Trans. Nonferrous Met. Soc. China 22(2012) 3034-3038
Influence of heat treatment on electrochemical properties of Ti1.4V0.6Ni alloy electrode containing icosahedral quasicrystalline phase
LIU Wan-qiang1,2, ZHANG Shan-shan1, WANG Li-min2
1. School of Materials Science and Engineering, Changchun University of Science and Technology, Changchun 130022, China;
2. State Key Laboratory of Rare Earth Resource Utilization, Changchun Institute of Applied Chemistry, CAS, Changchun 130022, China
Received 13 March 2012; accepted 27 June 2012
Abstract: The structures and electrochemical properties of the Ti1.4V0.6Ni ribbon before and after heat treatment are investigated systematically. The structure of the sample is characterized by X-ray powder diffraction analysis. Electrochemical properties including the discharge capacity, the cyclic stability and the high-rate discharge ability are tested. X-ray powder diffraction analysis shows that after heat treatment at 590 ��C for 30 min, all samples mainly consist of the icosahedral quasicrystal phase (I-phase), Ti2Ni phase (FCC), V-based solid solution phase (BCC) and C14 Laves phase (hexagonal). Electrochemical measurements show that the maximum discharge capacity of the alloy electrode after heat treatment is 330.9 mA��h/g under the conditions that the discharge current density is 30 mA/g and the temperature is 30 ��C. The result indicates that the cyclic stability and the high-rate discharge ability are all improved. In addition, the electrochemical kinetics of the alloy electrode is also studied by electrochemical impedance spectroscopy (EIS) and hydrogen diffusion coefficient (D).
Key words: quasicrystal; Ti1.4V0.6Ni alloy; electrochemical properties; cyclic stability; high-rate discharge; discharge capacity
1 Introduction
The quasicrystalline phase formation is observed in 1984 by SHECHTMAN et al. Many kinds of quasicrystalline materials have been discovered and investigated extensively during the past three decades. I-phase may have better physical properties for hydrogen-storage applications since some of its atoms have an adequate affinity for hydrogen. The hydrogen desorption performance of Ti45Zr38Ni17 I-phase is investigated by using high temperature XRD. The result validates the potential utility of Ti-based I-phase alloy [1]. The Ti45Zr38Ni17 I-phase can absorb a large amount of hydrogen, up to about two hydrogen atoms per metal atom, while the Ti41.5Zr41.5Ni17 I-phase alloy can reach up to three hydrogen atoms per metal atom [2,3]. The electrochemical maximum hydrogen storage capacity of Ti45Zr35Ni17Cu3 I-phase alloy of 240 mAh/g was reported at the discharge current density of 30 mA/g [4-6]. HU et al [7] systematically investigated the electrochemical hydrogen storage properties of Ti-V-Ni-Co I-phase alloys [7]. Our previous works reported that the electrochemical performance of Ti1.4V0.6Ni I-phase alloy is improved when AB3-type hydrogen storage alloy is added to Ti1.4V0.6Ni I-phase alloy [8]. Up to the present, few investigations of hydrogen storage properties have been focused on the other Ti-based I-phase alloys.
At present, the Ti-based Laves phase alloys [9,10] and the V-based solid solution alloys [11,12] are regarded as promising materials for electrodes of the Ni�CMH rechargeable batteries due to their higher discharge capacity. The V-based solid solution with BCC structure could absorb and desorb a large amount of hydrogen in the alkaline electrolyte. For example, the C14 Laves phase or the TiNi phase exhibits high electrocatalytic activity [13,14]. AKIBA and IBA [15] found the Laves phase alloys with a large amount of hydrogen capacity (above 2% in mass fraction) and fast hydrogen absorption and desorption kinetics at appropriate condition [15]. Some studies have discovered that a well-designed multiphase hydrogen storage alloys might greatly improve their electrochemical properties [13-16]. OVSHINSKY and FETCENRO [16] pointed out that some multielement, multiphase disordered hydrogen storage alloys provided the electrode surface with exceptionally good electrochemical catalytic activity.
In this work, multiphase Ti-V-Ni hydrogen storage alloy containing I-phase is formulated, and the structures and electrochemical properties of the alloy is investigated.
2 Experimental
Blend of Ti, V and Ni (99.9% purity) corresponding to the nominal composition of Ti1.4V0.6Ni was prepared by arc-melting under argon atmosphere on a water cooled copper hearth. The ingots were turned over and remelted three times to ensure the homogeneity. Then they were melt-spun onto a copper wheel rotating at 34 m/s to produce ribbon. The phase transformation temperature of the ribbon was investigated in an alumina pan under a purified argon atmosphere with a heating rate of 0.17 K/s by a differential scanning calorimeter (DSC). Alloy ribbons were mechanically crushed into fine powders of 0.038-0.075 ��m for testing. The powders were treated at 590 ��C for 30 min under vacuum. Structure of each heat treated sample was examined by XRD analysis with Cu K�� radiation on D/Max-rB X-ray diffractometer using JAD5 software.
The metal hydride electrode as the working electrode was prepared by mixing 0.15 g alloy powder and 0.75 g carbonyl nickel powder, then cold-pressing the mixture to form pellets under a pressure of 15 MPa with 10 mm in diameter and about 1.5 mm in thickness. For electrochemical measurements, the charge-discharge test was conducted in a half-cell consisting of a metal-hydride electrode, a Ni(OH)2/NiOOH counter electrode, and a Hg/HgO reference electrode in a 6 mol/L KOH solution electrolyte, with DC-5 battery testing instrument under computer control. During the charge/discharge test, the electrodes were fully charged (the over-charged ratio approximately 30%) at a current density of 60 mA/g for 6 h, and then discharged to -0.6 V vs Hg/HgO at 30 mA/g and 303 K. After every charge/discharge, the rest time was 5 min. For evaluating the high-rate dischargeability, discharge capacities of the alloy electrode at different discharge current densities were measured. The electrochemical impedance spectroscopy (EIS) analysis was taken at the 50% depth of discharge (DOD) using a Solartron 1287 Potentiostat/ Galvanostat and a Solartron 1255B frequency response analyzer with Z-PLOT software for WINDOWS. In this work, potentiostatic discharge technique was selected to estimate the hydrogen diffusion coefficient through a given electrode. After being fully charged and followed by 30 min opencircuit lay-aside, the tested electrodes were discharged with 500 mV potential-step for 3600 s on the EG & G PARC Model 273 Potentiostatic/ Galvanostat, using the M352 corrosion software.
3 Results and discussion
3.1 Alloy structures
Figure 1 shows the DSC curve of the melt-spun Ti1.4V0.6Ni alloy ribbon. The DSC trace is obtained at a heating rate of 0.17 K/s from 400 to 800 ��C. The crystallization takes place through a small sharp exothermic peak reaction for this alloy ribbon, and the main DSC exothermic peak shows a straight base line. The tardy peak of the exothermic reaction is due to the transformation from the I-phase to the C14 Laves phase, and the transfer peak temperature ranges from 564 ��C to 613 ��C.

Fig. 1 DSC curve of original melt-spun Ti1.4V0.6Ni alloy ribbon
Figure 2 shows the XRD patterns of the Ti1.4V0.6Ni alloy. It is found that the Ti1.4V0.6Ni alloy consists of I-phase, face centered cubic (FCC) phase with Ti2Ni- type structure in space group R-3m and V-based solid solution phase with body centered cubic (BCC) structure in space group Im3m. After heat treatment, the alloy also contains the C14 Laves phase with MgZn2 type hexagonal structure in space group P63/mmc except for I-phase and V-based solid solution phase. It can be found that the peak intensity of I-phase becomes weak, which means that the transformation from the I-phase to the C14 Laves phase occurs.

Fig. 2 XRD patterns of original (a) and treated Ti1.4V0.6Ni (b) alloys
3.2 Discharge capacity
Figure 3 shows the discharge capacity as a function of the cycle number at a current density of 30 mA/g and 30 ��C for the original and the heat treated alloy electrodes. The maximum electrochemical capacities of the above alloys are summarized in Table 1. The original Ti1.4V0.6Ni alloy and the heat treated alloy give a discharge capacity of 276.3 and 330.9 mA��h/g, respectively. This can be explained by that the heat treated alloy contains the C14 Laves phase with MgZn2 type, which acts as a catalyst and a micro-current collector, Ti2Ni phase (FCC), V-based solid solution phase (BCC) and I-phase which is activated to reversibly absorb and desorb a considerable amount of hydrogen in KOH electrolyte. The discharge capacity of the treated alloy is higher than that of the untreated one, and the cyclic stability of the alloy electrode is little improved.
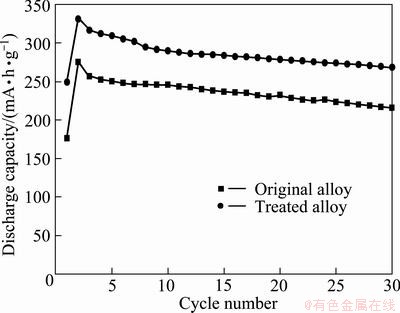
Fig. 3 Discharge capacity as function of cycle number for original and treated Ti1.4V0.6Ni alloy electrodes
Table 1 High-rate discharge abilities of original and treated Ti1.4V0.6Ni alloy electrodes

3.3 High-rate discharge ability and electrochemical kinetics
Figure 4 shows the high-rate discharge ability (HRD), which denotes the discharge properties of the fully activated alloys at large discharge current densities. The HRD is an important kinetic property for the metal hydride electrode. To evaluate the HRD, the charging current density is kept constant of 60 mA/g, the discharge capacities are noted as Ci at different discharge current densities. The HRD is defined as the ratio of the Ci to the maximum discharge capacity C30 when the discharge current density is 30 mA/g. It can be observed that the HRD of the treated alloy is superior to that of the original alloy under the same discharge condition, as summarized in Table 1. For example, the HRD increases from 74.2% to 79 % after heat treatment at the discharge current density of 240 mA/g. The significant improvement of the HRD of the heat treated alloy is probably attributed to the C14 Laves phase which acts as a catalyst.

Fig. 4 High-rate dischargeability of original and treated Ti1.4V0.6Ni alloy electrodes
Figure 5 shows the EIS curves of the original and the treated Ti1.4V0.6Ni alloy electrodes at 50% depth of discharge (DOD) and the equivalent circuit. It can be seen clearly that all the EIS curves consist of two semicircles and a straight line. The semicircle in the high frequency region relates to the contact resistance between the current collector and the alloy pellet, and in the low frequency region corresponds to the charge reaction resistance [17]. On the basis of the circuit and fitting program Z-VIEW [18], Rct is obtained. The charge-transfer resistance Rct is shown in Table 2. It can be seen that the Rct decreases from 0.218 �� to 0.175 �� by heat treatment, which results in a great improvement of the reactive activity, thus, the discharge capacity increases dramatically.

Fig. 5 EIS curves of original and treated Ti1.4V0.6Ni alloy electrodes (a) and equivalent circuit (b)
Figure 6 shows the semi-logarithmic curves of anodic current vs time response for the original and the treated Ti1.4V0.6Ni alloys. According to the model of ZHENG et al [19], the diffusion coefficient of the hydrogen atoms in the bulk alloy can be estimated through the slope of the linear region of the corresponding plots by the following formula:
 (1)
(1)
where D is the hydrogen diffusion coefficient; a is the radius of the spherical particle; i is the diffusion current density; C0 is the initial hydrogen concentration in the bulk electrode; Cs is the hydrogen concentration on the surface of alloy particles; d is the density of the hydrogen storage materials; t is the discharge time. With the average particle radius of 15 ��m, the hydrogen diffusion coefficients D in the bulk alloy electrodes are calculated by Eq. (1) and listed in Table 2, respectively.

Fig. 6 Semi-logarithmic plots of anodic current vs time response of original and treated Ti1.4V0.6Ni alloy electrodes
Table 2 Electrochemical behaviours of original and treated Ti1.4V0.6Ni alloy electrodes
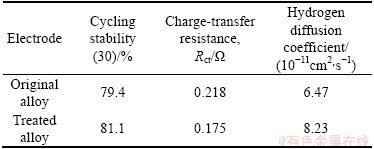
It can be seen that D increases from 6.47��10-11 to 8.23��10-11 cm2/s, indicating that the hydrogen diffusion rate is enhanced by heat treatment, which is consistent with that of the high-rate of discharge ability. This suggests that the diffusion process is dominated by the electrochemical reaction. As mentioned above, the treated alloy facilitated atomic hydrogen transportation. From the above results, it is believed that the kinetics of hydriding Ti1.4V0.6Ni alloy is greatly improved by heat treatment. Generally speaking, the kinetic properties of the hydride electrode are mainly controlled by the charge transfer resistance on the electrode surface and by the hydrogen diffusion resistance inside the electrode. In other words, the HRD is influenced by the hydrogen diffusion coefficient. In this work, D is increased markedly. The variation is virtually consistent with the HRD of the alloy electrode.
4 Conclusions
The structures and electrochemical properties of the original and the treated Ti1.4V0.6Ni alloy electrodes were investigated systematically. The powders were treated at 590 ��C for 30 min under vacuum. The result of the XRD shows that the original alloy consists of the I-phase, V-based solid solution phase with BCC structure and face centered cubic phase with Ti2Ni-type structure; however, the treated alloy includes the above phases as well as the C14 Laves phase with a hexagonal structure. The electrochemical properties such as discharge capacity, cyclic stability and high-rate discharge ability (HRD) are improved by heat treatment. The treated alloy electrodes exhibit higher hydrogen diffusion coefficient and lower charge-transfer resistance in comparison with the untreated one. This means that the heat treated alloy facilitates the formation of metal�Chydride and hydrogen diffusion within the bulk alloy. The C14 Laves phase acts as a catalyst and a micro-current collector, and improves the electrochemical characteristics of the alloy electrode.
References
[1] STROUD R M, VIANO A M, GIBBONS P C, KELTON K F. Stable Ti-based quasicrystal offers prospect for improved hydrogen storage [J]. Applied Physics Letters, 1996, 69: 2998-3000.
[2] GIBBONS P C, HENNING R G, HUETT V T, KELTON K F. Ti-Zr-Ni and Ti-Hf-Ni quasicrystals and approximants as hydrogen storage alloys [J]. Journal of Non-crystalline Solids, 2004, 334-335: 461-465.
[3] KIM J Y, HENNING R, HUETT V T, GIBBONS P C, KELTON K F. Hydrogen absorption in Ti-Zr-Ni quasicrystals and 1/1 approximants [J]. Journal of Alloys and Compounds, 2005, 404-406: 388-391.
[4] LIU Bao-zhong, WU Yao-ming, WANG Lin-min. Electrochemical properties of amorphous and icosahedral quasicrystalline Ti45Zr35Ni17Cu3 powders [J]. Journal of Power Sources, 2006, 159(2): 1458-1463.
[5] LIU Bao-zhong, WU Yao-ming, WANG Lin-min. Kinetic and electrochemical properties of icosahedral quasicrystalline Ti45Zr35Ni17Cu3 powders [J]. International Journal of Hydrogen Energy, 2006, 31(10): 1394-1400.
[6] LIU Bao-zhong, LIU Dong-ming, WU Yao-ming, WANG Lin-min. Hydrogen absorption in Ti45Zr35Ni17Cu3 amorphous and quasicrystalline alloy powders [J]. International Journal of Hydrogen Energy, 2007, 32(13): 2429-2433.
[7] HU Wen, NIU Xiao-dong, WATADA M, KAWABE Y, WU Yao-ming, WANG Li-dong, WANG Li-min. Electrochemical hydrogen storage in Ti1.6V0.4Ni1-xCox icosahedral quasicrystalline alloys [J]. Chem Phys Chem, 2010, 11(1): 295-300.
[8] LIU Wan-qiang, WANG Xin-lu, HU Wen, KAWABE Y, WATADA M, WANG Li-min. Electrochemical performance of TiVNi- quasicrystal and AB3-type hydrogen storage alloy composite materials [J]. International Journal of Hydrogen Energy, 2011, 36(1): 616-620.
[9] LEE H H, LEE K Y, LEE J Y. The Ti-based metal hydride electrode for Ni-MH rechargeable batteries [J]. Journal of Alloys and Compounds, 1996, 239(1): 63-70.
[10] YU J S, LEE S M, CHO K, LEE J Y. The cycle life of Ti0.8Zr0.2V0.5Mn0.5-xCrxNi0.8 (x=0 to 0.5) alloys to metal hydride electrodes of Ni-metal hydride rechargeable battery [J]. Journal of the Electrochemical Society, 2000, 147(6): 2013-2017.
[11] TSUKAHARA M, TAKAHASHI K, MISHIMA T, SAKAI T, MIYAMURA H, KURIYAMA N, UEHARA I. Metal hydride electrodes based on solid-solution type alloy TiV3Nix (0��x��0.75) [J]. Journal of Alloys and Compounds, 1995, 226(1-2): 203-207.
[12] IWAKURA C, CHOI W K, MIYAUCHI R, INOUE H. Electrochemical and structural characterization of Ti-V-Ni hydrogen storage alloys with BCC structure [J]. Journal of the Electrochemical Society, 2000, 147(7): 2503-2506.
[13] TSUKAHARA M, TAKAHASHI K, MISHIMA T, ISOMURA A, SAKAI T. V-based solid solution alloys with Laves phase network: Hydrogen absorption properties and microstructure [J]. Journal of Alloys and Compounds, 1996, 236(1-2): 151-155.
[14] TSUKAHARA M, TAKAHASHI K, MIYAUCHI T, ISOMURA A, SAKAI T. Vanadium-based solid solution alloys with three-dimensional network structure for high capacity metal hydride electrodes [J]. Journal of Alloys and Compounds, 1997, 253: 583-586.
[15] AKIBA E, IBA H. Hydrogen absorption by laves phase related BCC solid solution [J]. Intermetallics, 1998, 6: 461-470.
[16] OVSHINSKY S R, FETCENKO M A. Development of high catalytic activity disordered hydrogen-storage alloys for electrochemical application in nickel-metal hydride batterie [J]. Applied Physics A, 2001, 72(2): 239-244.
[17] GAO Xiao-peng, YANG Hua-bin, ZHOU Zuo-xiang. Electrochemical properties of the Zr(W0.4Ni0.6)2.4 hydrogen storage alloy electrode [J]. Journal of Alloys and Compounds, 1996, 235(2): 225-231.
[18] KURIYAMA N, SAKAI T, MIYAMURA H, UEHARA I, ISHIKAWA H. Electrochemical impedance and deterioration behavior of metal hydride electrodes [J]. Journal of Alloys and Compound, 1993, 202: 183-197.
[19] ZHENG Gang, POPOV B N, WHITE R E. Electrochemical determination of the diffusion-coefficient of hydrogen through an LaNi4.25Al0.75 Electrode in alkaline aqueous-solution [J]. Journal of the Electrochemical Society, 1995, 142(8): 2695-2698.
�ȴ�����������ʮ��������Ti1.4V0.6Ni�Ͻ�缫�ĵ绯ѧ���ܵ�Ӱ��
����ǿ1,2������1,2��������2
1. ����������ѧ ���Ͽ�ѧ�빤��ѧԺ������ 130022��
2. �й���ѧԺ ����Ӧ�û�ѧ�о�����ϡ����Դ�����ù����ص�ʵ���ң����� 130022
ժ Ҫ���о����ȴ���ǰ��Ti1.4V0.6Ni�Ͻ�Ľṹ�͵绯ѧ���ܡ�����X���߷�ĩ����(XRD)���������Ͻ�Ľṹ���绯ѧ�������ŵ�������ѭ���ȶ��Ժ߱��ʷŵ����ܵȡ�XRD���������������590 ��C�ȴ���yeated allloyhe 30 min�ĺϽ���Ҫ��������ʮ�������ࡢTi2Ni(FCC)�ࡢV��������(BCC)��C14Laves��(Hex)���绯ѧ������ʾ���ȴ�������30 ��C�ͷŵ�����ܶ�Ϊ30 mA/g�������£��Ͻ�缫�����ŵ������ɴ�330.9 mA��h/g������ѭ���ȶ��Ժ߱��ʷŵ�����Ҳ�õ����ơ����⣬ͨ���绯ѧ�迹�ͺϽ��ڲ������ɢϵ���о��˺Ͻ�缫�Ķ���ѧ���ܡ�
�ؼ��ʣ�����Ti1.4V0.6Ni�Ͻ𣻵绯ѧ���ܣ�ѭ���ȶ��ԣ��߱��ʷŵ磻�ŵ�����
(Edited by YANG Hua)
Foundation item: Project (20112216120001) supported by the Doctoral Program of Tertiary Education of the Ministry of Education of China; Project (21215141) supported by the Natural Science Foundation of Jilin Province, China; Project (20921002) supported by the Foundation for Innovative Research Groups of the National Natural Science Foundation of China; Projects (21073179, 61106050) supported by the National Natural Science Foundation of China; Project (BE2012047) supported by Scientific and Technological Supporting Program of Jiangsu Province of China and GS Yuasa Corporation of Japan; Project (11KZ38) supported by and Scientific and Technological Pillar Project of Changchun, China
Corresponding author: LIU Wan-qiang; Tel: +86-15904408948; E-mail: wqliu1979@126.com
DOI: 10.1016/S1003-6326(11)61567-7