��Һ��ɺ����������������ܶƲ����Ժ���Ӳ�ȵ�Ӱ��
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2018���10��
�������ߣ�S. MAHDAVI S. R. ALLAHKARAM
����ҳ�룺2017 - 2027
�ؼ��ʣ��ܶƲ㣻�����������Ǿ���ʮ����������ƣ�����������Ч�ʣ���Ӳ��
Key words��cobalt coating; pulse electrodeposition; saccharin; sodium dodecyl sulfate; characterization; current efficiency; microhardness
ժ Ҫ�������������������������ܻ����Һ���Ʊ������ܶƲ㣬�о���Һ��ɺ͵���������Ե���Ч���Լ��Ʋ���ò���ṹ��Ӳ�ȵ�Ӱ�죬ȷ����ѳ����������о����֣������Һ��������Ũ�Ⱥ�ʮ�����������(SDS)�����ԶƲ���Ӳ�ȵ�Ӱ��ɺ��Բ��ƣ����Ե��������Ч�ʵ�Ӱ���������ڵ������Һ�м����Ǿ�����ʹ�����ܶѽṹ(hcp)�ܱ�Ĥ�ľ����ߴ��С����Ӳ����ߣ����Ե���Ч��������Ӱ�졣�ں���SDS���Ǿ��Ķ�Һ�л�õĶƲ������⻬��ȱ�ݸ��١��������������Ч�ʺ���Ӳ�Ⱦ����ֵ�����ܶȺ�ռ�ձȵı仯���仯�����ŵ����ܶȺ�ռ�ձȵ�������⻬�ĶƲ��Ϊ��״�������ߴ������ȡ��Ҳ��֮�ı䡣
Abstract: Nanocrystalline cobalt coatings were produced from cobalt sulfate based electrolytes by using pulse current electrodeposition technique. The effects of bath composition and electrodeposition condition on current efficiency, morphology, structure and hardness of the coatings were investigated and the optimum deposition condition was determined. It was found that increment of cobalt sulfate concentration and sodium dodecyl sulfate (SDS) concentration in the bath had a negligible effect on microhardness of the coatings, while they were effective on electrodeposition current efficiency. Adding saccharin to electrodeposition bath decreased crystallite size of hexagonal close-packed (hcp) cobalt films and increased their microhardness without significant effect on current efficiency. Smoother and less defective coatings were also obtained from baths containing SDS and saccharin. The results revealed that both the current efficiency and microhardness were changed by variation of peak current density and duty cycle. Besides change of smooth morphology of the coatings to needle-shaped one, crystallite sizes and preferred orientation also varied with increasing the current density and duty cycle.
Trans. Nonferrous Met. Soc. China 28(2018) 2017-2027
S. MAHDAVI1, S. R. ALLAHKARAM2
1. Research Center for Advanced Materials, Faculty of Materials Engineering, Sahand University of Technology, 5133511996, Tabriz, Iran;
2. School of Metallurgy and Materials Engineering, College of Engineering, University of Tehran, Tehran, Iran
Received 7 October 2017; accepted 4 April 2018
Abstract: Nanocrystalline cobalt coatings were produced from cobalt sulfate based electrolytes by using pulse current electrodeposition technique. The effects of bath composition and electrodeposition condition on current efficiency, morphology, structure and hardness of the coatings were investigated and the optimum deposition condition was determined. It was found that increment of cobalt sulfate concentration and sodium dodecyl sulfate (SDS) concentration in the bath had a negligible effect on microhardness of the coatings, while they were effective on electrodeposition current efficiency. Adding saccharin to electrodeposition bath decreased crystallite size of hexagonal close-packed (hcp) cobalt films and increased their microhardness without significant effect on current efficiency. Smoother and less defective coatings were also obtained from baths containing SDS and saccharin. The results revealed that both the current efficiency and microhardness were changed by variation of peak current density and duty cycle. Besides change of smooth morphology of the coatings to needle-shaped one, crystallite sizes and preferred orientation also varied with increasing the current density and duty cycle.
Key words: cobalt coating; pulse electrodeposition; saccharin; sodium dodecyl sulfate; characterization; current efficiency; microhardness
1 Introduction
Hard chromium electrodeposits have been extensively used in engineering applications due to their resistance to wear and corrosion. These coatings are generally electrodeposited from baths containing hexavalent chromium, which produce large volumes of toxic waste and cause health and environmental problems. Low plating efficiency, high hydrogen reduction rate and non-uniformity of thickness of the coatings are other drawbacks of chromium electrodeposition. Therefore, Cr(VI) plating should be replaced by alternative processes and coatings [1-5]. Nickel and cobalt electrodeposits are two suitable candidates that can be considered as possible substitutes for hard chromium films [1-4], among which cobalt exhibits higher hardness and better tribological behavior [2,6].
Application of cobalt is not limited to hard chromium replacement, and it is also used in heterogenic catalysts, chemical sensors, solid oxide fuel cells, energy storage systems, magnetic devices, etc., due to its superior properties [7-12]. Mechanical, tribological, corrosion and magnetic properties of electrodeposits are strongly affected by microstructure and morphology of the coatings, which themselves are a function of bath composition and deposition conditions [2,7-9]. Direct current is widely used for depositing metal coatings due to its simplicity [2,13]. However, application of the alternative pulse electrodeposition technique has increased recently due to its ability in producing nanocrystalline deposits with adjusting the pulse parameters [2,14]. SU et al [2] have studied the effect of deposition technique on characteristics and properties of cobalt coatings. They showed that the nanocrystalline cobalt films deposited by pulse electrodeposition had smoother surface, higher hardness along with better wear and corrosion resistance than the coatings produced by direct currents. According to WANG et al [6], polycrystalline cobalt coatings deposited by using direct current had lower hardness and wear resistance than the nanocrystalline ones obtained with pulse electro- deposition. They also found that crystallite size was decreased by raising the pulse peak current density.
In spite of some research on pulse electrodeposition of cobalt coatings, there are limited studies about the effects of bath composition and pulse electrodeposition parameters on characteristics of cobalt coatings. In the present work, pulse electrodeposition technique was used to produce cobalt coatings from cobalt sulfate based baths. As additives in the electrodeposition bath have a great influence on quality of cobalt films [8,11], different baths containing various amounts of sodium dodecyl sulfate and saccharin were prepared. The aim of this work is to comprehensively investigate the effect of bath composition (i.e. cobalt sulfate, SDS and saccharin concentration) and deposition condition (i.e. current density and duty cycle) on current efficiency, morphology, microstructure and hardness of cobalt coatings.
2 Experimental
2.1 Preparation of substrate
316L stainless steel plates having the nominal chemical composition of Fe, 16.80 Cr, 10.05 Ni, 2.09 Mo, 1.32 Mn, 0.35 Si, 0.26 Cu, 0.11 V, 0.10 Co and 0.028 C (mass fraction, %) and dimensions of 30 mm �� 30 mm �� 2 mm were used as the substrate. The substrates were abraded with different grades of emery papers from 280 to 2000 grit and ultrasonically cleaned in acetone at room temperature for 5 min. Degreasing was done in an alkaline solution containing NaOH, Na2CO3 and Na3PO4��12H2O at 70 ��C for 15 min. For removal of surface oxide layer and preventing it from forming again, the substrates were cathodically treated in a solution containing hydrochloric acid and cobalt chloride at room temperature for 2 min, according to ASTM B254. The samples were transferred to the electrodeposition bath immediately after activation.
2.2 Bath composition and electrodeposition condition
Cobalt sulfate based electrolytes in a double electrode cell were used for electroplating experiments. Different bath compositions and electrodeposition conditions have been presented in Table 1. Analytical grade chemicals and distilled water were used for preparation of the electrolytes. The pH was adjusted by adding 1 mol/L NaOH or 1 mol/L H2SO4 solution to the bath. Pure cobalt plate was used as the anode. The exposed surface area of the anode and its distance from the 316L cathode were 9 cm2 and 4 cm, respectively.
2.3 Characterization
Morphologies of the electrodeposited coatings were investigated by a CAMSCAN MV2300 scanning electron microscope. Philips X��Pert Pro X-ray diffracto- meter with Cu K�� (��=1.542  ), step size of 0.02�� and step duration of 0.4 s was used to analyze structure of the as-deposited coatings. The Williamson-Hall equation was employed to estimate crystallite size of the deposits. Preferred orientation was also studied by using the relative texture coefficient (RTC, T(hkl)), which is given by the following expression [15,16]:
), step size of 0.02�� and step duration of 0.4 s was used to analyze structure of the as-deposited coatings. The Williamson-Hall equation was employed to estimate crystallite size of the deposits. Preferred orientation was also studied by using the relative texture coefficient (RTC, T(hkl)), which is given by the following expression [15,16]:
 (1)
(1)
where I(hkl) and  are the diffraction intensities of (hkl) lines in the XRD patterns of the deposits and randomly oriented cobalt powder sample (JCPDS No. 5-0727), respectively. In this investigation, only four reflection lines of (100), (002), (101), and (110) for Co were considered.
are the diffraction intensities of (hkl) lines in the XRD patterns of the deposits and randomly oriented cobalt powder sample (JCPDS No. 5-0727), respectively. In this investigation, only four reflection lines of (100), (002), (101), and (110) for Co were considered.
2.4 Hardness test
Hardness measurements were carried out on an AMSLER D-6700 Vicker��s microhardness testing machine, using different applied loads of 0.1-2 N for 15 s. The mean values of at least five measurements conducted on different areas of each sample were considered. Due to little thickness of some coatings, the following equation was used to remove the effect of substrate on the obtained hardness values [17,18]:
 (2)
(2)
Table 1 Bath composition and deposition condition of cobalt coatings

where Hf is the real hardness of the coating, A is the large scale hardness (i.e. hardness of the substrate), t is the thickness of the coating, and Bc and Bs are the slopes of the measured hardness diagram versus d-1 (d is the average length of the diagonal) for coated samples and substrate, respectively.
3 Results and discussion
3.1 Effect of cobalt sulfate concentration
Electrodeposition of cobalt coatings at different concentrations of cobalt sulfate was performed in simple baths containing none of SDS and sodium saccharin, while the peak current density and duty cycle were 100 mA/cm2 and 10%, respectively. Figure 1 shows the effect of cobalt sulfate concentration on current efficiency and microhardness of Co coatings. At low concentrations of cobalt sulfate (less than 250 g/L), the current efficiency increases rapidly, while further increasing of cobalt sulfate concentration has a negligible effect on that. Cobalt ion concentration around the cathode is multiplied by increasing the cobalt sulfate concentration in the bath. Therefore, the probability of metal ion depletion near the cathode decreases. Moreover, reduction potential of cobalt becomes nobler with increasing its ion concentration in the bath [12,19,20]. These factors result in decreased hydrogen evolution and higher current efficiencies.

Fig. 1 Effect of cobalt sulfate concentration on current efficiency and microhardness of coatings
Plateau shape of the current efficiency diagram at elevated cobalt sulfate concentrations can be due to the increased columbic forces between the adjacent ions, which results in high viscosity of the bath and low ionic mobility [21]. It is also clear from Fig. 1 that cobalt sulfate concentration of the bath has a slight effect on the hardness of the resultant coatings. However, the hardness of all the coatings is significantly higher than that of the steel substrate (HV 193).
According to these results, the bath containing 250 g/L cobalt sulfate can be suitable for electro- deposition of cobalt films.
3.2 Effect of SDS concentration
SDS as an anionic surfactant does not interfere with electrodeposition of metal, which can cause to a brittle coating [22]. For investigation of SDS concentration effects on characteristics and hardness of the coatings, saccharin concentration, peak current density and duty cycle were kept constant in all the tests at 0 g/L, 100 mA/cm2 and 10%, respectively. Effect of bath SDS concentration on current efficiency and hardness of cobalt coatings is exhibited in Fig. 2. SDS concentration has a minor effect on microhardness of the obtained coatings, while current efficiency decreases with increasing SDS concentration of the bath. Current efficiency in a bath without SDS is 90%, while it reaches 75% in a bath containing 1 g/L SDS. This also leads to the reduction of the coatings thickness from about 11 to 9 ��m. In fact, SDS molecules cover the cathode surface and inhibit cobalt reduction reaction, resulting in lower current efficiencies [23-25].

Fig. 2 Effect of bath SDS concentration on current efficiency and microhardness of cobalt coatings
XRD patterns of cobalt coatings deposited from baths containing 0.2 and 1 g/L SDS are presented in Fig. 3. According to these patterns, hexagonal close- packed (hcp) structure of cobalt films is not affected by SDS concentration of the bath. Broad peaks in these patterns indicate fine crystallite sizes. The average crystallite sizes for the coatings obtained from the baths with 0.2 and 1 g/L SDS are 49 and 53 nm, respectively.
Variation of relative intensities of diffraction peaks can be expressed through the value of the relative texture coefficient. T(hkl) values of three diffraction lines of (100), (002) and (101) were calculated according to Eq. (1). Effect of SDS concentration on RTC values is inconsiderable. T(002) values for the coatings obtained from baths containing 0.2 and 1 g/L SDS are 63% and 68%, respectively, indicating that preferred orientation of both coatings is mostly [001] and majority of grains are oriented with their basal plane aligned parallel to the surface of the deposit [26]. Negligible effect of SDS on the coatings microhardness (Fig. 2) can be due to the similar structures as well as comparable crystallite size and preferred orientation of cobalt films.

Fig. 3 XRD patterns of cobalt coatings electrodeposited from baths containing 0.2 (a) and 1 g/L (b) SDS
Surfactants are surface active agents that adsorb on the cathode and lower the surface tension at H2-sample and H2-liquid boundaries, which causes easier detachment of hydrogen bubbles and improvement of surface quality [25,27]. Investigation of appearance and morphology of as-deposited cobalt coatings also reveals that surface quality is improved by adding SDS to the electrodeposition bath (Fig. 4). The least defective cobalt film is obtained from a bath with 0.2 g/L SDS. Density of pores and defects partly increases in the presence of higher concentrations of SDS in the bath. This can be attributed to the formation of micelles by surfactants, which reduces their efficiency.
Considering current efficiency and microscopy results, 0.2 g/L may be the proper concentration of SDS for cobalt electrodeposition.
3.3 Effect of sodium saccharin concentration

Fig. 4 SEM micrographs from surface of cobalt coatings obtained from baths containing 0 (a), 0.2 (b) and 1 g/L (c) SDS
Saccharin is an effective stress reducer, which often helps to improve surface quality and produce bright deposits [28,29]. Effect of sodium saccharin on current efficiency and microhardness of the coatings was examined in baths containing constant concentrations of cobalt sulfate (250 g/L) and SDS (0.2 g/L), while the peak current density and duty cycle were 100 mA/cm2 and 10%, respectively. Figure 5 shows that microhardness significantly increases, while current efficiency negligibly decreases, with enhancing the bath sodium saccharin concentration. Increasing of hardness can be due to the reduction of the coatings crystallite sizes [28,29], which is confirmed by XRD results. X-ray diffraction pattern of the coating obtained from a bath containing 0.5 g/L sodium saccharin is exhibited in Fig. 6. The hcp structure of cobalt film has not been changed by adding sodium saccharin to the bath. However, crystallite size is decreased. The calculated crystallite size for this sample is 36 nm, which is lower than that produced in a bath without saccharin (49 nm). Saccharin increases cathode surface overpotential, leading to smaller critical nucleus size [30]. Therefore, the probability of nucleation is rapidly increased and the crystallite size is refined by formation of more nuclei. Moreover, saccharin molecules and/or their decomposed sulfide-containing species can adsorb on the cathode surface during the deposition process, which retards diffusion of ad-ions toward the active growth sites. This causes difficulties in grain growth, and as a result higher nucleation rate and finer crystallites [30,31]. The adsorbed saccharin molecules can also act as an obstacle against effective movement of dislocations and possibly enhance the hardness of the deposits [30].
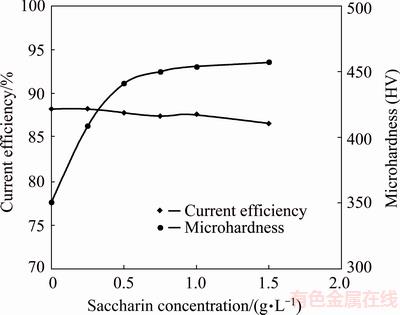
Fig. 5 Effect of bath sodium saccharin concentration on current efficiency and microhardness of cobalt coatings

Fig. 6 XRD pattern of cobalt coating electrodeposited from a bath containing 0.5 g/L sodium saccharin
It is also clear from Fig. 5 that the hardness increases rapidly when saccharin concentration of the bath is below 0.5 g/L, but it is almost constant at higher concentrations. Cathodic overpotentials tend to a constant value and saccharin adsorption on the cathode surface is saturated by increasing saccharin concentration of the bath [32]. Therefore, hardness values do not considerably change at high concentrations of sodium saccharin.
SEM micrograph from the surface of the cobalt film obtained from a bath with 0.5 g/L sodium saccharin is shown in Fig. 7. This coating is smoother than that obtained from baths without sodium saccharin (Fig. 4). Adsorption of saccharin at the active growth sites inhibits metal ion reduction at these places and results in smooth and shiny coating [32,33].

Fig. 7 SEM micrograph from surface of cobalt coating produced from a bath with 0.5 g/L sodium saccharin
According to the obtained results and by considering that high saccharin concentration increases impurity content and decreases ductility and strength of the coatings, the optimum amount of saccharin in cobalt electrodeposition bath can be 0.5 g/L.
3.4 Effect of peak current density
Effect of peak current density on cathodic current efficiency and deposition rate of cobalt coatings is shown in Fig. 8. Duty cycle was 10% in all the electrodeposition processes. According to Fig. 8, increasing the current density from 25 to 120 mA/cm2 enhances the current efficiency from about 55% to 92%. This means that cobalt ion reduction is facilitated, and reduction rate is increased by raising the current density within this range. However, current efficiency is decreased to about 46% by further increment of peak current density to 300 mA/cm2, representing sever hydrogen evolution reaction at high current densities [12,34,35]. High reduction rate of hydrogen ions also enhances hydroxyl ion concentration and solution pH near the cathode surface, resulting in the formation of hydroxide compounds, which prevent cobalt ion reduction on the cathode surface [14]. Therefore, in spite of the increment of deposition rates, current efficiency decreases at high current densities.
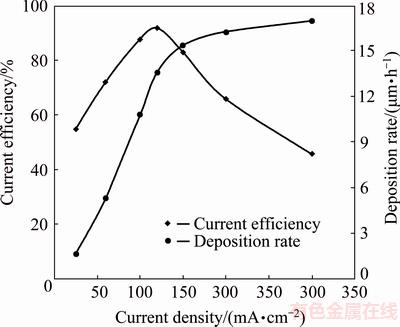
Fig. 8 Effect of current density on current efficiency and deposition rate of cobalt coatings
Figure 9 shows the effect of peak current density on the hardness of cobalt coatings. Microhardness has an ascending trend at low current densities. The maximum hardness (HV 448) is obtained at 120 mA/cm2. Enhancement of microhardness with raising the current density is due to the reduction of the coatings crystallite sizes as a result of higher cathodic overpotentials, and thus faster nucleation rates [6,36,37]. This is also confirmed by comparing XRD patterns of cobalt coatings deposited at 25 and 100 mA/cm2 (Figs. 10(a) and 6) with calculated crystallite sizes of 43 and 36 nm, respectively.

Fig. 9 Effect of current density on microhardness of cobalt coatings
By considering XRD patterns of the coatings produced at 200 and 300 mA/cm2 (Figs. 10(b) and (c)), the mean crystallite sizes for these films are 19 and 10 nm, respectively. In spite of smaller crystallite sizes of these coatings as compared to those deposited at low current densities, it is clear from Fig. 9 that microhardness drops off sharply at high current densities. This can be somewhat attributed to their open and porous morphologies.
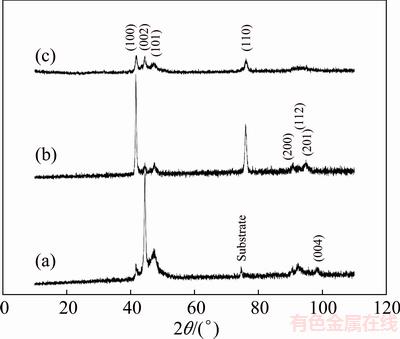
Fig. 10 XRD patterns of cobalt coatings electrodepositing at 25 (a), 200 (b) and 300 mA/cm2 (c)
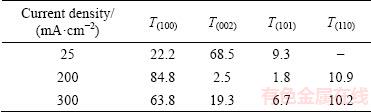
Preferred orientation also changes with variation of current density. RTC values of (100), (002), (101) and (110) planes of cobalt coatings electrodeposited at 25, 200 and 300 mA/cm2 are presented in Table 2. T(002) is severely decreased, while T(100) and T(101) are increased, by raising the current density. Variation of [001] texture to [100] can be due to the adsorption of different species (e.g. hydroxide compounds) on the cathode surface at high current densities [38]. These changes may be another reason for significantly different hardness of cobalt coatings obtained at 200 and 300 mA/cm2 with those produced at lower current densities (Fig. 9).
Table 2 Relative texture coefficient (RTC) values of (100), (002), (101) and (110) diffraction lines of Co coatings deposited at different current densities
As expected, different preferred orientations result in dissimilar appearance and morphology. Cobalt coatings are bright and shiny below 120 mA/cm2, but they become opaque and dark at higher current densities, as the coating deposited at 300 mA/cm2 has burned and totally dark appearance. Effect of current density on morphology of the as-deposited coatings is shown in Fig. 11. Smooth morphology at low current density is changed to needle-shaped by increasing the electro- deposition current density to 200 mA/cm2. Micro-cracks are also formed by further increment of current density to 300 mA/cm2 (Fig. 11(c)). This may be due to the formation of large amounts of hydride and hydroxide compounds, and as a result the decrement of ductility, at this current density.

Fig. 11 SEM micrographs from surface of cobalt coatings obtained at 25 (a), 200 (b) and 300 mA/cm2 (c)
Considering the obtained results, 120 mA/cm2 is chosen as the optimum current density for cobalt electrodeposition in this work.
3.5 Effect of pulse duty cycle
Mass transport plays a key role in deposition rate, morphology and properties of electrodeposits. Mass transport in the electrodeposition process depends significantly on the pulse limiting current density (JL), while JL itself is a function of pulse parameters, especially duty cycle. Limiting current density is reduced by increasing duration of pulse-on time. For achieving an appropriate coating, pulse-on time should be short enough to stay below the JL, but on the other hand, it should also be sufficiently long so that full charging of electrical double layer can occur [39,40]. Effect of the duty cycle on current efficiency and deposition rate of cobalt coatings is shown in Fig. 12. At low duty cycle (5%, pulse-on time of 5 ms), there is not enough pulse-on time for fully charging the double layer during each cycle, and thus current efficiency is small. At higher duty cycles (10% and 20%, pulse-on time of 10 and 20 ms, respectively), electrical double layer can be fully charged, while JL is still larger than the exerted current density. Therefore, current efficiency rises with duty cycle. However, further increasing the duty cycle decreases the JL to smaller values than the exerted current density. In this case, cobalt ion concentration around the cathode surface is reduced, and hydrogen evolution is intensified, resulting in an increment of pH and formation of hydroxide compounds as well as a drop of current efficiency.

Fig. 12 Effect of pulse duty cycle on current efficiency and deposition rate of cobalt coatings

Fig. 13 Effect of pulse duty cycle on microhardness of cobalt coatings

Fig. 14 XRD patterns of cobalt coatings electrodeposited at duty cycles of 5% (a), 20% (b) and 40% (c)
Effect of the duty cycle on microhardness of cobalt coatings is shown in Fig. 13. The microhardness decreases with enhancing pulse-on time, which can be due to the increment of crystallite size as also reported by other researchers [41,42]. This is also confirmed by calculation of crystallite sizes from XRD patterns of cobalt coatings obtained at different duty cycles (Fig. 14). Crystallite sizes are promoted from 25 to 63 nm by increasing the duty cycle from 5% to 40%. In fact, growth opportunity of the formed nuclei at a long pulse- on time is more than that at a short one.
It is also clear from Fig. 14 that all the coatings have hcp structure, but with different preferred orientations. T(100) increases at the expense of T(002) reduction (Table 3) with the enhancement of the duty cycle, which can be another reason for lower microhardness values at high duty cycles.
Table 3 Relative texture coefficient (RTC) values of (100), (002), (101) and (110) diffraction lines of Co coatings deposited at different duty cycles

SEM micrographs of cobalt coatings deposited at 5%, 10%, 20% and 40% duty cycles are shown in Fig. 15. At duty cycles of 5% and 10%, the surface of cobalt films is smooth and shiny. Uniform thickness of the coating at 10% duty cycle is also clear from cross sectional image (Fig. 15(c)). However, at duty cycle of 20%, the surface of the coating becomes somewhat opaque and defective (Fig. 15(d)). Higher magnification images from defective areas reveal the formation of needle-shaped morphology at these zones (Figs. 15(e) and (f)). Formation of this morphology can be due to variation of preferred orientation with duty cycle (Table 3), because higher T(100) encourages development of needle-shaped morphologies. At higher duty cycles, [100] preferred orientation is dominant and morphology of cobalt coatings is totally needle-shaped (Figs. 15(g) and (h)).
According to the obtained results, duty cycles of 5%, 10% or 20% may be chosen as the optimum value for electrodeposition of cobalt coatings. However, 10% duty cycle can be the best choice because of relatively high deposition rate, current efficiency and hardness, as well as smooth and defect-free morphology and appearance of the coatings obtained at this duty cycle.
4 Conclusions
1) An increase in cobalt sulfate concentration in the bath resulted in higher current efficiencies, while had almost no effect on hardness values.
2) Current efficiency decreased from about 90% to 75% with raising bath SDS concentration from 0 to 1 g/L. Although SDS had a negligible effect on hardness and structure of cobalt coatings, the least defective morphology was obtained from a bath with 0.2 g/L SDS.
3) Hexagonal close-packed structure of cobalt coatings was not changed by adding saccharin to the bath, but coatings with smoother morphology, finer crystallites and higher hardness values were obtained from baths containing sodium saccharin.
4) Current efficiency and hardness of the coating deposited at 120 mA/cm2 were about two times of those for the film obtained at 300 mA/cm2. Crystallite sizes decreased, preferred orientation changed from [002] to mostly [100], and smooth morphology converted to needle-shaped with rising pulse peak current density from 25 to 300 mA/cm2.
5) While there was an optimum duty cycle to obtain the maximum current efficiency, hardness of coatings decreased from HV 473 to HV 201 with increasing duty cycle from 5% to 40%. Variation of preferred orientation resulted in totally different morphologies at different duty cycles.
6) Shiny and hard cobalt coating was deposited with a high current efficiency from a bath containing 250 g/L cobalt sulfate, 0.2 g/L SDS and 0.5 g/L saccharin, while current density and duty cycle were 120 mA/cm2 and 10%, respectively.

Fig. 15 SEM micrographs of cobalt coatings deposited at 5% (a), 10% (b, c), 20% (d-f) and 40% (g, h) duty cycles
References
[1] Weston d p, Shipway p h, Harris s j, Cheng m k. Friction and sliding wear behaviour of electrodeposited cobalt and cobalt�Ctungsten alloy coatings for replacement of electrodeposited chromium [J]. Wear, 2009, 267: 934-943.
[2] Su f, Liu c, Zuo q, Huang p, Miao m. A comparative study of electrodeposition techniques on the microstructure and property of nanocrystalline cobalt deposit [J]. Materials Chemistry and Physics, 2013, 139: 663-673.
[3] Aledresse A, Alfantazi A. A study on the corrosion behavior of nanostructured electrodeposited cobalt [J]. Journal of Materials Science, 2004, 39: 1523-1526.
[4] Wasekar N P, Sundararajan G. Sliding wear behavior of electrodeposited Ni-W alloy and hard chrome coatings [J]. Wear, 2015, 342-343: 340-348.
[5] Mahdavi s, Allahkaram s r. Composition, characteristics and tribological behavior of Cr, Co-Cr and Co-Cr/TiO2 nano-composite coatings electrodeposited from trivalent chromium based baths [J]. Journal of Alloys and Compounds, 2015, 635: 150-157.
[6] Wang L, Gao Y, Xu T, Xue Q. A comparative study on the tribological behavior of nanocrystalline nickel and cobalt coatings correlated with grain size and phase structure [J]. Materials Chemistry and Physics, 2006, 99: 96-103.
[7] Banbur-Pawlowska S, Mech K, Kowalik R, Zabinski P. Analysis of electrodeposition parameters influence on cobalt deposit roughness [J]. Applied Surface Science, 2016, 388: 805-808.
[8] Frank A C, Sumodjo P T A. Electrodeposition of cobalt from citrate containing baths [J]. Electrochimica Acta, 2014, 132: 75-82.
[9] Schiavi P G, Altimari P, Zanoni R, Pagnanelli F. Morphology-controlled synthesis of cobalt nanostructures by facile electrodeposition: Transition from hexagonal nanoplatelets to nanoflakes [J]. Electrochimica Acta, 2016, 220: 405-416.
[10] Li M, Wang Z, Reddy R G. Cobalt electrodeposition using urea and choline chloride [J]. Electrochimica Acta, 2014, 123: 325-331.
[11] Patnaik P, Padhy S K, Tripathy B C, Bhattacharya I N, Paramguru R K. Electrodeposition of cobalt from aqueous sulphate solutions in the presence of tetra ethyl ammonium bromide [J]. Transactions of Nonferrous Metals Society of China, 2015, 25: 2047-2053.
[12] Zhou J, Wang S, Song X. Electrodeposition of cobalt in double-membrane three-compartment electrolytic reactor [J]. Transactions of Nonferrous Metals Society of China, 2016, 26: 1706-1713.
[13] Patnaik P, Tripathy B C, Bhattacharya I N, Paramguru R K, Mishra B K. Effect of tetra propyl ammonium bromide during cobalt electrodeposition from acidic sulfate solutions [J]. Metallurgical and Materials Transactions B, 2015, 46: 1252-1256.
[14] Lajevardi S A, Shahrabi T. Effects of pulse electrodeposition parameters on the properties of Ni-TiO2 nanocomposite coatings [J]. Applied Surface Science, 2010, 256: 6775-6781.
[15] Yang Y, Cheng Y F. Electrolytic deposition of Ni-Co-SiC nano-coating for erosion-enhanced corrosion of carbon steel pipes in oils and slurry [J]. Surface and Coatings Technology, 2011, 205: 3198-3204.
[16] Zhang Z, Jiang C, Fu P, Cai F, Ma N. Microstructure and texture of electrodeposited Ni-ZrC composite coatings investigated by Rietveld XRD line profile analysis [J]. Journal of Alloys and Compounds, 2015, 626: 118-123.
[17] Korsunsky A M, Constantinescu A. Work of indentation approach to the analysis of hardness and modulus of thin coatings [J]. Materials Science and Engineering A, 2006, 423: 28-35.
[18] Korsunsky A M, McGurk M R, Bull S J, Page T F. On the hardness of coated systems [J]. Surface and Coatings Technology, 1998, 99: 171-183.
[19] Sharma I G, Alex P, Bidaye A C, Suri A K. Electrowinning of cobalt from sulphate solutions [J]. Hydrometallurgy, 2005, 80: 132-138.
[20] Huang J H, Simard C K, Alfantazi A M. Electrowinning of cobalt from a sulfate-chloride solution [J]. Canadian Metallurgical Quarterly, 2004, 43: 163-172.
[21] Abdel Rahman H H, Moustafa A H E, Abdel Magid S M K. High rate copper electrodeposition in the presence of inorganic salts [J]. International Journal of Electrochemical Science, 2012, 7: 6959-6975.
[22] Weston D P, Zhu Y Q, Zhang D, Miller C, Kingerley D G, Carpenter C. Co-electrodeposition of inorganic fullerene (IF-WS2) nano-particles with cobalt from a gluconate bath with anionic and cationic surfactants [J]. Electrochimica Acta, 2011, 56: 6837-6846.
[23] Mafi I R, Dehghanian C. Comparison of the coating properties and corrosion rates in electroless Ni�CP/PTFE composites prepared by different types of surfactants [J]. Applied Surface Science, 2011, 257: 8653-8658.
[24] GOmez E, PanE S, Alcobe X, VallEs E. Influence of a cationic surfactant in the properties of cobalt�Cnickel electrodeposits [J]. Electrochimica Acta, 2006, 51: 5703-5709.
[25] Elansezhian R, Ramamoorthy B, Nair P K. The influence of SDS and CTAB surfactants on the surface morphology and surface topography of electroless Ni-P deposits [J]. Journal of Materials Processing Technology, 2009, 209: 233-240.
[26] Mahdavi S, Allahkaram S R. Characteristics of electrodeposited cobalt and titania nano-reinforced cobalt composite coatings [J]. Surface and Coatings Technology, 2013, 232: 198-203.
[27] Du L, Xu B, Dong S, Yang H, Wu Y. Preparation, microstructure and tribological properties of nano-Al2O3/Ni brush plated composite coatings [J]. Surface and Coatings Technology, 2005, 192: 311-316.
[28] Manhabosco T M, M��ller I L. Influence of saccharin on morphology and properties of cobalt thin films electrodeposited over n-Si(100) [J]. Surface and Coatings Technology, 2008, 202: 3585-3590.
[29] Cotell C M, Sprague J A, Smidt F A Jr. ASM metals handbook (5): Surface engineering [M]. 9th ed. USA: ASM International, 1994.
[30] Yu J K, Wang M Z, Li Q, Yang J, Liu L. Effects of saccharin on microstructure and property of electro-deposited Ni-Fe alloys [J]. Transactions of Nonferrous Metals Society of China, 2009, 19: 805-809.
[31] Hassani S, Raeissi K, Azzi M, Li D, Golozar M A, Szpunar J A. Improving the corrosion and tribocorrosion resistance of Ni-Co nanocrystalline coatings in NaOH solution [J]. Corrosion Science, 2009, 51: 2371-2379.
[32] Rashidi A M, Amadeh A. Effect of electroplating parameters on microstructure of nanocrystalline nickel coatings [J]. Journal of Materials Science and Technology, 2010, 26: 82-86.
[33] Li Y, Jiang H, Wang D, Ge H. Effects of saccharin and cobalt concentration in electrolytic solution on microhardness of nanocrystalline Ni-Co alloys [J]. Surface and Coatings Technology, 2008, 202: 4952-4956.
[34] Cho K W, Kwon H S. Effects of electrodeposited Co and Co-P catalysts on the hydrogen generation properties from hydrolysis of alkaline sodium borohydride solution [J]. Catalysis Today, 2007, 120: 298-304.
[35] Gabe D R. The role of hydrogen in metal electrodeposition processes [J]. Journal of Applied Electrochemistry, 1997, 27: 908-915.
[36] Rashidi A M, Eivani A R, Amadeh A. Application of artificial neural networks to predict the grain size of nano-crystalline nickel coatings [J]. Computational Materials Science, 2009, 45: 499-504.
[37] Savall C, Godon A, Creus J, Feaugas X. Influence of deposition parameters on microstructure and contamination of electrodeposited nickel coatings from additive-free sulphamate bath [J]. Surface and Coatings Technology, 2012, 206: 4394-4402.
[38] Vicenzo A, Cavallotti P L. Growth modes of electrodeposited cobalt [J]. Electrochimica Acta, 2004, 49: 4079-4089.
[39] Marlot A, Kern P, Landolt D. Pulse plating of Ni-Mo alloys from Ni-rich electrolytes [J]. Electrochimica Acta, 2002, 48: 29-36.
[40] Chandrasekar M S, Pushpavanam M. Pulse and pulse reverse plating��Conceptual, advantages and applications [J]. Electrochimica Acta, 2008, 53: 3313-3322.
[41] Yang Y, Cheng Y F. Fabrication of Ni�CCo�CSiC composite coatings by pulse electrodeposition��Effects of duty cycle and pulse frequency [J]. Surface and Coatings Technology, 2013, 216: 282-288.
[42] Pavlatou E A, Spyrellis N, Influence of pulse plating conditions on the structure and properties of pure and composite nickel nanocrystalline coatings [J]. Russian Journal of Electrochemistry, 2008, 44: 745-754.
S. MAHDAVI1, S. R. ALLAHKARAM2
1. Research Center for Advanced Materials, Faculty of Materials Engineering, Sahand University of Technology, 5133511996, Tabriz, Iran;
2. School of Metallurgy and Materials Engineering, College of Engineering, University of Tehran, Tehran, Iran
ժ Ҫ�������������������������ܻ����Һ���Ʊ������ܶƲ㣬�о���Һ��ɺ͵���������Ե���Ч���Լ��Ʋ���ò���ṹ��Ӳ�ȵ�Ӱ�죬ȷ����ѳ����������о����֣������Һ��������Ũ�Ⱥ�ʮ�����������(SDS)�����ԶƲ���Ӳ�ȵ�Ӱ��ɺ��Բ��ƣ����Ե��������Ч�ʵ�Ӱ���������ڵ������Һ�м����Ǿ�����ʹ�����ܶѽṹ(hcp)�ܱ�Ĥ�ľ����ߴ��С����Ӳ����ߣ����Ե���Ч��������Ӱ�졣�ں���SDS���Ǿ��Ķ�Һ�л�õĶƲ������⻬��ȱ�ݸ��١��������������Ч�ʺ���Ӳ�Ⱦ����ֵ�����ܶȺ�ռ�ձȵı仯���仯�����ŵ����ܶȺ�ռ�ձȵ�������⻬�ĶƲ��Ϊ��״�������ߴ������ȡ��Ҳ��֮�ı䡣
�ؼ��ʣ��ܶƲ㣻�����������Ǿ���ʮ����������ƣ�����������Ч�ʣ���Ӳ��
(Edited by Bing YANG)
Corresponding author: S. R. ALLAHKARAM; E-mail: akaram@ut.ac.ir
DOI: 10.1016/S1003-6326(18)64846-0

