Trans. Nonferrous Met. Soc. China 25(2015) 3498-3504
Corrosion of in-situ grown MgAl-LDH coating on aluminum alloy
Fen ZHANG1, Chang-lei ZHANG1, Liang SONG2, Rong-chang ZENG1, Zhen-guo LIU1, Hong-zhi CUI1
1. College of Materials Science and Engineering, Shandong University of Science and Technology, Qingdao 266590, China;
2. Qingdao Institute of Bioenergy and Bioprocess Technology, Chinese Academy of Sciences, Qingdao 266101, China
Received 24 November 2014; accepted 5 February 2015
Abstract: MgAl-layered double hydroxides (LDH) coatings were fabricated by the in-situ hydrothermal treatment method on the AA5005 aluminum alloy. The characteristics of the coatings were investigated by XRD, FT-IR, SEM and EDS. The effect of the pH value of the solution on the formation of the LDH coatings was studied. The optimum pH value of the solution was 10.0. The corrosion resistance of the LDH coatings was studied using potentiodynamic polarization tests and electrochemical impedance spectrum (EIS). The results demonstrate that the LDH coatings, characterized by platelets vertically to the substrate surface possess excellent corrosion resistance. The influence of the hydrothermal crystallization time on the corrosion resistance was evaluated. Prolonging the crystallization time can increase the corrosion resistance of the obtained LDH coatings. The anticorrosion mechanism of the LDH coatings was discussed.
Key words: aluminum alloy; layered double hydroxide; coating; corrosion
1 Introduction
Aluminum and its alloys have been extensively used outdoors because they are easy to handle and represent an important category of materials due to their high technological value and wide range of industrial applications, especially in the fields of transport, building, electrical engineering, aircraft and aerospace. However, the heterogeneous microstructure of aluminum alloys makes them particularly liable to localized corrosion, limiting their applications especially in marine environments. For many decades, chromate conversion coatings have been used for protection of aluminum alloys. However, it is well-known that the hexavalent chromate compounds are toxic and were found to induce unacceptable environment and health related hazards [1,2]. Up to now, various approaches have been proposed to develop environmentally friendly alternatives, including anodizing [3-5], conversion coatings [6-8], sol-gel synthesis [9,10], polymer coatings [11], magnetron sputtering [12,13] and self-assemble [14,15].
Layered double hydroxides (LDH) are a promising class of protection coatings for metals. LDH can be expressed by the general formula 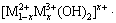
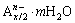 , where the cations M2+ and M3+ occupy the octahedral holes in a brucite-like layer, and the anion An- is located in the hydrated interlayer galleries [16,17]. Recently, extensive studies have been focused on the potential applications of LDH to protect metals such as magnesium alloys [18], aluminum alloys [19], and steel [20]. In the previous studies [21,22], MgAl-LDH coatings intercalated with
, where the cations M2+ and M3+ occupy the octahedral holes in a brucite-like layer, and the anion An- is located in the hydrated interlayer galleries [16,17]. Recently, extensive studies have been focused on the potential applications of LDH to protect metals such as magnesium alloys [18], aluminum alloys [19], and steel [20]. In the previous studies [21,22], MgAl-LDH coatings intercalated with  and
and 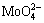 have been obtained by the co-precipitation and hydrothermal treatment method on AZ31 substrate, which possessed the ion-exchange, self-healing ability and improved anticorrosion properties. Also, this conversion coating is environmentally friendly and low cost.
have been obtained by the co-precipitation and hydrothermal treatment method on AZ31 substrate, which possessed the ion-exchange, self-healing ability and improved anticorrosion properties. Also, this conversion coating is environmentally friendly and low cost.
To date, two main methods have been developed for preparing the LDH coatings. One is the in-situ method, and the other is the two-step method, in which the LDH powder precursor is firstly synthesized by the co- precipitation method and then the coating is fabricated using a certain process. ZHANG et al [23] obtained ZnAl-LDH�CNO3 and ZnAl-LDH-laurate using the in-situ method on the porous anodic alumina/aluminum (PAA/Al) substrates. The existence of the LDH coating enhanced the anticorrosion properties of the aluminum metal substrate. BUCHHEIT et al [24] reported that Al-Zn-decavanadate hydrotalcite compounds dispersed as a particulate additive in organic resins lead to potent corrosion inhibition of an underlying aluminum alloy substrate. VEGA et al [25] synthesized ZnAl-vanadate hydrotalcite on aluminum alloy using the co- precipitation and air-spraying method. WANG et al [26] synthesized the 8-hydroxyquinolate intercalated MgAl- hydrotalcite coating on pure aluminum sheet by the chemical conversion treatment.  et al [27] found that the addition of hydrotalcite to sol-gel films improved the corrosion resistance of coated AA2024-T3 alloy in salt spray. ZHELUDKEVICH et al [28] observed the corrosion inhibition effect by adding the LDH powder to an epoxy resin applied on aluminum alloy. Those above coatings fabricated using the two-step method improved the corrosion resistance of their substrates; however, the adhesion of the coating to the substrate was much poor. Furthermore, the LDH coating preparation processes described above are complicated and have not yet achieved the desired level of corrosion resistance. The in-situ method expected to fabricate the LDH coatings on aluminum alloys with high corrosion resistance and good adhesion to the substrate using a simple technological process remains a considerable challenge. The present work aims to prepare the MgAl-LDH coatings on aluminum alloy using the one-step method. The effect of the pH value of the solution on the formation of the LDH coatings was investigated. Besides, the influence of the hydrothermal crystallization time on the corrosion resistance was evaluated.
et al [27] found that the addition of hydrotalcite to sol-gel films improved the corrosion resistance of coated AA2024-T3 alloy in salt spray. ZHELUDKEVICH et al [28] observed the corrosion inhibition effect by adding the LDH powder to an epoxy resin applied on aluminum alloy. Those above coatings fabricated using the two-step method improved the corrosion resistance of their substrates; however, the adhesion of the coating to the substrate was much poor. Furthermore, the LDH coating preparation processes described above are complicated and have not yet achieved the desired level of corrosion resistance. The in-situ method expected to fabricate the LDH coatings on aluminum alloys with high corrosion resistance and good adhesion to the substrate using a simple technological process remains a considerable challenge. The present work aims to prepare the MgAl-LDH coatings on aluminum alloy using the one-step method. The effect of the pH value of the solution on the formation of the LDH coatings was investigated. Besides, the influence of the hydrothermal crystallization time on the corrosion resistance was evaluated.
2 Experimental
2.1 Fabrication of MgAl-LDH coatings
The material used in this study was AA5005 aluminium alloy. Prior to coating, the alloy sheet was ground to 1500 grit SiC paper, then immersed in 0.5% aqueous NaOH solution for 1 min in order to remove the oxidation on the surface, and finally ultrasonically cleaned in ethyl alcohol for 10 min. 0.007 mol Mg(NO3)2��6H2O and 0.042 mol NH4NO3 were dissolved in 70 mL deionized water, and 1.0% ammonia solution was used to adjust the solutoin pH. The above solution was then transferred to a Teflon-lined autoclave in which the pretreated aluminium alloy was immersed. The Teflon-lined autoclave was then heated at 398 K for different time. The as-prepared samples were rinsed with de-ionized water and dried under ambient conditions.
2.2 Characterization
The structures of the LDH coatings were examined by using an X-ray diffractometer (XRD) (D/Max 2500PC) with a Cu target (l=0.1542 nm). Fourier- transform infrared (FT-IR) spectrum was obtained on a TENSOR-27 spectrophotometer using the KBr pellet technique. The morphologies of the surface and cross-section of the LDH coatings were observed with a field-emission scanning electronic microscope (FE-SEM, Hitachi S-4800). All samples were sputtered with gold. The adhesion of the LDH coating on the surface of the aluminum alloy substrate was tested according to the method reported in Ref. [29]. Potentiodynamic polarization curves and electrochemical impedance spectra (EIS) in 3.5% NaCl solution were obtained in a cell using a Princeton potentiostat (model 2273). A classical three-electrode system was used with the sample as the working electrode (1 cm2), a saturated calomel electrode (SCE) as the reference electrode, and a platinum plate as the counter electrode. The samples were immersed in the medium for 20 min before the electrochemical tests. The polarization curves were recorded with a sweep rate of 2 mV/s. EIS measurements were acquired from 100 kHz down to 10 mHz using a 5 mV amplitude perturbation.
3 Results and discussion
3.1 Characterization of MgAl-LDH coatings
The influence of the solution pH on the formation of MgAl-LDH coatings was investigated by XRD. Figure 1 illustrates the XRD patterns of the obtained LDH coatings with the same hydrothermal treatment period of 8 h under different pH conditions. As presented in Figs. 1(b) and (c), there are almost no characteristic peaks of the LDH phase when the solution pH values of 8 and 9 were adopted. While Figs. 1(d)-(f) show characteristic reflections of an LDH phase [30] when pH values of 9.5, 10.0 and 10.5 were used. In addition to the characteristic peaks of the bare aluminum alloy substrate, the XRD patterns of MgAl-LDH coatings showed two reflections below 25�� which can be assigned to (003) and (006) reflections of the LDH phase. It is noted that the intensity of the peaks of the LDH phase is the strongest when the solution pH of 10.0 was used. The LDH phase presents a lower crystallinity while a larger pH of 10.5 was adopted.
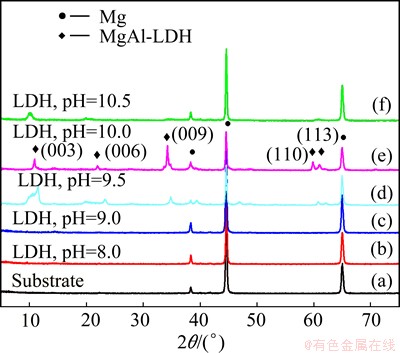
Fig. 1 XRD patterns of LDH coatings formed on aluminium alloy depending on solution pH of hydrothermal treatment
The solid crystallinity is enhanced by using the solution with a higher pH. The pH of the solution plays a key role in the formation of MgAl-LDH coatings by the in-situ synthesis method. The optimized pH value of the solution is 10.0, which is consistent with the result obtained by the coprecipitation method [31]. It is well-known that when aluminum substrates are placed in solutions with high pH values, the surface is partially dissolved, which generated aluminum oxides [29]. Combination of Al3+ ions released from the alumina with Mg2+ ions coming from the solution will then afford MgAl-LDH coating.
The XRD patterns (Fig. 2) demonstrate the effect of the hydrothermal treatment period on the formation of the MgAl-LDH coatings with the optimized solution pH of 10.0. As shown in Fig. 2, for LDH coatings with different crystallization periods, the XRD patterns (Figs. 2(b) and (c)) show an increase of reflection intensity with the increase of crystallization period.

Fig. 2 XRD patterns of LDH coatings formed on aluminium alloy depending on hydrothermal treatment period
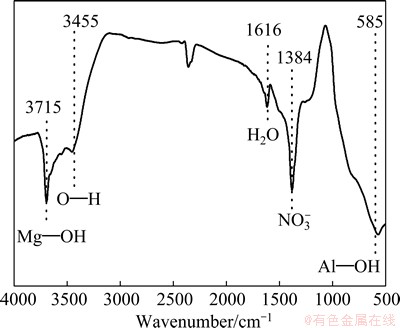
Fig. 3 FT-IR spectrum of LDH powder scraped from as-prepared MgAl-LDH coating treated for 8 h
Figure 3 shows the FT-IR spectrum of the powder scraped from the as-prepared MgAl-LDH coating. The band with the maximum peak at 1384 cm-1 confirmed the presence of NO3- anions that are intercalated in the interlayer [32]. NO3- anions are readily exchanged with other anions [33], giving the potential ability to further functionalize the coating by intercalation of appropriate anions. The absorption band at 3715 cm-1 corresponds to Mg��O��H stretching vibrations due to the magnesia octahedron structure of LDH. The absorption band at around 3455 cm-1 corresponds to O��H because of the presence of the surface absorption water and interlayer water [19]. The band at about 1616 cm-1 can be ascribed to the bending vibration of crystal water. Additionally, the band observed at about 585 cm-1 is mainly due to Al��OH lattice vibrations [34].
The SEM morphologies of the as-prepared LDH coated samples treated for 8 h are shown in Fig. 4. As can be seen in Fig. 4(a), the LDH coating is compact over the entire aluminum alloy substrate. Higher- magnification SEM images of the coating (Figs. 4(b) and (c)) clearly show that most of the LDH microcrystals have a curved platelet morphology. It is worth noting that some LDH intercalated compounds reported earlier also show a curved platelike/sheet morphology [35,36], formed by the cooperative organization of intercalated anions and cationic metal hydroxide layers, whereas the microstructure obtained here is different from that in Refs. [35,36]. The cross-section view (Fig. 4(d)) shows that a continuous layer of the LDH coating grows vertically on the substrate with a thickness of about 12 ��m.
Strong adhesion of anticorrosion coatings to the metal surface is essential if the coatings are useful in practice. As can be seen in Fig. 5, there is no delamination or peeling occurred on the cross-cutting surface, indicating that the LDH coatings have a strong adhesion to the metal substrate.
3.2 Corrosion resistance of LDH coatings
Potentiodynamic polarization is a commonly used technique that was employed to investigate the corrosion resistance of the LDH coatings. Figure 6 presents the potentiodynamic polarization curves of the LDH-coated samples as well as the aluminium alloy substrate. In a typical polarization curve, lower corrosion current density (Jcorr) corresponds to lower corrosion rate and better corrosion resistance. The current densities of the LDH-coated samples are 2.19��10-6, 1.20��10-6 and 0.983��10-6 A/cm2 for hydrothermal treatment periods of 4, 8 and 12 h, respectively, which are much lower than that of the substrate (4.27��10-5 A/cm2). Jcorr for the LDH-coated samples decreases more than by one order of magnitude compared with that of the aluminum alloy substrate. This suggests that the presence of the LDH coating considerably increases the chemical resistance of the aluminum alloy substrate. The significant reduction of the Jcorr implies that the LDH coatings can help to decrease the corrosion rate of the substrate. Also, it can be concluded that prolonging the hydrothermal treatment time leads to an increase of the anticorrosion property.

Fig. 4 SEM images of top (a, b, c) and cross-section (d) views of MgAl-LDH coating treated for 8 h on aluminum alloy surface

Fig. 5 Polarized light microscope image for MgAl-LDH coating treated for 8 h on aluminum alloy surface after adhesion test by ASTM standard D 3359-02 method B

Fig. 6 Tafel polarization curves in 3.5% NaCl solution of bare aluminum alloy, and LDH coatings on aluminum alloy with different crystallization time
Besides, it is worth noting that the breakdown potential (��b) of the coating is another parameter reflecting the corrosion resistance property besides the corrosion current. ��b of the aluminum alloy is -0.72 V (vs SCE), while the value of the LDH coating-4 h sample is -0.67 V (vs SCE). In particular, as for the LDH coating-8 h sample, there are two obvious ��b (-0.39 V, 0.01 V (vs SCE)) and one obvious passivation zone in the anodic branch of polarization curve. A reasonable explanation for this result is that the LDH coatings have a self-healing ability [21].
In order to further provide the information on the corrosion inhibition effect of the LDH coatings, EIS was carried out to analyze the corrosion resistance of the coating. The Bode plots and the Nyquist plots of the samples are presented in Figs. 7(a) and (b), respectively. Because the dimension of the Nyquist plot for AA5005 aluminum alloy is too small to be discerned in comparison with that of the alloy with hydrothermal treatment, an enlarged plot is inserted.
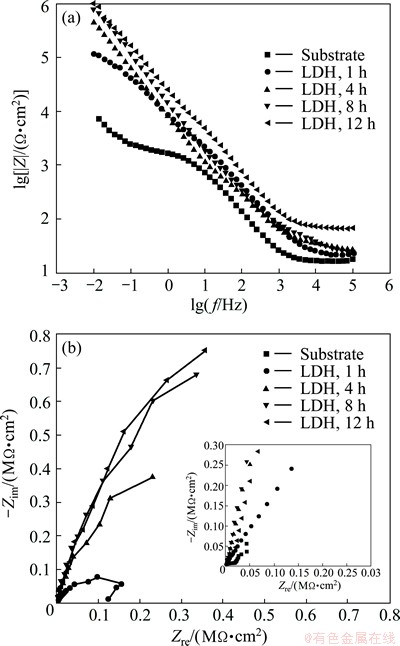
Fig. 7 Bode (a) and Nyquist (b) plots for MgAl-LDH coatings on aluminum alloy and bare aluminum alloy electrodes immersed in 3.5% NaCl solution
It can be observed from the Bode plots that the LDH-coated samples exhibit larger impedance at low frequency compared with the bare aluminum alloy sample (Fig. 7(a)). It is also noted that the low frequency impedance is considerably high, approximately 106 ����cm2, for the LDH-coated sample treated for 8 h and 12 h. Concurrently, it can be seen from the Nyquist plot (Fig. 7(b)) that the largest radii of curvature for the samples treated for 8 h and 12 h indicate that those samples possess higher corrosion protection ability. These results demonstrate that the impedance of the LDH-coated samples increases with prolonging hydrothermal treatment time, leading to the full crystallization of the LDH coating in accordance with the XRD results presented in Fig. 2. While when the crystallization time exceeds 8 h, the increase of the corrosion resistance is less obvious.
3.3 Protection ability of LDH coatings
Figure 8 shows the surface morphologies of the original LDH-coated sample and the LDH-coated sample immersed in 3.5% NaCl solution for 7 d. After immersion for 7 d, no noticeable changes were observed in the immersed LDH coating (Fig. 8(b)) and most of the area of the sample still exhibits a platelet structure of curved hexagonal morphology, similar to the original sample (Fig. 8(a)). However, some plates collapsed after immersion (marked by the arrow in Fig. 8(b)), which may be due to the dissolution of the platelet. It can be observed from the inset of Fig. 8 that the original LDH coating (Fig. 8(a)) is mainly composed of Mg, Al, O and C elements, with no Cl signal. However, Cl signals (Fig. 8(b)) were observed on the same sample after immersion, which indicates that the LDH coating exhibits ion exchange effect by absorbing Cl- and Na+ from NaCl solution and the interlayer of the LDH is able to retain Cl- and Na+ in the LDH structure.

Fig. 8 SEM images of original LDH coating-8 h sample (a) and immersed sample (b) (Insets are corresponding EDS spectra)
Besides, it is noted that the peak position of (003) shifts to a large angle of approximately 0.2�� (Fig. 9(b)), indicating that Cl- ions are intercalated by ion exchange. The improvement in the corrosion performance of the LDH coating can also be attributed to the adsorption and retention of corrosive Cl- ions and the release of NO3- ions. TEDIM et al [37] proved that nitrate-containing LDHs are effective chloride nanotraps due to the ion exchange process; therefore, the LDHs can delay coating degradation and the initiation of corrosion. Due to the ion exchange effect, Cl- can hardly reach the metal substrate. The ion exchange reaction of the LDH coating on the aluminum alloy in chloride-containing solution can be expressed as follows:
LDH-NO3-+Cl-��LDH-Cl-+NO3- (1)
This confirms that MgAl-LDH coating is able to provide corrosion protection.
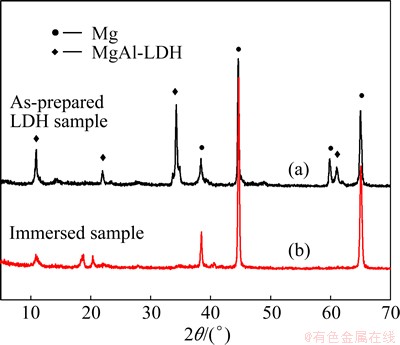
Fig. 9 XRD spectra of LDH coating-8 h sample before and after immersion in 3.5% NaCl solution for 7 d
4 Conclusions
1) The nitrate intercalated MgAl-LDH coating with curved platelet structure was fabricated by the in-situ hydrothermal treatment method. The coating consists of compact, homogeneous and well-crystallized nanostructure.
2) The corrosion current density of the LDH coatings is decreased by one order of magnitude compared with that of the AA5005 alloy. The as-prepared LDH coating can provide an effective protection to aggressive species.
3) The corrosion protection ability of the LDH coatings may be due to ion exchange effect and competitive adsorption for chloride ions on the alloy surface.
References
[1] ZUBILLAGA O, CANO F J, AZKARATE I, MOLCHAN I S, THOMPSON G. E, SKELDON P. Anodic films containing polyaniline and nanoparticles for corrosion protection of AA2024T3 aluminium alloy [J]. Surface and Coatings Technology, 2009, 203: 1494-1501.
[2] BOISIER G, LAMURE A,  N, PORTAIL N, VILLATTE M. Corrosion protection of AA2024 sealed anodic layers using the hydrophobic properties of carboxylic acids [J]. Surface and Coatings Technology, 2009, 203: 3420-3426.
N, PORTAIL N, VILLATTE M. Corrosion protection of AA2024 sealed anodic layers using the hydrophobic properties of carboxylic acids [J]. Surface and Coatings Technology, 2009, 203: 3420-3426.
[3] ARENAS M A, CONDE A, de DAMBORENEA J J. Effect of acid traces on hydrothermal sealing of anodising layers on 2024 aluminium alloy [J]. Electrochimica Acta, 2010, 55: 8704-8708.
[4] DU Nan, WANG Shuai-xing, ZHAO Qing, SHAO Zhi-song. Effects of boric acid on microstructure and corrosion resistance of boric/sulfuric acid anodic film on 7050 aluminum alloy [J]. Transactions of Nonferrous Metals Society of China, 2012, 22(9): 1655-1660.
[5] VENUGOPAL A, PANDA R, MANWATKAR S, SREEKUMAR K, KRISHNA L. RAMA, SUNDARARAJAN G. Effect of micro arc oxidation treatment on localized corrosion behaviour of AA7075 aluminum alloy in 3.5% NaCl solution [J]. Transactions of Nonferrous Metals Society of China, 2012, 22(3): 700-710.
[6] JOHANSEN HERBERT D, BRETT CHRISTOPHER M A, MOTHEO ARTUR J. Corrosion protection of aluminium alloy by cerium conversion and conducting polymer duplex coatings [J]. Corrosion Science, 2012, 63: 342-350.
[7] TIWARI A, HIHARA L H. High performance reaction-induced quasi-ceramic silicone conversion coating for corrosion protection of aluminium alloys [J]. Progress in Organic Coatings, 2010, 69: 16-25.
[8] SHI Hong-wei, HAN En-hou, LIU Fu-chun, KALLIP S. Protection of 2024-T3 aluminium alloy by corrosion resistant phytic acid conversion coating [J]. Applied Surface Science, 2013, 280: 325-331.
[9] DALMORO V, DOS SANTOS J H Z, ARMELIN E, ALEM N C, AZAMBUJA D S. A synergistic combination of tetraethylorthosilicate and multiphosphonic acid offers excellent corrosion protection to AA1100 aluminum alloy [J]. Applied Surface Science, 2013, 273: 758-768.
[10] VOEVODIN N N, KURDZIEL J W, MANTZ R. Corrosion protection for aerospace aluminum alloys by modified self- assembled nanophase particle (MSNAP) sol�Cgel [J]. Surface and Coatings Technology, 2006, 201: 1080-1084.
[11] SHABANI-NOOSHABADI M, GHOREISHI S M, BEHPOUR M. Electropolymerized polyaniline coatings on aluminum alloy 3004 and their corrosion protection performance [J]. Electrochimica Acta, 2009, 54: 6989-6995.
[12]  H, STOCK H R. Improving the corrosion protection of aluminium alloys using reactive magnetron sputtering [J]. Corrosion Science, 2005, 47: 953-964.
H, STOCK H R. Improving the corrosion protection of aluminium alloys using reactive magnetron sputtering [J]. Corrosion Science, 2005, 47: 953-964.
[13] DIESSELBERG M, STOCK H R, MAYR P. Corrosion protection of magnetron sputtered TiN coatings deposited on high strength aluminium alloys [J]. Surface and Coatings Technology, 2004, 177-178: 399-403.
[14] QU Jun-e, CHEN Geng, WANG Hai-ren, NIE De-jian. Effect of water content on corrosion inhibition behavior of self-assembled TDPA on aluminum alloy surface [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(10): 3137-3144.
[15] ZUO Ke, WANG Xin, LIU Wei, ZHAO Yue. Preparation and characterization of Ce-silane-ZrO2 composite coatings on 1060 aluminum [J]. Transactions of Nonferrous Metals Society of China, 2014, 24(6): 1474-1480.
[16] MIYATA S, OKADA A. Synthesis of hydrotalcite-like compounds and their physico-chemical properties-the systems Mg2+�CAl3+�CSO4 2- and Mg2+�CAl3+�CCrO42- [J]. Clays and Clay Minerals, 1977, 25: 14-18.
[17] MA Ren-zhi, LIU Zhao-ping, LI Liang, IYI N, SASAKI T. Exfoliating layered double hydroxides in formamide: A method to obtain positively charged nanosheets [J]. Journal of Materials Chemistry, 2006, 16: 3809-3813.
[18] LIN Jun-kai, JENG Kai-li, UAN Jun-yen. Crystallization of a chemical conversion layer that forms on AZ91D magnesium alloy in carbonic acid [J]. Corrosion Science, 2011, 53: 3832-3839.
[19] CHEN Hong-yun, ZHANG Fa-zhi, FU Shan-shan, DUAN Xue. In situ microstructure control of oriented layered double hydroxide monolayer films with curved hexagonal crystals as superhydrophobic materials [J]. Advanced Materials, 2006, 18: 3089-3093.
[20] MONTEMOR M F, SNIHIROVA D V, TARYBA M G, LAMAKA S V, KARTSONAKIS I A, BALASKAS A C, KORDAS G C, TEDIM J, KUZNETSOVA A, ZHELUDKEVICH M L, FERREIRA M G S. Evaluation of self-healing ability in protective coatings modified with combinations of layered double hydroxides and cerium molybdate nanocontainers filled with corrosion inhibitors [J]. Electrochimica Acta, 2012, 60: 31-40.
[21] ZENG Rong-chang, LIU Zhen-guo, ZHANG Fen, LI Shuo-qi, CUI Hong-zhi, HAN En-hou. Corrosion of molybdate intercalated hydrotalcite coating on AZ31 Mg alloy [J]. Journal of Materials Chemistry A, 2014, 2: 13049-13057.
[22] ZHANG Fen, LIU Zhen-guo, ZENG Rong-chang, LI Shuo-qi, CUI Hong-zhi, SONG Liang, HAN En-hou. Corrosion resistance of Mg�CAl-LDH coating on magnesium alloy AZ31 [J]. Surface and Coatings Technology, 2014, 258: 1152-1158.
[23] ZHANG Fa-zhi, ZHAO Li-li, CHEN Hong-yun, XU Sai-long, EVANS D G, DUAN Xu. Corrosion resistance of superhydrophobic layered double hydroxide films on aluminum [J]. Angewandte Chemie International Edition, 2008, 47: 2466-2469.
[24] BUCHHEIT RUDOLPH G, GUAN Hong, MAHAJANAM S, WONG F. Active corrosion protection and corrosion sensing in chromate-free organic coatings [J]. Progress in Organic Coatings, 2003, 47: 174-182.
[25] VEGA J M, GRANIZO N, de la FUENTE D, SIMANCAS J, MORCILLO M. Corrosion inhibition of aluminum by coatings formulated with Al-Zn�Cvanadate hydrotalcite [J]. Progress in Organic Coatings, 2011, 70: 213-219.
[26] WANG Li-da, ZHANG Kai-yue, HE Hao-ran, SUN Wen, ZONG Qiu-feng, LIU Gui-chang. Enhanced corrosion resistance of MgAl hydrotalcite conversion coating on aluminum by chemical conversion treatment [J]. Surface and Coatings Technology, 2013, 235: 484-488.
[27]  Characterization of hybrid sol�Cgel coatings doped with hydrotalcite-like compounds to improve corrosion resistance of AA2024-T3 alloys [J]. Progress in Organic Coatings, 2010, 67: 152-160.
Characterization of hybrid sol�Cgel coatings doped with hydrotalcite-like compounds to improve corrosion resistance of AA2024-T3 alloys [J]. Progress in Organic Coatings, 2010, 67: 152-160.
[28] ZHELUDKEVICH M L, POZNYAK S K, RODRIGUES L M, RAPS D, HACK T, DICK L F, NUNES T, FERREIRA M G S. Active protection coatings with layered double hydroxide nanocontainers of corrosion inhibitor [J]. Corrosion Science, 2010, 52: 602-611.
[29] BEVING D E, MCDONNELL A M P, YANG Wei-shen, YAN Yu-shan. Corrosion resistant high-silica-zeolite MFI coating: One general solution formulation for aluminum alloy AA-2024-T3, AA-5052-H32, AA-6061-T4, and AA-7075-T6 [J]. Journal of the Electrochemical Society B, 2006, 153: 325-329.
[30] VIALAT P, LEROUX F, TAVIOT-GUEHO C, VILLEMURE G, MOUSTY C. Insights into the electrochemistry of (CoxNi(1-x))2Al�C NO3 layered double hydroxides [J]. Electrochimica Acta, 2013, 107: 599-610.
[31] CAVANI F, TRIFIR F, VACCARI A. Hydrotalcite-type anionic clays: Preparation, properties and applications [J]. Catalysis Today, 1991, 11: 173-301.
[32] GUO Xiao-xiao, XU Sai-long, ZHAO Li-li, LU Wei, ZHANG Fa-zhi, EVANS D G., DUAN Xu. One-step hydrothermal crystallization of a layered double hydroxide/alumina bilayer film on aluminum and its corrosion resistance properties [J]. Langmuir, 2009, 25: 9894-9897.
[33] WILLIAMS G R,  D. Towards understanding, control and application of layered double hydroxide chemistry [J]. Journal of Materials Chemistry, 2006, 16: 3065-3074.
D. Towards understanding, control and application of layered double hydroxide chemistry [J]. Journal of Materials Chemistry, 2006, 16: 3065-3074.
[34] ARAMENDIA A M, BORAU V, JIMENEZ C, MARINAS J M, RUIZ J R, URBANO F J. Comparative study of Mg/M(III) (M=Al, Ga, In) layered double hydroxides obtained by coprecipitation and the sol�Cgel method [J]. Journal of Solid State Chemistry, 2002, 168: 156-161.
[35] XU Zhi-ping, BRATERMAN P S. High affinity of dodecylbenzene sulfonate for layered double hydroxide and resulting morphological changes [J]. Journal of Materials Chemistry, 2003, 13: 268-273.
[36] OGAWA M, ASAI S. Hydrothermal synthesis of layered double hydroxide-deoxycholate intercalation compounds [J]. Chemistry of Materials, 2000, 12: 3253-3255.
[37] TEDIM J, KUZNETSOVA A, SALAK A N, MONTEMOR F, SNIHIROVA D, PILZ M, ZHELUDKEVICH M L, FERREIRA M G S. Zn�CAl layered double hydroxides as chloride nanotraps in active protective coatings [J]. Corrosion Science, 2012, 55: 1-4.
���Ͻ����ԭλ�Ʊ�MgAl-LDHͿ�㼰����ʴ��
�� ��1���Ų���1���� ��2�����ٲ�1�������1����֥1
1. ɽ���Ƽ���ѧ ���Ͽ�ѧ�빤��ѧԺ���ൺ 266590��
2. �й���ѧԺ �ൺ������Դ������о������ൺ 266101
ժ Ҫ������ԭλˮ�ȷ������Ͻ�AA5005�����Ʊ�þ����״˫��������(LDH)Ϳ�㣻����X�������䡢����Ҷ���⡢ɨ����������Լ��������о�Ϳ��Ļ�ѧ�ɷ֡�������ò�����ࣻ������ҺpHֵ���γ�LDH��Ӱ�졣�������������ҺpHֵΪ10.0ʱ�����õ���LDHͿ��ᾧ��á�ͨ������λ�绯ѧ�����Լ��绯ѧ�迹�о�ˮ�ȷ�Ӧʱ���LDHͿ����ʴ���ܵ�Ӱ�졣����ˮ�ȷ�Ӧʱ����ӳ���Ϳ�����ʴ����ߣ�����ˮ�ȷ�Ӧʱ��ﵽ8 hʱ���ټ����ӳ���Ӧʱ�䣬����ʴ����߲����ԡ����⣬��̽����LDHͿ�����ʴ������
�ؼ��ʣ����Ͻ𣻲�״˫�������Ϳ�㣻��ʴ
(Edited by Xiang-qun LI)
Foundation item: Project (20133718120003) supported by the Doctoral Program Foundation of State Education Ministry, China; Project (BS2013CL009) supported by the Scientific Research Foundation of Shandong for Outstanding Young Scientist, China; Projects (13-1-4-217-jch, 13-1-4-188-jch) supported by the Applied Basic Research Foundation of Qingdao, China; Project (2014TDJH104) supported by the SDUST Research Fund, China
Corresponding author: Fen ZHANG; Tel: +86-532-86081226; E-mail: zhangfen2011@hotmail.com
DOI: 10.1016/S1003-6326(15)63987-5