Article ID: 1003-6326(2005)06-1297-06
High temperature brazing of diamond tools
YAO Zheng-jun(姚正军)1, SU Hong-hua(苏宏华)1,
FU Yu-can(傅玉灿)2, XU Hong-jun(徐鸿钧)2
(1. College of Materials Science and Technology,
Nanjing University of Aeronautics and Astronautics, Nanjing 210016, China;
(2. College of Mechanical and Electrical Engineering,
Nanjing University of Aeronautics and Astronautics, Nanjing 210016, China)
Abstract: A new brazing technique of diamond was developed. Using this new technique optimum chemical and metallurgical bonding between the diamond grits and the carbon steel can be achieved without any thermal damages to diamond grits. The results of microanalysis and X-ray diffraction analysis reveal that a carbide layer exists between the diamond and the matrix, which consists of Cr3C2, Cr7C3 and Cr23C6. Performance tests show that the brazed diamond core-drill has excellent machining performance. In comparison with traditional electroplated diamond core-drill, the brazed diamond core-drill manufactured using the new developed technique has much higher machining efficiency and much longer operating life.
Key words: high temperature brazing; microanalysis; diamond tool; machining test CLC
number: TG74; TG454 Document code: A
1 INTRODUCTION
The demand for superhard and precision machining and grinding tools has been increased because of strict control of machining tolerances for advanced and superalloy components in addition to their application in cutting and grinding of stones, concrete structures or composite materials. Previous work[1-5] has reported the development and applications of these tools, alloys and composites which can be suitable for machining, joining of dissimilar materials, and in other fields. Diamond is a very interesting alternative tool material, owing to its outstanding properties. The hardest material known, quite unreactive with most chemical products, is also able to dissipate very efficiently the heat produced during machining operations, owing to its very high room temperature thermal conductivity. Generally, diamond grit powders are embedded in the tool surfaces by electroplating, chemical bonding and other processes, which may have limitations to achieve an adequate level of strength. Recent work[5-9] reported the progress and use of active brazing alloys based on Ag, Cu and Ni matrix to bond diamond grits on the tool surface, providing better performance as compared with electroplating or chemical bonding. However, this will depend on the wettability and interfacial bond between the active brazing alloy matrix and diamond grit particles. This aspect of microstructure and reaction mechanism has not been fully studied for the Ni based and other active brazing alloys, however being essential for development of better quality tools. This paper describes a new brazing technique of diamond using Ni based brazing alloy. The morphology of brazed diamond grits and the interface structure between diamond grits and the matrix alloy are also presented.
2 EXPERIMENTAL
A brazing technique of diamond was developed in this study. The composition of brazing alloys was Ni-10Cr-3.5Si-2.0B(mass fraction, %) and its solidus temperature was 960℃. The segments of a brazing diamond specimen were prepared by the following procedures. Initially, synthetic diamond grits with particle sizes ranging from 40 to 50 mesh were stuck on the surface of 45 carbon steel substrate by a kind of adhesive. The brazing powder was then sprinkled on the diamond layer and the thickness of the brazing powder layer was about 2mm. Brazing was carried out at 1050℃ in vacuum, with pressures ranging from 1.33×10-3 to 1.33×10-4Pa. The samples were heated at a rate of 15℃/min to 1050℃. All the specimens were held at 1050℃ for 4min then furnace-cooled to room temperature.
The structure of the interface between the diamond grits and the brazing alloy was studied by electrolyzing the metal matrix from the specimen in a special acid. The brazing alloy was removed from the brazed layer, and diamond grits were left behind. The morphology of the diamond grits was examined using scanning electron microscopy(SEM). The distribution of different elements in the brazed layer was analyzed by using an electron probe microanalyzer(KEVEX, EDS). Structural analysis of the interfacial phases between the diamond grits and the bonding matrix was carried out using X-ray diffraction(XRD).
Performance tests of two different diamond core-drills, fabricated using the technique developed in this study and the traditional electroplated processing, respectively, were examined by a series of machining tests. The machine tool used for tests was a powered vertical driller(Z5125 model). During tests, an invariable press, 176N, was applied and the cutting fluid was water. The spindle rotating speed was kept constant at 4000r/min and the cutting depth was kept at a constant level (10mm). The machining object which presents in this paper was granite, and its thickness and hardness were 20mm and 7-8, respectively.
3 RESULTS AND DISCUSSION
3.1 Morphological observation and structural analysis
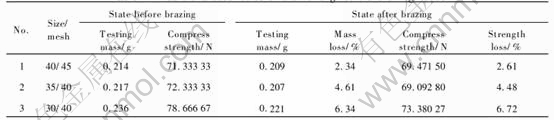
Using the optimum brazing technology that we have been developed, a favorable chemical and metallurgical bonding between the diamond grits and the carbon steel matrix can be achieved. Fig.1 shows the SEM micrograph of diamond grits brazed on the carbon steel matrix. The morphology of the single diamond grit and the bonding matrix are clearly shown in Fig.2, from which it can be obviously seen that the bonding between the diamond and the matrix is very good. In order to exhibit the brazing effect, the compress strength and mass loss of the diamond grits were measured, and the results are listed in Table1. From Table 1, we can see both the strength and the mass loss of the diamond grits via vacuum furnace brazing are very little, and the thermal damage of diamond can also be ignored(Fig.3).

Fig.1 SEM micrograph showing diamond grits and matrix alloy

Fig.2 SEM micrograph showing bonding between diamond grit and matrix alloy
Pull-out of diamond grits usually predominates as a cause for tool failure. This phenomenon frequently occurs in the performances of all sorts of diamond tools used widely at present[4, 8, 10]. As the interface between diamond grits and metal matrix withstands the moment strong impact force to which the diamond grits are subjected, a strong interfacial cohesion arising from an interfacial reaction is the basic requirement for enhancing tool life. In order to create the chemical metallurgical bonding between diamond grits and metal matrix, the addition of a low concentration of an active ele- ment, such as chromium, titanium, or vanadium,
Table 1 Basic characteristics of diamond grits via brazing route
is necessary[11]. In this study, about 10%Cr(mass fraction) is added to Ni-based brazing alloy. The previous investigations[12, 13] suggested that the catalytic conversion of diamond into graphite (or carbon) can take place in the presence of elements such as Fe, Ni, and Co. However no graphite/carbon layer was observed between diamond grits and the matrix alloy in the present investigation. This is probably due to the lower temperature and shorter holding period for brazing in the present work.
The interface layer between the diamond grits and the matrix alloy has been revealed by SEM observation, as shown in Fig.4, in which the profiles of C, Si, Cr, Ni distribution in the layer are also shown. The high Cr concentration in the interface layer implies that the interface layer mainly consists of Cr carbides.
Fig.5 shows the surface morphology of a diamond grit, from which Ni-based metallic constituents was leached away by electrolysis process. It seems that the diamond grit is covered by a network layer. Compositional analysis using EDS indicates that this layer is rich in Cr and C. The result of XRD analysis shown in Fig.6 reveals that this layer consists of Cr3C2, Cr7C3 and Cr23C6 phases, and the quantity of Cr23C6 phase is very little. More enlarged SEM images, shown in Figs.7, 8 and 9, exhibit that the carbide in the outer layer of this network shows rod-shaped morphology (Fig.8) and the carbide in the inside layer has an irregular plate-shaped morphology. Microanalysis by EDS indicates that the composition of rod-shaped phase is close to that of Cr7C3 and the ratio of Cr/C in the plate-shaped phase is close to that of Cr3C2.
Several investigations[11, 14-16] have proposed that there are two kinds of prominent effects on the diamond high temperature brazing tools with the addition of an active element in brazing alloy. Firstly, these active elements form transitiona
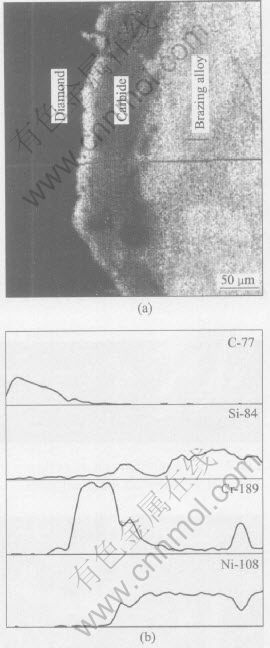
Fig.4 SEM micrograph showing interface layer between diamond grit and matrix alloy(a), and profiles showing distributions of C, Si, Cr and Ni in interface layer(b)
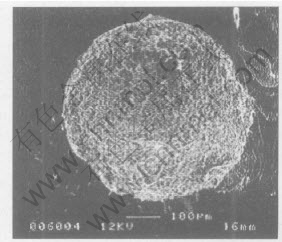
Fig.5 SEM micrograph showing carbide layer covering diamond grit
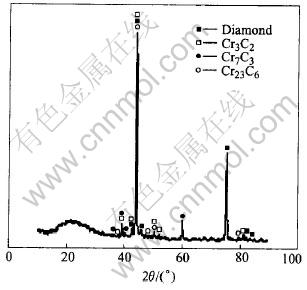
Fig.6 XRD pattern taken from brazing diamond

Fig.7 SEM micrograph of carbides in surface layer on diamond grits
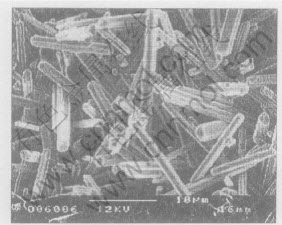
Fig.8 SEM micrograph showing outer layer carbide
carbide layers between the diamond grits and the metal matrix. The carbide layer behaves as a diffusing barrier to avoid excessive reaction of the matrix with the diamond grits, and a buffer zone to reduce thermal stress between diamond grits and matrix. Secondly, the chemical and metallurgical

Fig.9 SEM micrograph showing inside layer carbide
bonding between the diamond grits and the metal matrix can develop. All of these can make the diamond tools have a higher cutting efficiency and a longer cutting life[16-18]. The results of the present work are well consistent with the previous investigations.
3.2 Drilling performance contrast
The machining tests of two different diamond core-drills, fabricated using the technique developed in the study and the traditional electroplated processing, respectively, have been carried out through marbles, granites, ceramics, glass and armoring composite, and the results are summarized in Table 2. It can be seen that the mean drilling time of single hole for electroplated and brazed core-drill is 178s and 27s, respectively, indicating that the work efficiency of brazed diamond core-drill is about five times higher than that of electroplated one. Furthermore, the total number of drilled holes with electroplated and brazed core-drill is 7 and 151, respectively, which means the operating life of a brazed core-drill can be 21.6 times of that of electroplated one.
Fig.10 and Fig.11 show the surface morphologies of a brazed diamond core-drill before and after machining, respectively. After drilling 145 holes, the surface of the drill does not have many changes. From the surface of the drill no many diamond grits are pulled out. The failure type of this kind of drill is normal abrasion of diamond grits.
In comparison with Fig.10 and Fig.11, Figs.12 and 13 show the surface morphologies of an electroplated core-drill before and after machining.
Table 2 Results of drilling performance of brazed and electroplated core-drills

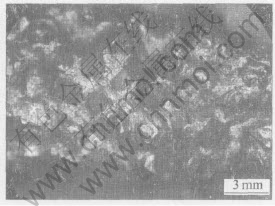
Fig.10 Optical micrograph showing surface of brazed diamond core-drill before performance test

Fig.11 Optical micrograph showing surface of brazed diamond core-drill after performance test
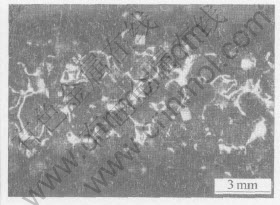
Fig.12 Optical micrograph showing surface of electroplated diamond core-drill before performance test

Fig.13 Optical micrograph showing surface of electroplated diamond core-drill after performance test
It can be obviously seen that the drill is damaged seriously after 5 holes drilling, and the failure type of the drill is typical pullout of diamond grits.
4 CONCLUSIONS
1) Using the brazing technique developed in the present work, the optimum chemical and metallurgical bonding between the diamond grits and the carbon steel can be achieved without any thermal damages to diamond grits.
2) The results of microanalysis and X-ray diffraction analysis reveal that a carbide layer exists between the diamond and the matrix, which consists of Cr3C2, Cr7C3 and Cr23C6.
3) The results of performance tests show that the brazed diamond core-drill has excellent machining performance. In comparison with the traditional electroplated diamond core-drill, the brazed diamond core-drill manufactured using the new developed technique has much higher machining efficiency and much longer operating life.
REFERENCES
[1]Frehn F, Notter A T. Grinding hardenable steel bonded carbide alloys with diamond wheels [J]. Ind Diamond Rev, 1979, 39(6): 195-202.
[2]Meszaros M, Vadasdi K. Process and equipment for electrochemical etching of diamond-containing Co-Wc tools and recovery of diamond from used steel tools [J]. International Journal of Refractory Metals and Hard Materials, 1996, 14(4): 229-234.
[3]LIU Sha, YI Dan-qing, YU Zhi-ming, et al. Chemical pretreatments at surface of WC-6% Co for diamond coatings [J]. Trans Nonferrous Met Soc China, 2002,12(3): 396-399.
[4]Hintermann H E, Chattopadhyay A K. New generation superabrasive tool with monolayer configuration [J]. Diamond and Related Materials, 1992, 1(12): 1131-1143.
[5]Sung C M. Brazed diamond grid: a revolutionary design for diamond saws [J]. Diamond and Related Materials, 1999, 8(8-9): 1540-1543.
[6]Khalid F A, Klotz U E, Elsener H R, et al. On the interfacial nanostructure of brazed diamond grits [J]. Scripta Materialia, 2004, 50(8): 1139-1143.
[7]Wang C Y, Clausen R. Marble cutting with single point cutting tool and diamond segments [J]. International Journal of Machine Tools and Manufacture, 2002, 42(9): 1045-1054.
[8]SUN Yu-chao, LIU Yi-bo, WANG Qin-sheng. Diamond tools and Fundamentals of Metallurgy [M]. Beijing: China Building Marerials Industry Press, 1999.(in Chinese)
[9]ZHANG Qi-yun, ZHUANG Hong-shou. Manual of Brazing and Soldering [M]. Beijing: China Machine Press, 1999.(in Chinese)
[10]Dwan J D. Production of diamond impregnated cutting tools [J]. Powder Metallurgy, 1998, 41(2): 84-86.
[11]Liao Y S, Luo S Y. Effects of matrix characteristics on diamond composites [J]. Journal of Materials Science, 1993, 28(5): 1245-1251.
[12]Hsieh Y Z, Lin S T. Diamond tool bits with iron alloys as the binding matrices [J]. Materials Chemistry and Physics, 2001, 72(2): 121-125.
[13]Xiao B, Xu H J, Wu Z B. Investigation on brazing of single-layer superabrasive wheel [J]. Key Engineering Materials, 2001, 202-203: 155-158.
[14]Chen S M, Lin S T. Brazing diamond grits onto a steel substrate using copper alloys as the filler metals [J]. Journal of Materials Engineering and Performance, 1996, 5(6): 761-766.
[15]Chattopadhyay A K, Chollet L, Hintermann H E. Experimental investigation on induction brazing of diamond with Ni-Cr hardfacing alloy under argon atmosphere [J]. Journal of Materials Science, 1991, 26(18): 5093-5100.
[16]Palavra A, Fernandes A J S, Serra C, et al. Wettability studies of reactive brazing alloys on CVD diamond plates [J]. Diamond and Related Materials, 2001, 10(3-7): 775-780.
[17]Chattopadhyay A K, Chollet L, Hintermann H E. Induction brazing of diamond with Ni-Cr hardfacing alloy under argon atmosphere [J]. Surface and Coatings Technology, 1991, 45(1-3): 293-298.
[18]Huang S F, Tsai H L, Lin S T. Effects of brazing route and brazing alloy on the interfacial structure between diamond and bonding matrix [J]. Materials Chemistry and Physics, 2004, 84(2-3): 251-258.
(Edited by YUAN Sai-qian)
Foundation item: Project(50175052) supported by the National Natural Science Foundation of China
Received date: 2005-06-14; Accepted date: 2005-10-20
Correspondence: YAO Zheng-jun, Associate Professor, PhD; Tel: +86-25-84892952; E-mail: yaozj@nuaa.edu.cn