
Synthesis and characterization of layered Li(Ni1/3Mn1/3Co1/3)O2
cathode materials by spray-drying method
LIU Zhi-min(������), HU Guo-rong(������), PENG Zhong-dong(���Ҷ�),
DENG Xin-rong(������), LIU Ye-xiang(��ҵ��)
School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China
Received 14 April 2006; accepted 21 December 2006
Abstract: Spherical Li(Ni1/3Mn1/3Co1/3)O2 was prepared via the homogenous precursors produced by solution spray-drying method. The precursors were sintered at different temperatures between 600 and 1 000 �� for 10 h. The impacts of different sintering temperatures on the structure and electrochemical performances of Li(Ni1/3Mn1/3Co1/3)O2 were compared by means of X-ray diffractometry(XRD), scanning electron microscopy(SEM), and charge/discharge test as cathode materials for lithium ion batteries. The experimental results show that the spherical morphology of the spray-dried powers maintains during the subsequent heat treatment and the specific capacity increases with rising sintering temperature. When the sintering temperature rises up to 900 ��, Li(Ni1/3Mn1/3Co1/3)O2 attains a reversible capacity of 153 mA?h/g between 3.00 and 4.35 V at 0.2C rate with excellent cyclability.
Key words: spherical Li(Ni1/3Mn1/3Co1/3)O2 powder; synthesis; characterization; spray-drying method; lithium ion battery
1 Introduction
LiCoO2 is widely used in commercial lithium-ion batteries. During the past decade, lithium-ion batteries with LiCoO2 and graphite have been developed to a quite high level. However, the toxicity and high cost of LiCoO2 basically restrict its application in future. Several cathode materials, such as LiMn2O4, LiNiO2, LiMnO2, LiNixCo1-xO2 and Li(NixCo1-2xMnx)O2 have been intensively proposed as possible alternatives to LiCoO2[1�C8]. Li(Ni1/3Mn1/3Co1/3)O2 can be regarded as a special case in which x is 1/3 in Li(NixCo1-2xMnx)O2 system[9]. It was reported that Li(Ni1/3Mn1/3Co1/3)O2 showed a specific capacity of 160 mA?h/g over 2.5- 4.4 V[10]. Due to the stable cycling performances, safety and good rate capability of Li(Ni1/3Mn1/3Co1/3)O2, this material is considered to be one of the best candidates of positive electrode material to replace LiCoO2.
Selecting synthetic method plays a very important role in preparing phase-pure, spherical and homogeneous Li(Ni1/3Mn1/3Co1/3)O2. Solid-state reaction at high temperature is the traditional method for synthesizing cathode materials. However, homogenous composition oxide particles with good morphology are difficult to obtain. Li(Ni1/3Mn1/3Co1/3)O2 is usually synthesized by the mixed hydroxide method[11-12], in which a M(OH)2 (M=Ni, Co, Mn) precursor is precipitated from a metal nitrate/vitriol solution and is then reacted with a lithium salt at elevated temperature. This is not difficult in practice, but pH value, temperature, flow conditions, concentrations, and other factors that influence the crystal structure, purity, and physical properties of the products, must be carefully controlled for optimum electrochemical performance[12].
Recent studies and development have shown that spray drying can produce fine spherical particles with high homogeneity and good morphology[13-17]. In this study, spray-drying method was used to produce the precursor, followed by the calcination at different temperatures to prepare Li(Ni1/3Mn1/3Co1/3)O2 powder. The effects of different sintering temperatures on the structure and electrochemical performances of samples were also studied.
2 Experimental
The samples of target compositions Li(Ni1/3Mn1/3- Co1/3)O2 with excess amounts of lithium between 0 and 5% were synthesized by the following method. The stoichiometric amounts of LiNO3, Co(NO3)2��6H2O, Ni(NO3)2��6H2O and Mn(NO3)2��4H2O were used as the starting materials and dissolved in distilled water. Polyvinyl alcohol(PVA) was dissolved in a separate container and used as a polymeric carrier in a mixed cation nitrate solution, which involved the formation of complexes between the cations and the hydroxyl side groups of PVA, thereby limiting the fractional precipitation of metal nitrate salts. Two kinds of aqueous solutions were mixed into a brown transparent solution, which was fed into a spray drying instrument to produce homogenous precursors. Then it was atomized via a nozzle with two flows at an air pressure of 0.4 MPa, and was dried by hot air. The overall flow rate of solution must be controlled at 10 L/h. The inlet hot air temperature reached 350 ��, and the exit hot air temperature was 150 ��. The as-prepared precursor powders were finally sintered in air at temperatures between 600 and 1 000 �� for 10 h.
The thermal evolution of the mixed precursor was studied by Mettler Toledo TGA/SDTA851e analysis. The samples were characterized by XRD using a Rigaku Minflex automated powder diffractometer and Cu K�� radiation. The scan electron microscope(SEM) study of the samples was performed on JSM-5600LV electron microscope.
The charge/discharge cycling tests were performed using the CR2032 coin-type cell that consisted of a cathode and lithium metal anode separated by a Celgard 2300 porous polypropylene film. For the fabrication of the positive electrode, 20 mg of Li(Ni1/3Mn1/3Co1/3)O2 was accurately weighted and mixed with 12 mg of conductive binder (8 mg of teflonized acetylene black and 4 mg of graphite). The mixture was pressed onto a stainless screen and dried at 170 �� for 5 h under vacuum oven. Lithium foil was used as the negative electrode. The electrolyte was 1 mol/L LiPF6 in ethylene carbonate/ dimethyl carbonate (EC/DMC, volume ratio of EC to DMC is 1?2). The cells were assembled in a glove box filled with dried argon gas.
3 Results and discussion
Fig.1 shows the XRD pattern of the precursor spray- drying at 350 ��. Because of the short time and low temperature of the spray-drying process, the crystal of precursor powers is hardly formed. There is little Bragg reflection visible in the pattern originating from the precursor except a nickel nitrate hydroxide Ni(NO3)2��2Ni(OH)2 and Co3O4 are illegibly observed. These compositions are associated with a PVA-nitrate interaction by which  groups are replaced by OH�� groups and partial hydrolyze and decompose in the precursor.
groups are replaced by OH�� groups and partial hydrolyze and decompose in the precursor.

Fig.1 XRD pattern of Li(Ni1/3Mn1/3Co1/3)O2 precursor
The TG and DTA curves of the precursor are shown in Fig.2. Because of PVA-nitrate interaction and complex composition, the reaction mechanism during sintering is difficult to be analyzed. The first mass loss in the temperature range below 160 �� on the TG curve is due to the removal of the absorbing and structural water from each chemical component, which is accompanied by a strong endothermic peak. The second mass loss occurs in the temperature region below 240 ��. In the DTA curve, there are two peaks. These peaks are related to the decomposition and synthesis among the mixed precursor. The steeper endothermic peak evidenced by DTA at 250 �� is ascribed to LiNO3 melting. The mass loss of the starting materials is almost terminated at 600 ��, indicating that the organic compounds and chemical component can be removed and the decomposition finished completely at temperatures lower than 600 ��. From the thermal analysis, Li(Ni1/3Mn1/3Co1/3)O2 can be obtained at a temperature around 600 ��.
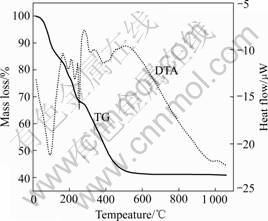
Fig.2 TG and DTA curves of precursor formed by spray-drying method
After sintering the precursor at 600, 700, 800, 900 and 1 000 �� for 10 h, the XRD patterns of the compounds Li(Ni1/3Mn1/3Co1/3)O2 are shown in Fig.3. The layered Li(Ni1/3Mn1/3Co1/3)O2 crystal peaks can be observed from all samples sintered at different tempera- tures. But many other peaks of Li2CO3 are also found in the sample sintered at 600 ��. Li2CO3 formation is related to the improved dispersion of Li (or very fine LiNO3 crystals) in the PVA matrix and the PVA combustion. This phenomenon has already been observed in the LiNi1-x-yCoxAlyO2 synthesis by spray-drying process[17]. The XRD patterns of the powders sintered at 700, 800, 900 and 1 000 �� are Li(Ni1/3Mn1/3Co1/3)O2 similar to that of LiCoO2 (��-NaFeO2 type, space group  and no impure peaks appear. The relative intensities of the diffraction peaks enhance with increasing sintering temperature. Cation mixing is known to deteriorate the electrochemical performance of the layered compounds. The integrated intensity ratio of the lines (003) to (104) (R) in the XRD patterns is shown to be a measure of the cation mixing and R��1.2 is an indication of undesirable cation mixing[18]. The lattice constants, a and c, c/a ratio, V, and the intensity ratio I003/I104 of the hexagonal unit cell are listed in Table 1. With the temperature increased from 600 to 900 ��, the intensity ratio of I003 to I104 increases from 1.060 1 to 1.214 8. When the materials are calcined at 1 000 ��, this ratio decreases to 1.192 2. The best results come from the materials calcined at 900 �� and show little evidence of ion mixing.
and no impure peaks appear. The relative intensities of the diffraction peaks enhance with increasing sintering temperature. Cation mixing is known to deteriorate the electrochemical performance of the layered compounds. The integrated intensity ratio of the lines (003) to (104) (R) in the XRD patterns is shown to be a measure of the cation mixing and R��1.2 is an indication of undesirable cation mixing[18]. The lattice constants, a and c, c/a ratio, V, and the intensity ratio I003/I104 of the hexagonal unit cell are listed in Table 1. With the temperature increased from 600 to 900 ��, the intensity ratio of I003 to I104 increases from 1.060 1 to 1.214 8. When the materials are calcined at 1 000 ��, this ratio decreases to 1.192 2. The best results come from the materials calcined at 900 �� and show little evidence of ion mixing.

Fig.3 X-ray diffraction patterns of Li(Ni1/3Mn1/3Co1/3)O2 obtained at different temperatures
Table 1 Lattice parameters of Li(Ni1/3Mn1/3Co1/3)O2 prepared by sintering at different temperatures

The typical SEM photographs of the precursor and Li(Ni1/3Mn1/3Co1/3)O2 powders sintering at 900 �� are shown in Fig.4. From SEM photographs, the particles of the precursor are spherical with size distribution in the range of 5-20 ��m. The surface of every spherical particle is not very smooth with some wrinkles. The morphology of the Li(Ni1/3Mn1/3Co1/3)O2 powers is resulted from the precursor and agglomerated by the crystalline grains to spherical morphology. The spherical powders exhibit higher tap-density and less agglomeration than irregular particles[18], which is important to improve the volume energy of the powders.

Fig.4 SEM images of precursor and layered Li(Ni1/3Mn1/3Co1/3)O2 powders: (a) Precursor powders; (b) Li(Ni1/3Mn1/3Co1/3)O2 powders sintered at 900 ��
The initial charge��discharge curves of Li/Li(Ni1/3- Mn1/3Co1/3)O2 sintered at different temperatures for 10 h, at a discharge rate of 0.2C in the potential range from 3.00 to 4.35 V, are shown in Fig.5. The Li/Li(Ni1/3Mn1/3- Co1/3)O2 cell has a very smooth and monotonous voltage profile, similar to the voltage profiles reported by OHZUKU et al[9] and SHAJU et al[10]. The sintering temperature remarkably affects the electrochemical characteristics of the Li(Ni1/3Mn1/3Co1/3)O2. The discharge capacities increase linearly with increasing sintering temperature from 600 to 900 ��. As the sample is sintered at 900 ��, the observed discharge capacity is about 153 mA?h/g. This obtained capacity is comparable with that of the reported in Refs.[9,11-12]. However, when the sintering temperature reaches 1 000 ��, the discharge capacity decreases to 145 mA?h/g, which is attributed to the volatilization of lithium at high temperature. Fig.6 shows the cycling behavior of the samples sintered at different temperatures at a discharge rate of 0.2C. The sample sintered at 900 �� exhibits very stable cyclability with little capacities loss after the 40th cycle. The cycling performance becomes worse at other sintering temperatures.

Fig.5 Typical initial charge��discharge curves of Li/Li(Ni1/3- Mn1/3Co1/3)O2 cells

Fig.6 Cycle performance of samples sintered at different temperatures at discharge rate of 0.2C on Li/Li(Ni1/3Mn1/3- Co1/3)O2 cell
Figs.7 and 8 show the rate performance of the sample sintered at 900 �� with various current densities between potential limits of 3.00-4.35 V. The cell is charged at a current density of 30 mA/g (0.2C) before each discharge test. The first discharge capacity of the sample decreases from 153 to 98.9 mA?h/g when the discharge current density increases from 30 mA/g (0.2C) to 750 mA/g (5C), indicating that only 65% of initial discharge capacity can be retained.

Fig.7 Initial discharge curves of Li(Ni1/3Mn1/3Co1/3)O2 sintered at 900 �� with various current densities

Fig.8 Rate performance of Li(Ni1/3Mn1/3Co1/3)O2 sintered at 900 ��
4 Conclusions
1) Layered Li(Ni1/3Mn1/3Co1/3)O2 powders with high homogeneity, spherical morphology and high capacity are synthesized by solution spray-drying method. Li2CO3 is detected by XRD when the precursor is prepared at low temperature. The Li2CO3 formation is related to the drying conditions of the precursor and the PVA combustion step.
2) The sintering temperature strongly affects the structure and the electrochemical performance. The layered Li(Ni1/3Mn1/3Co1/3)O2 powders synthesized at 900 �� for 10 h exhibit high discharge capacity and good cycling properties.
3) Spray-drying method is considered to be a promising alternative way to synthesize oxide electrode materials with homogeneous and spherical morphology for lithium-ion secondary batteries.
References
[1] GAO Y, DAHN J R. Synthesis and characterization of Li1+xMn2-xO4 for Li-ion battery applications [J]. J Electrochem Soc, 1996, 143(1): 100-119.
[2] ZHECHEVA E, STOYANOVA R, GOROVA M, LAVELA P, TIRADO J L. Co/Mn distribution and electrochemical intercalation of Li into LiMn2-yCoyO4 spinels, 0��y��1 [J]. Solid State Ionics, 2001, 140(1/2): 19-33.
[3] OHZUKU T, UEDA A, NAGAYAMA M. Electrochemistry and structural chemistry of LiNiO2(R3m) for 4 volt secondary lithium cells [J]. J Electrochem Soc, 1993, 140(7): 1862-1870.
[4] AMMUNDSEN B, PAULSEN J. Novel lithium-ion cathode materials based on layered manganese oxides [J]. Advanced Materials, 2001, 13: 943-949.
[5] CEDER G, MISHRA S K. Stability of orthorhombic and monoclinic-layered LiMnO2 [J]. Electrochem Solid State Lett, 1999, 2(11): 550-552.
[6] ROUGIER A, SAADOUNE I, GRAVEREAU P, WILLMANN P, DELMAS C. Effect of cobalt substitution on cation in LiNi1-xCoxO2 electrode materials [J]. Solid State Ionics, 1996, 90(1/4): 83-90.
[7] CHANG C C, SCARR N, KUMTA P N. Synthesis and electrochemical characterization of LiMO2 (M=Ni, Ni0.75Co0.25) for rechargeable lithium ion batteries [J]. Solid State Ionics, 1998, 112(3/4): 329-344.
[8] LIU Z L, YU A S, LEE Y J. Synthesis and characterization of LiNi1-x-yCoxMnyO2 as the cathode materials of secondary lithium batteries [J]. J Power Sources, 1999, 81/82: 416-419.
[9] OHZUKU T, MAKIMURA Y. Layered lithium insertion material of LiCo1/3Ni1/3Mn1/3O2 for lithium-ion batteries [J]. Chem Lett, 2001, 7: 642-643.
[10] SHAJU K M, CHOWDARI B V R. Performance of layered Li(Ni1/3Co1/3Mn1/3)O2 as cathode for Li-ion batteries [J]. Electrochim Acta, 2002, 48(2): 145-151.
[11] CHEN Y, WANG G X, KONSTANTINOV K, LIU H K, DOU S X. Synthesis and characterization of LiCoxMnyNi1-x-yO2 as a cathode material for secondary lithium batteries [J]. J Power Sources, 2003, 119/121: 184-188.
[12] LEE M H, KANG Y J, MYUNG S T, SUN Y K. Synthetic optimization of Li(Ni1/3Co1/3Mn1/3)O2 via co-precipitation [J]. Electrochim Acta, 2004, 50(4): 939-948.
[13] LI Yang-xing, WAN Chun-rong, WU Yu-ping, JIANG Chang-yin, ZHU Yong-jun. Synthesis and characterization of ultrafine LiCoO2 powders by a spray-drying method [J]. J Power Sources, 2000, 85(1): 294-298.
[14] WU H M, TU J P, YUAN Y F, LI Y, ZHAO X B, CAO G S. Structural, morphological and electrochemical characteristics of spinel LiMn2O4 prepared by spray-drying method [J]. Scripta Materialia, 2005, 52(6): 513-517.
[15] WU H M, TU J P, YUAN Y F, LI Y, ZHAO X B, CAO G S. Electrochemical and ex situ XRD studies of a LiMn1.5Ni0.5O4 high-voltage cathode material [J]. Electrochimica Acta, 2005, 50(20): 4104-4108.
[16] WEN Z Y, GU Z H, HUANG S H, YANG J H, LIN Z X, YAMAMOTO O. Research on spray-dried lithium titanate as electrode materials for lithium ion batteries [J]. J Power Sources, 2005, 146(1/2): 670-673.
[17] DUVIGNEAUD P H, SEGATO T. Synthesis and characterisation of LiNi1-x-yCoxAlyO2 cathodes for lithium-ion batteries by the PVA precursor method [J]. Journal of the European Ceramic Society, 2004, 24(6): 1375-1380.
[18] OHZUKU T, UEDA A, NAGAYAMA M, YIWAKOSHI, KOMORI H. Comparative study of LiCoO2, LiNi1/2Co1/2O2 and LiNiO2 for 4 volt secondary lithium cells [J]. Electrochim Acta, 1993, 38(19): 1159-1167.
Corresponding author: LIU Zhi-min; Tel: +86-731-8860474; E-mail: syautolzm@163.com
(Edited by CHEN Wei-ping)