J. Cent. South Univ. (2017) 24: 1713-1719
DOI: https://doi.org/10.1007/s11771-017-3578-6
Elasticity under pressure and thermal property of Mg2La from first-principles calculations
NIU Xiao-feng(ţ����)1, 2, HUANG Zhi-wei(��־ΰ)3, HU Lei(����)1, 2,
WANG Han(����)1, 2, WANG Bao-jian(������)1, 2
1. College of Materials Science and Engineering, Taiyuan University of Technology, Taiyuan 030024, China;
2. Shanxi Key Laboratory of Advanced Magnesium-based Materials, Taiyuan 030024, China;
3. Southwest Technique and Engineering Institute, Chongqing 400039, China
 Central South University Press and Springer-Verlag GmbH Germany 2017
Central South University Press and Springer-Verlag GmbH Germany 2017
Abstract: The elastic properties, thermodynamic and electronic structures of Mg2La were investigated by using first-principles. The calculated results show that pressure affects the elastic constants of C11 more than that of C12 and C44. Specifically, higher pressure leads to greater bulk modulus (B), shear modulus (G), and elastic modulus (E). We predict B/G and anisotropy factor A based on the calculated elastic constants. The Debye temperature also increases with increasing pressure. Based on the quasi-harmonic Debye model, we examined the thermodynamic properties. These properties include the normalized volume (V/V0), bulk modulus (B), heat capacity (Cv), thermal expansion coefficient (��), and Debye temperature (��). Finally, the electronic structures associated with the density of states (DOS) and Mulliken population are analyzed.
Key words: first-principles; elastic properties; thermodynamics properties; electronic structure
1 Introduction
With good rigidity, high strength and low density, magnesium alloy has been commonly used in the microelectronics, and automobile industries [1, 2]. However, the low strength at high temperature limits its application [3]. Generally speaking, rare earths (RE) additions to magnesium alloy could effectively improve the elastic properties at high temperature and casting properties [4].
The rare earth metal element La added to magnesium alloy could effectively improve the effect of solid solution and aging. The C15-Mg2La alloy as a typical representative of the Mg�CLa alloy has a good high temperature creep resistance. Recently, the research had been carried out for Mg2La alloy [5�C7]. However, there are few studies that examined the elastic properties, thermodynamic and electronic structures of the Mg2La alloy under different pressures and temperatures. As we all know, pressure and temperature had a great effect on the physical properties of materials. Therefore, systematic investigation on elastic properties, thermodynamic and electronic structures of Mg2La alloy at different pressure and temperature could be used to predict the properties of solid theory and determine the basic parameters of material [8].
In this study, elastic properties, thermodynamic and electronic structures of Mg2La alloy are studied by using first-principles. Specifically, it explores the pressure dependences of the shear modulus G, Poisson ratio ��, elastic modulus E, Debye temperature ��, bulk modulus B, and anisotropy factor A. In addition, the thermodynamic properties at different temperature and pressure are studied by the model of quasi-harmonic Debye. Finally, the pressure dependences of that Mg2La alloy are calculated. The results provide a valuable assessment of some properties is difficult to measure by experimental methods.
2 Structure and computational methods
The Mg2La phase belongs to C15-type structure, and space group is Fd3m (No. 227). It has the highest point symmetry and the Pearson symbol cF24. At unit cell of Mg2La, Mg and La atoms take up the positions of the 8a (1/8, 1/8, 1/8) and 16d (1/2, 1/2, 1/2) shown in Fig. 1, respectively.
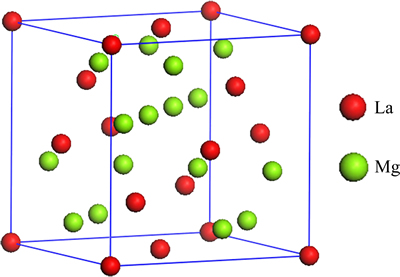
Fig. 1 Modeling crystal structure of Mg2La
We performed the first principles simulations based on density functional theory (DFT) [9] to calculate the electronic structures, elastic and thermodynamic properties of Mg2La [10]. The electron exchange potential energy was calculated by the GGA-PW91 [11, 12]. The kinetic energy cutoff was 400 eV [13]. The Monkhorste-Pack method [14] was used for sampling and integrating special points over the Brillouin zone (BZ), as 8��8��8 for Mg2La. The Pulay schemes [15, 16] were used for stress and energy calculations. The BFGS scheme was used for the structural optimizations. The settings for geometry optimizations were as follows: RMs force, stress, and displacement are 1.0��10-6 eV/ , 0.02 GPa, and 1.0��10-4
, 0.02 GPa, and 1.0��10-4  , respectively. The electronic structure and total energy were calculated by cell optimization with the SCF of 1.1��10-7 eV. The states of Mg 2p6 3s2 and La 5s25p65d16s2 were used in the current calculation [17]. The model of quasi-harmonic Debye [18] was used for computing the thermodynamic properties of the Mg2La alloy.
, respectively. The electronic structure and total energy were calculated by cell optimization with the SCF of 1.1��10-7 eV. The states of Mg 2p6 3s2 and La 5s25p65d16s2 were used in the current calculation [17]. The model of quasi-harmonic Debye [18] was used for computing the thermodynamic properties of the Mg2La alloy.
3 Discussions
3.1 Elastic properties
The elastic constants have a significant role in understanding the mechanical stability of the materials [19, 20]. Therefore, the research of elastic constants of Mg2La under different pressures has a great significance. Here, the elastic constants including C11, C12 and C44 of Mg2La are acquired by geometry optimization.
The calculated Mg2La phases in this work are cubic crystal. The elastic stability conditions are identified as[21]: 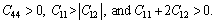 The elastic constants at zero pressure are listed in Table 1, together with theoretical values [6, 21, 22]. These elastic constants follow this stability conditions, indicating that the Mg2La phase is in elastic stability. Thus these findings suggest the rationality of the obtained elastic constants and the selected calculation conditions.
The elastic constants at zero pressure are listed in Table 1, together with theoretical values [6, 21, 22]. These elastic constants follow this stability conditions, indicating that the Mg2La phase is in elastic stability. Thus these findings suggest the rationality of the obtained elastic constants and the selected calculation conditions.
Figure 2 shows elastic constants under different pressures. The results show that C11, C12, C44 increased with the increase of pressure, among which the growth rate of C11 is more obvious than C12 and C44. This is because C11 expresses the length elasticity, while C12 and C44 express the shape elasticity. The strain can easily lead to the change in shape, but no change in length [23]. Hence, C12 and C44 are less impressible compared with C11.
Cauchy pressure (C11-C44) was introduced to predict the metallic or nonmetallic property of materials [24]. The positive value of C11-C44 represent material is metallic; otherwise, it is nonmetallic. The calculated results of Cauchy pressure (C11-C44) are as shown in Fig. 2(a). It is found that the Mg2La has a positive value of C11-C44, indicating that it is metallic material. Furthermore, the value of C11-C44 increases with higher pressure.
In addition, the shear moduli G, bulk moduli B, elastic moduli E, Poisson ratio ��, and anisotropy factor A of Mg2La were calculated via the Voigte-Reusse-Hill method (VRH) [25]. For cubic system, the calculation formulas as follows [26]:
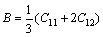 (1)
(1)
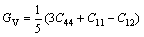 (2)
(2)
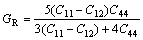 (3)
(3)
 (4)
(4)
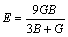 (5)
(5)
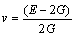 (6)
(6)
Table 1 Elastic constants Cij, elastic modulus, G/B, Poisson ratio �� and anisotropy factor A for Mg2La at 0 GPa
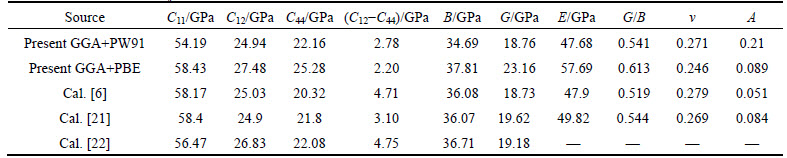
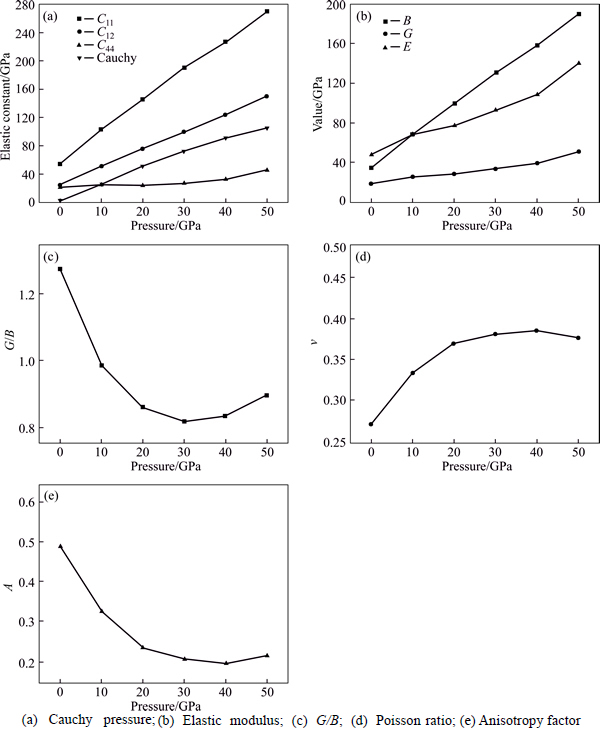
Fig. 2 Elastic constants of Mg2La under different pressures:
 (7)
(7)
The calculated results of G, B and E under different pressures are as shown in Fig. 2(b). It is found that G, B and E increase with additional pressure, indicating that the hardness can be improved with the pressure increasing. The G and B can measure resistance of material to shape and volume change, respectively, showing that the calculated B is much larger than G, which means that the resist tance volume change of Mg2La is much better than shape change.
The ratio of shear modulus to bulk modulus (G/B) was used to estimate the brittle and ductile of materials. Given G/B��0.57, the material is in brittleness, and theopposite is in ductility. The calculated value of G/B is shown in Fig. 2(c), we can found that the G/B of Mg2La is lower than 0.57, indicating that it is in ductility. The calculated G/B of Mg2La decrease with increasing pressure, illustrating that ductility can be improved with the higher pressure. The Poisson ratio �� is usually within the range of 0.25-0.5, which can be used to express the shear resistance of the material [27]. The calculated values of Mg2La at different pressures are in the range of 0.27-0.37 and increase with pressure increasing, indicating that the shear resistance of Mg2La is enhanced with increase of external pressure.
The anisotropy factor A has important significant on researching micro cracks of materials. A=1 indicates that the material is isotropic, and A being larger or less than 1 exhibits anisotropy [28]. The changes of anisotropy factor A with pressure are shown in Fig. 2(e). The A decreases with the pressure increasing. The values of A are listed in Table1. It is found that Mg2La can be considered elastically anisotropic material because of its A smaller than 1.0.
The Debye temperature �� provides some insights from the thermodynamic properties of elastic material [29]; it can distinguish between low and high temperature areas of the solid. For T>��, the material has energies of kBT. Otherwise, high-frequency materials should be predicted to be frozen [30]. The �� expressed as follows [31, 32]:
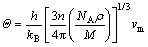 (8)
(8)
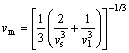 (9)
(9)
 (10)
(10)
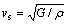 (11)
(11)
where kB is the Boltzmann constant; h is the Planck constant; n is the number of atoms per molecular formula; NA is the Avogadro number; M is the molecular weight; �� is the density; vl ,vm and vs are the longitudinal sound velocity, average sound velocity and the shear sound velocity, respectively.
The dependence of ��, vs vl, vm and �� with pressure ranging from 0 to 50 GPa is shown in Table 2, indicating that of �� increases with pressure increasing. The �� of Mg2La at 0 K and 0 GPa is 246.45 K, which agree well with the available theoretical values 272.81[7]. They are close to our result, indicating that the previous result is precise and the present calculated result is accurate.
3.2 Thermodynamic properties
The quasi-harmonic Debye was applied for studying the thermodynamic properties of the Mg2La under temperature between 0 and 1000 K and pressure between 0 and 50 GPa. The normalized volume V/V0 and bulk modulus B are listed in Fig. 3, showing that the calculated value of B obtained by the model of quasi-harmonic Debye is 35.42 GPa at 0 K and 0 GPa. This is in agreement with the previous findings of 36.07 GPa [21] and 36.71 GPa [22]. Furthermore, the volume decreases as a result of higher pressure, while the curve of V/V0 becomes steeper with the temperature increasing, indicating that the crystal structure of Mg2La becomes stable with higher pressure. The B decreases with the higher temperature and lower pressure. The hardness of Mg2La can be represented by bulk modulus. The changes of bulk modulus with pressure and temperature can be used to define the changes of hardness. Therefore, the hardness of Mg2La is reduced if the temperature is higher and the pressure is lower.
The heat capacity Cv at different temperatures and pressures are described in Fig. 4. These values of Cv decrease with the increase in pressure but increase with the increase in temperature, illustrating that effect of Cv on temperature is more sensitive on pressure. The curves of Cv increase rapidly under temperature below 500 K. The increasing law of Cv obeys Debye��s law [33]. The Cv is approximate to a constant value with the increase in temperature, and known as the Dulong-Petit limit [34]. However, the current heat capacity of Mg2La has no experimental values and theoretical calculations for comparison. Therefore, the calculated values in the work can provide the reference for the further experimental study.
The coefficient �� of thermal expansion at different temperatures and pressures is shown in Fig. 5. �� decreases slowly with the increase in pressure at temperature below 500 K, but it decreases quickly with increasing pressure at temperature beyond 500 K. For a given pressure, it can be found that �� increases rapidly at temperature below 500 K, while �� at 1000 K is slightly larger than that at 800 K, indicating that increasing rate of �� becomes slower at a high temperature.
Table 2 Pressure dependence of shear (vs ) and longitudinal sound velocity (vl), average wave velocity (vm ) and Debye temperature �� for Mg2La
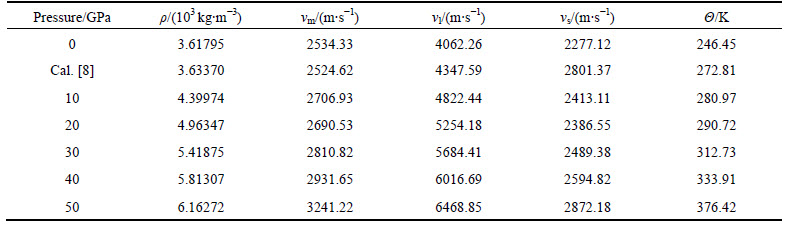
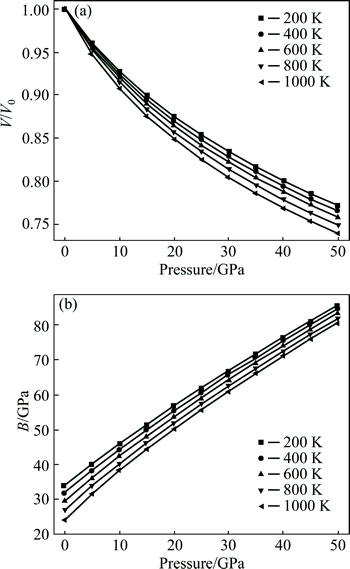
Fig. 3 Pressure dependence of normalized volume V/V0 (a) and bulk modulus B (b) of Mg2La under various temperatures

Fig. 4 Specific heat capacity of Mg2La as a function of temperature under different pressures
The Debye temperature �� of Mg2La at different pressures is shown in Fig. 6. It shows that �� based on the quasi-harmonic Debye model is 251.74 K at 0 K and 0 GPa, which is close to the calculated results (246.45 K and 272.81 K) derived from elastic data [7]. Furthermore,the �� increases markedly with increasing pressure, but decreases slowly with the increase in temperature. These results match with the general behavior of a decreasing Debye temperature with the higher temperature in intermetallic compounds [35]. The influence of temperature on the ��D is obviously smaller than that of pressure. We hope that the thermodynamic properties obtained by quasi-harmonic Debye model could give more insight into the physical properties of Mg2La.
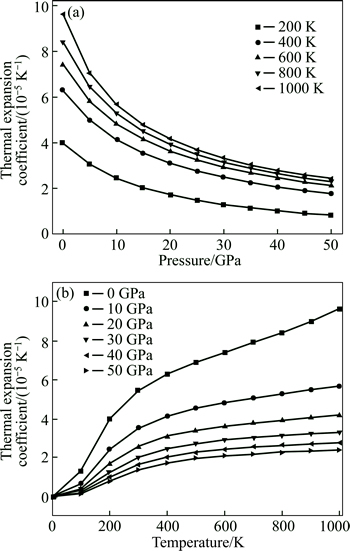
Fig. 5 Dependences of thermal expansion coefficient of Mg2La with pressure (a) and temperature (b)
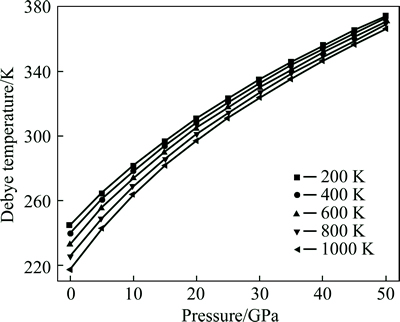
Fig. 6 Debye temperature of Mg2La as a function of pressure at different temperatures
3.3 Electronic structures
Both the local density of states (PDOS) and total density of states (TDOS) of Mg2La alloy are calculated for further investigating the bonding feature and the influence of pressure on the electronic structures. The PDOS of Mg2La is illustrated in Fig. 7(a), and TDOS is only plotted at pressure of 0, 30 and 50 GPa as shown in Fig. 7(b).
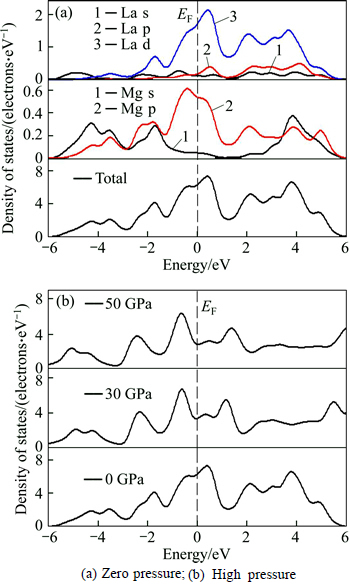
Fig. 7 DOS of Mg2La at different pressures:(Vertical dotted lines represent the Fermi level)
As shown in Fig. 7(a), we can found many energy states at Fermi level, indicating that Mg2La has metallic properties. Furthermore, the main bonding peaks are attributed to the valence electrons of Mg(s), Mg(p), La(s), La(p), and La(d) orbits normally located in energy range from -6 to 6 eV. Moreover, it is obvious that hybridization between Mg and La atoms forms covalent bonding feature.
TDOS at pressures of 0, 30 and 50 GPa is depicted for understanding the TDOS of Mg2La at different pressures shown in Fig. 7(b). The shape change of TDOS curves is slight, indicating that Mg2La maintains the stability of structure and has no structural changes at the pressure up to 50 GPa. Besides, TDOS decreases with external pressure increasing. It can be deduced that the variation of interaction potentials has happen in Mg2La because the interatomic distances shrink under pressure, which results in the reduction of the whole electronic energy levels.
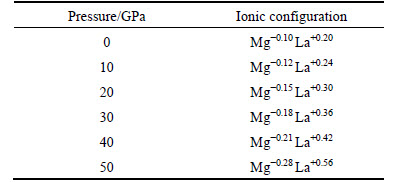
The charge populations and valence orbit track occupancy were also calculated for further understanding of the relative covalent bond and ionic bond of Mg2La at different pressures, as shown in Table 3. The (+) and (-) represent the gains and losses of electronic charge, respectively. The calculated values demonstrate that charges of Mg atom transfer to La atoms for Mg2La, and increase with increase of pressure. It��s worth noting that the absolute magnitudes of the atomic charges in the population analysis basically rely on basic settings. But the discovered charge transfer trend has important significance for better understanding of the bonding characteristics.
Table 3 Ionic configuration of Mg2La phases under pressure
4 Conclusions
This work studied the elastic, thermodynamic, and electronic properties of Mg2La at different pressures and temperatures based on the first-principle methods. We analyzed the bulk modulus, elastic constants, elastic modulus, Poisson ratio, shear modulus, and anisotropy factor of Mg2La at different pressures. The results show that the growth rate of C11 is significantly greater than that of C12 or C44, and the ductility can be improved with the increase of pressure. The bulk modulus, normalized volume, heat capacity, Debye temperature, and the coefficient of the thermal expansion are obtained by the model of quasi-harmonic Debye, and the results are explained and analyzed. Finally, The DOS and Mulliken population analysis are researched. It can be concluded that Mg2La is structurally stable and the charges of Mg atom transfer to La atoms and increase with the pressure increasing.
References
[1] POTZIES C, KAINERK U. Fatigue of magnesium alloys [J]. Advanced Engineering Materials,2004,6(6): 281-289.
[2] BLAWERT C, HORTN, KAINERK U. Automotive applications of magnesium and its alloys [J]. Transactions of the Indian Institute of Metals,2004,57(4): 397-408.
[3] NIE J F. Preface to viewpoint set on: Phase transformations and deformation in magnesium alloys [J]. Scripta Materialia, 2003, 48: 981-984.
[4] MORDIKE B L, EBERT T. Magnesium: Properties�C applications-potential [J]. Materials Science and Engineering A, 2001, 302: 37-45.
[5] WANG Yu-fei, ZHANG Wei-bing, WANG Zhi-zhong. First- principles study of structural stabilities and electronic characteristics of Mg-La intermetallic compounds [J]. Computational Materials Science,2007,41(1): 78-85.
[6] CHEN Qiu-yun, TAN Shi-yong, LAI Xin-chun. First-principles study of the elastic constants and optical properties of uranium metal [J]. Chin Phys B, 2012, 21(8): 1-8.
[7] CHEN Qiang, HUANG Zhi-wei, ZHAO Zu-de. First principles study on elastic properties, thermodynamics and electronic structural of AB2 type phases in magnesium alloy [J]. Solid State Communications, 2013,162(2): 1-7.
[8] BOUHEMADOU A , KHENATA R . Prediction study of structural and elastic properties under the pressure effect of M2GaC (M=Ti,V,Nb,Ta) [J]. Journal of Applied Physics, 2007, 102(4): 043528.
[9] SEGALL M D,LINDAN PHILIP J D,PROBERT M J, PICKARDC J,HASNIP P J,CLARK S J, PAYNE M C. First-principles simulation: Ideas, illustrations and the CASTEP code [J]. Physical, 2002, 14: 2717-2744.
[10] KOHN W, SHAM L. Self-consistent equations including exchaneg and correlation effect [J]. Physical Review, 1965, 140: 1133-1138.
[11] PACK J D , MONKHORST H J. Special points for Brillouin-zone integrations a reply [J]. Physical Review B,1977,16(12): 5188-5192.
[12] PERDEWJ P, BURKE K, ERNZERH M. Erratum: Generalized gradient approximation made simple [J]. Physical Review Lett,1996, 77(18): 3865-3868.
[13] NYLEN J. Structural relationships, phase stability and bonding of compounds PdSnn(n=2, 3, 4) [J]. Cheminform,2004,6(1): 147-155.
[14] MONKHORS H J, PACK J D. Special points for Brillouin-zone integrations [J]. Physical Review B, 1976, 13: 88-92.
[15] HAMMER B, HANSEN L B, NORSKOV J K. Improved adsorption energetics within density-functional theory using revised Perdew- Burke-Ernzerhof functionals[J]. Physical Review B, 1999, 59: 7413-7421.
[16] FISCHER T H, ALMLOF J. General methods for geometry and wave function optimization [J]. Journal of Physical Chemistry, 1992, 96(24): 9768-9774.
[17] FAN Chang-zeng, ZENG Song-yan, LI Li-xin, LIU Ri-ping. Potential super-hard Osmium di-nitride with fluorite structure: First-principles calculations [J]. Physical Review B, 2006, 74(12): 125118-125123.
[18] BLANCO M A, FRANCISCO E, LUANA V. GIBBS: Isothermal- isobaric thermodynamics of solids from energy curves using a quasi-harmonic Debye model [J]. Computer Physics Communications, 2004,158(1): 57-72.
[19] PUGHA S F. Relations between the elastic moduli and the plastic properties of polycrystalline pure metals [J]. The London, Edinburgh, and Dublin Philosophical Magazine and Journal of Science, 1954, 45: 823-843.
[20] MAYER B, ANTON H, BOTT E, METHFESSEL M, STICHT J. Ab-initio calculation of the elastic constants and thermal expansion coefficients of Laves phases [J]. Intermetallics,2003,11(1): 23-32.
[21] LU Jian-min, HU Qing-miao, YANG Rui. A comparative study of elastic constants of NiTi and NiAl alloys from first-principle calculations [J]. J Mater Sci Technol,2009,25(2): 215-218.
[22] WANG Chen-ju, GU Jian-bing, KUANG Xiao-yu. Accurate calculations of the high-pressure elastic constants based on the first- principles [J]. Chin Phys B, 2015, 24(8): 1-6.
[23] CIFTCI Y O, COLAKOGLU K, DELIGOZ E, BAYHAN U. First- Principles calculations on structure, elastic and thermodynamic properties of Al2X(X=Sc, Y) under pressure[J]. Mater Sci Technol, 2012, 28: 155-163.
[24] WANG Jing-yang, ZHOU Yan-chun .Dependence of elastic stiffness on electronic band structure of nanolaminateM2AlC(M=Ti,V,Nb, andCr) ceramics [J]. Physical ReviewB,2004,69(21): 1681-1685.
[25] HILL R. The elastic behavior of a crystalline aggregate [J]. Proceedings of the Physical Society,1952,65(5): 349-354.
[26] WU Zhi-jian, ZHAO Er-jun, XIANG, MENG Jian. Crystal structures and elastic properties of superhard IrN2 and IrN3 from first principles [J]. Physical Review B, 2007, 76(5): 15-29.
[27] BIRCH F. Finite strain isotherm and velocities for single-crystal and polycrystalline NaCl at high pressures and 300/sup 0/K [J]. Geophys. Res (United States),1978,83:b3(B3): 1257-1268.
[28] TVERGAARD V, HUTCHINSON J W. Microcracking in ceramics induced by thermal expansion or elastic anisotropy[J]. American Ceramic Society, 1988, 71: 157-166.
[29] KARKI B B, STIXRUDE L, CLARK S J, WARREN M C, ACKLAND G J. Structure and elasticity of MgO at high pressure [J]. American Mineralogist,1997,82(B6): 51-60.
[30] MAYER B, ANTON H, BOTT E, METHFESSEL M, STICHT J. Ab-initio calculation of the elastic constants and thermal expansion coefficients of Laves phases [J]. Intermetallics,2003,11(1): 23-32.
[31] DELIGOZ E, CIFTCI Y O, JOCHYM P T, COLAKOGLU K. The first principles study on PtC compound materials[J]. Materials Chemistry Physics,2008,111(1): 29-33.
[32] SIN��KO G V, SMIRNOV N A. Ab initio calculations of elastic constants and thermodynamic properties of bcc, fcc, and hcp Al crystals under pressure [J]. Journal of Physics Condensed Matter,2002,14(29): 6989-7005.
[33] DEBYE P. The theory of specific heat [J]. Annalen der Physik, 1912, 39: 789-839. (in Germany)
[34] FENG Ju-lia, XIAO Bing, ZHOU Ray, PAN Will. Thermal expansions of Ln2Zr2O7 (Ln=La, Nd, Sm, and Gd) pyrochlore[J]. Journal of Applied Physics,2012,111(10): 1-4.
[35] BUJARD P , WALKER E. Elastic constants of Cr3Si solid State [J]. Solid State Communications,1981,39(5): 667-669.
(Edited by YANG Hua)
Cite this article as: NIU Xiao-feng, HUANG Zhi-wei, HU Lei, WANG Han, WANG Bao-jian. Elasticity under pressure and thermal property of Mg2La from first-principles calculations [J]. Journal of Central South University, 2017, 24(8): 1713-1719. DOI: https://doi.org/10.1007/s11771-017-3578-6.
Foundation item: Project(51574176) supported by the National Natural Science Foundation of China; Project(143020142-S) supported by the Program for the Top Young Academic Leaders of Higher Learning Institutions of Shanxi Province (TYAL), China; Project(201603D421028) supported by the Key Research and Development Program of Shanxi Province (International Cooperative Project), China
Received date: 2016-03-23; Accepted date: 2016-07-09
Corresponding author: NIU Xiao-feng, PhD, Associate Professor; Tel: +86-13313518391; E-mail: niu.xiao.feng@126.com