
Improvement of corrosion resistance of
AZ91D magnesium alloy by gadolinium addition
ZHOU Xue-hua(��ѧ��)1, 2, WEI Zhong-ling(������)2, CHEN Qiu-rong(������)2, GAN Fu-xing(�ʸ���)1
1. School of Resource and Environmental Science, Wuhan University, Wuhan 430072, China;
2. Shanghai Institute of Microsystem and Information Technology,
Chinese Academy of Sciences, Shanghai 200050, China
Received 28 July 2006; accepted 15 September 2006
Abstract: Based on the previous investigation on beneficial introduction of holmium into magnesium alloy, the effect of gadolinium, an adjacent rare earth element, on corrosion resistance was examined. The corrosion behavior of two Mg-9Al-Gd alloys (Mg-9Al-0.45Gd and Mg-9Al-1.43Gd) was evaluated and compared with that of Mg-9Al alloy without Gd by means of specimen mass loss and hydrogen evolution in 3.5% NaCl solution saturated with Mg(OH)2. The Gd-containing alloys exhibit enhanced corrosion resistance with respect to the plain Mg-9Al alloy. The microstructures of Mg-9Al alloy and Mg-9Al-0.45 Gd alloy were observed by electron probe microanalysis (EPMA) and energy dispersion spectroscopy (EDS). The alloys with Gd addition show a microstructure characterized by �� phase solid solution, surrounded by minor amount of �� phase and more grain-like Gd-containing phase. To illustrate the involved mechanism their polarization curves were recorded. The electrochemical investigations reveal that Gd addition shifts the corrosion potential of the alloy towards active, as Gd containing phase is more active and hence less cathodic. As a result, the micro-galvanic corrosion is suppressed. Moreover corrosion product films formed on the Gd containing alloys are more compact and provide a better protective effectiveness than that on the alloy without Gd against corrosion. Repassivation measurements in mixture solution of 0.21 mol/L K2CrO4+0.6 mol/L NaCl also verify the beneficial role of Gd addition. Based on the present preliminary analysis, both the deposited Gd-containing phases and corrosion product films are believed to be responsible for the improved corrosion behaviour due to Gd addition.
Key words: AZ91D alloy; magnesium alloy; corrosion; gadolinium; rare earth
1 Introduction
Although magnesium has numerous attractive advantages such as low density, high specific strength, good machinability and excellent castability, its poor corrosion resistance limits its widespread applications [1-4]. Many researches have indicated that the addition of adequate alloying elements can enhance corrosion resistance of magnesium[5-7]. Among the alloying additions, rare earth (RE) elements are regarded as promising candidates for this purpose. As a result, a few alloys with improved performance have been developed.
The beneficial effect of RE on the corrosion resistance of Mg-Al alloys have been investigated by ROSALBINO et al[8], FAN et al[9] and ZHOU et al[10]. Alloying Mg-Al alloys with RE metals gave a significant improvement in their corrosion resistance. LUNDER and NISANCIOGLU[11] studied the electrochemical behaviour of the AE42 alloy (4% Al, 2% RE, mass fraction) by considering the role of the Al11RE3 phase which is mainly located at the grain boundaries of the Mg matrix. Based on the distribution of constituent phases the micro galvanic corrosion behavior was considered. The morphology and structure of the formed corrosion product on the alloys are another essential factors, which were investigated by NORDLIEN et al[12]. Not long ago ROSALBINO and his coworkers chose erbium as a representative of the heavy rare earths to study the corrosion behaviour of two Mg-Al-Er alloys and compare with that of the commercial AM60 alloy[8]. Obviously, most researches so far are focused on the effect of the mischmetal (a mixture of RE elements) instead of individual element.
Recently, we have remarkably improved the corrosion resistance of Mg-9Al alloy by adding elemental holmium [10]. To our knowledge, however, the effect of Gd addition has not been noticed yet, and such information is not available in literature. In addition, China is rich in RE, such study is of practical significance and will promote the enlarged application of RE resources.
2 Experimental
The actual compositions of the alloys studied were determined by Inductively Coupled Plasma �CAtomic Emission Spectrometry (ICP-AES) and the results are presented in Table 1. The specimens for electrochemical measurements were molded in epoxy resin with only one side exposed 1 cm2 as working surface. All samples were abraded and polished with 1000 grit SiC paper, degreased, washed and dried before tests. Most tests were carried out in 3.5% NaCl solution saturated with Mg(OH)2 except potential-pulse experiments in which the mixture solution of 0.21 mol/L K2CrO4+0.6 mol/L NaCl was used as oxidizing environment. During the experiments, the temperature of the test solution was maintained at 25 ��. Mass loss and hydrogen evolution were measured to evaluate the corrosion rate of the alloys. The electrochemical measurements were carried out in a three-electrode electrolytic cell containing 500 mL test solution by means of Solartron SI 1287 electrochemical interface. For potentiodynamic polari- zation curves the potential scanning at 0.33 mV/s started from -2 000 mV (vs SCE) and stopped at -1 400 mV (vs SCE). In potential-pulse experiments, the current decay was recorded after a potential pulse of 500 mV for 30 s was interrupted. The microstructure and the surface morphology of the Mg alloys were examined by EPMA-EDS analysis and field emission scanning electron microscope (FESEM), respectively.
Table 1 Chemical composition of pure gadolinium and studied magnesium alloys (mass fraction, %)
3 Results and discussion
3.1 Comparison of corrosion rates of alloys with and without Gd addition
All the specimens were characterized by general corrosion and no visible signs of localized corrosion was detected. As shown in Fig.1, on the basis of mass loss data, the average corrosion rate of the Gd-containing alloys is obviously less than that of the alloy without Gd addition, while there is no remarkable difference between the corrosion rate of Mg-9Al-0.45Gd and Mg-9Al-1.43Gd.
Alternatively, the corrosion rate of magnesium alloy was assessed by the hydrogen evolution. As shown in Fig.2, the hydrogen evolution volume of the Gd-containing alloys is significantly less than that of Mg-9Al alloy at any time. Moreover, the hydrogen evolution of Mg-9Al alloy ceaselessly increases with time, showing a steady corrosion rate. The hydrogen evolution volume of the Gd-containing alloys increases rapidly at beginning while then increase slowly, which indicates the slowdown of corrosion with time.

Fig.1 Average corrosion rates of Mg-9Al alloys with and without Gd addition in 3.5%NaCl solution saturated with Mg(OH)2 at 25 �� for 24 h

Fig.2 Accumulative hydrogen evolution volume for Mg-9Al alloys with and without Gd addition in 3.5%NaCl solution saturated with Mg(OH)2 at 25 �� as function of time
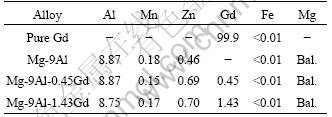
3.2 Effect of Gd addition on microstructure of Mg-9Al alloys
Mg-9Al alloy has a microstructure of Mg-based solid solution (�� phase) with discontinuous �� phase along the grain boundaries and some brighter isolated phases as illustrated in Fig.3(a). Such a microstructure is similar to that reported by SONG et al[4]. Corrosion of Mg-9Al alloy could be regarded as the consequence of micro-galvanic coupling of anodic �� phase with other cathodic phases. The microstructure of Mg-9Al-0.45Gd alloy is generally different from that of Mg-9Al alloy as shown in Fig.3(b). More grain-like new deposited phases appear and the �� phase decreases. The EDS analysis of Mg-9Al alloys with Gd addition indicates these new phases contain higher content of Gd, Al and Mn (see Table 2). Generally, the RE containing phases exhibit active potential[5]. So the Gd-containing deposited phases maybe have more active potential, which result in decrease of the micro-galvanic corrosion caused by its coupling with anodic Mg-based �� phase[13-16].
The influence of Gd addition can also be illustrated by polarization curves. As shown in Fig.4, pure Gd has higher corrosion potential and lower corrosion current density than Mg-9Al alloy. Moreover, Gd addition to Mg-9Al alloy shifts the corrosion potential towards more active. The polarization curves of the three alloys are not symmetrical, and their cathodic branch is much steeper than the anodic branch, which implies that the cathodic process of the alloys plays more important role in corrosion. The anodic branches of all the alloys are nearly similar, while the cathodic current density of the
Gd-containing alloys at the same potential are much lower than that of Mg-Al alloy without Gd addition, which shows the cathodic process of corrosion reaction is effectively suppressed by Gd addition. As discussed above, Gd addition to the magnesium alloy facilitates formation of less cathodic Gd-containing phases.
3.3 Effect of Gd addition on protection of corrosion product film
The corrosion product films of the three magnesium alloys under study after immersion in 3.5% NaCl solution saturated with Mg(OH)2 for 2 h are illustrated in Fig.5. The film of Mg-9Al alloy without Gd addition is looser and cracked (Fig.5(a)), while the films of Mg-9Al-0.45Gd (Fig.5(b)) and Mg-9Al-1.43Gd (Fig.5(c)) alloys are smoother, uniform and more compact. The corrosion product film of Mg-9Al alloys mainly consists of oxide and hydroxide of magnesium and less aluminum. They are usually loose and half-baked, but the corrosion product film of Mg-9Al-Gd alloys may be a mixture of oxide and hydroxide of magnesium and more aluminum. As expected, the compact corrosion product film would obstruct further corrosion underneath.

Fig.3 Metallographical morphologies of Mg-9Al(a) and Mg-9Al-0.45Gd(b)
Table 2 Chemical composition of constituent phases in Mg-9Al and Mg-9Al-0.45Gd alloys (mass fraction, %)
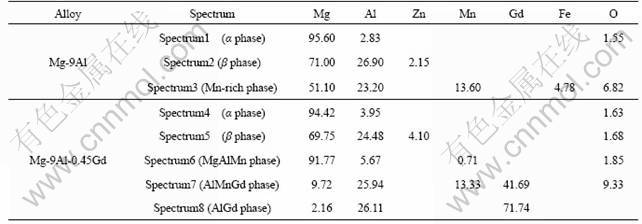

Fig.4 Polarization curves for pure Gd and Mg-9Al alloys with and without Gd addition after immersion in 3.5%NaCl solution saturated with Mg(OH)2 at 25 �� for 15 min
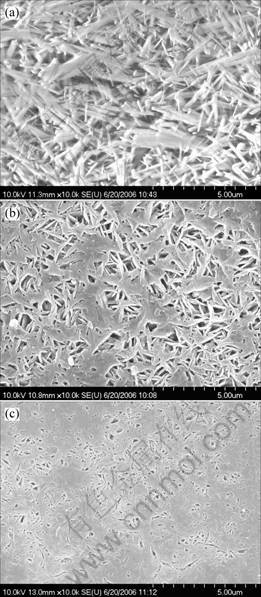
Fig.5 Surface morphologies of three alloys with and without Gd addition after immersion in 3.5% NaCl solution saturated with Mg(OH)2 for 2 h: (a) Mg-9Al; (b) Mg-9Al-0.45Gd; (c) Mg-9Al-1.43Gd
Besides, in this study repassivation process of the Mg-9Al alloys was compared by measuring current transients after initiating breakdown of the passive film, which is accomplished by applying an anodic potential pulse in 0.21 mol/L K2CrO4+0.6 mol/L NaCl solution. As illustrated in Fig.6, after an anodic potential pulse, the current fluctuates and descends slowly. The periodic current resurgence along with the longer decay time indicates the greater difficulty of its repassivation. Under the same condition, the current of the Gd-containing alloys decreases more rapidly and reaches a smaller and steady value. It suggests that Gd-addition can enhance the repassivation of Mg-9Al alloys.
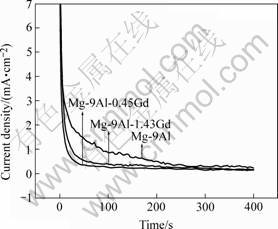
Fig.6 Repassivation behavior of Mg-9Al alloys with and without Gd addition in 0.21 mol/L K2CrO4+0.6 mol/L NaCl solution at their respective open-circuit potentials after anodic potential pulse
4 Conclusions
1) 0.45% or 1.43% Gd addition to Mg-9Al alloy can significantly improve its corrosion resistance.
2) Gd addition can optimize the microstructure of Mg-9Al alloys. Metallographically Mg-9Al-Gd alloys are composed of ��, �� phases as well as Gd-containing intermetallic phases. The latter generally contain Gd, Al, Mg and Mn etc and are easily oxidized and passivated. Furthermore, the Gd addition reduces the fraction of �� phase in alloys. As a result, the cathodic role of the deposited phases is drastically weakened and the micro-galvanic corrosion is obviously suppressed.
3) The corrosion product films on Gd-containing alloys�� surface are more compact and complete than that on the surface of Mg-9Al alloy without Gd addition. They are more protective.
Acknowledgements
The authors would like to thank Prof. XU Nai-xin, Prof. HUANG Yuan-wei, Prof. ZHANG Chen-dian, Mr. CHEN Kai-sheng, Mr. YUAN Feng, Mrs. DING Cui-hong for their help during the investigation. The authors are also indebted to the Corrosion Research Center of Shanghai Institute of Microsystem and Information Technology, Chinese Academy of Sciences for the experimental facilities and financial support.
References
[1] LINDSTR?M R, JOHANSSON L G, THOMPSON G E, SKELDON P, SVENSSON J E. Corrosion of magnesium in humid air [J]. Corrosion Science, 2004, 46: 1141-1158.
[2] SONG G, ATRENS A, STJOHN D, NAIRN J, LI Y. The electrochemical corrosion of pure magnesium in 1N NaCl [J]. Corrosion Science, 1997, 39: 855-875.
[3] INOUE H, SUGAHARA K, YAMAMOTO A, TSUBAKINO H. Corrosion rate of magnesium and its alloys in buffered chloride solutions [J]. Corrosion Science, 2002, 44: 603-610.
[4] SONG G, ATRENSA A, DARGUSCHA M. Influence of microstructure on the corrosion of die cast AZ91D [J]. Corrosion Science, 1999, 41: 249-273.
[5] WU Guo-hua, FAN Yu, GAO Hong-tao, ZHAI Chun-quan, ZHU Yan-ping. The effect of Ca and rare earth elements on the microstructure, mechanical properties and corrosion behavior of AZ91D[J]. Mater Sci Eng A, 2005, 408: 255-263.
[6] LUNDER O, LEIN J E, NISANCIOGLU K. Effect of Mn additions on the corrosion behaviour of mould-cast magnesium ASTM AZ91 [J]. Corrosion, 1987, 43: 291-295.
[7] SONG G, STJOHN D. The effect of zirconium grain refinement on the corrosion behaviour of magnesium-rare earth alloy MEZ [J]. Journal of Light Metals, 2002, 2: 1-16.
[8] ROSALBINO F, ANGELINI E, DE NEGRI S, SACCONE A, DELWNO S. Effect of erbium addition on the corrosion behaviour of Mg-Al alloys [J]. Intermetallics, 2005, 13: 55-60.
[9] FAN Yu, WU Guo-hua, ZHAI Chun-quan. Influence of cerium on the microstructure, mechanical properties and corrosion resistance of magnesium alloy [J]. Mater Sci Eng A, 2006, 433: 208-215.
[10] ZHOU Xue-hua, HUANG Yuan-wei, WEI Zhong-ling, CHEN Qiu-rong, GAN Fu-xing. Improvement of corrosion resistance of AZ91D magnesium alloy by holmium addition [J]. Corrosion Science, 2006, 48: 4223-4233.
[11] LUNDER O, NISANCIOGLU K. Electrochemical behavior of intermetallic phases in Mg-Al cast alloy[A]. COSTA J M, MERCER A D. Proceedings of 10th European Corrosion Congress[C]. London: The Institute of Materials, 1993.
[12] NORDLIEN J H, NISANCIOGLU K, ONO S, MASUKO N. Morphology and structure of water-formed oxides on ternary MgAl alloys[J]. J Electrochem Soc, 1997, 144: 461-466.
[13] LUNDER O, AUNE T K, NISANCIOGLU K. The role of Mg17Al12 phase in the corrosion of Mg alloy AZ91 [J]. Corrosion, 1989, 45: 741-748.
[14] SONG G, ATRENS A, WU Xian-liang, ZHANG Bo. Corrosion behaviour of AZ21, AZ501 and AZ91 in sodium chloride [J]. Corrosion Science, 1998, 40: 1769-1791.
[15] ZHANG Tao, LI Ying, WANG Fu-hui. Roles of �� phase in the corrosion process of AZ91D magnesium alloy [J]. Corrosion Science, 2006, 48: 1249-1264.
[16] MATHIEU S, RAPIN C, STEINMETZ J, STEINMETZ P. A corrosion study of the main constituent phases of AZ91 magnesium alloys [J]. Corrosion Science, 2003, 45: 2741-2755.
(Edited by LONG Huai-zhong)
Foundation item: Project(045211028) supported by Shanghai Science & Technology Development Fund, China
Corresponding author: ZHOU Xue-hua; Tel: +86-21-62511070-8966; E-mail: xuehua_zhou@163.com