
Microstructure evolution during reheating of extruded Mg-Gd-Y-Zr alloy into semisolid state
GAO Lei(高 磊)1, 2, LIANG Song-mao(梁松茂)1, CHEN Rong-shi(陈荣石)1, HAN En-hou(韩恩厚)1
1. State Key Laboratory for Corrosion and Protection, Institute of Metal Research,
Chinese Academy of Sciences, Shenyang 110016, China;
2. Graduate School of Chinese Academy of Sciences, Beijing 100039, China
Received 13 May 2010; accepted 25 June 2010
Abstract: The microstructure evolution of an extruded Mg-8.57Gd-3.72Y-0.54Zr (mass fraction, %, GW94) alloy during reheating into the semisolid state was investigated using optical microscopy (OM), scanning electron microscopy (SEM), and energy dispersive X-ray spectroscopy (EDX). Typical semisolid microstructure with globular solid particles distributed in the liquid matrix is obtained over 600 °C. The solid content of (Gd+Y) in the primary α-Mg particles decreases with increasing the semisolid temperature. With the prolongation of isothermal holding time, the liquid fraction does not change significantly, while the grains grow up and spheroidize. Three methods used to determine the liquid fraction as a function of temperature, namely quantitative metallography on quenched microstructures, cooling curve thermal analysis, and thermodynamic calculations were further compared.
Key words: semisolid; Mg-Gd-Y alloy; partial remelting; microstructural evolution
1 Introduction
Magnesium alloys offer lightweight alternatives to conventional metallic alloys, and structural applications in the automotive and aerospace industry[1]. Semisolid metal (SSM) processing is recognized as a technology offering several potential advantages over casting and forging, such as reduction of macro-segregations and porosity[2]. However, magnesium alloys currently used in SSM processing are restricted to a few commercial alloys such as AZ91, AM50 and AM60[3-5]. A new magnesium alloy with a composition of Mg-8.57Gd-3.72Y-0.54Zr (mass fraction, %, GW94) amenable to semisolid forming has been developed by a combination of thermodynamic and experimental approaches[6]. The alloy was designed for high temperature applications and exhibited superior creep behavior, acceptable castability and excellent corrosion resistance[7].
One of the most challenging aspects in SSM processing is to determine the actual volume fraction of the liquid at the processing temperature[8]. The liquid fraction has great impact on the SSM slurry viscosity and the subsequent filling of the mold in the casting stage. Different methods are reported in the literature for measuring the liquid fraction, among which the most commonly used are quantitative metallography, thermal analysis, and application of thermodynamic data[8-10].
In this work, the microstructure evolution of an extruded GW94 alloy during reheating into the semisolid state is presented and explained by metallurgical considerations. Three methods, namely quantitative metallography, cooling curve thermal analysis, and thermodynamic calculation are used to determine the evolution of liquid fraction as a function of temperature. The discrepancies between the results obtained by different methods are further discussed.
2 Experimental
Strain-induced melt activation (SIMA) process[11] was performed to prepare the semisolid slurry. The feedstock material used in the present study was an extruded Mg-8.57Gd-3.72Y-0.54Zr (mass fraction, %, GW94) alloy. Feedstock material production started from permanent mold casting billets. These billets of 106 mm in diameter were extruded down to 29 mm in diameter at 440 °C, corresponding to an area reduction R of 13.4.
For cooling curve thermal analysis (CCA), samples of about 25 mm in diameter and 50 mm in length were cut from the cast ingots. The samples were remelted in a steel crucible in an electrical resistance furnace and cooled to room temperature in air. The temperature was monitored by thermocouples to obtain cooling curve. The liquid fraction versus temperature curves were obtained by Newton method[12].
Quenching experiments were used to ‘freeze’ the microstructure in the semisolid state and permit the evaluation of the liquid fractions using quantitative metallography. Quenching test was conducted on small cylindrical samples of 15 mm in diameter and 12 mm in height. The specimens were heated into the semisolid state in a vertical resistance furnace. A K-type thermocouple placed in a hole of 3 mm in diameter and 5 mm in depth in the center of the sample ensured accurate temperature measurement and feedback control. The same thermocouple was used in all experiments. The temperature accuracy was controlled within ±0.5 °C. Heating time to the semisolid temperature range was between 7 and 10 min. Samples were rapidly quenched into cold water after holding for 0, 5, 10, 15 and 30 min, respectively.
The quenched samples were ground, polished and then etched in a solution of 5% nitric acid in ethanol. Microstructure investigations were conducted using optical microscope (OM), scanning electron microscope (SEM), and energy dispersive X-ray spectroscope (EDX). Quantitative metallography was performed by the image analysis software ImageJ. More than three samples were used for each quenching condition to assure the reproduction of the data.
3 Results and discussion
3.1 Feedstock materials
The microstructure of the extruded feedstock material GW94 is shown in Fig.1. Dynamic recrystallization during the extrusion process has transformed the coarse, dendritic microstructure of the permanent mold casting billet into a fine and equiaxed grain structure[6]. It can be seen that the microstructure is homogeneous with some extrusion strips and the mean grain size is about 40 μm. The broken second-phase particles (i.e. the black particles in Fig.1) scatter in the matrix.
Fig.2 shows a typical cooling curve of GW94 alloy with average cooling rate of 0.64 °C/s. The first derivate of the cooling curve (dT/dt) was determined to enhance slope changes that are related to the solidification reactions for the different phases, and to facilitate the determination of the critical solidification characteristics of the alloy[12-13]. The liquidus and solidus of the GW94 alloy determined from Fig.2 are 650 °C and 540 °C, respectively. In this temperature range, quenching experiments were performed. It should be noticed that the results are slightly different from the differential scanning calorimetry (DSC) analysis[6]. This may be due to the fact that the heterogeneous nucleation of primary Mg in CCA tests shifts the liquidus temperature up[14].

Fig.1 Optical microstructure of extruded GW94 alloy
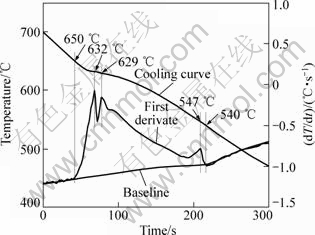
Fig.2 Representative cooling curve of GW94 alloy
3.2 Influence of temperature
Fig.3 shows the microstructure of GW94 alloy after reheating to different temperatures and 5 min isothermal holding. At 580 °C, the solidus temperature is exceeded. The primarily melted phase is mainly distributed at triple junctions and selected grain boundaries while other regions are still fully solid (Fig.3(a)). Moreover, the grain boundaries become illegible. Further remelting occurs at 590 °C (Fig.3(b)). Liquid has appeared not only along grain boundaries but also as tiny liquid pockets within the grain. A typical semisolid structure is seen after quenching from 600 °C (Fig.3(c)), i.e. globular solid particles distributed in the liquid matrix. Examples of more advanced melting at 610, 620, and 630 °C, are depicted in Figs.3(d), (e) and (f), respectively. Liquid inclusions can be found within the grains. These inclusions are unfavorable as they do not contribute to the ability of the material to flow and SSM processing has therefore to be done at increased temperature[15]. In addition, these liquid inclusions become fewer but bigger with increasing the temperature. Some pores can also be detected throughout the microstructure, which can be attributed to solidification shrinkage after quenching.
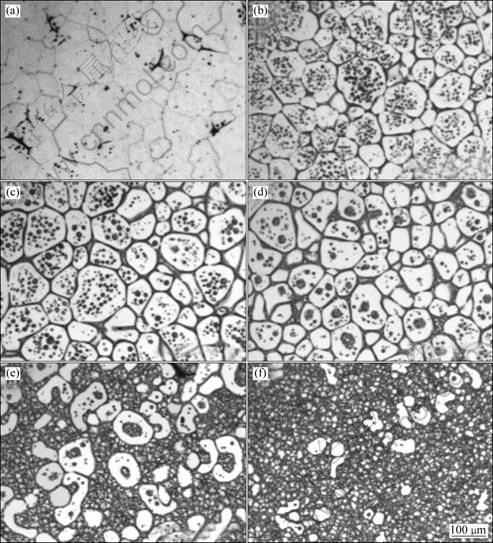
Fig.3 Microstructures of GW94 after reheating to different temperatures and 5 min isothermal holding: (a) 580 °C; (b) 590 °C; (c) 600 °C; (d) 610 °C; (e) 620 °C; (f) 630 °C
Typical SEM microstructures of the sample quenched from 610 °C and 620 °C are shown in Fig.4. It consists of liquid and primary magnesium particles. The former liquid transforms into quasi eutectics and, due to rapid solidification after immersion into water, the dendrite size is rather small. The quasi eutectics consist of small α-Mg grains interconnected by the Mg24(Y,Gd)5 phase. In addition to the change in proportion between the solids and quasi eutectics, there are also subtle differences in the internal microstructure of both constituents, particularly the unmelted α-Mg. Fig.5 shows the SEM/EDX analysis result of the solute content (Gd+Y) in the primary α-Mg particles. It can be seen that the solute content decreases with increasing the semisolid temperature, suggesting that back diffusion in the solid occurs during isothermal holding process. This diffusion process leads to the increased level of homogenization, as indicated by the error bar at each temperature.
3.3 Influence of holding time
Microstructure evolution of GW94 during isothermal holding at 610 °C is illustrated in Fig.6. The sample, which was immediately quenched after it had reached 610 °C (Fig.6(a)), shows a relatively small solid particle size with d≈71 μm. After 5 min holding time (Fig.6(b)), grains grow into a steady range (d≈111 μm) which can keep in further holding time. The solid grains grow to 129 μm for holding time of 30 min (Fig.6(e)). The driving force for coarsening in the semisolid state is the reduction in interfacial free energy[16]. Coarse microstructures tend to have poor semisolid formability and mechanical strength. However, most primary magnesium particles are likely to remain spherical during growth. Plenty of very small liquid inclusions develop in the early stage of remelting. These inclusions become fewer but bigger with increasing the holding time in the semisolid state. Quantitative metallurgy indicates that the liquid fraction does not change significantly with soaking time.

Fig.4 SEM images of GW94 after reheating to 610 °C (a) or 620 °C (b) and 5 min isothermal holding
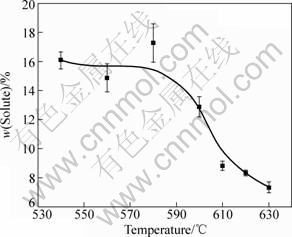
Fig.5 SEM/EDX results showing solute content of (Gd+Y) in primary α-Mg particles
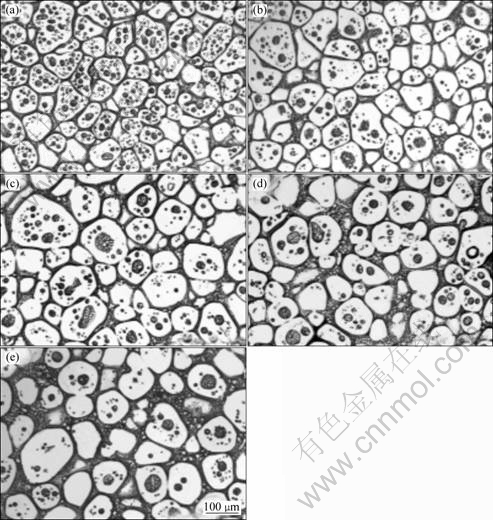
Fig.6 Microstructures of GW94 after reheating to 610 °C and isothermal holding for different time: (a) 0 min; (b) 5 min; (c) 10 min; (d) 15 min; (e) 30 min
3.4 Liquid fraction evolution
Fig.7 shows the liquid fraction measured by quantitative metallography from the quenching experiments. The error bars shown at each temperature indicate the average of the standard deviations of all the tests at a given temperature for soaking time from 0, 5, 10, 15 to 30 min. It can be seen that the volume fraction of liquid does not change significantly with soaking time, which can be further controlled on an industrial scale.
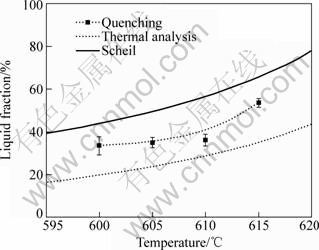
Fig.7 Evolution of liquid fraction obtained from quenching experiments, cooling curve thermal analysis, and calculated data from Scheil model
Liquid fractions were also calculated by a commercial thermodynamic software package called Pandat together with the recent thermodynamic assessment of Mg-Gd-Y system[17]. The results of the Pandat calculation using the Scheil model, as well as the cooling curve thermal analysis, are also plotted and compared with the experimental data in Fig.7. The discrepancies between the results obtained by different methods are mainly caused by variations in thermal histories[8]. It is shown that the liquid fraction obtained from the cooling curve thermal analysis falls behind that of others. This may be due to the heterogeneous nucleation of primary Mg in CCA tests[14]. Fig.7 also shows that the liquid fractions obtained from the quenching experiments are lower than the calculated data. This result is expected since the Scheil equation assumes no diffusion in the solid phase[18]. However, during the isothermal holding process, there is sufficient time for back diffusion (Fig.5) in the solid since the solid particles are in the semisolid range for several minutes before being rapidly quenched. The increased level of homogenization results in decreased liquid fraction at a given temperature[9]. Furthermore, as reported by several investigators[9-10, 16], the liquid fractions obtained from quantitative metallography of quenched samples are normally lower than the true liquid fractions present at given temperature because the quenching is not sufficiently rapid for the liquid content to be entirely ‘frozen’. During the quenching, some liquid deposits onto the solid, appearing to be ‘solid’ in the quenched microstructure[19]. The coarsening of the primary α-Mg particles during quenching process is further responsible for the liquid fraction differences[16].
4 Conclusions
1) Typical semisolid microstructure of Mg-Gd-Y-Zr alloy with globular solid particles distributed in the liquid matrix is obtained over 600 °C. With the prolongation of isothermal holding time, the liquid fraction does not change significantly, while the solid grains grow up and spheroidize.
2) The liquid inclusions become fewer but bigger with increasing the temperature and holding time in the semisolid state. Diffusion in the primary solid leads to the homogenization of the microstructure and further affects the liquid fraction.
3) Three methods, namely quantitative metallo- graphy, cooling curve thermal analysis, and thermodynamic calculation are used to determine the evolution of liquid fraction as a function of temperature. The discrepancies among the results obtained by different methods are mainly caused by variations in thermal histories and by coarsening of the primary α-Mg particles during the quenching process.
References
[1] MORDIKE B L, EBERT T. Magnesium properties-applications- potential [J]. Mater Sci Eng A, 2001, 302: 37-45.
[2] FAN Z. Semisolid metal processing [J]. Int Mater Rev, 2002, 47: 49-85.
[3] CZERWINSKI F, ZIELINSKA-LIPIEC A, PINET P J, OVERBEEKE J. Correlating the microstructure and tensile properties of a thixomolded AZ91D magnesium alloy [J]. Acta Mater, 2001, 49: 1225-1235.
[4] BLANDIN J J, GIUNCHI D, SUERY M, EVANGELISTA E. Effect of thermal treatments on mechanical behaviour of thixoformed magnesium alloy [J]. Mater Sci Technol, 2002, 18: 333-340.
[5] KLEINER S, BEFFORT O, WAHLEN A, UGGOWITZER P J. Microstructure and mechanical properties of squeeze cast and semi-solid cast Mg-Al alloys [J]. J Light Met, 2002, 2: 277-280.
[6] GAO L, CHEN R S, HAN E H. Thixoformability of a Mg-Gd-Y magnesium alloy and its mechanical properties [J/OL]. doi: 10.1179/026708309X12578491814834.
[7] GAO L, CHEN R S, HAN E H. Microstructure and strengthening mechanisms of a cast Mg-1.48Gd-1.13Y-0.16Zr (at.%) alloy [J]. J Mater Sci, 2009, 44: 4443-4454.
[8] NAFISI S, EMADI D, GHOMASHCHI R. Semi solid metal processing: The fraction solid dilemma [J]. Mater Sci Eng A, 2009, 507: 87-92.
[9] TZIMAS E, ZAVALIANGOS A. Evaluation of volume fraction of solid in alloys formed by semisolid processing [J]. J Mater Sci, 2000, 35: 5319-5329.
[10] WANNASIN J, CANYOOK R, BURAPA R, SIKONG L, FLEMINGS M C. Evaluation of solid fraction in a rheocast aluminum die casting alloy by a rapid quenching method [J]. Scr Mater, 2008, 59: 1091-1094.
[11] YOUNG K P, KYONKA C P, COURTOIS. Fine grained metal composition: US4415374 [P]. 1983-11-05.
[12] LIANG S M, CHEN R S, BLANDIN J J, SUERY M, HAN E H. Thermal analysis and solidification pathways of Mg-Al-Ca system alloys [J]. Mater Sci Eng A, 2008, 480: 365-372.
[13] GAO L, LIANG S M, CHEN R S, HAN E H. Correlation of recalescence with grain refinement of magnesium alloys [J]. Trans Nonferrous Met Soc China, 2008, 18: s288-s291.
[14] NAFISI S, GHOMASHCHI R. Grain refining of conventional and semi-solid A356 Al-Si alloy [J]. J Mater Process Technol, 2006, 174: 371-383.
[15] KLEINER S, BEFFORT O, UGGOWITZER P J. Microstructure evolution during reheating of an extruded Mg-Al-Zn alloy into the semisolid state [J]. Scr Mater, 2004, 51: 405-410.
[16] ATKINSON H V, LIU D. Microstructural coarsening of semi-solid aluminium alloys [J]. Mater Sci Eng A, 2008, 496: 439-446.
[17] GUO C P, DU Z M, LI C R. A thermodynamic description of the Gd-Mg-Y system [J]. CALPHAD, 2007, 31: 75-88.
[18] FLEMINGS M C. Solidification processing [M]. New York: McGraw-Hill, 1974.
[19] TZIMAS E, ZAVALIANGOS A. Evolution of near-equiaxed microstructure in the semisolid state [J]. Mater Sci Eng A, 2000, 289: 228-240.
(Edited by YANG Bing)
Foundation item: Project(2007CB613704) supported by the National Basic Research Program of China
Corresponding author: CHEN Rong-shi; Tel: +86-24-23926646; Fax: +86-24-23894149; E-mail: rongshichen@yahoo.com
DOI: 10.1016/S1003-6326(09)60343-5