
Effect of crystallization on corrosion resistance of Cu52.5Ti30Zr11.5Ni6 bulk amorphous alloy
TAN Chao-Gui(檀朝桂), JIANG Wen-juan(蒋文娟),
WU Xue-qing(吴学庆), WANG Xiu-feng(王秀锋), LIN Jian-guo(林建国)
Faculty of Material and Photoelectronic Physics, Xiangtan University, Xiangtan 411105, China
Received 20 September 2006; accepted 25 May 2007
Abstract: The effects of crystallization on the corrosion resistance of a Cu52.5Ti30Zr11.5Ni6 bulk amorphous alloy in 1 mol/L HCl, and 6 mol/L NaOH solutions were studied. The amorphous alloy was identified by differential thermal analysis(DSC) and by X-ray diffraction(XRD). The partially and fully crystallized alloys were prepared by controlling the annealing temperatures at 738 and 873 K for 1 and 12 min, respectively, and the corrosion resistances of those annealed alloys were compared with that of the amorphous alloy by immersion test and potentiodynamic measurements in 1 mol/L HCl and 6 mol/L NaOH solutions. The results show that the partially crystallized alloy exhibits high corrosion resistance, whereas full crystallization results in deteriorated corrosion resistance compared with that of the as-cast amorphous alloy.
Key words: amorphous materials; crystallization; corrosion behavior; polarization
1 Introduction
In the past decade, multi-component bulk metallic glasses(BMGs) have attracted large interest due to their unique properties[1-2]. Recently, quite a number of alloy families based on Zr, Pd, Fe, La, Cu and Mg have been developed, which exhibit an excellent glass- forming ability, and thus can be cast in bulk with dimension ranging from several millimeters to centimeters at cooling rates of less than 100 K/s[1-4]. Among the systems developed, Cu-based bulk metallic glasses are particularly interesting because of the combination of good mechanical property, less price and potential for industry applications. JOHNSON et al[5] developed firstly Cu47Zr11Ti34Ni8 BMG with a size of 4mm in thickness, opening the door to the possibility of fabrication of Cu-based bulk metallic glass. Subsequently, INOUE et al[6-7] and ZHANG et al[8] reported a new BMG of Cu60Zr30Ti10 with critical size of 3 mm in diameter, which exhibited a very good mechanical property with compressive/tensile yield strengths up to 1.9 GPa. The glass formation ability and mechanical properties can be further enhanced by the addition of a small amount of Sn, Be and Y with maximum sample size up to d5 mm and tensile strengthen over 2.5 GPa. However, the drawback of Cu- based BMGs originates from the chemical instability[9], which limits their applications.
It is well known that amorphous alloys have comparatively higher corrosion resistance than conventional crystalline alloys due to their chemical homogeneity and being free from defects such as grain boundaries, precipitates and segregations, which are more susceptible to chemical attacks[10]. Apart from the homogeneity and structure, the compositions of amorphous alloys also affect the corrosion resistance of the alloys[11]. The effect of the compositional changes on the corrosion resistance has been widely studied for amorphous alloys, such as Fe-based[12-14], Zr-based [15-16] alloys. It was found that additions of valve- metals, such as Cr, Mo, Ta, Ti or Nb, improve the corrosion resistance of the alloys a lot since they promote the spontaneous passivation of the alloys. Recently, there were also studies focusing on the effect of partially crystallization of the amorphous alloys such as Fe-based[17-18], Mg-based[19] and Zr-based ones[20- 21], on their corrosion behavior. However, some studies indicated that the partially crystallized alloys exhibit improved corrosion resistance[17-19], while others had the opposite conclusion[20-21]. Limited corrosion studies of Cu-based bulk metallic glasses have been conducted in recent years[22-23], and most of them reported the difference in corrosion resistance caused by additional elements[24-27]. The work on the effect of crystallization on the corrosion resistance of Cu-based alloys is rare and incomplete. The aim of the present work is therefore to fill in the gap by studying the corrosion and passivation behavior of different structural states, i.e. amorphous, partially and fully crystallized, respectively, of the Cu52.5Ti30Zr11.5Ni6 alloy obtained by controlled annealing of the alloy.
2 Experimental
Cu-based amorphous alloy ingots with nominal composition of Cu52.5Ti30Zr11.5Ni6 were prepared by arc melting the mixtures of the constituent elements of high purities in an argon atmosphere and cast into 5 mm diameter rods by copper-mold suction casting. The samples were evacuated in quartz tubes back-filled with argon, and annealed at the temperatures of 738 and 873 K for 1 and 12 min respectively. The heating and cooling rates were 40 and 200 K/min. The structure of the samples were examined with XRD using Cu Kα radiation at tube voltage of 40 kV and tube current of 30 mA. The extent of crystallization was also estimated by DSC measurement of the enthalpy change of the crystallization peak.
Corrosion behaviors of the metallic glasses were evaluated by immersion tests and potentiodynamic polarization measurements. The specimens were mechanically polished in cyclohexane with SiC paper down to 600 grit, and polished with 0.5 μm alumina powders. They were then degreased in acetone, dried in air and further exposed to air for 24 h before the tests. Electrolytes used were 1 mol/L HCl and 6 mol/L NaOH aqueous solutions, respectively, prepared from reagent grade chemicals and deionized water. Corrosion rates were estimated from the mass loss after immersion in the solutions for durations from one day to a few weeks. Potentiodynamic polarization curves were measured with a Solartron model 1287 potentiostat and a three-electrode cell. The counter electrode was graphite and the reference electrode was saturated calomel (SCE, φ(SHE)=0.241 V). The samples were immersed in the electrolyte for 20 min before measurements were done in order to stabilize the open-circuit potentials of the samples. Polarization curves were then measured with the cell open to air at a sweep rate of 5 mV/min at 298 K.
3 Results and discussion
In order to obtain partially and fully crystallized samples, DSC was employed to accurately perform the heat treatments of the samples. Fig.1 shows the DSC curve of the as-cast Cu-based alloy. It is observed that the crystallization of the as-spun sample takes place with two exothermic peaks, and there is a slight glass transition (Tg=688 K) before the occurrence of the first one. It is noticeable that the glass transition at low heating rate for this alloy before the occurrence of the first one is undetectable. The first annealing temperature, i.e. 738 K, is chosen above the onset temperature of crystallization (Tx=734 K) and the second temperature, i.e. 873 K, is chosen after the second large exothermic peak. The annealing treatments for 1 and 12 min lead to partial and full devitrifications, respectively. The decreasing magnitude of the exothermic peaks with increasing annealing time indicates a decreasing amount of the amorphous phase.
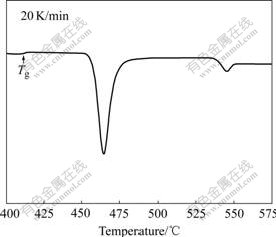
Fig.1 DSC curve of as-cast Cu-based alloy
Fig.2 shows the XRD patterns of the as-cast and annealed samples. For the as-cast sample, the typical broad peak without any distinct crystalline peak indicates a fully amorphous structure. The XRD patterns for the annealed samples show well-defined peaks. Only Cu10Zr7 crystallites are present in the amorphous matrix after annealing at 738 K for 1 min. However, Cu3Ti and Ni3Zr are also found when the matrix is annealed at 873 K for 12 min.
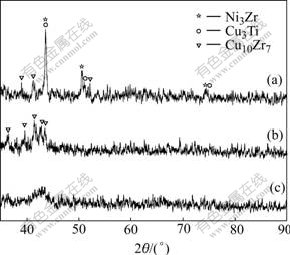
Fig.2 XRD patterns of cast as-cast and annealed samples: (a) Annealed at 873 K for 12 min; (b) Annealed at 738K for 1 min; (c) As-cast
The corrosion rates of as-cast and annealed samples in 6 mol/L NaOH solution at 298 K are shown in Fig.3. It is found that the partially crystallized sample shows superior corrosion resistance in this alkaline solution. Especially, the corrosion rate of partially crystallized samples is almost half that of the as-cast samples, while the fully crystallized sample exhibits the worst corrosion resistance.
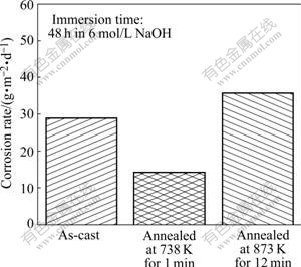
Fig.3 Corrosion rates of as-cast and annealed samples in 6 mol/L NaOH solution
Fig.4 shows the polarization curves for the samples in 1 mol/L HCl solution. The polarization behaviors are similar to each other except that of the fully crystallized sample. The fully crystallized sample exhibits a process with active passive transition, and there is a wide passive region at the end of the corrosion as well as the other two samples. While the amorphous and partially crystallized alloys suffer from active dissolution immediately and then exhibit a spontaneously passivated behavior, possibly because of the unstable surface films of the alloys in the chloride-ions-containing solutions. From the polarization curves in Fig.4 and the data in Table 1, the partially crystallized sample shows a little improvement in the corrosion resistance compared with that of the as-cast sample, viz., the higher φcorr, the lower critical passivating current density(Jp).
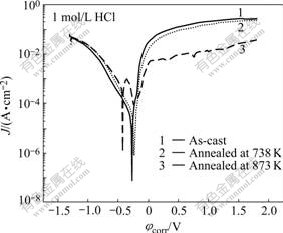
Fig.4 Potentiodynamic polarization curves of as-cast and annealed samples in 1 mol/L HCl solutions at 298 K open to air
Table 1 Results of potentiodynamic polarization measurements
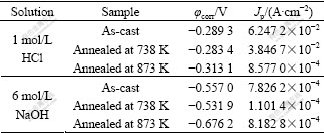
Their polarization curves in 6 mol/L NaOH solution are also shown in Fig.5. All of them show current plateau, associated with a passive film formation on the alloy surface in Fig.5. The three alloys are spontaneously passivated with significantly low passive current density of the order 10-4 A/cm2. There is a large extent between pitting potential and corrosion potential(φcorr), which means that the alloys have high pitting resistance[17] in 6 mol/L NaOH solution. In general, these features indicate a rapid formation of protective anodic films with low electronic conductivity, which is a typical behaviour of valve-metals, e.g. Al, Zr, Hf[18]. However, when anodic voltage becomes larger, the surface films of the alloys dissolve and transform to porous surface film, i.e. trans-passive dissolution. Moreover, they show active cathodic reaction as the cathode current density changes rapidly with potential. Among the three samples, the partially crystallized sample annealed at 738 K shows the best corrosion resistance, viz., the highest φcorr, and the lowest Jp, as shown in Table 1. The results of the immersion tests and the potentiodynamic polarization tests are in good agreement. They also demonstrate that the corrosion resistance of the as-cast amorphous alloy can be improved by controlled annealing, which induces nucleation of crystallites in the amorphous matrix. The mechanism of such improvement is still unknown. But some researchers proposed that the presence of the amorphous and crystalline interface may promote the diffusion of passive elements of the alloys for faster formation of the protective passive film[19, 28]. When the annealing time is longer, the crystallites grow up and lead to deterioration in the corrosion resistance of the alloy. It may be due to the formation of compositional heterogeneities and more boundary regions, therefore increasing the susceptibility to the localized corrosion.
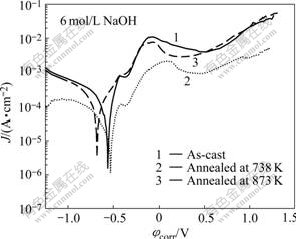
Fig.5 Potentiodynamic polarization curves of as-cast and annealed samples in 6 mol/L NaOH solutions at 298 K open to air
4 Conclusions
The effect of structure evolution on the corrosion resistance of the amorphous Cu52.5Ti30Zr11.5Ni6 alloy was investigated by using immersion tests and potentiodynamic polarization measurements in 1 mol/L HCl and 6 mol/L NaOH solutions, respectively. The results show that the partially crystallized sample has higher corrosion resistance than that of the amorphous alloys, while the fully crystallized sample shows deterioration in the corrosion resistance.
References
[1] JOHNSON W L. Bulk glass forming metallic alloys: Science and technology [M]. America: MRS Bull, 1999.
[2] INOUE A. Bulk amorphous alloys: Practical characteristics and applications [M]. Switzerland: Trans Tech Publications Inc, 1998.
[3] LEONHARD A, XING L Q, HEILMAIER M, GEBERT A, ECKERT J, SCHULTZ L. Effect of crystalline precipitations on the mechanical behavior of bulk glass forming Zr-based alloys [J]. Nanostruct Mater, 1998, 10: 805-817.
[4] INOUE A. Bulk amorphous and nanocrystalline alloys with high functional properties [J]. Mater Sci Eng A, 2001, A304/306: 1-10.
[5] GLADE S C, L?FFLER J F, BOSSUYTS, JOHNSON W L. Crystallization of amorphous Cu47Ti34Zr11Ni8 [J]. J Appl Phys, 2001, 89: 1573-1579.
[6] INOUE A, ZHANG W, ZHANG T, KUROSAKA K. High-strength Cu-based bulk glassy alloys in Cu-Zr-Ti and Cu-Hf-Ti ternary systems [J]. Acta Mater, 2001, 49: 2645-2652.
[7] INOUE A, ZHANG W, ZHANG T, KUROSAKA K. Cu-based bulk glassy alloys with high tensile strength of over 2000 MPa [J]. J Non-Cryst Solids, 2002, 304: 200-209.
[8] ZHANG Q S, ZHANG H F, Deng Y F, DING B Z, HU Z Q. Bulk metallic glass formation of Cu-Zr-Ti-Sn alloys [J]. Scripta Mater, 2003, 49: 273-278.
[9] YAMAMOTO T, ZHANG T, INOUE A. Formation, thermal stability, mechanical properties and corrosion resistance of Cu-Zr-Ti-Ni-Nb bulk glassy alloys [J]. Mater Trans, 2003, JIM 44: 1147-1152.
[10] HASHIMOTO K, FROMENT (ED.) M, Passivity of metals and semiconductors [M]. Amsterdam: Elsevier Science Publishers, 1983.
[11] TURN J C JR, LATANISION R M. Corrosion rate measurements and calculations: Calculation of error limits and transport effects [J]. Corrosion Science, 1983, 39: 271-283.
[12] PANG S, ZHANG T, ASAMI K, INOUE A. New Fe-Cr-Mo-(Nb, Ta)-C-B glassy alloys with high glass-forming ability and good corrosion resistance [J]. Mater Trans, 2001, JIM 42: 376-380.
[13] PANG S, ZHANG T, ASAMI K, INOUE A. Oxidation mechanism of copper at 62-1 073 K [J]. Mater Trans, 2002, JIM 43: 2137-2151.
[14] MARIANO N A, SOUZA C A C, MAY J E, KURI S E. Influence of Nb content on the corrosion resistance and saturation magnetic density of Fe-Cu-Nb-Si-B alloys [J]. Mater Sci Eng A, 2003, 354: 1-5.
[15] PANG S, ZHANG T, ASAMI K, INOUE KIMURA A H. Corrosion behavior of Zr-(Nb-)Al-Ni-Cu glassy alloys [J]. Mater Trans, 2000, JIM 41: 1490-1495.
[16] RAJU V R, KUHN U, WOLFF U, SCHNEIDER F, ECKERT J, REICHE R, GEBERT A. Corrosion behaviour of Zr-based bulk glass-forming alloys containing Nb or Ti [J]. Mater Lett, 2002, 57: 173-177.
[17] QIN F X, ZHANG H F, CHEN P, CHEN F F, QIAO D C, HU Z Q. Corrosion behavior of bulk amorphous Zr55Al10Cu30Ni5-xPdx alloys [J]. Mater Lett, 2004, 58: 1246-1250.
[18] LOHRENGEL M M. Thin anodic oxide layers on aluminium and other valve metals: High field regime [J]. Mater Sci Eng R, 1993, R11: 243-294.
[19] SOUZA C A C, KIMINAMI C S. Crystallization and corrosion resistance of amorphous FeCuNbSiB [J]. J Non-Cryst Solids, 1997, 219: 155-159.
[20] SZEWIECZEK D, TYRLIK-HELD J, PASZENDA Z. Corrosion investigations of nanocrystalline iron based alloy [J]. J Mater Process Technol, 1998, 78: 171-176.
[21] ONG M S, LI Y, BLACKWOOD D J, NG S C, KAM C H. Effect of heat treatment on the corrosion behaviour of amorphous Mg-18at% Ni alloy [J]. J Alloys Compd, 1998, 279: 252-258.
[22] KAMACHI MUDALI U, SCUDINO S, KUHN U, ECKERT J, GEBERT A. Polarisation behaviour of the Zr57Ti8Nb2.5Cu13.9Ni11.1Al7.5 alloy in different microstructural states in acid solutions [J]. Scripta Mater, 2004, 50: 1379-1384.
[23] ZANDER D, KOSTER U. Corrosion of amorphous and nanocrystalline Zr-based alloys [J]. Mater Sci Eng A, 2004, A375/377: 53-59.
[24] QIN C L, ASAMI K, ZHANG T, INOUE A. Corrosion behavior of Cu-Zr-Ti-Nb bulk glassy alloys [J]. Mater Trans, 2003, JIM 44: 749-753.
[25] ASAMI K, QIN C L, ZHANG T, INOUE A. Effect of Ag addition on the glass-forming ability and thermal stability of Mg-Cu-Y alloys [J]. Mater Sci Eng A, 2004, A375/377: 759-762.
[26] LIU B, LIU L. The effect of microalloying on thermal stability and corrosion resistance of Cu-based bulk metallic glasses [J]. Mater Sci Eng A, 2006, A415: 286-290.
[27] ASAMI K, QIN C L, ZHANG T, INOUE A. Effect of additional elements on the corrosion behavior of a Cu-Zr-Ti bulk metallic glass [J], Mater Sci Eng A, 2004, A375/377: 235-239.
[28] SOUZA C A C, POLITI F S, KIMINAMI C S. Influence of structural relaxation and partial devitrification on the corrosion resistance of Fe78B13Si9 amorphous alloy [J]. Scripta Mater, 1998, 39: 329-334.
Foundation item: Project(50371072) supported by the National Natural Science Foundation of China; Project(05FJ4048) supported by the Provincial Science and Technology Department Foundation of Hunan, China
Corresponding author: TAN Chao-Gui; Tel: +86-732-8292195; Fax: +86-732-8292486; E-mail: guichaotan@yahoo.com
(Edited by HE Xue-feng)