�ȴ���������Y���Ӷ�VO2����ṹ������¶ȵ�Ӱ��
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2020���4��
�������ߣ����� ����� �Ž� ��� ���ֲ� ������
����ҳ�룺1038 - 1045
�ؼ��ʣ�VO2���壻ˮ�Ⱥϳɣ��ȴ�����Y���ӣ�����¶�
Key words��VO2 powders; hydrothermal synthesis; heat treatment; Y-doping; phase transition temperature
ժ Ҫ����ˮ�Ⱥϳɷ��Ʊ�VO2���壬�о��ȴ���������Y���Ӷ�VO2�ṹ������¶ȵ�Ӱ�졣XRD��SEM��TEM����������ȴ����¶ȶ�VO2ǰ����ľ���ת��Ӱ�������������¶ȣ�������VO2(B)��VO2(M)ת�䡣Y���Ӷ�VO2�ṹ����Ӱ�죬Y3+����ռ��V4+�������λ�ã��γ�YVO4�����壬�Ӷ�����VO2�ľ���������Y���ӵ��µľ����������ֹ�����ľۼ�����ˣ�Y���Ӿ���һ����ϸ���������á�DSC���߱�����Y���ӿ��Խ���VO2(M)������¶ȣ���Y����Ũ��Ϊ9%(Ħ������)ʱ������¶ȿ���δ���ӵ�68.3 ��C���͵�61.3 ��C��
Abstract: The VO2 powders were prepared by hydrothermal synthesis. The effects of heat treatment conditions and Y-doping on the structure and phase transition temperature of VO2 were studied. The XRD, SEM and TEM results show that the heat treatment temperature has a significant effect on the crystal transformation of VO2 precursor. Increasing temperature is conducive to the transformation of precursor VO2(B) to ultrafine VO2(M). The Y-doping affects the structure of VO2. Y3+ can occupy the lattice position of V4+ to form YVO4 solid solution, which can increase the cell parameters of VO2. Due to the lattice deformation caused by Y-doping, the aggregation of particles is prevented, and the grain is refined obviously. DSC curves show that Y-doping can reduce the phase transition temperature of VO2(M). After adding 9 at.% Y, the phase transition temperature can be reduced from 68.3 to 61.3 ��C.
Trans. Nonferrous Met. Soc. China 30(2020) 1038-1045
Bin WANG1,2, Er-hu LI1, Jin-jing DU1, Jun ZHU1,2, Lin-bo LI1,2, Tian-tian ZHOU1
1. School of Metallurgy Engineering, Xi��an University of Architecture and Technology, Xi��an 710055, China;
2. Research Center of Metallurgical Engineering and Technology of Shaanxi Province, Xi��an 710055, China
Received 5 August 2019; accepted 18 February 2020
Abstract: The VO2 powders were prepared by hydrothermal synthesis. The effects of heat treatment conditions and Y-doping on the structure and phase transition temperature of VO2 were studied. The XRD, SEM and TEM results show that the heat treatment temperature has a significant effect on the crystal transformation of VO2 precursor. Increasing temperature is conducive to the transformation of precursor VO2(B) to ultrafine VO2(M). The Y-doping affects the structure of VO2. Y3+ can occupy the lattice position of V4+ to form YVO4 solid solution, which can increase the cell parameters of VO2. Due to the lattice deformation caused by Y-doping, the aggregation of particles is prevented, and the grain is refined obviously. DSC curves show that Y-doping can reduce the phase transition temperature of VO2(M). After adding 9 at.% Y, the phase transition temperature can be reduced from 68.3 to 61.3 ��C.
Key words: VO2 powders; hydrothermal synthesis; heat treatment; Y-doping; phase transition temperature
1 Introduction
VO2 is a typical thermally induced phase change compound. When temperature is up to ~68 ��C, its crystal structure will change, along with its electrical conductivity and optical transmittance [1-3]. Thermally induced phase transition is reversible and occurs in an extremely short time, which makes VO2 have excellent application value in fields of sensors [4,5], smart coating [6], and energy saving windows [7-9]. However, for bulk single-crystal VO2, the preparation cost is very high. Also, before and after the phase transition, there is a volume change of about 0.3%. In addition, after several phase transitions, structural fragmentation will be observed, which is not conducive to its practical application.
The VO2 powders can well withstand the stress caused by the volume expansion before and after phase transition. In addition, ultra-fine scale materials are more conducive to the photoelectric mutation performance, so the research on the synthesis of ultrafine VO2 powders is of great theoretical importance and applied value [10-12].
The phase transition temperature of 68 ��C is still relatively high for VO2 application in the field of energy-saving windows. To solve this problem, a series of researches have been carried out on element doping, including W [13,14], Mo [15,16], Sb [17], Zr [18], F [19], Cr [20], Nb [21], and Al [22]. Although there is no unified conclusion on the influencing mechanism of various elements doping on the structure and properties of VO2, it is generally acknowledged that introducing metal ions with higher valence state or larger radius into VO2 can reduce the phase transition temperature.
Rare earth ions are often used as dopants of photoelectric materials and photocatalytic materials, which play a significant role in improving the photoelectric properties. However, there are few reports on the preparation of VO2 using rare earth doping. It is reported that Eu-doping decreases the semiconductor-to-metal transition (SMT) temperature of monoclinic VO2 polycrystalline thin films from 68 to 47.5 ��C due to substitution of trivalent Eu3+ ions in VO2 lattice [23]. Similar phenomenon in SMT temperature of VO2 thin films with Gd3+-doping is also observed [24]. Considering the similarity of properties among rare earth elements, it can be inferred that Y3+ is a promising dopant. Based on this motivation, we prepared the Y3+-doped VO2 powders. The preparation of ultrafine powder by hydrothermal synthesis has the advantages of mild reaction conditions, easy control of chemical valence state and narrow particle size distribution [25]. Therefore, the ultrafine VO2 powders were prepared by the hydrothermal synthesis method. The effects of heat treatment conditions and rare earth element Y on the structure and phase transition temperature of VO2 powders were also investigated.
2 Experimental
0.01 mol oxalic acid (C2H2O4��2H2O, analy- tically pure) and 0.005 mol vanadium penoxide (V2O5, analytically pure) powders were mixed with 80 mL deionized water. The mixture was continuously stirred at 50 ��C until the suspension turned into dark blue. Then, 10 mL urea precipitant (H2NCONH2, analytically pure, 0.15 mol/L) and a certain proportion of Y2O3 dopant (analytically pure) were added. After full stirring, the mixed suspension was transferred into a closed poly- tetrafluoroethylene reactor with heat preservation at 190 ��C for 72 h. After the reaction was finished, the product was filtered and washed thoroughly to obtain precursor VO2(B). The precursor VO2(B) was placed in the tube furnace with heat treatment for a certain time under nitrogen atmosphere and then cooled down to ambient temperature in the furnace. The product was dispersed with ultrasonic treatment to obtain ultrafine VO2(M) powders.
X-ray diffractometer (D8 ADVANCE A25) was employed to analyze the phase composition. TEM and HRTEM images of the powders were obtained by using high-resolution transmission electron microscope (JEM-2100F STEM/EDS). Scanning electron microscope (SEM) (JSM-6390A) was applied to observing the microstructure. Laser particle size analyzer (Mastersizer 2000) was used to detect the particle size distribution of the powder particles. Differential scanning calorimetry (STA449F3) test technology was used to analyze the phase transition temperature of the ultrafine VO2(M) powders.
3 Results and discussion
3.1 Effect of heat treatment conditions on phase and structure of VO2
Figure 1 shows the XRD patterns of VO2 powders prepared at different heat treatment temperatures. It can be seen that the heat treatment temperature has a certain influence on the phase of VO2. The product is mainly monoclinic VO2(B) after a heat treatment at 400 ��C, which indicates that the precursor crystal form is not sufficiently transformed to the VO2(M) phase at this temperature.
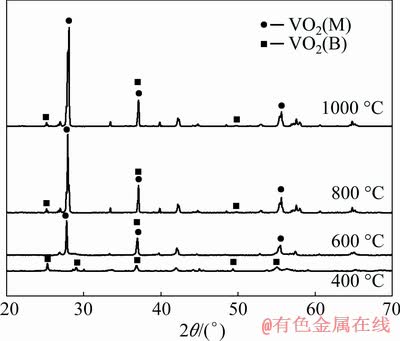
Fig. 1 XRD pattern of VO2 powders at different heat treatment temperatures
When the temperature is up to 600 ��C, VO2(M) diffraction peak can be detected, but with low crystallinity. When the temperature further increases to 800 and 1000 ��C, the diffraction peak strength is significantly enhanced with 2�� of 27.76��, 37.09�� and 55.63��, and the corresponding product is VO2(M). This indicates that appropriate enhancement in the heat treatment temperature can strengthen the crystal transformation from VO2(B) to VO2(M).
Figure 2 shows SEM images of VO2 powders prepared under different heat treatment conditions.

Fig. 2 SEM images of VO2 powders synthesized under different heat treatment conditions
It can be seen that the sample without heat treatment presents a typical VO2(B) nanosheet structure (Fig. 2(a)). After a heat treatment under 400 ��C for 5 h, the sample still shows obvious VO2(B) structure with irregular layered morphology (Fig. 2(b)). It is interesting to note that the sample presents granular structure but a few laminates exist in the matrix with a higher heat treatment temperature of 600 ��C for 5 h (Fig. 2(c)). When the heat treatment time is extended to 7 h at 600 ��C, the particle dispersion can be observed obviously (Fig. 2(d)). The possible reason for the morphology changes is that longer time at higher temperature of 600 ��C can promote the crystalline transformation of VO2. The higher temperature provides sufficient energy for the grain growth not only in a single direction, but in both two-dimensional and three-dimensional directions. The recrystallization process breaks original equilibrium and improves particle dispersion. When the temperature rises up to 800 ��C, the crystal structure is more fully transformed, and spherical grains can be observed (Fig. 2(e)). However, this temperature state cannot last very long time. When the heat treatment time is extended to 7 h at 800 ��C, obvious adhesion occurs among the particles, as shown in Fig. 2(f). For the heat treatment at 1000 ��C for 5 h, the grain transforms into spherical shape with a grain size of ~10 ��m (Fig. 2(g)). When holding time is extended to 7 h, the phenomenon of particle adhesion is also observed, along with the disappearance of partial grain boundaries (Fig. 2(h)).
3.2 Effect of Y-doping on structure and phase transformation of VO2
Y-doped VO2(M) powders were prepared with Y2O3 as dopant. XRD results of different samples are shown in Fig. 3. The diffraction peaks in Fig. 3 match the standard card JCPDS No. 17-0341 of YVO4 and the standard card JCPDS No. 43-1051 of VO2(M), indicating that Y3+ in the sample does not exist in the form of Y2O3, but reacts with VO2 to form the YVO4 phase. YVO4 belongs to the tetragonal crystal system, and the space group is I41/amd. Cell parameters a, b and c are 7.118, 7.118 and 6.289 nm, respectively. This is mainly because, after Y2O3 doping, Y3+ occupies the lattice position of V4+ in VO2, forming a substitutional solid solution. In addition, it is known from the results in Fig. 1 that, the characteristic peak of VO2(M) (011) crystal plane is at 2�� of ~27.7��. After doping 3, 6 and 9 at.% Y in the samples, the angular positions (2��) are shifted to 27.84��, 27.86�� and 27.92��, respectively. These small shifts to higher diffraction angles indicate the decrease in the crystal lattice spacing according to Bragg��s equation (d=��/(2sin ��), d is spacing, �� is wavelength of X-ray, and �� is diffraction angle). The diffraction peak positions imply that a certain doping level of Y can cause slight lattice distortion in VO2.
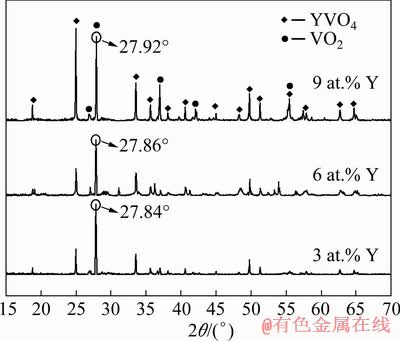
Fig. 3 XRD patters of VO2(M) powders doped with different contents of Y3+
In order to further analyze the lattice structure of the samples, TEM test was performed on the samples with 6 at.% Y doping, and the results are shown in Fig. 4. The clear lattice planes can be seen, indicating a good crystallinity of the obtained powders. The interplanar distance of (011) plane is 0.319 nm, which is consistent with the HRTEM image of VO2. This means that although there is a certain amount of YVO4, the sample is still mainly in the form of VO2 phase.

Fig. 4 TEM (a) and HRTEM (b) images for 6 at.% Y-doped VO2 powders

Fig. 5 SEM images of VO2 powders doped with different contents of Y
The microstructures of samples doped with different contents of Y are shown in Fig. 5. It can be seen that Y-doping plays a significant role in grain refinement. When the un-doped samples are treated at 800 ��C for 5 h, the particle size is 2-3 ��m, and that is less than 2 ��m after Y-doping. The particle size decreases obviously with increasing Y dosage. After adding 9 at.% Y, the particle size of about 500 nm can be obtained. This is probably because of the lattice deformation caused by Y-doping during the recrystallization process with heat treatment. The lattice deformation results in barrier accumulation among grain boundaries, preventing aggregation of particles. It slows down the grain growth, and finally the morphology with smaller grain size can be obtained. Interestingly, increasing Y dosage up to 12 at.%, the particle size of VO2 no longer decreases significantly. It is mainly due to the appearance of excess Y3+, which results in failure of some Y3+ entering into VO2 lattice effectively.
The EDS results of VO2 with different Y-doping levels are shown in Table 1, indicating that the measured value is coincident with the experimental design value basically, except the 12 at.% adding level. For this level, partial Y cannot enter the main structure of VO2, which results in lower measured value.
Figure 6 shows particle size distribution diagram of Y-doped VO2 powder samples. It can be seen that the particle size of each sample is substantially normally distributed. After adding 3, 6, 9 and 12 at.% Y, the average particle sizes of VO2 powder samples drop from 886.1 to 683.8, 469.4, 277.6 and 321.3 nm, respectively. These particle size distribution data are similar to the SEM results of Fig. 5. This indicates that Y-doping has an obvious grain refinement effect on VO2 powder. 9 at.% Y has the most significant effect of grain refinement on VO2 powder samples, and smaller average particle size along with a narrow size- distributed region can be observed.
Table 1 EDS results of Y-doped VO2 powders
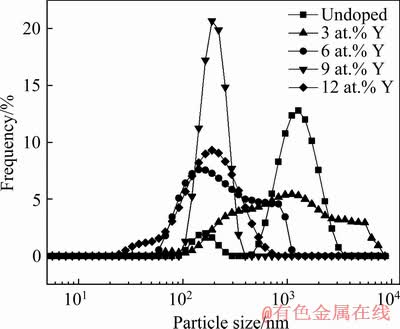
Fig. 6 Particle size distribution of Y-doped VO2 powders
Differential scanning calorimetry (DSC) was applied to investigating the contents of Y on the phase transition temperature of VO2(M) samples, and the results are shown in Fig. 7.
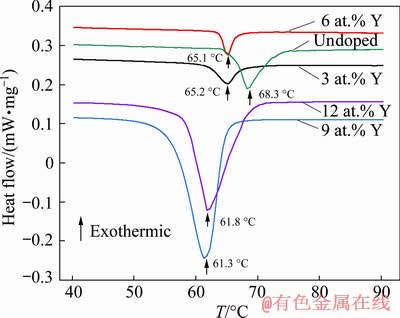
Fig. 7 DSC curves of VO2 powders doped with different contents of Y
It can be seen from Fig. 7 that obvious endothermic peaks corresponding to the phase transitions for all the tested samples can be detected during environmental heating process.
Within the doping range of 9 at.%, the transformation temperature decreases with increasing Y-doping amount. When adding 3 at.% Y, the phase transition temperature of VO2 reduces from 68.3 to 65.2 ��C. Further increasing Y content up to 9 at.%, the phase transition temperature drops down to 61.3��C. The main reason is that the Y-doping changes the energy level structure of VO2. The introduction of impurity elements and the difference in radii of Y and V ions destroy the stable phase structure of V4+-V4+. As the amount of Y increases, lattice deformation increases, thereby reducing the phase transition temperature of VO2. When Y-doping level is up to 12 at.%, the phase transition temperature of VO2 does not change significantly. And the value of phase transition temperature is 61.8 ��C, which is close to that of 9 at.% Y-doped VO2. This is mainly due to the excessive Y addition, which leads to partial Y failure to achieve effective doping. In addition, the heat absorption peaks of each DSC curve are integrated, and the phase change enthalpy values of the un-doped samples, the samples doped with 3, 6, 9 and 12 at.% Y were 13.5, 6.9, 5.6, 42.3 and 36.3 J/g, respectively. Therefore, an appropriate increase in the doping amount of Y is beneficial to improving the phase change performance of VO2.
4 Conclusions
(1) Appropriate enhancement in the heat treatment temperature is conducive to the crystal transformation process of VO2(B)��VO2(M). The crystal transformation of VO2 heat-treated at 800 ��C for 5 h under nitrogen atmosphere is more complete, and the grains are spherical.
(2) For VO2(M) powders doped with Y2O3, Y3+ will occupy the lattice position of V4+ to form the substitutional solid solution of YVO4 and increase the cell parameters of VO2.
(3) The lattice deformation of VO2 caused by Y-doping prevents the particles from accumulating, thus refining the grains.
(4) Due to the difference in ionic radius, Y-doping destroys the stable phase structure of V4+-V4+, thereby reducing the phase transition temperature. When the Y-doping content is 9 at.%, the phase transition temperature can be reduced from 68.3 to 61.3 ��C.
References
[1] ZHANG S X, CHOU J Y, LAUHON L J. Direct correlation of structural domain formation with the metal insulator transition in a VO2 nanobeam [J]. Nano Letters, 2009, 9(12): 4527-4532.
[2] LEE S, IVANOV I N, KEUM J K, LEE H N. Epitaxial stabilization and phase instability of VO2 polymorphs [J]. Scientific Reports, 2016, 6: 19621-19628.
[3] WANG S F, LIU M S, KONG L B, LONG Y, JIANG X C, YU A B. Recent progress in VO2 smart coatings: Strategies to improve the thermochromic properties [J]. Progress in Materials Science, 2016, 81: 1-54.
[4] MICOCCI G, SERRA A, TEPORE A, CAPONE S, RELLA R, SICILIANO P. Properties of vanadium oxide thin films for ethanol sensor [J]. Journal of Vacuum Science & Technology, 1997, 15(1): 34-38.
[5] AZHARUDEEN A M, KARTHIGA R, RAJARAJAN M, SUGANTHI A. Enhancement of electrochemical sensor for the determination of glucose based on mesoporous VO2/PVA nanocomposites [J]. Surfaces and Interfaces, 2019, 16: 164-173.
[6] CHANG T C, CAO X, DEDON L R, LONG S W, HUANG A B, SHAO Z W, LI N, LUO H J, JIN P. Optical design and stability study for ultrahigh-performance and long-lived vanadium dioxide-based thermochromic coatings [J]. Nano Energy, 2018, 44: 256-264.
[7] LIANG Z H, ZHAO L, MENG W F, ZHONG C, WEI S B, DONG B H, XU Z X, WAN L, WANG S M. Tungsten-doped vanadium dioxide thin films as smart windows with self-cleaning and energy-saving functions [J]. Journal of Alloys and Compounds, 2017, 694: 124-131.
[8] LIU C, BALIN I, MAGDASSI S, ABDULHALIM I, LONG Y. Vanadium dioxide nanogrid films for high transparency smart architectural window applications [J]. Optics Express, 2015, 23(3): 124-32.
[9] HUANG Z L, CHEN C H, LV C H, CHEN S H. Tungsten-doped vanadium dioxide thin films on borosilicate glass for smart window application [J]. Journal of Alloys and Compounds, 2013, 564: 158-161.
[10] CAO J, ERTEKIN E, SRINIVASAN V, FAN W, HUANG S, ZHENG H, YIM J W L, KHANAL D R, OGLETREE D F, GROSSMAN J C, WU J. Strain engineering and one-dimensional organization of metal-insulator domains in single-crystal vanadium dioxide beams [J].��Nature Nanotechnology, 2009, 4(11): 732-737.
[11] XIAO X D, ZHANG H, CHAI G Q, SUN Y M, YANG T, CHENG H L, CHEN L H, MIAO L, XU G. A cost-effective process to prepare VO2 (M) powder and films with superior thermochromic properties [J]. Materials Research Bulletin, 2014, 51: 6-12.
[12] LI S Y, NIKLASSON G A, GRANQVIST C G. Nano- thermochromics: Calculations for VO2 nanoparticles in dielectric hosts show much improved luminous transmittance and solar energy transmittance modulation [J]. Journal of Applied Physics, 2010, 108(6): 61-64.
[13] CAO C X, GAO Y F, LUO H J. Pure single-crystal rutile vanadium dioxide powders: Synthesis, mechanism and phase-transformation property [J]. The Journal of Physical Chemistry C, 2011, 112(48): 18810-18814.
[14] LI J, LIU C Y, MAO L J. The character of W-doped one-dimensional VO2(M) [J]. Journal of Solid State Chemistry, 2009, 182(10): 2835-2839.
[15] ZHANG Y F, ZHANG J C, ZHANG X Z, HUANG C, ZHONG Y L, DENG Y. The additives W, Mo, Sn and Fe for promoting the formation of VO2 (M) and its optical switching properties [J]. Materials Letters, 2013, 92: 61-64.
[16] MAI L Q, CHEN W, XU Q, PENG J F, ZHU Q Y. Mo-doped vanadium oxide nanotubes: Microstructure and electrochemistry [J]. Chemical Physics Letters, 2003, 382(1): 307-312.
[17] GAO Y F, CAO C X, DAI L, LUO H J, KANEHIRA M, DING Y, WANG Z L. Phase and shape controlled VO2 nano-structures by antimony doping [J]. Energy & Environmental Science, 2012, 5(9): 8708-8713.
[18] SHEN N, CHEN S, CHEN Z, LIU X L, CAO C X, DONG B R, LUO H J, LIU J J, GAO Y F. The synthesis and performance of Zr-doped and W-Zr-codoped VO2 nanoparticles and derived flexible foils [J]. Journal of Materials Chemistry A, 2014, 2(36): 15087-15093.
[19] ZHANG Y F, ZHANG J C, ZHANG X Z, DENG Y, ZHONG Y L, HUANG C, LIU X, LIU X H, MO S B. Influence of different additives on the synthesis of VO2 polymorphs [J]. Ceramics International, 2013, 39(7): 8363-8376.
[20] JI C H, WU Z M, WU X F, WANG J, LIU X C, GOU J, ZHOU H X, YAO W, JIANG Y D. Terahertz transmittance and metal- insulator phase transition properties of M2 phase VO2 films induced by Cr doping [J]. Applied Surface Science, 2018, 455: 622-628.
[21] WANG Z L, ZHANG R, CHEN X H, FU Q S, LI C L, YUAN S L, ZHAO X J, TAO H Z. Nb doping effect in VO2 studied by investigations of magnetic behavior [J]. Ceramics International, 2018, 44(7): 8623-8627.
[22] WAN J Y, REN Q H, WU N N, GAO Y F. Density functional theory study of M-doped (M=B, C, N, Mg, Al) VO2 nanoparticles for thermochromic energy-saving foils [J]. Journal of Alloys and Compounds, 2016, 662: 621-627.
[23] CAO X, WANG N, MAGDASSI S, MANDLER D, LONG Y. Europium doped vanadium dioxide material: Reduced phase transition temperature, enhanced luminous transmittance and solar modulation [J]. Science of Advanced Materials, 2014, 6(3): 58-561.
[24] GU D E, ZHOU X, SUN Z H, JIANG Y D. Influence of gadolinium-doping on the microstructures and phase transition characteristics of VO2 thin films [J]. Journal of Alloys and Compounds, 2017, 705: 64-69.
[25] ZHAO W H, WEI Z Q, WU X J, ZHANG X D, ZHANG L, WANG X. Microstructure and photocatalytic activity of Ni-doped ZnS nanorods prepared by hydrothermal method [J]. Transactions of Nonferrous Metals Society of China, 2019, 29(1): 157-164.
�� ��1,2�������1���Ž�1���� ��1, 2�����ֲ�1, 2��������1
1. ���������Ƽ���ѧ ұ��ѧԺ������ 710055��
2. ����ʡұ�̼����о����ģ����� 710055
ժ Ҫ����ˮ�Ⱥϳɷ��Ʊ�VO2���壬�о��ȴ���������Y���Ӷ�VO2�ṹ������¶ȵ�Ӱ�졣XRD��SEM��TEM����������ȴ����¶ȶ�VO2ǰ����ľ���ת��Ӱ�������������¶ȣ�������VO2(B)��VO2(M)ת�䡣Y���Ӷ�VO2�ṹ����Ӱ�죬Y3+����ռ��V4+�������λ�ã��γ�YVO4�����壬�Ӷ�����VO2�ľ���������Y���ӵ��µľ����������ֹ�����ľۼ�����ˣ�Y���Ӿ���һ����ϸ���������á�DSC���߱�����Y���ӿ��Խ���VO2(M)������¶ȣ���Y����Ũ��Ϊ9%(Ħ������)ʱ������¶ȿ���δ���ӵ�68.3 ��C���͵�61.3 ��C��
�ؼ��ʣ�VO2���壻ˮ�Ⱥϳɣ��ȴ�����Y���ӣ�����¶�
(Edited by Wei-ping CHEN)
Foundation item: Projects (51404183, 51504177) supported by the National Natural Science Foundation of China
Corresponding author: Jin-jing DU; Tel: +86-17792350559; E-mail: dujinzi@xauat.edu.cn
DOI: 10.1016/S1003-6326(20)65275-X

