J. Cent. South Univ. Technol. (2010) 17: 1-6
DOI: 10.1007/s11771-010-0001-y
Improved oxidation resistance of chemical vapor reaction SiC coating modified with silica for carbon/carbon composites
YANG Xin(����)1, ZOU Yan-hong(����)2, HUANG Qi-zhong(������)1,
SU Zhe-an(���ܰ�)1, CHANG Xin(����)1, ZHANG Ming-yu(�����)1, XIAO Yong(Ф��)1
1. State Key Laboratory of Powder Metallurgy, Central South University, Changsha 410083, China;
2. Materials Science and Engineering Post-doctoral Research Center, Central South University,
Changsha 410083, China
? Central South University Press and Springer-Verlag Berlin Heidelberg 2010
Abstract: To protect carbon/carbon (C/C) composites from oxidation, a SiC coating modified with SiO2 was prepared by a complex technology. The inner SiC coating with thickness varying from 150 to 300 ��m was initially coated by chemical vapor reaction (CVR): a simple and cheap technique to prepare the SiC coating via siliconizing the substrate that was exposed to the mixed vapor (Si and SiO2) at high temperatures (1 923-2 273 K). Then the as-prepared coating was processed by a dipping and drying procedure with tetraethoxysilane as source materials to form SiO2 to fill the cracks and holes. Oxidation tests show that, after oxidation in air at 1 623 K for 10 h and thermal cycling between 1 623 K and room temperature 5 times, the mass loss of the CVR coated sample is up to 18.21%, while the sample coated with modified coating is only 5.96%, exhibiting an obvious improvement of oxidation and thermal shock resistance of the coating. The mass loss of the modified sample is mainly contributed to the reaction of C/C substrate with oxygen diffusing through the penetrating cracks formed in thermal shock tests.
Key words: carbon/carbon composites; coating; oxidation; SiC
1 Introduction
Carbon/carbon (C/C) composites are considered as one of the most advanced materials for their excellent properties at high temperatures. Due to their key properties of very high strength and strength retention at temperatures above 2 273 K, lightweight and high thermal stability [1-2], they are widely used as high temperature structural materials in fusion reactors, engines, rockets and aerospace vehicles [3-4]. However, their easy oxidation above 723 K in an oxidizing environment greatly limits their applications in aerospace industry [5-6]. Therefore, the technology of reliable oxidation protection is crucial to exert their full potential. To overcome this serious defect, much attention has been paid to this work. It is generally recognized that coating is one of the most efficient ways that can protect carbon materials from oxidation at high temperatures [7] because it can directly cut off the diffusion channels for oxygen to attack the matrix.
To prepare coatings on C/C composites, ceramics such as SiO2, MoSi2, Al2O3, SiC and Si3N4 have been extensively used [8-14], and among these possible materials, SiC is determined to be one of the most ideal coating materials due to its good mechanical properties, excellent anti-oxidation property, low thermal expansion coefficient (CTE) and good physical-chemical compatibility with carbon materials [15-16]. However, up to now, much work has been devoted to technologies of pack cementation [17], chemical vapor deposition (CVD) [18], laser-induced chemical decomposition (LICD) [19] and slurry-sintering [20] to prepare SiC coating on C/C composites, while little work has been performed on developing new techniques with potential applications. With respect to these developed technologies, CVD and LICD are regarded as expensive techniques that are difficult to achieve a perfectly dense structure. Though pack cementation and slurry method are simple and low cost techniques that can be conducted in laboratory, their reliability of obtaining a uniform coating still needs further verification. Furthermore, their applications can be easily restricted for their disadvantages of treating parts with complex shape and big size.
The aim of this work was to develop a new technology with potential applications that can offer long time anti-oxidation protection for C/C composites. A complex technology with two-step technique for preparing a dense SiC coating on C/C composites was developed. The inner coating was firstly prepared by chemical vapor reaction (CVR) and then modified with SiO2 to seal cracks and holes. The effect of SiO2 modification on the oxidation behavior of SiC coated C/C composites was discussed.
2 Experimental
The C/C composites used in this work were 2.5- dimensional materials with a density of 1.72 g/cm3 produced by chemical vapor infiltration in our laboratory. Small specimens (12 mm��10 mm��10 mm) used as substrates were cut from the obtained C/C composites. The specimens were hand-polished with 600 grit SiC paper, followed by ultrasonic washing in ethanol and then dried at 393 K for 2 h. The materials used in the CVR were Si sheets (industrial reagent, ��99.4%) and SiO2 powders (industrial reagent, ��99.2%). Before the CVR, they were initially washed by dilute hydrochloric acid to remove the impurities, and then dried at 373 K for a sufficient time. After being placed in a graphite crucible, they were heated to generate vapor at temperature above their melting points. Meanwhile, the as-prepared specimens were exposed to the mixed vapor to form the coatings. The whole process was conducted at the temperature in the range of 1 923-2 273 K for 1- 3 h, followed by a natural cooling course. Details for preparing the inner layer CVR SiC coating were reported in Ref.[21].
The modification of the obtained coating was carried out as follows: the coated samples were firstly immersed in tetraethoxysilane (TEOS) solution for 3 h, and then withdrawn to dry at 423 K for 2 h. In this process, SiO2 was formed via the following chemistry reactions:
Si(OC2H5)4(l)+4H2O(l)=Si(OH)4(l)+4C2H5OH(l) (1)
Si(OH)4(l)=SiO2(s)+2H2O(l) (2)
The process was repeated 3-5 times to fill the cracks and holes in the primal coating. The isothermal oxidation tests of the as-coated specimens were performed at 1 623 K in an electrical furnace in air. Meanwhile, the specimens were also endured thermal shock cycles from 1 623 K to room temperature, when they were taken out of the furnace directly into air. Cumulative mass changes of the samples after every thermal cycle were measured by the precision balance and reported as a function of time. The surface and cross-section morphologies of the coatings were observed with scanning electron microscopy (SEM). X-ray diffraction (XRD) and energy dispersive spectroscopy (EDS) were also used to identify crystalline structures and analyze element distribution in the coating, respectively.
3 Results and discussion
3.1 Microstructure of CVR coating
Fig.1 shows the XRD pattern of the coating prepared by CVR. It can be seen that there are only diffraction peaks for SiC in the as-prepared coating, while any peaks corresponding to carbon and silicon are not detected. Thus, we can deduce that the obtained coating is composed of SiC.
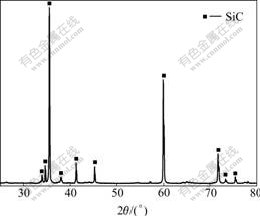
Fig.1 XRD pattern of as-prepared coating obtained by CVR
Typical surface morphologies and the spot EDS analyses of the prepared CVR coating are displayed in Fig.2. The EDS results further confirm the formation of a SiC coating and are in good agreement with the XRD analysis. Moreover, it is clear that the coating surface is composed of SiC micro-crystals together with large SiC grains that are homogenously distributed and randomly oriented. Obviously, the coating surface is very rough, and some cracks and holes can also be found, indicating the formation of a porous structure. These defects offer the diffusion channels for oxygen to attack the substrate and may lead to the degradation of the oxidation protective ability of the coating. So, to improve the anti-oxidation property of the coating, it is necessary to explore a modification process to seal these cracks and holes.

Fig.2 SEM images and EDS analysis of obtained SiC coating on C/C composites: (a) Cracks; (b) Holes in SiC coating; (c) EDS analysis of spot A in Fig.2(a)
Fig.3 shows SEM image of the cross-section of the as-prepared coating. From Fig.3, it can be seen that there are no holes visible in the coating, and also continuous interface is found between the coating and the substrate, which reveals good compatibility and adherence between them. Moreover, it is evident that the obtained coating has an uneven thickness distribution, varying from 150 to 300 ��m. This result can be explained by the structural difference of C/C composites. As C/C composites are of porous structures consisting of holes, pyrolytic carbon and carbon fibers, the great diversity of silicon vapor diffusivities through these parts leads to the ununiformity of the coating thickness. According to different structures of the C/C substrate shown in Fig.3, three zones are present in the as-formed coating. In the zone where carbon fibers in C/C composites are perpendicular to the surface, the coating has the thinnest thickness less than 150 ?m. Higher coating penetration occurs where carbon fibers are parallel to the surface, which results in the increment of the coating thickness up to 240 ?m. As for the transition zone where carbon fibers change direction, the pyrolytic carbon appears in this area and then the coating with thickness of 300 ?m is formed. The same results can also be found in the work of FU et al [22].

Fig.3 SEM image of cross-section of as-prepared SiC coating
3.2 Improved structure of modified SiC coating
Surface SEM image of the coating with modification shows the formation of a dense and crack-free surface (Fig.4), which is expected to provide better oxidation protection than monolayer SiC coating. As revealed in Figs.4(a) and (b), it can be seen that the coating consists of two kinds of crystalline particles characterized by EDS as SiC and SiO2, respectively. By spot EDS analysis, the zones with low and high oxygen content corresponding to SiO2 coated SiC crystals and SiO2 sealed areas can be distinguished easily, which implies that the formed SiO2 particles have a sufficient amount and can cover continuously on the entire coating surface. As the formed SiO2 particles have a very small size less than 5 ?m, so they could efficiently fill the cracks and holes in the primary coating.

Fig.4 SEM surface images and area EDS analysis of SiC coating modified with SiO2: (a) SiO2-coated SiC crystals; (b) Formed SiO2 particles; (c) EDS analysis of spot 1 in Fig.4(a); (d) EDS analysis of spot 2 in Fig.4(b)
3.3 Oxidation behaviors
The isothermal oxidation curves of the samples at 1 623 K in air are shown in Fig.5. For a comparison, bare C/C composites without any coating were also tested. From Fig.5, we find that the mass loss of the bare C/C composites increases rapidly with increased oxidation time, and after 2 h oxidation, the C/C composites are almost burnt away due to the attack by oxygen. While as concerned with the sample directly coated with SiC coating, the mass loss is only 18.21% after 10 h oxidation in air. However, for the SiC coated sample modified with SiO2, a very small mass loss of 5.96% is observed for up to 10 h exposure, indicating that the modification process significantly improves the oxidation resistance of the SiC coated C/C composites. Meanwhile, the thermal shock resistance of the modified coating was also investigated in the oxidation tests. After enduring 5 thermal cycles from 1 623 K to room temperature, no spallation or peeling was found. Therefore, it can be inferred that the coating has excellent thermal shock resistance, which may be attributed to the excellent bonding ability of the coating prepared with CVR.
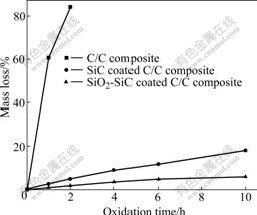
Fig.5 Isothermal oxidation curves of different samples in air at 1 623 K
After 10 h oxidation at 1 623 K in air, the coating surface morphologies were observed with scanning electron microscopy (Fig.6). It was found that a smooth glass layer was formed on the coating surface due to the oxidation of SiC crystals. The formation of SiO2 glass can reduce the oxidation rate and act as a sealant to fill the defects in the coating. From Fig.6, it can be found that some holes were present on oxidized coating surface, which provided pathways for oxygen to attack the substrate and resulted in the mass loss of the coated samples. Moreover, compared with the modified sample, it was noted that a great number of holes with diameters of 10-100 ��m were visible on the monolayer SiC coating surface. The formation of these holes can be explained as follows: because the formed SiO2 glass has a very low oxygen permeability and evaporation rate, the oxidation process of the coated sample is mainly controlled by the rate of oxygen diffusion through the defects in the coating. During the oxidation test, inward diffusion of oxygen and outward diffusion of CO or CO2 will occur simultaneously along the defects. According to FERGUS and WORRELL [23], once the pressure of the outward diffusion gas exceeds 0.1 MPa, the gas product can bubble through the SiO2 film because of the low viscosity of glass layer at 1 623 K. So, after the gas bubbles break up, a big hole forms. The similar evidence can also be found in the work of HUANG et al [17]. As fewer defects are contained in the modified sample, thus, fewer holes are formed on the coating surface, and as a result the oxidation resistance of the coating improves obviously. Additionally, some micro-cracks are also found on the modified coating surface (Fig.6(b)). Due to the quick cooling of the modified sample from 1 623 K to room temperature in the oxidation test, penetrating cracks were inevitably formed in the coating. Though these penetrable cracks can be self-sealed when the sample was heated to high temperature again, actually, they may not be self-healed completely because of the poor fluidity of the SiO2 glass at 1 623 K. Furthermore, in the thin areas where the volume of SiO2 glass was not enough to seal the transverse cracks, oxygen began to diffuse through these defects and directly attack the substrate. As shown in Fig.7, a small cavity beneath a transverse crack was observed for a modified sample after five cycles of thermal shock treatment. Therefore, it can be concluded that these transverse cracks play a more important role in the oxidation of the modified sample than holes though elimination of these cracks and holes in the coating needs further research. To further improve the oxidation resistance of the SiC coated C/C composites, additional research focused on preparing multi-layer coating with excellent self-healing ability is necessary.

Fig.6 Surface morphologies of coated samples after oxidation at 1 623 K for 10 h: (a) SiC coated sample; (b) SiC coated sample modified with SiO2

Fig.7 Cross-section micrograph of modified SiC coating after oxidation and thermal cycling
4 Conclusions
(1) A SiC coating modified with SiO2 can be prepared on C/C composites by the CVR combined with a modification process using TEOS as source materials. The dense structure of the modified coating is contributed to the filling of SiO2 in the SiC coating.
(2) The modification has intense effect on the oxidation resistance of the SiC coating. Compared with the monolayer SiC coating, the modified coating exhibits significantly improved anti-oxidation property for C/C composites. After oxidation in air at 1 623 K for 10 h and thermal cycling between 1 623 K and room temperature 5 times, the mass loss of the CVR coated sample is up to 18.21%, while that of the sample coated with modified coating is only 5.96%.
(3) The mass loss of the modified sample is mainly attributed to the reaction of C/C matrix with oxygen diffusing through the penetrating cracks formed in thermal shock tests.
References
[1] FU Qian-gang, LI He-jun, SHI Xiao-hong, LI Ke-zhi, WANG Chuang, HUANG Min. Double-layer oxidation protective SiC/glass coatings for carbon/carbon composites [J]. Surface and Coatings Technology, 2006, 200(11): 3473-3477.
[2] YUAN Yi-dong, ZHANG Fu-kuan, ZHOU Wan-cheng. Preparation and characteristics of C/C composite brake disc by multi-cylindrical chemical vapor deposition processes [J]. Journal of Central South University of Technology, 2005, 12(4): 400-402.
[3] KIM J I, KIM W J, CHOI D J, PARK J Y, RYU W S. Design of a C/SiC functionally graded coating for the oxidation protection of C/C composites [J]. Carbon, 2005, 43(8): 1749-1757.
[4] ZHU Y C, OHTANI S, SATO Y, IWAMOTO N. The improvement in oxidation resistance of CVD-SiC coated C/C composites by silicon infiltration pretreatment [J]. Carbon, 1998, 36(7/8): 929-935.
[5] FU Qian-gang, LI He-jun, SHI Xiao-hong, LIAO Xiao-ling, LI Ke-zhi, HUANG Min. Microstructure and anti-oxidation property of CrSi2-SiC coating for carbon/carbon composites [J]. Applied Surface Science, 2006, 252(10): 3475-3480.
[6] HUANG Jian-feng, ZENG Xie-rong, LI He-jun, XIONG Xin-bo, SUN Guo-ling. ZrO2-SiO2 gradient multilayer oxidation protective coating for SiC coated carbon/carbon composites [J]. Surface and Coatings Technology, 2005, 190(2/3): 255-259.
[7] LI Guo-dong, XIONG Xiang, HUANG Bai-yun, HUANG Ke-long. Structural characteristics and formation mechanisms of crack-free multilayer TaC/SiC coatings on carbon-carbon composites [J]. Transactions of Nonferrous Metals Society of China, 2008, 18(2): 255-261.
[8] KOH Y H, KWON O S, HONG S H, KIM H E, LEE S K. Improvement in oxidation resistance of carbon by formation of a protective SiO2 layer on the surface [J]. Journal of the European Ceramic Society, 2001, 21(13): 2407-2412.
[9] KWON O S, HONG S H, KIM H. The improvement in oxidation resistance of carbon by a graded SiC/SiO2 coating [J]. Journal of the European Ceramic Society, 2003, 23(16): 3119-3124.
[10] ZHAO Juan, LIU Lang, GUO Quan-gui, SHI Jing-li, ZHAI Geng-tai. Oxidation protective behavior of SiC/Si-MoSi2 coating for different graphite matrix [J]. Materials Letters, 2006, 60(16): 1964-1967.
[11] YANG Xin, ZOU Yan-hong, HUANG Qi-zhong, SU Zhe-an, WANG Xiu-fei, ZHANG Ming-yu. Preparation and structure of MoSi2-Mo5Si3/SiC multi-coating for carbon/carbon composites [J]. Journal of Inorganic Materials, 2008, 23(4): 779-783. (in Chinese)
[12] HUANG Jian-feng, ZENG Xie-rong, LI He-jun, XIONG Xin-bo, HUANG Min. Mullite-Al2O3-SiC oxidation protective coating for carbon/carbon composites [J]. Carbon, 2003, 41(14): 2825-2829.
[13] FU Qian-gang, LI He-jun, SHI Xiao-hong, LI Ke-zhi, ZHANG Wei, HUANG Min. A SiC whisker-toughened SiC-CrSi2 oxidation protective coating for carbon/carbon composites[J]. Applied Surface Science, 2007, 253(8): 3757-3760.
[14] ZHU Y C, OHTANI S, SATO Y, IWAMOTO N. Formation of a functionally gradient (Si3N4+SiC)/C layer for the oxidation protection of carbon�Ccarbon composites [J]. Carbon, 1999, 37(9): 1417-1423.
[15] ZHU Qing-shan, QIU Xue-liang, MA Chang-wen. Oxidation resistant SiC coating for graphite materials [J]. Carbon, 1999, 37(9): 1475-1484.
[16] FU Qian-gang, LI He-jun, SHI Xiao-hong, LI Ke-zhi, SUN Guo-dong. Silicon carbide coating to protect carbon/carbon composites against oxidation [J]. Scripta Materialia, 2005, 52(9): 923-927.
[17] HUANG Jian-feng, ZENG Xie-rong, LI He-jun, XIONG Xin-bo, FU Ye-wei. Influence of the preparation temperature on the phase, microstructure and anti-oxidation property of a SiC coating for C/C composites [J]. Carbon, 2004, 42(8/9): 1517-1521.
[18] CHENG Lai-fei, XU Yong-dong, ZHANG Li-tong, YIN Xiao-wei. Preparation of an oxidation protection coating for C/C composites by low pressure chemical vapor deposition [J]. Carbon, 2000, 38(10): 1493-1498.
[19] SNELL L, NELSON A, MOLIAN P. A novel laser technique for oxidation resistant coating of carbon-carbon composites [J]. Carbon, 2001, 39(7): 991-999.
[20] ZHAO Juan, WANG Gui, GUO Quan-gui, LIU Lang. Microstructure and property of SiC coating for carbon materials [J]. Fusion Engineering and Design, 2007, 82(7): 363-368.
[21] LIU Xing-fang, HUANG Qi-zhong, SU Zhe-an, JIANG Jian-xian. Preparation of SiC coating by chemical vapor reaction [J]. Journal of the Chinese Ceramic Society, 2004, 32(7): 906-910. (in Chinese)
[22] FU Qian-gang, LI He-jun, SHI Xiao-hong, LI Ke-zhi, HUANG Min, SUN Guo-dong. Silicide coating for protection of C/C composites at 1 873 K [J]. Surface and Coatings Technology, 2006, 201(6): 3082-3086.
[23] FERGUS J W, WORRELL W L. Silicon-carbide/boron containing coatings for the oxidation protection of graphite [J]. Carbon, 1994, 33(4): 537-543.
Foundation item: Project(2006CB600901) supported by the National Basic Research Program of China; Project(50802115) supported by the National Natural Science Foundation of China
Received date: 2009-02-23; Accepted date: 2009-05-09
Corresponding author: HUANG Qi-zhong, Professor; Tel: +86-731-88836078; E-mail: qzhuang@mail.csu.edu.cn
(Edited by YANG You-ping)