J. Cent. South Univ. Technol. (2009) 16: 0212-0217
DOI: 10.1007/s11771-009-0036-0

Novel fluorescence sensor based on covalent immobilization of 3-amino-9-ethylcarbazole via electrostatically assembled gold nanoparticle layer
TAN Shu-zhen(谭淑珍), LONG Shu(龙 姝), XIA Jiao-yun(夏姣云),
CAO Zhong(曹 忠), ZHANG Ling(张 玲), GONG Fu-chun(龚福春)
(School of Chemistry and Biological Engineering, Changsha University of Science and Technology,
Changsha 410004, China)
Abstract: A novel technique of immobilizing indicator dyes by electrostatic adsorption and covalent bonding to fabricate optical sensors was developed. 3-Amino-9-ethylcarbazole (AEC) was attached to the outmost surface of quartz glass slide via aminosilanizing the slide, crosslinking chitosan, adsorbing Au nanoparticle, self-assembling HS(CH2)11OH, and coupling AEC. Thus, an AEC-immobilized optical sensor was obtained. The sensor exhibits a wide linear response range from 7.0×10-7 to 1.0×10-4 mol/L and a correlation coefficient of 0.995 9 for the detection of 2-nitrophenol. The detection limit and response time of the sensor are 1.0×10-7 mol/L and less than 10 s, respectively. The fluorescence intensity of the used sensor can be restored to the blank value by simply rinsing with blank buffer. A very effective matrix for immobilizing indicator dye is provided by the proposed technique, which is adaptable to other indicator dyes with amino groups besides AEC.
Key words: optical sensor; chitosan; Au nanoparticle; 3-amino-9-ethylcarbazole; 2-nitrophenol; indicator dye
1 Introduction
Optical sensors provide promising ways for solving analytical tasks because of their advantages of easy fabrication, high sensitivity and low cost. the technique of immobilizing indicator dyes plays an important role in development and design of optical sensors. The reported methods include physical entrapment [1-2], electrostatic adsorption [3-5] and covalent bonding [6-14]. Although physical entrapment is a very simple way and can give high yield at low cost, the durability of the sensing layer fabricated is rather poor since indicator dyes can easily leach out of the sensor matrix. The electrostatic adsorption and covalent bonding have considerable strength. Covalent bonding of indicator dyes has attracted research interest since it can prevent dye from leaching, resulting in an enhanced lifetime. A research group in Hunan University, China, reported that some indicator dyes can be covalently immobilized by UV photopolymerization [12-14]. The obtained sensors have high sensitivity, showing no dye-leaching. But it is usually difficult to control the thickness of the sensing layer in the preparing process, and sometimes it is not easy to restore the fluorescence intensity of the used sensor to the blank value by rinsing because indicator dyes or dye-analyte complexes are distributed in the whole matrix of the senor rather than on its outmost surface. To solve this problem, a new method of immobilizing indicator dyes was investigated.
STELLACCI et al [15] reported that a self-assembled layer of chromophores on a metal nanoparticle core can pack about 2 500 chromophores within a sphere with an diameter of less than 10 nm. They believe that metal nanoparticles are promising to be used as carriers for anchoring large numbers of chromophores, and may have an impact on ultrasensitive detection. In this work, we employed Au nanoparticles as bridges and carriers for anchoring indicator dyes on the surface of quartz glass slide. The overall fabricating procedure of the sensor was as follows: aminosilanization of the quartz glass slide, crosslink of chitosan by using glutaraldehyde, immobilization of Au nanoparticles by electrostatic adsorption, self-assembly of HS(CH2)11OH, and covalent bonding of indicator dyes with amino groups by using cyanuric chloride as coupling agent. 3-Amino-9- ethylcarbazole (AEC) was selected as an example of indicator dye. An AEC-immobilized sensor was obtained in this way and used to assay 2-nitrophenol.
2 Experimental
2.1 Reagents
3-Amino-9-ethylcarbazole (AEC, 90%) and (3-aminopropyl)trimethoxysilane (APS, 85%) were purchased from Acros Organics, USA. 11-Mercapto-1- undecanol (97%) and cyanuric chloride (99%) were purchased from Aldrich Chemical Co, USA. Chloroauric acid (HAuCl4) and chitosan (75%-85% deacetylation) were acquired from Sigma-Aldrich Inc., USA. Sodium citrate, sodium borohydride, 2-nitrophenol and the other materials were of analytical grade and obtained from Changsha Chemical Reagents, China. All reagents were used as received without further purification. Doubly distilled water was used throughout. Britton-Robinson (B-R) buffer solutions with various pH values were obtained by mixing appropriate amounts of phosphoric acid, acetic acid and boric acid, and pH was adjusted by sodium hydroxide. A stock solution of 2-nitrophenol (1.43×10-3 mol/L) was acquired by dissolving 2-nitrophenol in doubly distilled water. Working solutions of 2-nitrophenol with different concentrations were obtained by serial dilution of the stock solution with water.
2.2 Instrument
All fluorescence measurements were performed on a Perkin Elmer LS-55 luminescence spectrometer. The excitation and emission slits were set at 4.0 and 10.0 nm, respectively. Excitation and emission wavelengths of 242 and 393 nm were used, respectively. The AEC- immobilized sensor was placed in a quartz glass vessel containing solutions of 2-nitrophenol or blank B-R buffer. Between two assays, doubly distilled water was used to wash the sensor until its original blank fluorescence intensity was restored. A transmission electron micrograph (TEM) of Au nanoparticles was taken using an FEI Tecnai G2 20 electron microscope (Netherlands) equipped with Gatan 794 cooled multi-scan CCD TV camera for high-resolution imaging. For TEM study, the sample was prepared by adding drops of freshly prepared Au colloid solution on a carbon film supported on a Cu grid. Measurements of pH were performed with a Mettler Toledo Delta 320 pH meter.
2.3 Preparation of colloidal Au
All glasswares used in the following procedures were cleaned in a bath of freshly prepared solution (the volume ratio of HNO3 to HCl is 3?1), thoroughly washed with water and dried prior to use. Colloidal Au was prepared according to Refs.[16-17] with a slight modification. A 1 mL aliquot of HAuCl4 (1%, mass fraction) was dissolved in 90 mL of millipore purified water at room temperature under vigorous stirring. Then, 2 mL of 38.8 mmol/L sodium citrate was added and stirred for 2 min. 1 mL of 0.075% NaBH4 made freshly by dissolving the salt in 38.8 mmol/L sodium citrate was added to this solution, and the resultant colloid was stirred for another 5 min. This method produced an Au nanoparticle colloid with an average diameter of about 20 nm, as characterized by transmission electron micro- scopy.
2.4 Preparation of AEC-immobilized sensor
The overall procedure is schematically illustrated in Fig.1 and described in detail as follows.
(1) Cleaning of the quartz glass slide [18]. The slide was washed successively with 3% hydrofluoric acid, 10% hydrogen peroxide and doubly distilled water, and then dried at room temperature.
(2) Aminosilanization of the glass slide. The cleaned glass slide was submerged into ethanol containing 2% APS for 2 h, then taken out and rinsed successively with water and ethanol to remove the unbound materials from the surface. Thus, an APS monolayer was formed on the surface of the slide.
(3) Attachment of glutaraldehyde. The APS-coated slide was soaked in 5% glutaraldehyde aqueous solution for 2 h, then taken out, rinsed with water and dried.
(4) Crosslink of chitosan. 1 mL of acetic acid and 50 mL of chitosan aqueous solution (1%) were mixed and stirred for 1 h. A colloidal solution was formed. Then the slide was dipped in the colloidal solution. After 5 h, the slide was removed, rinsed with water and dried.
(5) Immobilization of Au nanoparticles. The chitosan-coated slide was immersed in the Au colloidal solution for 12 h, then removed, rinsed with water and dried.
(6) Self-assembly of HS(CH2)11OH. The Au-coated slide was soaked in HS(CH2)11OH ethanolic solution (1.0×10-3 mol/L) for 24 h, and then rinsed with ethanol.
(7) Covalent bonding of AEC. Cyanuric chloride (18.6 mg) and AEC (21.7 mg) were dissolved in 10 mL of acetone. After stirring at room temperature for 3 h, a clear solution was formed [19-20]. The HS(CH2)11OH- coated slide was submerged in this solution for 3 h, and then rinsed with acetone. Thus, the indicator dye, AEC, was immobilized onto the outmost surface of the slide, and the AEC-immobilized sensor was acquired.
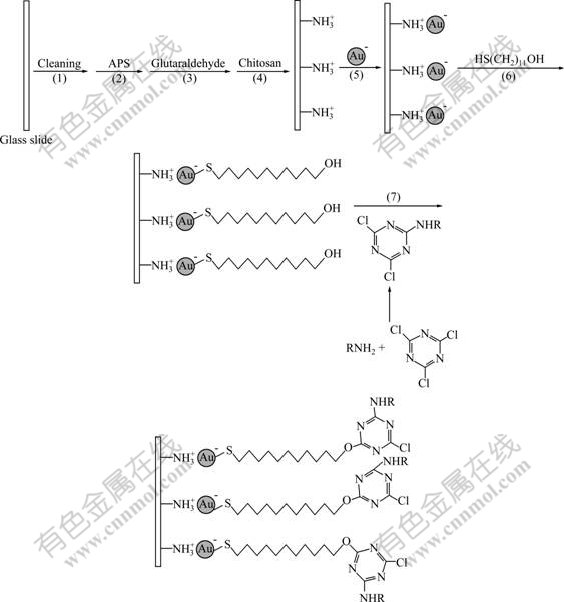
Fig.1 Fabricating procedure of AEC-immobilized sensor (RNH2: indicator dye with amino group)
3 Results and discussion
3.1 Preparation of AEC-immobilized sensor
A self-assembled layer of chromophores on a metal nanoparticle core allows for packing of large numbers of chromophores. Therefore, metal nanoparticles can be used as bridges and carriers of anchoring indicator dyes in the preparation of optical sensors. The application of Au nanoparticles has recently been the hot spot and foreland in some fields owing to their good chemical stability, biocompatibility and simple preparation [21]. In this work, Au nanoparticles were chosen to immobilize indicator dyes. In order to prevent the conglomeration of Au nanoparticles, chitosan was utilized to disperse them, and a thin film on the quartz glass slide was formed. Chitosan, a natural biopolymer, exhibits excellent film forming ability, and plenty amounts of amino groups in its molecule makes it possess positive charges by controlling pH [22]. Au nanoparticles, which are electronegative, can be easily entrapped into the chitosan film by electrostatic adsorption. Chitosan film will be not firm if it is immobilized on the slide by physical absorbtion. The best way is to fix chitosan by covalent bonding. Therefore, APS was used to aminosilanize the slide to introduce into amino groups. Then glutaraldehyde was picked as cross-linking agent to react with amino groups between chitason and APS. Thus, chitosan was covalently immobilized on the glass slide, and a firm film was formed. Subsequently, Au nanoparticles were embedded in the film by electrostatic adsorption.
Au nanoparticles will quench the fluorescence of indicator dye if the distance between them is very close [23-24]. To solve this problem, a long spacer, ―(CH2)11―, was set between Au nanoparticle and indicator dye when the dye was anchored. In order to realize this process, active hydroxyl groups were introduced to the surface of Au nanoparticles by self-assembly of thiol with a terminal hydroxyl group, while indicator dyes with amino groups were activated by using cyanuric chloride. Then, active hydroxyl groups on the surface of Au nanoparticles reacted with the secondary chlorine of cyanuric ring in the activated indicator dyes. The indicator dyes were immobilized to the slide via these steps. AEC was selected as an example of indicator dye with amino group, and an AEC-immobilized sensor was obtained.
3.2 Effect of pH value
The effect of pH value was investigated by measuring fluorescence intensity of the AEC- immobilized sensor in serial B-R buffer solutions with various pH values when the concentration of 2-nitrophenol was fixed at 1.43×10-5 mol/L. Experimental results show that the fluorescence intensity of the sensor remains almost constant in the pH range from 2 .2 to 6.5. In subsequent experiments, a B-R buffer solution with pH=3.2 was selected for the determination of 2-nitrophenol.
3.3 Response spectrum, linear response range, detec- tion limit of AEC-immobilized sensor to 2- nitrophenol
Fig.2 shows the fluorescence spectra of the AEC-immobilized sensors in a B-R buffer (pH=3.2) containing different amounts of 2-nitrophenol. the fluorescence of the sensor is quenched by 2-nitrophenol, which makes it possible assay 2-nitrophenol by using this sensor.

Fig.2 Fluorescent excitation/emission spectra of AEC- immobilized sensor exposed to 2-nitrophenol solutions with different concentrations: 1―0; 2―7.15×10-7 mol/L; 3― 1.43×10-6 mol/L; 4―2.86×10-6 mol/L; 5―5.72×10-6 mol/L; 6―8.58×10-6 mol/L; 7―1.14×10-5 mol/L; 8―1.43×10-5 mol/L; 9―2.15×10-5 mol/L; 10―2.86×10-5 mol/L; 11― 4.29×10-5 mol/L; 12―5.72×10-5 mol/L; 13―8.58×10-5 mol/L; 14―1.14×10-4 mol/L; 15―1.43×10-4 mol/L
The measuring principle is proposed according to Refs.[25-26]. While contacting with the sensor, 2-nitrophenol in the aqueous phase interacts with AEC in the solid phase. Supposing that a complexation equilibrium between 2-nitrophenol (NP) in the aqueous phase and AEC in the solid phase is established by forming a complex with a complexing ratio of m:n, the reaction equations can be represented as
mNP(aq)+nAEC(s) (NP)m(AEC)n(s) (1)
(NP)m(AEC)n(s) (1)
where K denotes the equilibrium constant of the reaction and is defined by
 (2)
(2)
The relative fluorescence intensity (α) is defined as
 (3)
(3)
where ct(AEC) and c(AEC)s are the total and uncomplexed concentrations of AEC in the solid phase, respectively, F and F0 are the fluorescence intensities of the sensors in the presence and absence of 2-nitrophenol (NP), respectively. By the law of mass action, one obtains
c(AEC)s+nc[(NP)m(AEC)n]s=ct(AEC) (4)
Combining Eqns.(2-4), one obtains
 (5)
(5)
Eqn.(5) is the theoretical basis of determining the concentration of 2-nitrophenol. Theoretical curves of α vs lg caq(NP) are shown in Fig.3. These curves were predicated by adjusting a complexing ratio (m:n) and equilibrium (K) constant. It can be seen that the curve corresponding to a 1:1 complexing ratio and K=4.4×104 gives the best fit to the experimental data (Fig.3, Curve 3). Therefore, a 1:1 complex was formed between 2-nitrophenol and AEC.

Fig.3 Theoretical curves of α vs lg caq(NP) by adjusting complexing ratio and equilibrium constant: 1―m:n=1:3, K= 5.8×1010; 2―m:n=1:2, K=4.38×107; 3―m:n=1:1, K= 4.4×104 (best fit); 4―m:n=2:1, K=2.0×109; 5―m:n=3:1, K=8.4× 1013 (○ denotes experimentally obtained data points)
Fig.4 shows the plot of experimental values of F0/F vs caq(NP), showing that a linear relationship between F0/F and caq(NP) exists in the range from 7×10-7 to 1×10-4 mol/L with following regression equation:
 (r=0.995 9) (6)
(r=0.995 9) (6)
Eqn.(6) provides quantitative basis for the determination of 2-nitrophenol. The detection limit, which is defined as the concentration of 2-nitrophenol when the signal change is three times the standard deviation (n=10) of the blank buffer signal, is 1.0×10-7 mol/L.

Fig.4 Plot of experimental values of F0/F vs concentration of 2-nitrophenol
3.4 Response time, reproducibility and reversibility of AEC-immobilized sensor
When the AEC-immobilized sensor contacts with 2-nitrophenol solutions with different concentrations, its fluorescence intensity will reach stable values within 10 s. The response time is very short. This may be attributed to the fact that AEC is immobilized onto the outmost surface rather than into the interior of the support matrix, and the diffusion limitation of the support matrix is avoided. The repeatability and reversibility of the AEC-immobilized sensor were evaluated by alternately measuring fluorescence intensity of 2-nitrophenol solution (1.43×10-5 mol/L) and blank B-R buffer solution (pH 3.2). Fig.5 shows that the prepared sensor can be restored easily by simply rinsing with blank buffer after it is used in 2-nitrophenol solution, and the reproducibility and reversibility of the sensor are enhanced.

Fig.5 Repeatability and reversibility of AEC-immobilized sensor (Sample was alternated from blank B-R buffer to solution of 1.43×10-5 mol/L 2-nitrophenol)
4 Conclusions
(1) A new strategy of immobilizing indicator dyes was proposed based on electrostatic adsorption and covalent bonding. This approach may be applicable to the other indicator dyes with amino groups besides AEC.
(2) Au nanoparticles are chosen as bridges and carriers of anchoring indicator dye. Chitosan, which is cross-linked to the aminosilanized glass slide by glutaraldehyde, is used to disperse Au nanoparticles and prevent their conglomeration. A long spacer, ―(CH2)11―, is set between Au nanoparticle and indicator dye so that the fluorescence intensity of the indicator dye cannot be quenched.
(3) AEC is selected as an example of indicator dye with amino group, and the AEC-immobilized sensor is obtained. The sensor is used to determine 2-nitrophenol with rapid response, good reproducibility and reversibility. The linear response to 2-nitrophenol covers the range from 7.0×10-7 to 1.0×10-4 mol/L with a detection limit of 1.0×10-7 mol/L.
References
[1] Zhang X B, Guo C C, Li Z Z, Shen G L, Yu R Q. An optical fiber chemical sensor for mercury ions based on a porphyrin dimer [J]. Analytical Chemistry, 2002, 74(4): 821-825.
[2] Zeng H H, Wang K M, Yu R Q. Development of an optode membrane for the determination of 2-nitrophenol based on fluorescence energy transfer [J]. Analytica Chimica Acta, 1994, 298(2): 271-277.
[3] Liu Y M, Pereiro-García R, Valencia-González M J, Diaz-Garcia M E, Sanz-Medel A. Evaluation of some immobilized room-temperature phosphorescent metal chelates as sensing materials for oxygen [J]. Analytical Chemistry, 1994, 66(6): 836-840.
[4] Igarashi S, Kuwae K, Yotsuyanagi T. Optical pH sensor of electrostatically immobilized porphyrin on the surface of sulfonated-polystyrene [J]. Analytical Sciences, 1994, 10(5): 821-823.
[5] Lu J Z, Zhang Z J. A reusable optical sensing layer for 2-nitrophenol based on the luminescence quenching of the Eu-thenoyltrifluoroacetone complex [J]. Analytica Chimica Acta, 1996, 318(2): 175-179.
[6] Christine M, David W, Fred M, Stanley K. Polymer modification of fiber optic chemical sensors as a method of enhancing fluorescence signal for pH measurement [J]. Analytical Chemistry, 1986, 58(7): 1427-1430.
[7] Hisamoto H, Manabe Y, Yanai H, Tohma H, Yamada T, Suzuki K. Molecular design, characterization, and application of multiinformation dyes for multidimensional optical chemical sensings (2): Preparation of the optical sensing membranes for the simultaneous measurements of pH and water content in organic media [J]. Analytical Chemistry, 1998, 70(7): 1255-1261.
[8] Xavier M P, Garcia-Fresnadillo D, Moreno-Bondi M C, Orellana G. Oxygen sensing in nonaqueous media using porous glass with covalently bound luminescent Ru(II) complexes [J]. Analytical Chemistry, 1998, 70(24): 5184-5189.
[9] Mohr G J, Tirelli N, Spichiger-Keller U E. Plasticizer-free optode membranes for dissolved amines based on copolymers from alkyl methacrylates and the fluoro reactand ETHT 4014 [J]. Analytical Chemistry, 1999, 71(8): 1534-1539.
[10] Ambrose T M, Meyerhoff M E. Optical ion sensing with immobilized thin films of photocrosslinked decyl methacrylate [J]. Analytica Chimica Acta, 1999, 378(1/3): 119-126.
[11] Niu C G, Li Z Z, Zhang X B, Shen G L, YU R Q. Covalently immobilized aminonaphthalimide as fluorescent carrier for the preparation of optical sensors [J]. Analytical and Bioanalytical Chemistry, 2002, 372(4): 519-524.
[12] Niu C G, Zeng G M, Chen L X, Shen G L, YU R Q. Proton “off-on” behaviour of methylpiperazinyl derivative of naphthalimide: A pH sensor based on fluorescence enhancement [J]. Analyst, 2004, 129(1): 20-24.
[13] Niu C G, Guan A L, Zeng G M, Liu Y G, Huang G H, Gao P F, Gui X Q. A ratiometric fluorescence halide sensor based on covalently immobilization of quinine and benzothioxanthene [J]. Analytica Chimica Acta, 2005, 547(2): 221-228.
[14] TAN S Z, NIU C G, JIANG J H, Shen G L, YU R Q. Optochemical sensor for an ornidazole assay using 1-amino-4- allyloxyanthraquinone as a fluorescent indicator [J]. Analytical Sciences, 2005, 21(8): 967-971.
[15] Stellacci F, Bauer C A, Meyer-Friedrichsen T, Wenseleers W, Marder S R, Perry J W. Ultrabright supramolecular beacons based on the self-assembly of two-photon chromophores on metal nanoparticles [J]. Journal of the American Chemical Society, 2003, 125(2): 328-329.
[16] Brown K R, Fox A P, Natan M J. Morphology-dependent electrochemistry of cytochrome c at Au colloid-modified SnO2 electrodes [J]. Journal of the American Chemical Society, 1996, 118(5): 1154-1157.
[17] Grabar K C, Freeman R G, Hommer M B, Natan M J. Preparation and characterization of Au colloid monolayers [J]. Analytical Chemistry, 1995, 67(4): 735-743.
[18] Yakushiji T, Sakai K, Kikuchi A, Aoyagi T, Sakurai Y, Okano T. Effects of cross-linked structure on temperature- responsive hydrophobic interaction of poly(N-isopropylacrylamide) hydrogel-modified surfaces with steroids [J]. Analytical Chemistry, 1999, 71(6): 1125-1130.
[19] Shakhsher Z M, Seitz W R. Optical detection of cationic surfactants based on ion pairing with an environment-sensitive fluorophor [J]. Analytical Chemistry, 1990, 62(17): 1758-1762.
[20] Zhang Z, Zhang Y, Ma W, Russell R, Shakhsher Z M, Grant C L, Seitz W R, Sundberg D C. Poly(vinyl alcohol) as a substrate for indicator immobilization for fiber-optic chemical sensors [J]. Analytical Chemistry, 1989, 61(3): 202-205.
[21] XIA Jin-lan, FU Jin-dian, NIE Zhen-yuan, SHEN Li. Preparation, optical properties and cell staining of water soluble amine-terminated PAMAM G 2.0-Au nanocomposites [J]. Journal of Central South University of Technology, 2005, 12(6): 641-646.
[22] WEI Wan-zhi, ZHAI Xiu-rong, ZENG Jin-xiang, GAO Yan-ping, GONG Shu-guo. New amperometric glucose biosensor by entrapping glucose oxidase into chitosan/nanoporous ZrO2/multiwalled carbon nanotubes nanocomposite film [J]. Journal of Central South University of Technology, 2007, 14(1): 73-77.
[23] Dubertret B, Calame M, Libchaber A J. Single-mismatch detection using gold-quenched fluorescent oligonucleotides [J]. Nature Biotechnology, 2001, 19(4): 365-370.
[24] Maxwell D J, Taylor J R, Nie S. Self-assembled nanoparticle probes for recognition and detection of biomolecules [J]. Journal of the American Chemical Society, 2002, 124(32): 9606-9612.
[25] Watanabe K, Nakagawa E, Yamada H, Hisamoto H, Suzuki K. Lithium ion selective optical fiber sensor based on a novel neutral ionophore and a lipophilic anionic dye [J]. Analytical Chemistry, 1993, 65(19): 2704-2710.
[26] Kurihara K, Ohtsu M, Yoshida T, Abe T, Hisamoto H, Suzuki K. Micrometer-sized sodium ion-selective optodes based on a “Tailed” neutral ionophore [J]. Analytical Chemistry, 1999, 71(16): 3558-3566.
Foundation item: Project(20775010) supported by the National Natural Science Foundation of China; Project(208095) supported by the Key Project of Ministry of Education, China; Project(07A006) supported by the Scientific Research Fund of Hunan Provincial Education Department, China; Project(07JJ3020) supported by Hunan Provincial Natural Science Foundation of China
Received date: 2008-07-16; Accepted date: 2008-09-22
Corresponding author: TAN Shu-zhen, Professor; Tel: +86-731-5258733; E-mail: tsz519@163.com
(Edited by CHEN Wei-ping)