J. Cent. South Univ. (2012) 19: 797-802
DOI: 10.1007/s11771-012-1074-6![]()
Thermodynamic analysis of simplified dual-pressure
ammonia-water absorption power cycle
HUA Jun-ye(����Ҷ), CHEN Ya-ping(����ƽ), LIU Hua-jin(�����), WU Jia-feng(��η�)
Key Laboratory of Energy Thermal Conversion and Control of Ministry of Education,School of Energy and Environment, Southeast University, Nanjing 210096, China
? Central South University Press and Springer-Verlag Berlin Heidelberg 2012
Abstract: A simplified dual-pressure ammonia-water absorption power cycle (DPAPC-a) using low grade energy resources is presented and analyzed. This cycle uses turbine exhaust heat to distill the basic solution for desorption. The structure of the cycle is simple which comprises evaporator, turbine, regenerator (desorber), absorber, pump and throttle valves for both diluted solution and vapor. And it is of high efficiency, because the working medium has large temperature difference in evaporation and small temperature difference in absorptive condensation, which can match the sensible exothermal heat resource and the cooling water simultaneously. Orthogonal calculation was made to investigate the influence of the working concentration, the basic concentration and the circulation multiple on the cycle performance, with 85-110 ��C heat resource and 20-32 ��C cooling water. An optimum scheme was given in the condition of 110 ��C sensitive heat resource and 20 ��C cooling water, with the working concentration of 0.6, basic concentration of 0.385, and circulation multiple of 5. The thermal efficiency and the power recovery efficiency are 8.06 % and 6.66%, respectively. The power recovery efficiency of the DPAPC-a is 28.8% higher than that of the steam Rankine cycle (SRC) and 12.7% higher than that of ORC (R134a) under the optimized situation.
Key words: absorption power cycle; ammonia-water; circulation multiple; ammonia concentration; Kalina cycle
1 Introduction
With the shortage of energy resources and the increased awareness of sustainable development and green environment protection, the utilization of low grade energy resources is concerned by more and more people. The low grade energy resources which are huge in quantities and varieties, including industrial waste heat, geothermal and solar energy, are promising alternative energy resources to fossil fuels. However, the selection of suitable working medium as well as power cycle is what should be considered primarily. As a traditional power circulation medium, water, with very low pressure, doesn��t work well for recovery of low-temperature heat sources, because the turbine will be of long blades and big size, as the consequence, huge equipment cost and friction loss. Also, the vacuum condition at turbine exhaust tends to cause air leakage into the system, which makes the reduction of heat transfer coefficient in condenser, thus the system must be equipped with air evacuation device, making the system more complex. There are two main approaches for solving this issue. One is to use organic Rankine cycle (ORC) [1-4] with low-boiling-point organic medium and the other is to use absorptive power cycle with ammonia-water solution. The main shortcoming of ORC lies in the difficulty of choosing suitable working medium. The ammonia-water medium has superior features over organic media. Both water and ammonia are natural substances, plentiful and inexpensive, and the mixture has superior thermophysical properties of variable temperature in phase change to match heat transfer processes with both heat source and cooling water.
Since KALINA proposed the first absorptive ammonia-water power cycle as the bottom cycle in combined power generation system [5], the ammonia- water power cycle has consistently drawn attention [6-14]. However, the current analysis for ammonia-water power cycle showed that it is still difficult for the practical application and few demonstration projects were built. Therefore, further efforts should be made to improve the cycle so that it could be driven by the low grade energy resources. The Kalina cycle and other triple-pressure cycles are suitable for high to medium temperature waste heat situation. For the low temperature waste heat, CHEN [14] proposed a dual-pressure ammonia-water absorption power cycle (DPAPC-a) [14], which is a simplified case of triple-pressure cycle at low-temperature heat source
domain, for the evaporation pressure could be merged with the desorption pressure.
The proposal of this DPAPC-a system will contribute to the high use of low temperature waste heat and can be as a widely used extension of ammonia-water absorption cycle.
2 DPAPC-a system
The dual-pressure ammonia-water absorption power cycle mainly has two cycle loops. For the heat resource below 120 ��C, the simplified loop of dual-pressure ammonia-water absorption power cycle (DPAPC-a), as shown in Fig. 1, is suitable, while for the heat resource of temperature from 120 ��C to 150 ��C, a somewhat complex loop (DPAPC-b) with an external preheater and a second recuperative heat exchanger are incorporated into the simplified cycle for desorption [15]. In this work, the thermodynamic analysis is focused on the simplified dual-pressure ammonia-water absorption power cycle (DPAPC-a).
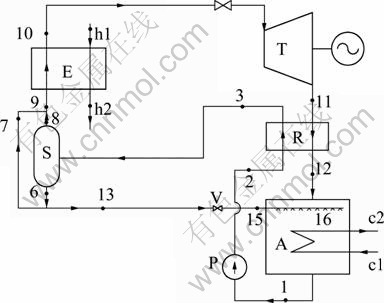
Fig. 1 Schematic diagram of simplified dual-pressure ammonia-water absorption power cycle (DPAPC-a) (E�� Evaporator; T��Turbine; R��Regenerator; A��Absorber; P�� Pump; V��Throttle valve; S��Separator; h1, h2-Inlet and outlet of heat resource; c1, c2-Inlet and outlet of cooling water)
The medium in the evaporator and the turbine is called the working solution. With the working solution vapor (10) expanding in the turbine, it outputs work. And then, the exhaust vapor (11) releases part of thermal energy but remains the same concentration in a heat exchanger called regenerator as wet vapor (12) until it is absorbed by the dilute solution (15) in an absorber, forming basic solution (1) at the solution outlet of the absorber, with latent heat transmitted away by the cooling water. Then, the basic solution is pumped to high pressure (2) and is heated to a two-phase state (3) by the turbine exhaust vapor in the regenerator to realize desorption. The working solution (9) is formed by mixing the rich ammonia vapor (8) with some dilute liquid solution (7) from the separator and then goes into the evaporator for heating, while the rest (13) of the dilute solution (6) goes into the absorber through a throttle valve.
There exists a mutual restrict relationship among the desorption pressure (high pressure), the absorption pressure (low pressure), as well as the working concentration and basic concentration, as shown in Fig. 2. The reduction of turbine back pressure will be beneficial to increasing the turbine output, but it is restricted by the heat transfer requirement of desorption. If the absorption pressure is decreased with other conditions remaining constant, then the basic solution at regenerator outlet will not achieve two-phase flow state for desorption, because the heat transfer difference is not sufficient between turbine exhaust vapor temperature (near the intersection point of the working concentration and the low pressure dew point line) and the desorption temperature (higher than the intersection point of the basic concentration and the high pressure bubble point line). Similarly, if other conditions remain unchanged and just the high pressure rises with raised heat source temperature, the basic solution at regenerator outlet will not achieve two-phase flow state for desorption with increased desorption temperature unless the low pressure is increased with increased basic concentration. Therefore, this kind of cycle loop is just applicable to the low waste heat source below 120 ��C. However, by using external heat source to realize desorption heating, the DPAPC-b can be applied to somewhat higher temperature heat resource.
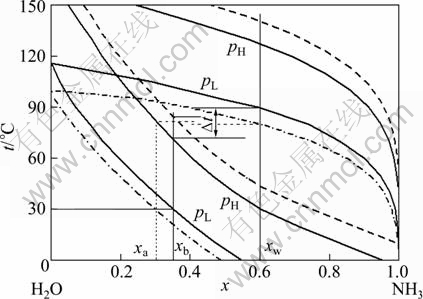
Fig. 2 Phase equilibrium diagram of DPAPC-a
There exist at least three independent concentration solutions: basic solution xb from the solution outlet of the absorber to the inlet of the separator, work solution xw from the inlet of the evaporator to the outlet of the work solution side of the regenerator, and dilute solution xa from the liquid outlet of the separator to the dilute solution inlet of the absorber. The ammonia-water absorption power cycle can adapt to the different temperature change requirements in evaporator and absorber, just through adjusting ammonia concentration, and thereby reduces exergy loss of the cycle.
3 Thermodynamic analysis and modeling
For thermal design optimization with computer programming, it needs a calculation program for physical properties of ammonia-water mixture. This work adopts Schulz state equations to establish the property parameter model. These equations with high precision in low temperature range are widely used in thermal calculation of ammonia-water absorption refrigeration systems. The liquid enthalpy of pure ammonia is set at 200 kJ/kg when temperature is 0 ��C. The calculation results under the application scope show that the maximum relative error for the liquid enthalpy of the mixture is 5.8%, while the maximum relative error for vapor enthalpy is 0.5%. Both of them comply with the engineering design accuracy.
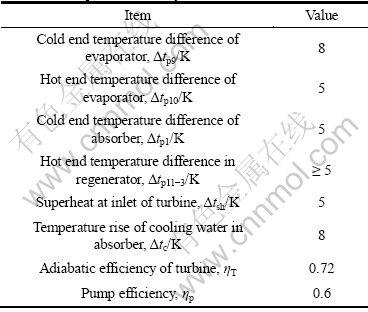
Table 1 lists pinch-point heat transfer temperature difference of equipment and temperature rise and efficiencies for the cycle parameter calculation. According to mass and energy conservation laws, ignoring all pressure drops and heat losses in heat exchangers and pipelines, the thermodynamic equations of state points of main components of the DPAPC-a system are listed in Table 2.
Table 1 Basic parameters of cycle
Based on the laws of thermodynamics, the main indicators used to evaluate this power circulation system are listed in Table 3.
The benchmark temperature t0 from Eq. (12) for waste heat recovery efficiency hh may take the ambience environment temperature or the minimum temperature that waste heat could be utilized, and t0=50 ��C is selected in this work. For waste heat power recovery system, the power recover efficiency h0 is the most important thermal performance index that measures the efficiency of the whole system, and it accords more with the essence of waste recycling system than the index of thermal efficiency. Because the higher thermal efficiency is sometimes at the cost of higher heat resource exhaust temperature, it may not recover more power in parameter optimization process.
Table 2 Thermodynamic formula of state points of main components

Table 3 Main efficiency indexes of cycle

4 Result and discussion
4.1 Circulation multiple
Circulation multiple f means the flow rate ratio of working media in absorber to that in evaporator. In the circumstances of given working concentration and basic concentration, f is only related to the desorption temperature t3. With other parameters unchanged, when f increases, desorption temperature of separation decreases, thus the waste heat recovery efficiency hh increases. Figure 3(a) shows that the input heat raises fast first and then levels off with the change of circulation multiple f, while the turbine power declines linearly. Figure 3(b) shows that the thermal efficiency h declines with the increase of circulation multiple f; however, the comprehensive index of the power recovery efficiency h0 increases fast first and then levels off with the increase of circulation multiple f. Considering generally the heat transfer performance and operation condition, the circulation multiple f is selected to be 5 in this work.
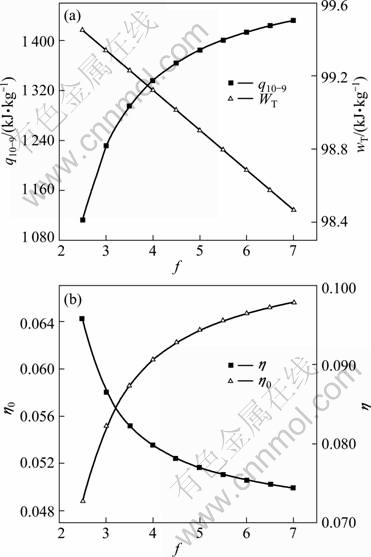
Fig. 3 Influence of circulation multiple f on thermal performance of cycle: (a) Absorbing heat q10-9 and turbine specific power wT; (b) Power recovery efficiency h0 and thermal efficiency h
4.2 Concentration
As mentioned before, the whole power cycle mainly has three medium concentrations: work concentration, basic concentration and dilute concentration, of which only two are independent with fixed circulation multiple f. Figure 4 shows the trend of the efficiency performances of the cycle with the changes of both basic medium concentration xb and work concentration xw with other parameters fixed and the circulation multiple f of 5.
From Figs. 4(a) and 4(b), it can be seen that with fixed working concentration xw, the thermal efficiency h and power recovery efficiency h0 increase linearly with the increase of basic concentration xb until they reach summit and then descend sharply, and that the curves of both thermal efficiency h and power recovery efficiency h0 with large working concentration xw have higher values under the calculation scope. This indicates that there exist optimal working concentration and basic concentration which make the system achieve the optimal state. By considering comprehensively the thermal performance and operation condition, the system working concentration xw is chosen as 0.6 and basic concentration xb is chosen as 0.384-0.385. Moreover, in order to keep the normal operation of the system equipment, the pressures of the desorption (high) and absorption (low) should also change accordingly, otherwise large humidity at turbine exhaust will cause turbine blades liable to water erosion. In this way, a constraint of turbine outlet vapor humidity less than 5% has been set in the thermal calculation program.
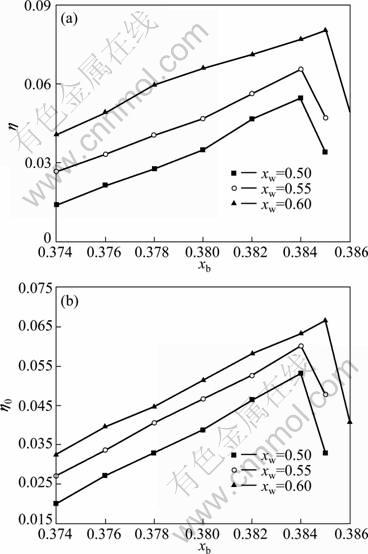
Fig. 4 Influence of concentrations on efficiencies of cycle: (a) Thermal efficiency h; (b) Power recovery efficiency h0
4.3 Heat source temperature and cooling water temperature
By selecting the circulation multiple f=5, the working concentration xw=0.6, under the heat source temperature from 85 ��C to 110 ��C, and the cooling water temperature from 20 ��C to 32 ��C, the thermal calculation is performed according to equations in Table 2. Some comparative results are shown as line-curves in Figs. 5(a) and 5(b) and more intuitively surface graphics in Figs. 6(a) and 6(b). Figures 5 and 6 show the influence of both heat source and cooling water temperature on the thermal properties of the DPAPC-a.
Obviously, as heat source temperature increases and/or cooling water temperature decreases, both the thermal efficiency and power recovery efficiency increase accordingly. The turbine inlet temperature is directly influenced by the waste heat source temperature, and from Fig. 2 it can be seen that the high pressure of the cycle pH will cause the change of the heat source temperature at fixed working concentration, and then the low pressure of the cycle pL or the basic concentration xb needs to be adjusted to fit heat transfer requirement. Thus, the calculation for each scheme also includes the optimization of basic concentration xb. From Figs. 5 and 6, it is also revealed that when the waste heat source temperature is lower than 90 ��C and at same time the cooling water temperature is higher than 28 ��C, there exists no effective point, which means that the thermal cycle could not operate in such condition.
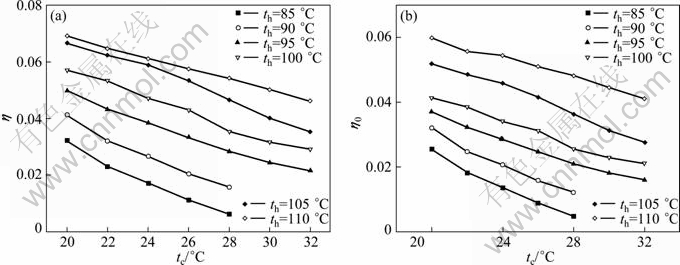
Fig. 5 Influence of temperatures of heat source and cooling water on efficiencies of cycle in line-curves: (a) Thermal efficiency h; (b) Power recovery efficiency h0
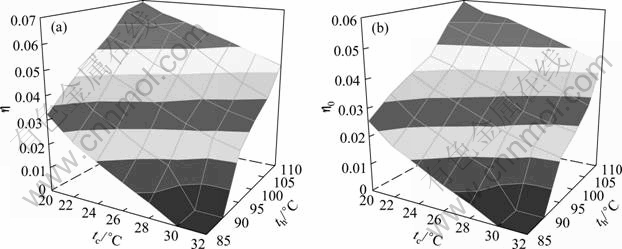
Fig. 6 Influence of temperatures of heat source and cooling water on efficiencies of cycle in surface graph: (a) Thermal efficiency h; (b) Power recovery efficiency h0
4.4 Optimal conditions
Under the heat source temperature of 110 ��C, and the cooling water temperature of 20 ��C, synthesizing all the parameter optimization analyses above, the optimized parameters of the DPAPC-a are obtained, as listed in Table 4.
4.5 Comparison of DPAPC-a with SRC and ORC
In order to have a clearer understanding with the economic features of DPAPC-a cycle, select the same temperatures of heat resource and cooling water to compare DPAPC-a with steam Rankine cycle (SRC) and organic Rankine cycle (ORC, medium R134a), with results shown in Table 5 and Fig. 6. From Table 5, the power recovery efficiency of DPAPC-a appears much higher than that of the other two cycles, with increments of 28.8% and 12.7% compared with SRC and ORC, respectively. Figure 6 shows the heat transfer of the working mediums in three cycles with both heat resource and cooling water, and reveals that the higher power recovery efficiency of DPAPC-a over that of the SRC and the ORC lies in its higher waste heat recovery efficiency with minimized heat transfer exergy loss in evaporation.
Table 4 Optimal thermodynamic properties of state-points of cycle
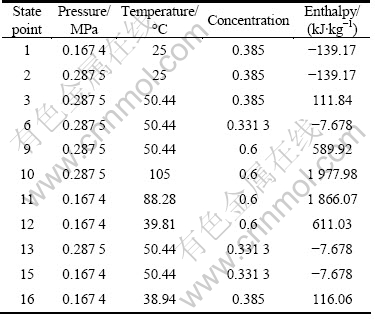
Table 5 Comparison of performance parameters of three kinds of cycles

![]()
Fig. 7 Heat transfer curves of three cycles
5 Conclusions
1) The simplified dual-pressure ammonia-water absorption power cycle (DPAPC-a) is suitable for power recovery from 85 ��C to 110 ��C of low grade sensitive heat resource. It is simple, but has higher efficiency and other feasible characteristics over the conventional SRC and ORC, because the ammonia-water mixture in the cycle has suitable temperature difference which matches the heat resource and cooling water, respectively.
2) For waste heat recovery system, the power recovery efficiency is a more reasonable index to evaluate the cycle efficiency feature, which is a comprehensive index of multiplying thermal efficiency by waste heat recovery efficiency. In fact, it is high waste heat recovery efficiency that leads to high power recovery efficiency of the DPAPC-a.
3) There exists a mutual restrict relationship among desorption pressure, absorption pressure, working concentration and basic concentration in the DPAPC-a.
4) As the circulation multiple increases, the power recovery efficiency of the cycle improves due to higher waste heat recovery efficiency, in spite of the decreased thermal efficiency.
5) The working concentration and the basic concentration of the ammonia-water medium of the cycle shall be matched to improve the performance.
References
[1] RADERMACHER R. Thermodynamic and heat transfer implications of working fluid mixtures in Rankine cycles [J]. Int J Heat Fluid Flow, 1989, 10(2): 90-102.
[2] MAGO P J, CHAMRA L M, SOMAYAJI C. Performance analysis of different working fluids for use in organic Rankine cycles [C]// Proceedings of the Institution of Mechanical Engineers, Part A: Journal of Power and Energy, 2007: 255-263.
[3] BORSUKIEWICZ G A, NOWAK W. Maximizing the working fluid flow as away of increasing power output of geothermal power plant [J]. Appl Therm Eng, 2007, 27(11/12): 2074-2078.
[4] RAYEGAN R, TAO Y X. A procedure to select working fluids for solar organic Rankine cycles (ORCs) [J]. Renew Energy, 2011, 3(6): 659-670.
[5] KALINA A I. Combined-cycle system with novel bottoming cycle [J]. J Eng Gas Turbines Power, 1984, 106(10): 737-742.
[6] GOSWAMI D Y, XU F. Analysis of a new thermodynamic cycle for combined power and cooling using low and mid temperature solar collectors [J]. Solar Energy Engineering, 1999, 121(2): 91-97.
[7] INFANTE FERREIRA C A, ZAMFIRESCU C, ZAVTSEV D. Twin screw oil-free wet compressor for a compression�Cabsorption cycle [J]. Refrigeration, 2006, 29(4): 556-565.
[8] ROY P, DESILETS M, GALANIS N, NESREDDINE H, CAYER E. Thermodynamic analysis of a power cycle using a low-temperature source and a binary NH3-H2O mixture as working fluid [J]. Int J Thermal Sci, 2010, 49(1): 48-58.
[9] ZAMFIRESCU C, DINCER I. Thermodynamic analysis of a novel ammonia�Cwater trilateral Rankine cycle [J]. Thermochim Acta, 2008, 477(1/2): 7-15.
[10] PADILLA R V, DEMIRKAYA G, GOSWAMI D Y, STEFANAKOS E, RAHMAN M. Analysis of power and cooling cogeneration using ammonia-water mixture [J]. Energy, 2010, 35(12): 1-9.
[11] LUO Chen-ding, ZHANG Na, CAI Rui-xian, LIU M. Sensitivity analysis of ammonia absorption power/refrigeration combined cycle [J]. Journal of Chinese Electrical Engineering Science, 2008, 28 (17): 1-7. (in Chinese)
[12] WAGAR W R, ZAMFIRESCU C, DINCER Z. Thermodynamic performance assessment of an ammonia�Cwater Rankine cycle for power and heat production [J]. Energy Conversation Manage, 2010, 51(12): 2501-2509.
[13] CHEN Ya-ping. Thermodynamic analysis of a modified Kalina cycle [J]. Journal of Southeast University, 1989, 19(4): 52-58. (in Chinese)
[14] CHEN Ya-ping. Thermodynamic analysis of a simplified Kalina cycle [J]. Thermal Power Generation, 1997, 47(4): 20-23. (in Chinese)
[15] LIU Hua-jin. Study on a modified ammonia-water absorption power cycle system for utilization of low-grade waste heat [D]. Nanjing: Southeast University, 2011. (in Chinese)
(Edited by YANG Bing)
Foundation item: Project(50976022) supported by the National Natural Science Foundation of China; Project(BY2011155) supported by Science and Technology Innovation and Transformation of Achievements of Special Fund of Jiangsu Province, China
Received date: 2011-07-26; Accepted date: 2011-11-14
Corresponding author: CHEN Ya-ping; Professor; PhD; Tel: +86-13851729402; E-mail: ypgchen@sina.com