Collecting performance of vegetable oils in scheelite flotation and differential analysis
��Դ�ڿ������ϴ�ѧѧ��(Ӣ�İ�)2019���4��
�������ߣ��³� ���ĺ� ������ �캣�� ����
����ҳ�룺787 - 795
Key words��scheelite; flotation; vegetable oil; synergistic effect; adsorption
Abstract: The composition of a collector directly affects its collecting performance in mineral flotation. In this study, three vegetable oils were used as the collectors, the flotation performance of scheelite and the differential analysis were studied through flotation experiments, zeta potential, contact angle measurement and Fourier transform infrared spectrum (FTIR) analysis. Flotation results show that the recovery of scheelite increases in the order of oleic acid
Cite this article as: JIA Wen-hao, QIN Wen-qing, CHEN Chen, ZHU Hai-ling, JIAO Fen. Collecting performance of vegetable oils in scheelite flotation and differential analysis [J]. Journal of Central South University, 2019, 26(4): 787�C795. DOI: https://doi.org/10.1007/s11771-019-4048-0.

J. Cent. South Univ. (2019) 26: 787-795
DOI: https://doi.org/10.1007/s11771-019-4048-0
JIA Wen-hao(���ĺ�), QIN Wen-qing(������), CHEN Chen(�³�),ZHU Hai-ling(�캣��), JIAO Fen(����)
School of Mineral Processing and Bioengineering, Central South University, Changsha 410083, China
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2019
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2019
Abstract: The composition of a collector directly affects its collecting performance in mineral flotation. In this study, three vegetable oils were used as the collectors, the flotation performance of scheelite and the differential analysis were studied through flotation experiments, zeta potential, contact angle measurement and Fourier transform infrared spectrum (FTIR) analysis. Flotation results show that the recovery of scheelite increases in the order of oleic acid
Key words: scheelite; flotation; vegetable oil; synergistic effect; adsorption
Cite this article as: JIA Wen-hao, QIN Wen-qing, CHEN Chen, ZHU Hai-ling, JIAO Fen. Collecting performance of vegetable oils in scheelite flotation and differential analysis [J]. Journal of Central South University, 2019, 26(4): 787�C795. DOI: https://doi.org/10.1007/s11771-019-4048-0.
1 Introduction
In flotation of tungsten ore, oleic acid (or sodium oleate) is the most widely used collector [1�C6]. Usually, the other reagents, called as synergists, are mixing used in order to enhance the collecting performance of oleic acid. On the one hand, different fatty acids can be used together. YANG [7] studied flotation performance of scheelite using different kinds of fatty acids and their mixtures, and the results showed that the addition of linoleic acid and dodecylic acid could increase the recovery of scheelite with oleic acid alone. FILIPPOV et al [8] found that due to a better organization of the adsorption layer, the flotation selectivity between scheelite and fluorite can be increased using new reagent formulations which contained oleate, linoleate and palmitic acid with a proper ratio. Nevertheless, the effect of the content of each component on flotation performance of scheelite was not studied, and the mixed collector with the optimal composition was not found. Another typical example is the mixture of oleic acid and oxidized paraffin soap, which have been successfully used in flotation practice of scheelite [9]. On the other hand, other different kinds of surfactants can also be used as the synergists of oleic acid, such as octyl hydroxamic acid [8], lauryl amine [11], polyoxyethylene ether [12�C14], and so on.
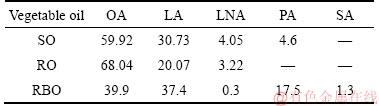
At present, oleic acids used in actual production of scheelite flotation are mainly the industrial oleic acids, which are the mixtures of oleic acid, linoleic acid, linolenic acid, palmitic acid and stearic acid. So they are suggested to exhibit better collecting performance than pure oleic acid due to the synergistic effect [15, 16]. Generally, the industrial oleic acid can be classified into vegetable oil and animal oil depending on the source, and vegetable oil is the most commonly used kind in view of the wide source and low price, including rice bran oil, rapeseed oil, corn oil and soybean oil. A large number of researches have been done on the collecting performance of vegetable oils in flotation of phosphate ore. LUO et al [15, 16] investigated the flotation performance of collophanite using cotton oil, soybean oil and rice bran oil as collector, and the results showed that the ratio of linoleic acid to oleic acid in vegetable oil had great influence on flotation results, and the larger the ratio, the higher the flotation recovery. GUIMARAES et al [17] showed that corn oil, soybean oil and rice bran oil had been successfully used as collectors in apatite flotation in Brazil. However, there is no systematic research on flotation performance of scheelite using different vegetable oils, and the reasons for the difference in flotation performance are not explained in theory. In addition, the compositions of vegetable oils are variable, even though they are derived from the same vegetable, the content of each component is different. Literatures reported that soybean oil contained 20%�C30% oleic acid, 50%�C60% linoleic acid, 0�C10% linolenic acid and palmitic acid, but rice bran oil contained about 40% oleic acid, 40% linoleic acid and 20% palmitic acid [17, 18]. Obviously, the collecting properties of vegetable oils would change along with their compositions. Nevertheless, the effect of the composition on flotation performance of scheelite as well as which component is helpful for scheelite flotation is unknown.
Therefore, in this study, we first investigated the flotation performance of scheelite using three different vegetable oils, then found the beneficial components for scheelite flotation by using the mixtures of oleic acid and other components, and analyzed the adsorption mechanism by ways of zeta potential, contact angle measurement and FTIR analysis.
2 Experimental
2.1 Materials
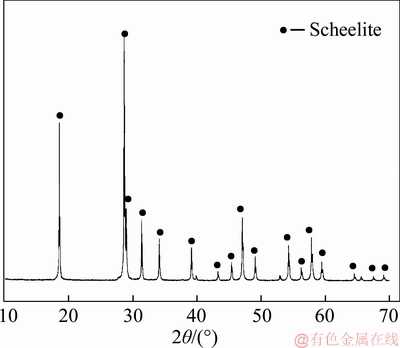
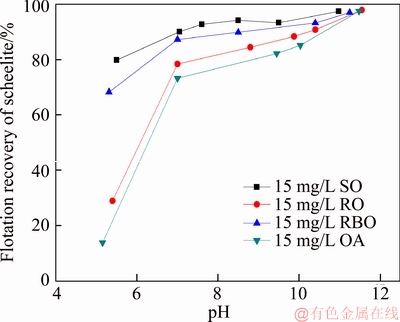
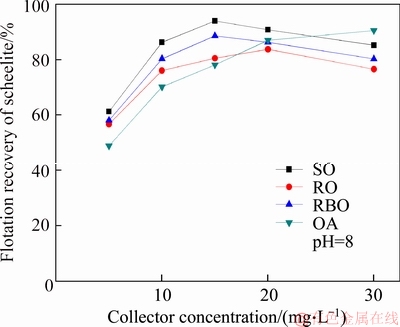
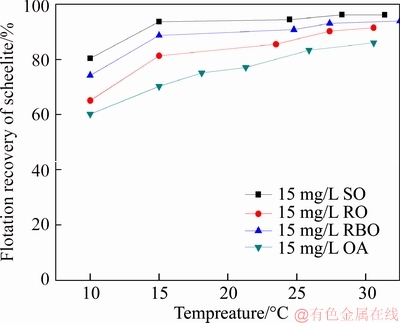
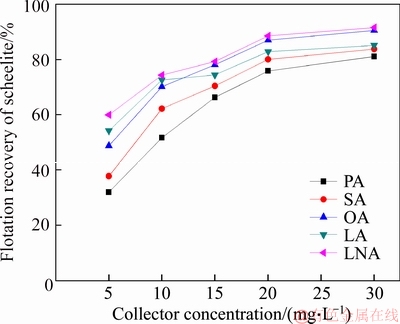
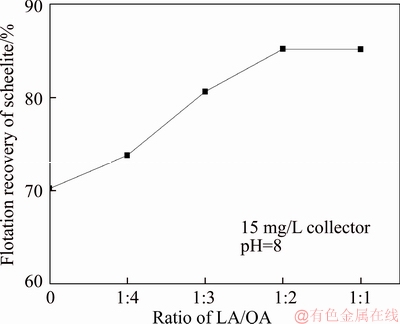
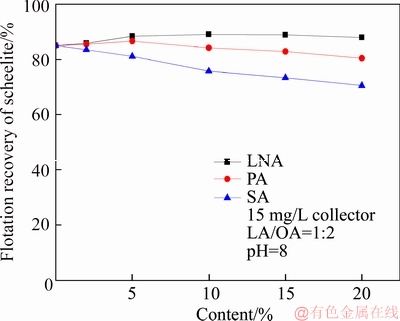
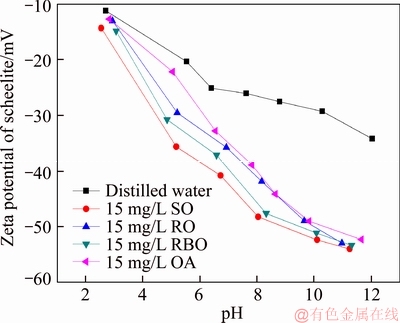
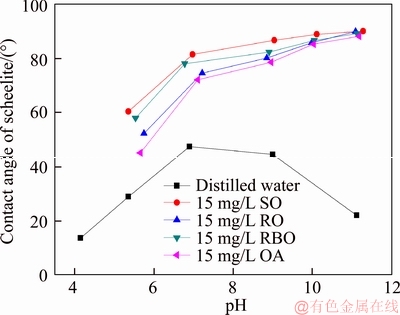
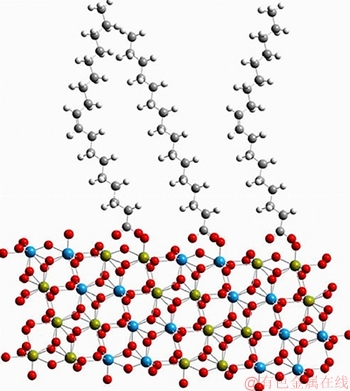
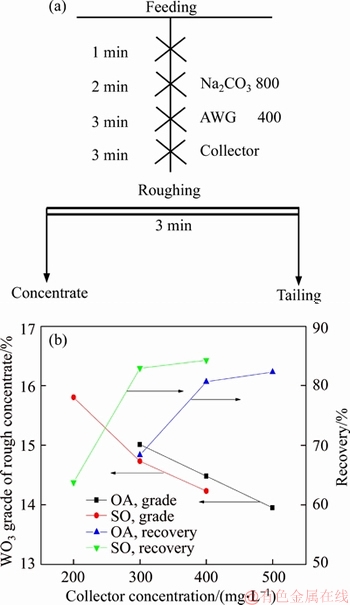
The pure sample of scheelite was obtained from Sichuan Province, China. Results of chemical analysis and XRD detection (Figure 1) prove that the WO3 grade is 77.33% with the scheelite purity of 95.91%, and the gangue mineral is mainly quartz. Through crushing, grinding and screening, the samples of 38 Figure 1 XRD pattern of pure scheelite sample 2.2 Flotation experiments For each test, 2 g scheelite samples and 30 mL distilled water were placed into a flotation cell of 40 mL, and hung from a XFG flotation machine (Jilin Exploration Machinery Factory, China) to carry out flotation tests. The flotation recovery of the floated product was used to characterize the floatability of scheelite. Except the temperature test, all experiments were conducted at normal temperature. Table 1 Fatty acids used in this study 2.3 Zeta potential measurement The mineral suspensions with 0.01% samples were first stirred well, adjusted to a required pH value, fully reacted with collectors, then extracted and injected into the electrode to measure the zeta potentials using a Malvern zeta sizer analyzer (England). The averaged value of three measured values for each test was recorded, and the repeatability was ��0.1 mN/m. 2.4 Contact angle measurement Atomic force microscope results show that the surface roughness of scheelite crystal is below 0.05 ��m [19], which does not affect the wettability of scheelite according to Busscher��s research [20]. The sample was fully conditioned with every collector, washed three times, and dried in a vacuum oven, then the contact angles of scheelite were measured using pendant-drop method on a GBX contact angle meter (France). A distilled water droplet of 3.5 ��L was introduced onto scheelite surface, and an averaged value of three measured values was recorded. When each test was completed, the sample was washed with acetone, polished and then washed with distilled water to obtain a fresh surface for the next measurement. The measurement accuracy is ��2��. 2.5 FTIR analysis The scheelite sample was conditioned with the collector for 10 min at pH 8, filtered, washed three times, and then dried in a vacuum oven at 40 ��C. The FTIR spectra of scheelite were analyzed using a Fourier transform infrared spectrometer (Shimadzu, Japan). 3 Results and discussion 3.1 Flotation performance of scheelite with different vegetable oils Three different vegetable oils of SO, RBO and RO were used as the collector, and the flotation performance of scheelite was investigated. Figure 2 presents the effect of pH on flotation recovery of scheelite using 15 mg/L collectors. With the increase of pH value, flotation recovery of scheelite gradually increases and scheelite exhibits good floatability in the pH range of 7�C11. Compared with the pure OA, the recovery of scheelite with vegetable oil is improved, and the increased value decreases with pH value rising. At around pH 5, the recovery of scheelite is about 80% with SO, while only 15% with pure OA. In addition, the recovery of scheelite increases in the order of RO Figure 2 Effect of pH on flotation recoveries of scheelite with 15 mg/L vegetable oils Figure 3 Effect of collector concentration on flotation recoveries of scheelite at pH 8 Figure 3 shows flotation recoveries of scheelite as a function of collector concentration at about pH 8. The results display two different variation trends of scheelite recovery. For pure OA, the recovery of scheelite gradually increases from about 48% to 90% when the concentration increases from 5 mg/L to 30 mg/L. While for vegetable oils, with the increase of collector concentration, the scheelite recovery increases till the maximum appears and then begins to decrease. The highest recovery of scheelite is about 94% with 15 mg/L SO, 88% with 15 mg/L RBO and 84% with 20 mg/L RO, respectively. Compared with pure OA, a lower concentration of the vegetable oil is required to obtain a similar scheelite recovery, showing the stronger collecting capability of the vegetable oil. Figure 4 presents the effect of temperature on flotation recovery of scheelite with 15 mg/L collector at pH 8. As temperature rises, the recovery of scheelite gradually increases. Compared with pure OA, the recoveries of scheelite are obviously improved in the presence of vegetable oils, especially at low temperatures (<15 ��C). When the temperature is 10 ��C, the recovery of scheelite is 80% for SO, but only 60% for pure OA. Hence, it is suggested that the three vegetable oils have stronger resistance to low temperature than pure OA. Figure 4 Effect of temperature on flotation recoveries of scheelite at pH 8 From the above results, it can be concluded that compared with pure OA, the three vegetable oils are more effective in scheelite flotation, and the recovery of scheelite increases in the order of RO Table 2 Main components of three vegetable oils (volume fraction, %) 3.2 Effect of collector composition on flotation performance of scheelite The chemical structures of different fatty acids affect their collecting performance, and then influence their mixtures. Using the main components of the vegetable oil as the collector, the effect of collector concentration on flotation recoveries of scheelite at pH 8 is shown in Figure 5. It can be seen that the recovery of scheelite increases with increasing the collector concentration, and it basically increases in the order of PA Figure 5 Effect of collector concentration on flotation recoveries of scheelite at pH 8 Figure 6 Effect of LA content on flotation recovery of scheelite at pH 8 The ratio of LA/OA was fixed at 1:2, and the total concentration of the mixed collector was 15 mg/L, the effect of the fraction of LNA, PA and SA on flotation performance of scheelite was investigated at pH 8. As shown in Figure 7, the recovery of scheelite is improved to 88.55% in the presence of 5% LNA, and no obvious increase is seen with a further increase of LNA content. For PA, a low content less than 5% can increase the recovery of scheelite, but a higher content of PA shows the opposite effect. However, the recovery of scheelite gradually decreases with the increase of SA content. Figure 7 Effect of LNA, PA and SA content on flotation recovery of scheelite Based on the above results, it can be indicated that the collecting abilities of different vegetable oils depend on their compositions (the kinds of fatty acids and their contents). In the case of the vegetable oil SO, the LA/OA ratio is close to 2:1, and the contents of LNA and PA are less than 5% which are beneficial for scheelite flotation, thus SO exhibits the best collecting capability. For the vegetable oil RBO, the high content of PA (>5%) and the presence of SA cause the scheelite recovery decrease in comparison with that using SO. Among the three vegetable oils, the lowest recovery of scheelite is observed using RO as the collector, which is attributed to the low content of LA and LNA. Therefore, the results indicate that when OA is used as the major collector, the additions of LNA and PA with appropriate content show synergistic effect in scheelite flotation, but SA deteriorates the scheelite flotation. The results well explain the difference in the flotation performance of scheelite using the three vegetable oils as collectors. At present, the synergistic effect of the mixed collector is explained from the following two aspects: surface property of the mixed surfactant and the adsorption on mineral surface. Researches have shown that the mixed fatty acids have a lower CMC value than single individuals, indicating a higher surface activity [18, 21]. So in this paper, the adsorption of the collectors on scheelite surface is mainly studied. 3.3 Zeta potential of scheelite surface The change in zeta potential of mineral surface before and after adding reagents can be used to analyze the adsorption mechanism of reagents on mineral surface. The effect of pH on zeta potential of scheelite is shown in Figure 8. It can be seen that scheelite is negatively charged when pH value is larger than 2, which agrees with other literatures [22, 23]. After adding collectors, the zeta potentials of scheelite shift negatively, which indicates the chemical adsorption of the collector on scheelite surface. Compared with pure OA, the zeta potentials of scheelite are decreased in the presence of vegetable oils, indicating a stronger adsorption of vegetable oils occur on scheelite surface. Moreover, the decreased value of zeta potential is in the increasing order of RO Figure 8 Effect of pH on zeta potential of scheelite surface 3.4 Contact angles of scheelite The contact angle of scheelite is affected by the preparation method, the roughness, the source and the measurement equipment [24]. GAO et al [25] showed that the static contact angle of scheelite is 73.1�� for {001} cleavage surface, 62.7�� for {112} cleavage surface, and 74.4�� for {112} crystal surface, respectively in distilled water. While FENG et al [26] showed the contact angle of scheelite was around 47 �� at pH 9 in water. Figure 9 shows the effect of pH on contact angle of scheelite using 15 mg/L collectors. The contact angle of scheelite sample used in this study is 45.6�� at pH 8 in distilled water. After reacted with collectors, the contact angle of scheelite increases, which is caused by the adsorption of the collector, thereby the hydrophobicity and floatability of scheelite increase. Furthermore, the increased value increases in the order of OA The three vegetable oils consist of different fatty acids with the same head groups (��COOH) and similar carbon chains, so it is considered that they have the similar dissolved species in aqueous solutions. Under the strong acid condition, the molecular species of RCOOH(aq) and RCOOH(l) dominate, the weak physical adsorption of the collectors occurs on scheelite surface, resulting in the poor floatability, low zeta potential and low contact angle. In neutral and alkaline conditions, the anionic species of RCOO�C, (RCOO�C)22�C and RCOOH��RCOO�C are present. At this moment, the chemical adsorption of fatty acids dominates, greatly increasing the hydrophobicity and flotation recovery of scheelite [26]. On the other hand, different fatty acids have different dissociation constants, so the same kind of dissolved species exists in aqueous solutions corresponding to different pH values. Flotation results show that the advantages of vegetable oils are more significant under the weak acid and neutral conditions, so it is inferred that another main component LA in vegetable oils may exist in the presence of the anionic species at lower pH value, thus increasing the surface properties of the vegetable oils and the adsorption on scheelite surface. Figure 9 Effect of pH on contact angle of scheelite surface 3.5 FTIR analysis Figure 10 shows the infrared spectra of scheelite before and after collector treatment at pH 8. In the addition of the collector OA or SO, several new bands are observed in the spectra of scheelite (Figures 10(b) and (c)). The new bands at 2855 cm-1 and 2926 cm-1 are due to ��CH3 and ��CH2 stretching [28], the band at 1701 cm�C1 is attributed to the stretching vibration of C=O, indicating the chemical adsorption of OA and SO on scheelite surface. Compared with the spectrum (b), the intensities of the new bands are enhanced in the spectrum (c), further confirming the stronger adsorption of SO on scheelite surface. As anionic collectors, electrostatic repulsion exists between two adjacent fatty acid molecules and dispersion force exists between their hydrocarbon chain. And the electrostatic repulsion is negative to adsorption of collectors while the dispersion force is favorable [29]. CAO et al [18] investigated the effect of fatty acid composition on phosphate flotation, and the results showed that the optimal composition of the mixed collector was determined to be 54 wt% HOl, 36 wt% LA and 10 wt% LNA, and the chemical reaction involved the carboxyl groups of fatty acids and Ca species at the apatite surface for each fatty acid in the mixed collector. In addition, FILIPPOV et al [8] suggested the organization of the adsorption layer of OA and PA on {112} surface which is dominant cleavage plane of scheelite, as shown in Figure 11. Unlike unsaturated fatty acid, hydrocarbon chain of PA is linear, and the combination of OA and PA can maximize the chain-chain lateral interactions and it is propitious to the formation of stable adsorption layer. Thus it is indicated that the vegetable oil SO has a better organization of the adsorption layer on the surface of scheelite. Figure 10 FTIR analysis of scheelite surface: Figure 11 Suggestion of organization of adsorption layer on {112} scheelite surface in presence of OA and PA (C=grey, H=white, O=red, Ca=yellow, W=blue) 3.6 Real scheelite ore flotation using vegetable oils The real ore samples were taken from Henan Province, China. Chemical analysis results show that the components are mainly SiO2 and CaO with the content of 49.23% and 24.87%, followed by Fe and Al2O3, but the grade of WO3 is very low, only 0.086%. In actual production of this ore, Petrov method is adopted to realize the beneficiation of scheelite. Preliminary tests have shown that flotation results of the roughing operation directly influence the quality of the final concentrate product, so the condition tests of roughing operation were carried out, and the flowsheet and the results are shown in Figure 12. With the increase of collector dosage, the WO3 grade of the rough concentrate gradually decreases, and the recovery increases. When the concentration is 400 g/t, the rough concentrate containing 1.423% WO3 with the recovery of 84.22% is obtained using SO as the collector, while the WO3 grade of the rough concentrate obtained with pure OA is 1.448% with the recovery of 80.61%. Therefore, it is shown that the vegetable oil SO is a superior collector for scheelite flotation. Figure 12 Flowchart (a) and results of real ore flotation tests (b) 4 Conclusions Vegetable oils, the mixtures of different fatty acids, are considered to be more efficient in the flotation of non-sulfide minerals than single oleic acid. In this paper, three vegetable oils were used as the collectors of scheelite, and the main conclusions are as follows. Compared with pure OA, the three vegetable oils have the advantages in wide pH range, low concentration and high resistance to low temperature, and the recovery of scheelite changes in the increasing order of OA Results of zeta potential, contact angle measurement and FTIR analysis indicate that compared with pure OA, the stronger adsorption of the vegetable oil occurs on scheelite surface due to the co-adsorption of the different compositions. For a raw ore with only 0.086% WO3, the rough concentrate containing 1.423% WO3 with the recovery of 84.22% is obtained using 400 g/t SO as the collector. In conclusion, the vegetable oils show better collecting performance in scheelite flotation due to the synergistic effect between different fatty acids, and the composition of the vegetable oils plays a key role in their collecting abilities. References [1] KARDANOV K H D, BOLYANSKII B M, GALICH V M, GERASIMOV YU V. Flotation of scheelite ore by different brands of oleic acid [J]. Tsvetnye Metally, 1975, 7: 96�C97. [2] LU Yong-xin, LI Chang-gen. Selective flotation of scheelite from calcium minerals with sodium oleate as a collector and phosphates as modifiers. II. Mechanism of the interaction between phosphate modifiers and minerals [J]. International Journal of Mineral Processing, 1983, 10: 219�C235. [3] LI Chang-gen,LU Yong-xin. Selective flotation of scheelite from calcium minerals with sodium oleate as a collector and phosphates as modifiers. I. Selective flotation of scheelite [J]. International Journal of Mineral Processing, 1983, 10(3): 205�C218. [4] FENG Bo, LUO Xian-ping, WANG Jin-qing, WANG Peng-cheng. The flotation separation of scheelite from calcite using acidified sodium silicate as depressant [J]. Minerals Engineering, 2015, 80: 45�C49. [5] MENG Qing-you, FENG Qi-ming, OU Le-ming. Flotation behavior and adsorption mechanism of fine wolframite with octyl hydroxamic acid [J]. Journal of Central South University, 2016, 23: 1339�C1344. [6] MENG Qing-you, FENG Qi-ming, OU Le-ming. Effect of temperature on floatability and adsorption behavior offine wolframite with sodium oleate [J]. Journal of Central South University, 2018, 25: 1582�C1589. [7] YANG Yao-hui. The combined effect of the fatty acids collectors in flotation of scheelite [D]. Changsha: Central South University, 2010. (in Chinese) [8] FILIPPOV L O, FOUCAUD Y, FILIPPOV I V, BADAWI M. New reagent formulations for selective flotation of scheelite from a skarn ore with complex calcium minerals gangue [J]. Minerals Engineering, 2018, 123: 85�C94. [9] QIU Ting-sheng, CHEN Xiang, WEN De-xin, LIAO De-hua. Flotation process and flow tests of a refractory scheetite [J]. Nonferrous Metals Science and Engineering, 2013, 4(5): 48�C53. (in Chinese) [10] GAO Yue-sheng, GAO Zhi-yong, SUN Wei, HU Yue-hua. Selective flotation of scheelite from calcite: A novel reagent scheme [J]. International Journal of Mineral Processing, 2016, 154: 10�C15. [11] WANG Jian-jun, GAO Zhi-yong, GAO Yue-sheng, HU Yue-hua, SUN Wei. Flotation separation of scheelite from calcite using mixed cationic/anionic collectors [J]. Minerals Engineering, 2016, 98: 261�C263. [12] ZHU Hai-ling, QIN Wen-qing, CHEN Chen, LIU Rui-zeng. Interactions between sodium oleate and polyoxyethylene ether and the application in the low-temperature flotation of scheelite at 283 K [J]. Journal of Surfactant and Detergent, 2016, 19(6): 1�C7. [13] ZHU Hai-ling, QIN Wen-qing, CHEN Chen, LIU Rui-zeng. Low-temperature collecting performance of mixed anionic- nonionic surfactants for scheelite flotation and its application [J]. Chinese Journal of Nonferrous Metals, 2016, 26(10): 2188�C2196. (in Chinese) [14] CHEN Chen, ZHU Hai-ling, SUN Wei, HU Yue-hua, QIN Wen-qing. Synergetic effect of the mixed anionic/non-ionic collectors in low temperature flotation of scheelite [J]. Minerals, 2017, 7: 87. [15] LUO Hui-hua, LI Cheng-xiu, TANG Jia-yan, WANG Ya-yun, CHEN Bing-yan. Composition and flotation performance of plant acidic oil [J]. Journal of Wuhan Institute of Technology, 2012, 34(12): 20�C24. (in Chinese) [16] LUO Hui-hua, TANG Jia-yan, LI Cheng-xiu, WANG Ya-yun, CHEN Bing-yan. Flotation performance of plant fatty acid on collophanite at normal temperature [J]. Journal of Wuhan Institute of Technology, 2013, 35(1): 17�C20. (in Chinese) [17] GUIMARAES R C, ARAUJO A C, PERES A E C. Reagents in igneous phosphate ores flotation [J]. Minerals Engineering, 2005, 18(2): 199�C204. [18] CAO Qin-bo, CHENG Jin-hua, WEN Shu-ming, LI Cheng-xiu, BAI Shao-jun, LIU Dan. A mixed collector system for phosphate flotation [J]. Minerals Engineering, 2015, 78: 114�C121. [19] HU Yue-hua, GAO Zhi-yong, SUN Wei. Anisotropic surface energies and adsorption behaviors of scheelite crystal [J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2012, 415: 439�C448. [20] BUSSCHER H J, van PELT A W J, de BOER P, de JONG H P, ARENDS J. The effect of surface roughening of polymers on measured contact angles of liquids [J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 1984, 9: 319�C331. [21] JIANG Qing-mei, DAI Zi-lin. Performance of Mixed Fatty Acids in Separating Scheelite from Fluorite and Calcite [J]. Ming and Metallurgical Engineering, 2012, 32(2): 42�C44, 48. (in Chinese) [22] GAO Zhi-yong, BAI Ding, SUN Wei, CAO Xue-feng, HU Yue-hua. Selective flotation of scheelite from calcite and fluorite using a collector mixture [J]. Minerals Engineering, 2015, 72: 23�C26. [23] YANG Fan, SUN Wei, HU Yue-hua, LONG Si-si. Cationic flotation of scheelite from calcite using quaternary ammonium salts as collector: Adsorption behavior and mechanism [J]. Minerals Engineering, 2015, 81: 18�C28. [24] KOWALCZUK P B, DRZYMALA J. Contact angle of bubble with an immersed-in-water particle of different materials [J]. Eng Chem Res, 2011, 50: 4207�C4211. [25] GAO Zhi-yong, SUN Wei, HU Yue-hua, LIU Xiao-wen. Surface energies and appearances of commonly exposed surfaces of scheelite crystal [J]. Transactions of Nonferrous Metals Society of China, 2013, 23: 2147�C2152. [26] FENG Bo, WANG Peng-cheng, WANG Jin-qing. Functions of sodium oleate in fine grain scheelite flotation [J]. Nonferrous Metals Engineering, 2015, 5(4): 44�C48. [27] WANG Dian-zuo, HU Yue-hua. Flotation solution chemistry [M]. Changsha, China: Hunan Science and Technology Press, 1988. (in Chinese) [28] CHEN Chen, ZHU Hai-ling, QIN Wen-qing, CHAI Li-yuan, JIA Wen-hao. Improving collecting performance of sodium oleate using a polyoxyethylene ether in scheelite flotation [J]. Journal of Central South University, 2018, 25(12): 2971�C 2978. [29] JENSEN J H, GORDON M S. An approximate formula for the intermolecular Pauli repulsion between closed shell molecules [J]. Molecular Physics, 1996, 89: 1313�C1325. (Edited by HE Yun-bin) ���ĵ��� ֲ�������ڰ��ٿ�ѡ�еIJ������ܼ�������� ժҪ�����ռ������ֱ��Ӱ�����ڿ��︡ѡ�еIJ������ܡ����о���������ֲ������Ϊ���ռ���ͨ����ѡ���顢����λ���ԡ��Ӵ��Dz����Լ�������ײ��Կ����˰��ٿ�ĸ�ѡ���ܼ������������ѡ���������������ٿ�Ļ����ʰ�������<������<����<���͵�˳���������������pH=5~8֮�䡣���ٿ�ѡ�����ʵIJ�����Ҫ��������Щ���ռ�����ɲ�ͬ������ġ������ᡢ�������ʮ����(<5%)������������������ٿ�IJ�����������ʮ�������������ٿ�ѡ������λ���ԡ��Ӵ��Dz����������������������������ֲ������Ϊ���ռ�ʱ�����ռ��ڰ��ٿ�����������ǿ�����ں�0.086%WO3��ԭ���ô���Ϊ���ռ������Եõ�WO3 ƷλΪ1.423%��������Ϊ84.22%�İ��ٴ־��� �ؼ��ʣ����ٿ�ѡ��ֲ�������Ч���ã����� Foundation item: Project(2016RS2016) supported by Provincial Science and Technology Leader Program, Hunan, China; Project(2017zzts807) supported by Postgraduate Innovative Research Projects of Central South University, China Received date: 2017-12-13; Accepted date: 2018-08-04 Corresponding author: CHEN Chen, PhD; E-mail: chenchen338@126.com; ORCID: 0000-0003-0370-4049