
ARTICLE
J. Cent. South Univ. (2019) 26: 1503-1509
DOI: https://doi.org/10.1007/s11771-019-4106-7
Hierarchical TiO2 nanorods with a highly active surface for photocatalytic CO2 reduction
CAO Mao-qi(曹茂启)1, 2, LIU Kang(刘康)1, ZHOU Hui-min(周惠敏)1, LI Hong-mei(李红梅)1,
GAO Xiao-hui(高小惠)1, QIU Xiao-qing(邱晓清)3, 4, LIU Min(刘敏)1, 5
1. Institute of Super-Microstructure and Ultrafast Process in Advanced Materials,
School of Physics and Electronics, Central South University, Changsha 410083, China;
2. School of Chemistry and Chemical Engineering, Qiannan Normal University for Nationalities,Duyun 558000, China;
3. School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China;
4. School of Materials Science and Energy Engineering, Foshan University, Foshan 528000, China;
5. State Key Laboratory of Powder Metallurgy, Central South University, Changsha 410083, China
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2019
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2019
Abstract: Photocatalytic carbon dioxide reduction reaction (CO2RR) has been considered as one of most effective ways to solve the current energy crisis and environmental problems. However, the practical application of photocatalytic CO2RR is largely hindered by lock of efficient catalyst. Here, hierarchical titanium dioxide (TiO2) nanostructures with a highly active {001} surface were successfully synthesized by a facile approach from metal Ti powders. The obtained hierarchical TiO2 nanostructures were composed of TiO2 nanorods, which have a diameter about 5–10 nm and a length of several micrometers. It is found that these nanorods have exposed {001} facets. On the other hand, these hierarchical TiO2 nanostructures have a good light-harvesting efficiency with the help of TiO2 nanorods component and large specific surface area. Therefore, these hierarchical TiO2 nanostructures exhibit a much better activity for photocatalytic CO2 reduction than that of commercial TiO2 (P25). This high activity can be attributed to the synergistic effects of active surface, efficient charge transfer along nanorods and good light harvesting in the nanorod-hierarchical nanostructures.
Key words: TiO2; {001} facets; hierarchical nanorods; CO2 photoreduction
Cite this article as: CAO Mao-qi, LIU Kang, ZHOU Hui-min, LI Hong-mei, GAO Xiao-hui, QIU Xiao-qing, LIU Min. Hierarchical TiO2 nanorods with a highly active surface for photocatalytic CO2 reduction [J]. Journal of Central South University, 2019, 26(6): 1503-1509. DOI: https://doi.org/10.1007/s11771-019-4106-7.
1 Introduction
The rapid combustion of fossil fuels caused a sharp increase of carbon dioxide (CO2) emission into the atmosphere, resulting in problems of global warming, climatic change and future energy shortage [1–6]. The conversion of greenhouse gas CO2 to high value added fuels of methane, formic acid and methanol, using solar energy triggered photocatalytic process, is considered a desirable way to help address these problems [1, 6]. The best- known semiconductor of titanium dioxide (TiO2) has attracted a lot of attention in photocatalysis with its excellent stability, nontoxicity and low price [7, 8]. It has been proved that the crystal structure, morphology, size and active surface of TiO2-based catalyst have great influences on its performance [7, 8].
Generally, CO2 photoreduction is a process with multi-steps including both activation and dissociation of CO2. The activation of CO2 on a given photocatalyst is the initial step and also the key step for CO2 photoreduction [6]. The surface of photocatalysts is one of the most important role in the reactions of photocatalytic because of their active sites for reactant adsorption and more reductive electrons for reactions [7, 8]. Experimental studies and theoretical calculations have demonstrated that anatase {001} facets are more active than {101} facets [9–13]. Therefore, TiO2 with exposed {001} facets should have high chemical activity, which is beneficial to improve their photocatalytic efficiency. However, the order of the average surface energies is γ{001}(0.90 J/m2)>γ{100} (0.53 J/m2)>γ{101} (0.44 J/m2 [9, 10]). According to common sense, the high surface energy facets diminish quickly in the process of crystal growth to minimization its surface energy. Therefore, the exposed {001} facets of TiO2 crystals are rarely obtained [11–13]. Moreover, most anatase TiO2 particles with {001} facets reported by now, have a typical truncated tetragonal bipyramid ship [11–13]. The {001} facets of anatase TiO2 nanorods with higher-surface-energy have not been reported in the experiment.
Here, a facile and novel method to obtain hierarchical TiO2 nanorods by a hydrothermal process is reported in this work. After hydrothermal reaction and wash process, the flower-like titanate nanostructures were gained. The obtained titanate nanostructures have a number of titanate nanoribbons. The corresponding anatase TiO2 nanorods without destroying the 3D hierarchical structure were obtained from high temperature heat treatment of the flower-like titanate nanostructures. Interestingly, after heat treatment, the titanate nanoribbons were broken into many TiO2 nanorods. It was found that the nanorods have exposed {001} facets. Moreover, these hierarchical TiO2 nanorods have a higher surface area and a better light adsorption efficiency. Thus, they exhibit enhanced photocatalytic activity for the CO2 photoreduction.
2 Experimental
The flower-like titanate nanostructures were obtained with the method of hydrothermal synthesis. In a typical synthesis, 0.1 g Ti powder was dispersed into 150 mL 9 mol/L H2O2/0.1 mol/L HNO3 aqueous solution and kept for 2 h in an oven maintained at 80 °C. An orange-yellow transparent titanium peroxo solution was obtained [14]. 12 mL of the above room temperature solution was mixed with 28 mL 40 wt% NaOH solution. The mixed solution was then placed in a Teflon-lined autoclave which was kept at 150 °C for 5 h. At last, the product was collected by centrifugation and thoroughly washed with high purity water (18 MΩ) and 0.1 mol/L HNO3 aqueous solution until the pH was about 7.0. The obtained product was dried at 80 °C over night in air. Corresponding hierarchical anatase TiO2 nanorods were synthesized by annealing the flower-like titanate nanostructures at 500 °C in air for 2 h (Figure 1).

Figure 1 Schematic illustration of fabrication process for flower-like titanate nanostructures and hierarchical TiO2 nanorods, obtained from Ti powders:
The crystal structures of the products were obtained by X-ray diffraction with Cu Ka as radiation (λ=1.54056  ). Scanning electron microscope (SEM) images were obtained by a field-emission SEM. Transmission electron microscopy (TEM) images were obtained by an FEI F-20 high-resolution TEM (HRTEM) at an accelerating voltage of 200 kV. The Brunauer– Emmett–Teller (BET) surface area and pore size distribution profiles were obtained from a Quantachrome Autosorb-1 B apparatus. About 100 mg of a sample was utilized for this purpose, which was degassed under vacuum (10–3 Pa) at 200 °C, prior to N2 adsorption.
). Scanning electron microscope (SEM) images were obtained by a field-emission SEM. Transmission electron microscopy (TEM) images were obtained by an FEI F-20 high-resolution TEM (HRTEM) at an accelerating voltage of 200 kV. The Brunauer– Emmett–Teller (BET) surface area and pore size distribution profiles were obtained from a Quantachrome Autosorb-1 B apparatus. About 100 mg of a sample was utilized for this purpose, which was degassed under vacuum (10–3 Pa) at 200 °C, prior to N2 adsorption.
The experiment of photocatalytic reduction CO2 was performed in a 220 mL capacity homemade capacity at room temperature. A thin film at the bottom of the reactor was obtained with 0.1 g sample suspended in 20 mL deionized water after ultra-sonicated and dried in an oven at 60 °C. The air in the reactor was removed by blowing ultra-pure nitrogen gas for 30 min resulting in the reaction system into an anaerobic condition. The necessary reactant of CO2 and H2O was produced in situ by a chemical reaction from 0.3 mL HCl with the concentration of 4 mol/L and 0.12 g NaHCO3. Then, the reactor was irradiated for 2 h with a Xe lamp as solar simulator. At the end of CO2 reduction reaction, 1 mL gas in the rector was taken out and analyzed by a gas chromatograph. The amount of methanol produced after the reaction was quantified as a major liquid product in the reaction.
3 Results and discussion
Figure 1 shows the schematic illustration of the fabrication process for the flower-like titanate nanostructures and the hierarchical TiO2 nanorods from Ti powders. First, Ti powders were dissolved in H2O2/HNO3. Then, the obtained solution hydrothermally reacted with NaOH solution to form flower-like titanate nanostructures, which were composed of many titanate nanoribbons. After high temperature treatment, the flower-like titanate nanostructures were transformed into their corresponding hierarchical TiO2 nanorods without destroying the 3D hierarchical structure. These hierarchical TiO2 nanorods were composed of numerous TiO2 nanorods, which have exposed {001} facets.
Figure 2(a) shows the XRD results of the obtained hierarchical flower-like titanate nanostructures and their corresponding hierarchical TiO2 nanostructures. For hierarchical titanate nanostructures, the observed 2θ values in the XRD at 9.1°, 24.6°, 28.0°, 48.3° and 62.2° can be attributed to (020), (110), (130), (200) and (002) faces of H2Ti3O7, respectively [15]. This XRD result suggests that the obtained hierarchical titanate nanostructure is a layered titanate [15]. After heating treatment, all the primary peaks are in good agreement with anatase TiO2 (JCPDS card No. 21-1272). The result and component are further confirmed by energy dispersive X-ray spectroscopy (EDS, Figure 2(b)).
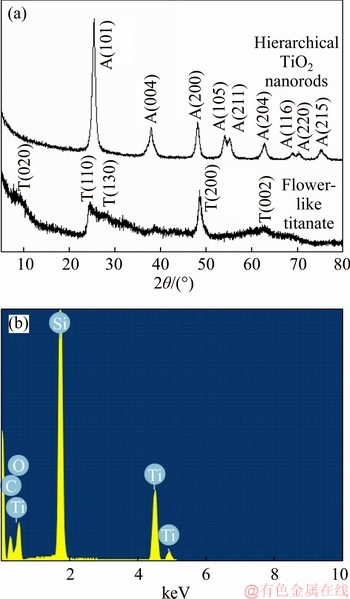
Figure 2 XRD patterns of flower-like titanate and hierarchical TiO2 nanorods (T—H2Ti3O7; A—anatase) (a) and EDS of hierarchical TiO2 nanorods (b)
Typical FE-SEM images of the hierarchical titanate and TiO2 nanostrucutres are shown in Figure 3. The as-prepared titanate products contain numerous flower-like titanate nanostructures. These flower-like titanate nanostructures have a diameter of 3–5 μm, and are composed of many thin titanate nanoribbons (Figures 3(a) and (b)). The TiO2 nanoribbons are 0.5–2 μm in length, about 100– 300 nm in width and several nanometers in thickness. Figures 3(c) and (d) show the FE-SEM images of the corresponding hierarchical TiO2 nanostructures. The high-temperature annealing process does not destroy the 3D hierarchical structure. However, the titanate nanoribbons are broken into many small TiO2 nanorods (inset of Figure 3(d)). It can be seen that these TiO2 nanorods have a diameter of 5–10 nm and a length of several micrometers.
Figure 4 shows the TEM images of the hierarchical titanate nanostructures. As shown in Figures 4(a) and (b), there are many thin nanoribbons in the hierarchical titanate nanostructures. The observed spacing between the lattice planes for titanate nanoribbons is about 0.36 nm, corresponding to d110 of H2Ti3O7 (HRTEM image of Figure 3(c) [15]). In fact, the two rings indexed to the (110) and (200) diffraction planes of H2Ti3O7 are shown in the inset of Figure 4(a). Thus, it can be confirmed that the obtained hierarchical titanate nanostructures are layered H2Ti3O7. These layered titanate nanoribbons are quite thin and easily broken into TiO2 nanrods after annealing. EDS analysis shows that the flower-like titanate nanostructures are chemically composed of Ti and O elements (note that H is not detectable with EDS, C and Cu elements are originated from TEM grid, Figure 4(d), proving the complete ionic exchange from Na+ to H+.

Figure 3 FE-SEM images:(Inset shows FE-SEM image of constituent TiO2 nanorods)

Figure 4 Typical TEM image and SAED pattern of flower-like titanate nanostructures (a), TEM image of titanate nanoribbons (b), HRTEM of a single titanate nanoribbon (c) and EDS spectra of flower-like titanate nanostructures (d)
Figure 5 shows the TEM images of the hierarchical TiO2 nanostructures. As shown in Figure 5(a), the TEM image of the hierarchical TiO2 nanorods is similar to that of the hierarchical titanate nanostructures. It is proved that the TiO2 product maintained the 3D hierarchical structure.However, as shown in Figure 5(b), it clearly shows that the hierarchical TiO2 nanostructures are composed of a large number of TiO2 nanorods. The diameter of the nanorods is 5–10 nm. The result indicates that the nanorods originate from nanoribons. The possible reason is that the nanoribbons are too thin to withstand the high temperature treatment and broken into nanorods during the high temperature treatment. Figure 5(c) shows the HRTEM image of a single TiO2 nanorod. The lattice spacing of 0.35 nm corresponds to the {101} planes, while the lattice spacing of 0.47 corresponds to the {002} planes [12, 13]. The angle labeled in the inset corresponding fast-Fourier transform (FFT) image is 68.3°, which is identical to the theoretical value for the angle between the {101} and {001} facets in an anatase crystal [12, 13]. The results indicate that the nanorods exposed with {001} facets.
Next, we measured the surface area. A type IV curve with a hysteresis loop of the hierarchical TiO2 nanostructure was obtained by nitrogen adsorption- desorption isotherm (Figure 6). The BET surface area of the hierarchical TiO2 nanorods is 186.8 m2/g, which is much larger than that of the P25 TiO2 nanoparticles, 51.3 m2/g. The pore volume of the hierarchical TiO2 nanostructures is 0.47 cm3/g, which is also much larger than that of the P25 TiO2 nanoparticles, 0.08 cm3/g. The significant increase of the pore volume can be attributed to the formation of highly porous nanostructures. Table 1 shows the BET surface areas and the pore volumes of the hierarchical TiO2 nanorods and P25 TiO2 nanoparticles.

Figure 5 TEM images of hierarchical TiO2 nanorods (a, b) and HRTEM image of an individual nanorod (c) (Inset shows corresponding fast-Fourier transform pattern)
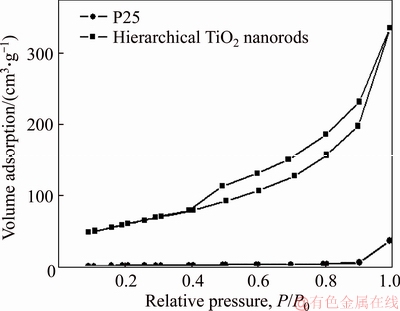
Figure 6 Nitrogen adsorption/desorption isotherms of hierarchical TiO2 nanorods and P25
Table 1 BET surface areas (SBET) and pore volumes (V) of hierarchical TiO2 nanorods and P25

The UV–Vis absorption spectra of the hierarchical TiO2 nanostructures and P25 TiO2 nanoparticles are shown in Figure 7. The absorption of the hierarchical TiO2 nanostructures is stronger than that of P25. One of the most possible reasons is the highly porous structure of hierarchical TiO2 nanostructures resulting in the harvesting of light [16, 17].

Figure 7 UV-Vis spectra of hierarchical TiO2 nanorods and P25
The photocatalytic CO2 reduction activity on the hierarchical TiO2 nanostructures was evaluated under UV light irradiation. No hydrocarbon product was detected in the absence of either photocatalyst or light irradiation, revealing that the hydrocarbon products were produced by a photocatalytic reaction on the photocatalyst. Figure 8 represents a comparison of photocatalytic methanol and methane production rates by hierarchical TiO2 nanostructures and P25 as photocatalysts. Hierarchical TiO2 nanostructures exhibited obvious photocatalytic activity for methanol and methane generation. The rates for methanol and methane are 2.8 and 3.6 μmol/(g·h), respectively. These results are comparable with the previous reported results [1, 3]. As the most active commercial photocatalyst, P25 only show a very low methanol and methane generation rate of 0.6 and 0.8 μmol/(g·h), respectively. It can be seen that the activity of hierarchical TiO2 nanostructures is around 5 times compared with that of P25. The high photocatalytic activity of hierarchical TiO2 nanostructures was attributed to the exposed {001} faces, which offer active sites for CO2 activation. The hierarchical structure enhanced the light scattering and adsorption, providing more sites for reactions. Further, the nanorods benefit the charge transfer along the rod direction, which reduced the electron-hole recombination.

Figure 8 Comparison of CH3OH and CH4 production rates over hierarchical TiO2 nanorods and P25 under light irradiation for 2 h
4 Conclusions
A new process for the fabrication of hierarchical TiO2 nanorods is reported in this work. The as synthesized flower-like titanate nanostructures consisted of many untrathin nanorribons and can be easily transformed into hierarchical TiO2 nanostructures without destroying their 3D hierarchical structures by a moderate annealing process. Interestingly, after heat treatment, the thin nanoribbons are transformed into many well crystallinity nanorods with exposed {001} facets. Due to their active facets, these hierarchical TiO2 nanorods show 5 times of CO2 photoreduction performance compared with P25. This approach is also the reference in the synthesis of other materials with special hierarchical nanostructures.
References
[1] LINSEBIGLER A L, LU G Q, YATES J T. Photocatalysis on TiO2 surfaces: Principles, mechanisms, and selected results [J]. Chem Rev, 1995, 95: 735-758.
[2] HE H N, GAN Q M, WANG H Y, XU G L, ZHANG X Y, HUANG D, FU F, TANG Y G, AMINE K, SHAO M H. Structure-dependent performance of TiO2/C as anode material for Na-ion batteries [J]. Nano Energy, 2018, 44: 217-227.
[3] ZHANG Q, HE H N, HUANG X B, YAN J, TANG Y G, WANG H Y. TiO2@C nanosheets with highly exposed (001) facets as a high-capacity anode for Na-ion batteries [J]. Chem Eng J, 2018, 332: 57-65.
[4] XUE X, SUN D, ZENG X G, HUANG X B, ZHANG H H, TANG Y G, WANG H Y. Two-step carbon modification of NaTi2(PO4)3 with improved sodium storage performance for Na-ion batteries [J]. Journal of Central South University, 2018, 25(10): 2320-2331.
[5] LIU A Q, LIU K, ZHOU H M, LI H M, QIU X Q, YANG Y, LIU M. Solution evaporation processed high quality perovskite films [J]. Sci Bull, 2018, 63: 1591-1596.
[6] CENTI G, PERATHONER S. Opportunities and prospects in the chemical recycling of carbon dioxide to fuels [J]. Catal Today, 2009, 148: 191-205.
[7] PAN J, LIU G, LU G Q, CHENG H M. On the true Photoreactivity Order of {001}, {010}, and {101} facets of anatase TiO2 crystals [J]. Angew Chem Int Ed, 2011, 50: 2133-2137.
[8] ROY N, SOHN Y, PRADHAN D. Synergy of low-energy {101} and high-energy {001} TiO2 crystal facets for enhanced photocatalysis [J]. ACS Nano, 2013, 7: 2532-2540.
[9] LAZZERI M, VITTADINI A, SELLONI A. Erratum: Structure and energetics of stoichiometric TiO2 anatase surfaces [Phys. Rev. B 63, 155409 (2001)] [J]. Phys Rev B, 2002, 65: 119901.
[10] DIEBOLD U. The surface science of titanium dioxide [J]. Surf Sci Rep, 2003, 48: 53-229.
[11] SELLONI A. Fluorine-containing species can cause titania to crystallize with an unusually large fraction of reactive {001} facets [J]. Nat Mater, 2008, 7: 613-615.
[12] YANG H G, SUN C H, QIAO S Z, ZOU J, LIU G, SMITH S C, CHENG H M, LU G Q. Anatase TiO2 single crystals with a large percentage of reactive facets [J]. Nature, 2008, 453: 638-641.
[13] LIU M, PIAO L Y, LU W M, JU S T, ZHAO L, ZHOU C L, YAN Z J, WANG W J. Anatase TiO2 single crystals with exposed {001} and {110} facets facile synthesis and enhanced photocatalysis [J]. Chem Coumm, 2010, 46: 1664-1666.
[14] KAKIHANA M, TADA M, SHIRO M, PETRYKIN V, OSADA M, NAKAMURA Y. Structure and Stability of water soluble (NH4)8[Ti4(C6H4O7)4(O2)4]8H2O [J]. Inorg Chem, 2001, 40: 891-894.
[15] MAO Y B, WONG S S. Size- and shape-dependent transformation of nanosized titanate into analogous anatase titania nanostructures [J]. J Am Chem Soc, 2006, 128: 8217-8226.
[16] AO Y, FU D, YUAN C. A simple method for the preparation of titania hollow sphere [J]. Catal Commun, 2008, 9: 2574-2577.
[17] YU J G, LIU S W, YU H G. Microstructures and photoactivity of mesoporous anatase hollow microspheres fabricated by fluoride-mediated self-transformation [J]. J Catal, 2007, 249: 59-66.
(Edited by FANG Jing-hua)
中文导读
具有高活性晶面的分级二氧化钛纳米棒光催化二氧化碳还原
摘要:光催化二氧化碳还原被认为是能够同时解决能源和环境问题的最有效方式之一。但高效的二氧化碳还原催化剂的缺乏限制其实际应用。在本文中,我们成功合成了具有{001}高活性晶面的分级二氧化钛纳米棒结构,纳米棒的直径为5-10 nm,长度为几个微米。分级纳米棒结构使得其具有更大的比表面积,进而大大促进了光吸收。具有有效电荷传输、更大比表面积及更强光吸收的分级结构二氧化钛纳米棒与商业的P25二氧化钛相比具有更强的二氧化碳光催化还原性能。
关键词:二氧化钛;{001}晶面;分级纳米棒;二氧化碳光还原
Foundation item: Project(21872174) supported by the National Natural Science Foundation of China; Projects(2017CX003, 20180018050001) supported by the Innovation-Driven Plan in Central South University, China;Project supported by State Key Laboratory of Powder Metallurgy in Central South University, China; Project(JCYJ20180307151313532) supported by Shenzhen Science and Technology Innovation Project, China; Project supported by the Thousand Youth Talents Plan of China; Project supported by the Hundred Youth Talents Program of Hunan, China
Received date: 2018-11-01; Accepted date: 2019-02-25
Corresponding authors: LI Hong-mei, Lecturer; Tel: +86-13787202527; E-mail: hongmeili@csu.edu.cn; QIU Xiao-qing, PhD, Professor; Tel: +86-13860639566; E-mail: xq-qiu@csu.edn.cn