mL sterile deionized-water. Thereafter, the supernatant was decanted and diluted into a series of dilution sequences (1 mL supernatant was added to double-deionized water) [16]. A 0.1 mL of 10-4, 10-5, 10-6 and 10-7 dilution sequence was spread individually on Chashi agar plates, in which 100 mg/L Cd, 150 mg/L Pb and 150 mg/L Zn were contained. The medium was prepared by adding 30 g sucrose, 1 g K2HPO4, 3 g NaNO3, 0.5 g MgSO4, 0.5 g KCl, 0.01 g FeSO4 and 1 L H2O. The pH value of Chashi liquid medium was adjusted to 6.7 using 0.1 mol/L HCl. The strains were incubated in electro-thermal incubator at 30 �� for 7 d. Subsequently, the above strains were isolated and purified, and then innoculated into modified Chashi liquid medium, which was prepared by 90 g glucose, 0.05 g KH2PO4, 3 g NaNO3, 0.5 g MgSO4, 0.5 g KCl, 0.01 g FeSO4 and 1 L H2O. Medium pH was adjusted to 6.7 using 0.1 mol/L HCl. From the third day, the pH value of the liquid medium was monitored so that a strain would be found out, which can degrade the pH value of the liquid medium. The degradation of the pH value means that many organic acids were produced during the oxidation of the sugar [17].
2.3 Identification of strain
A single colony of pH-degrade strain from a fresh liquid medium was extracted and the strain species was identified by sequencing 18SrDNA and ITS, of which DNA was extracted using method of PhCH2Cl [18]. Strain��s 18SrDNA gene primers and ITS gene primers were used for polymerase chain reaction (PCR) amplification of 18SrDNA gene and ITS gene. 18SrDNA gene primers are P18sf:5��-AACCTGGTTGATCCTGC- CAGT-3�� and P18sr:5��-CGACGGGCGGTGTGTAC-3��, ITS4 gene primer is 5��-TCCTCCGCTTATTGATATGC- 3��, and ITS5 gene primer is 5��-GGAAGTAAAAGTA- ACAAGG-3��. The PCR products were purified using the TLA quick Midi Purification Kit. The purified PCR products were sent to Wuhan Biological Company for sequencing. The sequences were initially analyzed at NCBI server (http://www.ncbi.nlm.nih.gov/) using BLAST tools and corresponding sequences were downloaded.
Table 1 Physico-chemical characteristics of contaminated soil

2.4 Selection of medium and carbon source
Carbon source kind and content are key factors for strain to produce organic acids. Different glucose and sucrose contents were used for P. Chrysogenum to produce organic acids in this work. Modified Chashi liquid medium was employed in which carbon sources were glucose (60, 90 and 120 g/L) and sucrose (60, 90 and 120 g/L) individually. 49 mL of modified Chashi liquid medium was put in 250 mL autoclaved conical flasks with 2.5 g autoclaved soil ( e.g. soil:water=5% (w/��)) to bioleach. 1 mL spore suspension (about 108 spores/mL) was added aseptically to each conical flask, and then was put in a rotary shaking incubator at 30 �� and 120 r/min for 15 d. Sterile deionized water was added every other day in order to keep mass balance of each conical flask. The number of spores was counted using a haemocytometer and standardized to approximately 7.5��108 mL-1. The bioleaching ratios of different carbon sources after 15 d were compared by determining Pb, Zn, Cd and Cu concentrations in filtrates. Sterile experimental conditions were achieved with autoclave, each flask contained modified Chashi liquid medium at 115 �� for 30 min prior to inoculation, and the contaminated soil was autoclaved separately. Each carbon source was run in triplicate.
2.5 Selection of nitrogen source of liquid medium
In order to find out a proper nitrogen source for P. Chrysogenum, the nitrogen source of modified Chashi medium was changed into (NH4)2SO4, NH4NO3, peptone, beef extract or yeast extract individually. Nitrogen content in the modified Chashi liquid medium was equivalent to that in Chashi liquid medium (3 g/L NaNO3). The bioleaching ratios of different nitrogen sources after 15 d were compared by determining Pb, Zn, Cd and Cu concentrations in filtrates. All other operation sequences were the same as those in the selecting of carbon source. Each nitrogen source was run in triplicate.
2.6 Selection of pH value of liquid medium
49 mL modified Chashi liquid medium with 2.5 g autoclaved soil (e.g. soil:water=5% (w/��)) was used to bioleach in 250 mL autoclaved conical flasks. 1 mL spore suspension (about 108 mL-1) was added aseptically to each conical flask. The pH value of above medium was adjusted to 3, 5, 6.7, 7 and 9. The method of bioleaching was the same as the above, and then the largest bioleaching ratio of modified Chashi liquid medium at that pH value was selected. Each pH value level was run in triplicate.
2.7 Selection of temperature of bioleaching
Soil was bioleached using the same liquid medium as the above at 20, 30 and 40 ��. The ratios of bioleaching at different temperatures were compared by determining the metal concentrations in filtrates using ICP-OES. Each temperature level was run in triplicate.
2.8 Comparison between different methods of bioleaching
2.8.1 One-step bioleaching experiments
The one-step bioleaching process was carried out by putting 49 mL modified liquid medium (90 g/L glucose, pH 7.0) into 250 mL autoclaved conical flasks with 2.5 g autoclaved soil. 1 mL of spore suspension (about 108 mL-1) was added to these conical flasks individually. 50 mL modified Chashi liquid medium with 2.5 g autoclaved soil without inoculation was used as control. All flasks were incubated in a rotary shaking incubator at 120 r/min and 20 �� for 15 d. All experiments were run in triplicate.
2.8.2 Two-step bioleaching experiments
In the two-step bioleaching process, 1 mL of spore suspension ( about 108 mL-1) was first innoculated in 49 mL of modified Chashi liquid medium (90 g/L glucose, pH 7.0) in 250 mL autoclaved conical flask without adding any soil (the first step). Five days later, 5% (w/��) autoclaved soil was added to the conical flask (the second step). Bioleaching was carried out by tumbling the mixture in a rotary shaking incubator at 120 r/min and 20 �� for 10 d [7]. All the experiments were run in triplicate.
Sterile experimental conditions were achieved by putting each flask containing modified Chashi liquid medium in autoclave at 115 �� for 30 min prior to inoculation, and soil was autoclaved separately. The hyphae were sought out. The soil was washed with deionized water three times and then heavy metal contents were analyzed.
2.9 Analysis of organic acids
Organic acids were analyzed using high performance liquid chromatography (HPLC) (HP 1100 series). Zorbax C18 column (250 mm��4.6 mm) was used with a mobile phase containing 0.01 mol/L KH2PO4- H3PO4, 3% methanol (volume fraction) in bi-deionized water adjusted to pH 2.6 with H3PO4 at a flow rate of 0.5 ��L/min, and with diode array detector (DAD) at 210 nm for the determination of organic acids [19].
2.10 Analytical methods
The statistical analysis was processed by Excel 2003 and SAS system.
3 Results
3.1 Isolation, selection, acclimation and identification of P. Chrysogenum
In order that a resistant indigenous strain was obtained, soil dilution was prepared and then spread on Chashi agar medium. Consequently, seven strains grew on Chashi plates. Thereafter, strains�� colonies were separated and further purified. After five days, seven strains were inoculated on Chashi agar medium which contained 200 mg/L Cd, 400 mg/L Pb and Zn for further acclimation. And then, only five of the seven strains grew, the five strains were transplanted to modified Chashi medium, and the liquid medium pH was determined. Finally, one strain was found, by which the pH value of the liquid medium was degraded. The degradation of pH value meant that many organic acids were produced. These organic acids were then to be used to remove soil heavy metals [20-23].
The strain with the ability of producing acids and resisting heavy metal was selected for identification using the 18S rDNA gene sequence and ITS gene sequence. The 18S rDNA sequence size was 1 069 gene bps, and ITS4 and ITS5 sequences sizes were 559. Its similarity of gene sequence of 18S rDNA and ITS5 showed 100% with P. Chrysogenum, and that of ITS4 showed 99% with P. Chrysogenum. The morphology of strain F1 was shown in Fig. 1, which was very similar with the P. Chrysogenum. Phylogenetic tree obtained from ITS5 sequence comparisons of 559 bps showed the relationship between members of the family Penicillium and the strain F1 stored in our laboratory (Fig. 2). The bootstrap neighbor-joining tree (random number generator seed of 57, trails of 1 000) was constructed with Clustal X version 2.0.

Fig. 1 Micrograph of P. Chrysogenum
Thus, according to the gene sequence, morphology and phylogenetic tree, the strain was identified as Penicillium Chrysogenum.
3.2 Selection of carbon source
Different carbon sources were selected to culture P. Chrysogenum in order to make P. Chrysogenum produce the most organic acids [24]. When glucose and sucrose were used as carbon sources, bioleaching ratios were compared, and the bioleaching efficiency of glucose as carbon source was better than that with sucrose as carbon source (Fig. 3). When the glucose content was 90 g/L, the bioleaching ratios were 1.9% of Pb, 64.8% of Cd, 24.6% of Cu and 41.8% of Zn. 1.3% of Pb, 58.3% of Cd, 15.0% of Cu and 35.6% of Zn were bioleached in 60 g/L glucose as carbon source; 2.4% of Pb, 60.6% of Cd, 10.2% of Cu and 43.2% of Zn in 120 g/L glucose as carbon source.
3.3 Selection of nitrogen source
(NH4)2SO4, NH4NO3, peptone, yeast extract and beef extract were used respectively instead of NaNO3 in modified Chashi liquid medium. The bioleaching ratios of different nitrogen sources were compared. Different metals showed different bioleaching ratios in the same nitrogen source in Fig. 4. 1.6% of Pb, 38.2% of Zn, 85.5% of Cd and 22.8% of Cu were bioleached in (NH4)2SO4 as nitrogen source of liquid medium. 20% of Pb, 52.4% of Zn, 63.0% of Cd and 35.5% of Cu were bioleached in NaNO3 as nitrogen source of liquid medium. 1.8% of Pb, 25.9% of Zn, 40.6% of Cd and 23.1% of Cu were bioleached in NH4NO3 as nitrogen source of liquid medium. 6.1% of Pb, 12.8% of Zn, 31% of Cd and 36.7% of Cu were bioleached in pepton as nitrogen source of liquid medium. The bioleaching ratios of Pb, Zn, Cd and Cu of yeast extract and beef extract as nitrogen source of liquid medium were smaller than those of pepton as nitrogen source of liquid medium. This also showed that the bioleaching ratios of inorganic nitrogen sources namely NaNO3, NH4NO3 and (NH4)2SO4 were larger than those of organic nitrogen sources namely peptone, yeast extract and beef extract in Fig. 4. In the view of inorganic nitrogen sources, the bioleaching ratio of NaNO3 as nitrogen source was larger than that of (NH4)2SO4 and NH4NO3 as nitrogen source.
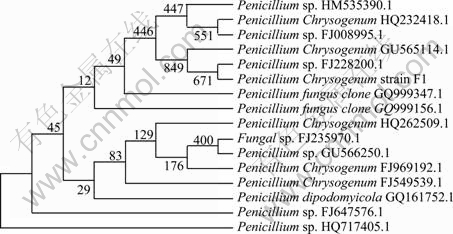
Fig. 2 Phylogenetic tree derived from ITS
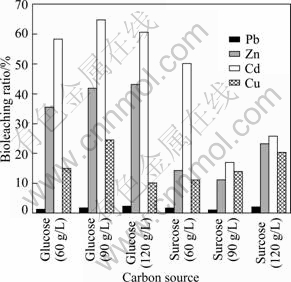
Fig. 3 Bioleaching ratios of different carbon sources
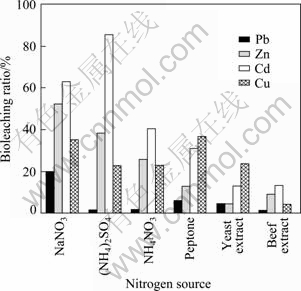
Fig. 4 Bioleaching ratios of different nitrogen sources
3.4 Selection of pH value of medium
Different pH value levels of Chashi liquid medium were used to bioleach the contaminated soil. The pH value level was set at 3, 5, 6.7, 7 and 9. The bioleaching ratios are given in Table 2. The order of bioleaching ratios of Pb was pH 7> pH 3> pH 6.7> pH 9> pH 5; The order of bioleaching ratios of Zn was pH 7>pH 3> pH 5> pH 9>pH 6.7; The order of bioleaching ratios of Cd was pH 7=pH 6.7>pH 3=pH 5>pH 9; The order of bioleaching ratios of Cu was pH 7>pH 3>pH 9>pH 5> pH 6.7. The bioleaching ratios of all the metals at pH 7 were the largest than any other pH value.
3.5 Selection of temperature of bioleaching
Bioleaching ratios of all the metals at 20 �� were larger than those at 30 �� and 40 �� ( Fig. 5 ). The order of bioleaching ratios at different temperatures was 20 �� >30 ��>40 ��. It was because that 20 �� was the most suitable temperature for P. Chrysogenum to grow, and more organic acids were produced and more heavy metals were removed from the soil. P. Chrysogenum didn��t grow at 40 ��, so the ratios of bioleaching at 40 �� were the lowest.
Table 2 Bioleaching ratios at different pH values
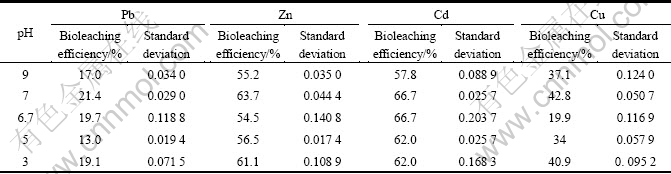
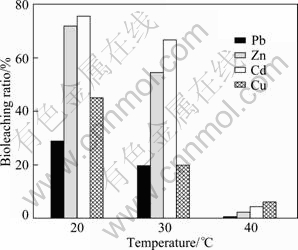
Fig. 5 Bioleaching ratios at different temperature levels
3.6 Bioleaching contaminated soil using P. Chrysogenum
Various metals showed different leaching efficiencies by different ways. Bioleaching ratios were compared in the one-step with those in the two-step (Fig. 6). Zn and Cd showed approximative bioleaching efficiencies in the two methods: Bioleaching ratios of Zn were 70% in the one-step and 71.8% in the two-step; bioleaching ratios of Cd both in the one-step and in the two-step were 74%. Bioleaching ratios of Pb and Cu showed larger values in the two-step than in the one-step, 49.9% of Pb and 61.3% of Cu in the two-step but 36.9% of Pb and 43% of Cu in the one-step process.
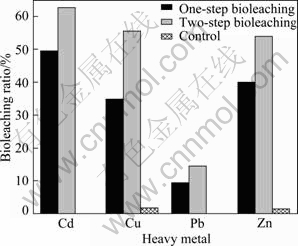
Fig. 6 Bioleaching ratios of P. Chrysogenum by different methods
3.7 Kinds of organic acids
After bioleaching, gluconic acid, pyruvic acid and citric acid were determined. The four organic acids were all observed in the bioleaching solution. Their retention time is shown in Fig. 7.
4 Discussion
Glucose is a more suitable carbon source for P. Chrysogenum to produce more organic acids than any contents of sucrose. Therefore, more organic acids are produced by glucose as carbon source than sucrose [25]. This is because glucose is monose which is directly oxidized into organic acids [26]. Sucrose is disaccharide and oxidized monose firstly, secondly monose is oxidized into organic acids. Thus, not all the sucroses can be oxidized thoroughly. From the cost and bioleaching efficiency, 90 g/L glucose is the most suitable content when P. Chrysogenum is used to bioleach for fifteen days.
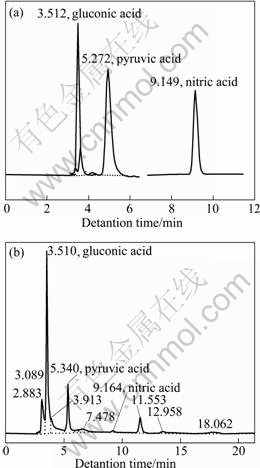
Fig. 7 Kinds of organic acids: (a) Standard; (b) Sample
Inorganic nitrogen sources are better for bioleaching than organic nitrogen. Organic nitrogen is fitter for the growth of P. Chrysogenum than inorganic nitrogen. The production of organic acid is prevented by the rapid growth of P. Chrysogenum. NaNO3 as nitrogen source is better for bioleaching than (NH4)2SO4 and NH4NO3 as nitrogen source. It may be because different forms of nitrogen in ![]() and
and ![]() influence the metabolism of P. Chrysogenum [27], or because the existence of
influence the metabolism of P. Chrysogenum [27], or because the existence of ![]() influences the content of heavy metal ions in filtrate.
influences the content of heavy metal ions in filtrate.
Neutral solution is the most suitable pH level than alkaline solution and acidic solution. It is the most suitable pH value for P. Chrysogenum to grow. Furthermore, there are no precipitates produced in alkaline solution, and no influence on the growth by acid solution. Room temperature is the best temperature for bioleaching than 30 �� and 40 ��, for room temperature is the best temperature for the growth of P. Chrysogenum and for the metabolism of P. Chrysogenum.
Gluconic acid, pyruvic acid and citric acid are produced when glucose is oxided by P. Chrysogenum. Degradation of pH value and solubilization of heavy metal are resulted from the production of gluconic acid, pyruvic acid and citric acid. Other kinds of organic acids would be determined to confirm the pathway of P. Chrysogenum metabolism. Different bioleaching efficiencies are shown by different metals in the same method. Different bioleaching efficiencies are shown by one metal in different methods. This may be because of their chemical forms and their different kinds of salts in the soil. Thus, sequential extraction should be used to study the chemical forms of heavy metal before or after bioleaching.
5 Conclusions
It is indicated that indigenous P. Chrysogenum is effective in removing heavy metal from contaminated soil. Glucose as carbon source, NaNO3 as nitrogen source, pH 7 and room temperature are the best conditions for P. Chrysogenum to bileach.
References
[1] CHAI Li-yuan, HUANG Shun-hong, YANG Zhi-hui, PENG Bin. Cr(VI) remediation by indigenous bacteria in soils contaminated by chromium-containing slag [J]. Journal of Hazardous Materials, 2009, 167: 516-522.
[2] PARK D H, YUN Y S, PARK J M. Use of dead fungal biomass for the detoxi?cation of hexavalent chromium: Screening and kinetics [J]. Process Biochemistry, 2005, 40: 2559-2565.
[3] PARVAZE A W, MOHAMMAD S K, ALMAS Z D. Chromium reduction, plant growth�CPromoting potentials, and metal solubilization by bacillus sp. isolated from alluvial soil [J]. Current Microbiology, 2007, 54: 237-243.
[4] WANG Wei, WANG Jun-qin, YANG Jie, ZHANG Wei. Preliminary study on conditions of isolating and cultivating arsenic-oxidizing strains [J]. Chinese Journal of Endemiology, 2006, 25: 96-98.
[5] KITJA C, ANCHARID A, SOMBOON T. Isolation and characterization of arsenic resistant bacteria from tannery wastes and agricultural soils in Thailand [J]. Annals of Microbiology, 2009, 59: 649-656.
[6] REN Wan-xia, LI Pei-jun, GENG Yong, LI Xiao-jun. Biological leaching of heavy metal from a contaminated soil by Aspergillus niger [J]. Journal of Hazardous Materials, 2009, 167: 164-169.
[7] MULLIGAN C N, KAMAL M, GIBBS B F. Bioleaching of heavy metals from a low-grade mining ore using Aspergillus niger [J]. Journal of Hazardous Materials, 2004, 110: 77-84.
[8] AMIRI F, YAGHMAEI S, MOUSAYI S M. Bioleaching of tungsten-rich spent hydrocracking catalyst using Penicillium simplicissimum [J]. Bioresource Technology, 2010, 102: 1567-1573.
[9] CUI De-jie, ZHANG Yu-long. Current situation of soil contamination by heavy metal and research advances on the remediation techniques [J]. Chinese Journal of Soil Science, 2004, 35: 366-370. (in Chinese)
[10] WU Gang, KANG Hu-biao, ZHANG Xiao-yang, SHAO Hong-bo, CHU Li-ye, RUAN Cheng-jiang. A critical review on the bio-removal of hazardous heavy metal from contaminated soils: Issues, progress, eco-environmental concers and opportunities [J]. Journal of Hazardous Materials, 2010, 174: 1-8.
[11] DOMEN Le?tan, LUO Chun-ling, LI Xiang-dong. The use of chelating agents in the remediation of metal-contaminated soils: A review [J]. Environmental Contamination, 2008, 153: 3-13.
[12] AMIRI F, YAGHMAEI S, MOUSAVI S M. Bioleaching of tungsten-rich spent hydrocracking catalyst using Penicillium simplicissimum [J]. Bioresource Technology, 2010, Z18: 1-4.
[13] Soil quality-Determination of lead, cadmium-KI-MIBK extraction flame atomic adsorption spectrophotometry [S]. GB/T 17140��1997. (in Chinese)
[14] Soil quality-Determination of copper, zinc-KI-MIBK extraction flame atomic adsorption spectrophotometry [S]. GB/T 17138��1997. (in Chinese)
[15] Chinese CRM/RM Information Center. Method of determination of soil pH value [EB/OL]. www.gbw114.org.
[16] XU Guang-hui, ZHEN Hong-yuan. The handbook of analysis methode for soil microorganism [M]. Agricultural Publwashing House, 1986: 57-58. (in Chinese)
[17] HUANG Xiu-li. Microbiology [M]. Beijing: Academic Press, 2009: 96. (in Chinese)
[18] ZHU Heng, QU Feng, ZHU Li-huang. Extracting fungi DNA for molecular biology analysis using CHCl3 [J]. Acta Mycologica Sinica, 1994, 13: 34-40.
[19] CATHERINE N M, MAHTAB K, BERNA F G. Bioleaching of heavy metals from a low-grade mining ore using Aspergillus niger [J]. Journal of Hazardous Materials, 2004, 110: 77-84.
[20] WALID A L, GHANEM K M, EHAB R E H. Citric acid production by a novel Aspergillus niger isolate: I. Mutagenesis and cost reduction studies [J]. Bioresource Technology, 2007, 98: 3464-3469.
[21] WALID A L, KHALED M G, EHAB R E H. Citric acid production by a novel Aspergillus niger isolate: II. Optimization of process parameters through statistical experimental designs [J]. Bioresource Technology, 2007, 98: 3470-3477.
[22] ZANDRA A, EMMA J, von KRONHELM T, ALLARD B, van HEES P. Remediation of metal contaminated soil by organic metabolites from fungi I-production of organic acids [J]. Water Air Soil Contamination, 2010, 205: 215-226.
[23] ZANDRA A, BERT A. Remediation of metal contaminated soil by organic metabolites from Fungi II- metal redistribution [J]. Water Air Soil Contamination, 2010, 207: 5-18.
[24] HEATH E C, HENRY K. Biochemistry of filamentous Fungi. II: The quantitative significance of an ��oxidative pathway�� during the growth of Penicillium chrysogenum [J]. Journal of Biological Chemistry, 1956, 71: 174-180.
[25] HOCKENHULL D J D, MARY H B, WALKER A D, WICKIN G D. Organic acid metabolism of Penicillium Chrysogenum I. Lactate and Acetate [J]. Biochemistry, 1954, 56: 73-82.
[26] DEFIEBRE C W, KNIGHT S G. The oxidation of glucose by Penicillium Chrysogenum [J]. Biochemistry, 1953, 66: 170-172.
[27] AZAM F, IFZAL M. Microbial populations immobilizing NH4+-N and NO3--N differ in their sensitivity to sodium chloride salinity in soil [J]. Soil Biology & Biochemistry, 2006, 38: 2491-2494.
(Edited by YANG Bing)
Foundation item: Project(2009ZX07212-001-01) supported by Major Science and Technology Program for Water Pollution Control and Treatment of China; Project(50925417) supported by the National Natural Science Funds for Distinguished Young Scholar in China; Projects(50830301, 51074191) supported by the National Natural Science Foundation of China
Received date: 2011-07-08; Accepted date: 2011-12-25
Corresponding author: YANG Zhi-hui, Professor, PhD; Tel: +86-731-88830875; E-mail: yangzh@csu.edu.cn

