Modeling flame propagation speed and quenching distance of aluminum dust flame with spatially random distribution of particles
��Դ�ڿ������ϴ�ѧѧ��(Ӣ�İ�)2017���1��
�������ߣ�Mehdi Bidabadi Alireza Khoeini Poorfar
����ҳ�룺81 - 89
Key words��flame propagation speed; random particle distribution; quenching distance; random discrete combustion; aluminum
Abstract: In this research combustion of aluminum dust particles in a quiescent medium with spatially discrete sources distributed in a random way was studied by a numerical approach. A new thermal model was generated to estimate flame propagation speed in a lean/rich reaction medium. Flame speed for different particle diameters and the effects of various oxidizers such as carbon dioxide and oxygen on flame speed were studied. Nitrogen was considered the inert gas. In addition, the quenching distance and the minimum ignition energy (MIE) were studied as a function of dust concentration. Different burning time models for aluminum were employed and their results were compared with each other. The model was based on conduction heat transfer mechanism using the heat point source method. The combustion of single-particle was first studied and the solution was presented. Then the dust combustion was investigated using the superposition principle to include the effects of surrounding particles. It is found that larger particles have higher values of quenching distance in comparison with smaller particles in an assumed dust concentration. With the increase of dust concentration the value of MIE would be decreased for an assumed particle diameter. Considering random discrete heat sources method, the obtained results of random distribution of fuel particles in space provide closer and realistic predictions of the combustion physics of aluminum dust flame as compared with the experimental findings.
J. Cent. South Univ. (2017) 24: 81-89
DOI: 10.1007/s11771-017-3411-2

Mehdi Bidabadi, Alireza Khoeini Poorfar
Combustion Research Laboratory, School of Mechanical Engineering, Iran University of
Science and Technology (IUST), Narmak, 16846-13114, Tehran, Iran
 Central South University Press and Springer-Verlag Berlin Heidelberg 2017
Central South University Press and Springer-Verlag Berlin Heidelberg 2017
Abstract: In this research combustion of aluminum dust particles in a quiescent medium with spatially discrete sources distributed in a random way was studied by a numerical approach. A new thermal model was generated to estimate flame propagation speed in a lean/rich reaction medium. Flame speed for different particle diameters and the effects of various oxidizers such as carbon dioxide and oxygen on flame speed were studied. Nitrogen was considered the inert gas. In addition, the quenching distance and the minimum ignition energy (MIE) were studied as a function of dust concentration. Different burning time models for aluminum were employed and their results were compared with each other. The model was based on conduction heat transfer mechanism using the heat point source method. The combustion of single-particle was first studied and the solution was presented. Then the dust combustion was investigated using the superposition principle to include the effects of surrounding particles. It is found that larger particles have higher values of quenching distance in comparison with smaller particles in an assumed dust concentration. With the increase of dust concentration the value of MIE would be decreased for an assumed particle diameter. Considering random discrete heat sources method, the obtained results of random distribution of fuel particles in space provide closer and realistic predictions of the combustion physics of aluminum dust flame as compared with the experimental findings.
Key words: flame propagation speed; random particle distribution; quenching distance; random discrete combustion; aluminum
1 Introduction
Dust cloud explosion hazard has been a threat to mankind and people��s properties for the last 150 years [1]. Wherever dust is produced, explosion can happen if there are enough amounts of oxidizer, dust concentration and an ignition source with sufficient energy to ignite the dust cloud. Accordingly, modeling and investigating of dust cloud ignition and combustion assist us to manage and avoid explosions. Nowadays with improvement of powder technology and increase of powder usage in industries and laboratories, the need for preventive methods has become more important. Many combustible dusts allow a flame to propagate through the fuel particles if dispersed as a cloud in air and ignited, in a manner similar to (though not identical to) the propagation of flames in premixed fuel oxidant gases. Many studies have been published regarding combustion of organic and inorganic dust particles within the last few years. MOSTAFAVI et al [2] performed experimental research and thermo-gravimetric analysis for lycopodium dust particles. SOLTANINEJAD et al [3] carried out a study in order to analyze one-dimensional dust flame propagation in cloud of fuel mixture. The combustion of metallic particles used in a variety of industrial applications is a highly exothermic event. Basic mechanisms of combustion of such two-phase mixtures are not well understood because of two major problems facing combustion of dust particle research. The first is the complex nature of processes involving the physical and chemical properties of the fuel. The second relates to the size, shape and spatial distribution of the particles. Interest in aluminum combustion is associated with fire and explosion safety and the fact that metals are important components of modern propellants. The heat released due to combustion of metals especially aluminum, has high value and high volumetric energy density. Combustion of dust particles in air has been a serious safety concern for the mining, manufacturing, and energy production industries. ROCKWELL RANGWALA [4] analyzed a premixed dust�Cair flame, under the conditions where a homogeneous gas-phase reaction front can exist, and discussed four different possible types of flames. BIDABADI et al [5] conducted an analytical study in order to determine the role of Lewis and Damk hler numbers on the premixed flame propagation through micro-organic dust particles.
hler numbers on the premixed flame propagation through micro-organic dust particles.
Aluminum dust particles are widely used in various applications, such as hydrogen production, propulsion and pyrotechnic aluminum compounds because of the high burning rate of dust particles and enhanced combustion efficiency. Aluminum powder is a very reactive metal and its oxidation can occur in either thermite or dust explosions. Many researchers have done experiments and analytical studies in order to specify the characteristics of aluminum dust combustion on burning time, ignition temperature, burning velocity, quenching distance, activation energy, flammability limit, etc.
PALECKA et al [6] studied quenching distances for laminar flames in hybrid methane�Caluminum fuel mixtures experimentally measured at different concentrations of aluminum suspensions. BELYAEV et al [7] investigated the combustion of aluminum and introduced an equation to estimate the burning time of aluminum as follows:
 (1)
(1)
where DP is the particle diameter; �� is the burning time; ak is a constant parameter.
FRIEDMAN et al [8] and MACEK [9] reported burning time data for combustion of aluminum particle. Their results suggest an exponent of 1.2-1.5 for the particle diameter in Eq. (1). DAVIS [10] found that an exponent of 1.8 fits his experimental well. DREIZIN et al [11] studied combustion of aluminum with a particle diameter of 150 ��m in air at room temperature and standard pressure of 1.013��105 Pa. Their report shows a significant decrease in the aluminum burning time with the increase of oxygen concentration. To consider the effect of different oxidizers, BROOKS et al [12] suggested an effective oxidizer Eq. (2) which includes the concentrations of O2, CO2 and H2O.
 (2)
(2)
BECKSTEAD [13] suggested that, with the coefficients of 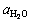 and
and 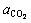 at 0.6 and 0.22 respectively, an agreement between different studies can be observed. The BECKSTEAD��s burning time equation for combustion of aluminum particles has been used in this investigation, and it is stated as follows:
at 0.6 and 0.22 respectively, an agreement between different studies can be observed. The BECKSTEAD��s burning time equation for combustion of aluminum particles has been used in this investigation, and it is stated as follows:
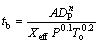 (3)
(3)
where A=0.0244 for n=1.5; A=0.00735 for n=1.8; DP is the particle diameter; tb is the burning time. SHOSHIN et al [14] suggested that the burning time of aluminum particles is a function of particle diameter:
tb=310DP (4)
where DP is the particle diameter and �� is the burning time. This equation has also been used in the current study for the generated thermal model in estimation of flame propagation speed of combustion of an aluminum dust cloud by employing different burning time models.
The particle diameter also affects the ignition temperature. MARINO [15] determined the ignition temperature of aluminum particles at different diameters. He derived an equation that fits his data best and the ignition temperature was obtained as a function of particle diameter as follows:
Tig=34.5DP+789.1 (5)
There are two general approaches to model dust combustion: the continuous or macroscopic approach and the discrete or microscopic approach. From the microscopic viewpoint, the propagation of the flame front is inherently unsteady, as it migrates from particle to particle [16, 17]. The spatial distribution of particles strongly influences the flame propagation [18]. In contrast, in the traditional continuous or macroscopic approach to model particles in a gaseous suspension, the discrete nature of the heat sources is averaged to yield a mean propagation speed. Continuous models of dust combustion cannot usually capture the lean flammability limit concentration, quenching distances or the threshold particle diameter, dust concentration and heat release. When the sources are regularly distributed in space, discreteness introduces a limit and propagation beyond this limit is only possible in a system with randomly distributed sources through local fluctuations of source concentration, which influences the combustion behavior [19]. From microscopic point of view, a collection of particles in gas phase medium forms a discrete domain and particles are distributed completely randomly. The burning mechanism for micron-sized aluminum particles is within a diffusion-controlled regime; however, aluminum particles also undergo heterogeneous combustion in oxygen due to the oxide coating present on the particles.
A new thermal model has been generated to estimate the flame front speed for micron-sized aluminum dust particles under various dust concentrations and particle sizes. Also quenching distance and minimum ignition energy have been investigated as a function of dust concentration in a random reaction medium. In the present study the thermal model considers the role of the randomly distributed particles in space in different oxidizers. This discrete heat source method provides a dust combustion model, from ignition process to the final state, including steady flame propagation, flame quenching and explosion. The present thermal model uses a novel approach for simulation of combustion of inorganic dust particles. The model is based on conduction heat transfer mechanism.
2 Discrete thermal model
2.1 Uniform distribution
The combustion of dust clouds is a complex process, involving particle heating, evaporation, intermixing with oxidizer, ignition, burning and quenching of particles. Particle size and dust concentration clearly play very important roles, but heat transfer is the dominant phenomenon. In the present discrete approach, dust particles are assumed to be uniformly dispersed in air as shown schematically in Fig. 1.

Fig. 1 Spatial distribution of particle in a uniform dust cloud (Here layer n-1 represents burnt products, layer n burning, and layer n+1, preheating)
When the ignition system provides the minimum amount of energy to the dust cloud, the temperature of some particles is increased to the ignition temperature. As these particles start to ignite, they act as heat sources in the dust cloud system and cause the temperature of the surrounding particles to rise. The temperature rise in the other particles is calculated as the sum of thermal effects from the burned and burning particles. The combustion process will proceed to the neighbor particles when the particle��s temperature reaches ignition temperature, as shown in Fig. 1. BIDABADI [20] showed that the ratio of the temperature difference caused by conduction to the temperature difference caused by radiation in aluminum particles is 501. This ratio is also known as Stark number which corresponds to the relative role of heat transfer by conduction to that by radiation. The radiative heat transfer vanishes for the limiting case of a transparent medium. This ratio has a large value for aluminum; therefore, one can neglect the effects of radiation heat transfer on combustion of aluminum dust cloud. In the present study, both conduction and radiation heat transfer mechanisms have been considered and modelled.
The temperature increase of particles in the preheated zone as a result of only the conduction heat transfer mechanism is expressed based on the superposition principle. The thermal model generated in this study for the uniform particle distribution type, is based on heterogeneous combustion in three dimensions. The model relies on the following assumptions:
1) Each particle is spherical in shape, and the associated flame diameter remains constant and equal to the particle diameter [21].
2) Combustion is assumed to take place in a quiescent reaction medium. Therefore, convection effects are not considered and the radiation heat transfer in combustion of aluminum dust cloud is negligible, and only conduction heat transfer is considered.
3) The thermal properties of the medium and particles are assumed a constant value.
4) There is an equal and constant spacing between the particles in uniform model.
5) A constant rate of energy release is considered during the combustion of a single particle.
6) The micron-sized aluminum particle is assumed to burn in a diffusion-controlled regime corresponding to infinitely fast reaction rate relatively to diffusion.
The combustion process is represented by a one-step irreversible reaction of the form vF+ where F, O2 and P denote fuel, oxygen and product, respectively, and the quantities vF,
where F, O2 and P denote fuel, oxygen and product, respectively, and the quantities vF,  and vP denote the respective stoichiometric coefficients.
and vP denote the respective stoichiometric coefficients.
The heat transfer in single-particle combustion and the temperature distribution throughout the domain is modeled as a purely conductive process in spherical coordinates, for the temperature rise Ta=T-T�� relative to the ambient temperature T��:
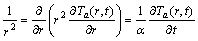 (6)
(6)
The initial and boundary conditions of the above equation are also shown below:
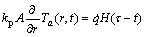 at r=rp, Ta(��, t)=0, Ta(r, 0)=0 (7)
at r=rp, Ta(��, t)=0, Ta(r, 0)=0 (7)
where H is the Heaviside function and defined as below:
 (8)
(8)
and 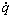 is the rate of heat release of a single particle during the burning time. The rate of heat release is assumed to behave as [22]:
is the rate of heat release of a single particle during the burning time. The rate of heat release is assumed to behave as [22]:
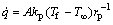 (9)
(9)
where A is the surface area of particle; kp is the heat transfer coefficient of the particle.
The solution of Eq. (6) for temperature rise was obtained through the whole domain by BIDABADI et al [23] as
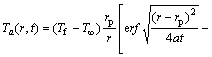
 (10)
(10)
 (11)
(11)
where Erfc is the complementary error function which is defined as
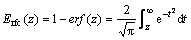 (12)
(12)
Ta is the space-time distribution of temperature around a single burning particle and beyond; Ts is the total effect of burning and burned particles which is indicative of the temperature of medium fluid around a particle in the preheated zone; r is the radial distance; rp is particle radius; t is time; �� is the burning time. The solution for any point in the domain is assumed to be given by the linear superposition of the solutions for all burning and burned particles.
T��=300 K, and Tf=3700 K are the ambient and adiabatic flame temperatures, respectively [24]. Table 1 lists some properties of the aluminum particles.
Table 1 Properties of aluminum particle

The spacing between the target particle and each surrounding particle placed at node numbers i, j, k is given by:
 (13)
(13)
where L is the spacing between two adjacent layers and determined from the mass dust concentration cd:
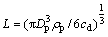 (14)
(14)
where Dp is particle diameter and ��p is particle density. The resulting flame propagation speed is defined as the ratio of the space between two adjacent layers to the difference of their ignition times [25].
 (15)
(15)
The ignition time of a single particle in a layer is assumed to be representative of its layer ignition time.
Every experimental apparatus in dust combustion uses facility as an igniter. To study the effect of this component, energy release is assumed to be sudden and it is presented by Dirac delta function in this research. The igniter��s distance to the nearest layer is assumed to be L and energy release occurs in a plane parallel to layers. The energy equation of igniter presented in Cartesian coordinate is given by
 (16)
(16)
The boundary conditions of this equation are:
 (17)
(17)
where Q is the energy released from igniter and considered to be the minimum energy needed to ignite first layer and ��(t) is Dirac delta function.
Solving Eq. (16) yields:
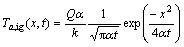 (18)
(18)
2.2 Random distribution
The model discussed in the previous section is limited to a system with particles distributed uniformly and statically in a reacting medium. In reality, dust particles may not be distributed regularly in space, so that a random placement with a given mean distance can be more realistic. Therefore, in order to create a reaction medium with randomly distributed particles, the number of nodes, Ne, uniformly distributed within the medium, is considered. Each empty node shows the location where a particle can be placed. The particles in the model are randomly distributed, generating a dust cloud with N particles. The domain size is constant and equal to the one in the uniform distribution model. The variable �� defines the fraction of filled locations:
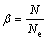 (19)
(19)
For uniform distribution of particles, ��=1, and for random distribution, ��<1.
In order to maintain the same overall mass concentration as that in the uniform distribution model, the new distance between two adjacent layers, Lr or Lrandom, should be presented as given below:
 (20)
(20)
Therefore, the total volume is  and the total particle mass is N��pVp, and the total mass concentration per unit volume is maintained. The random model employs the same governing equations used in the case of uniformly distributed particles.
and the total particle mass is N��pVp, and the total mass concentration per unit volume is maintained. The random model employs the same governing equations used in the case of uniformly distributed particles.
2.3 Quenching distance
In this section, combustion of aluminum dust particles is studied in a narrow channel with infinite length and constant width with aluminum particles distributed randomly in space. The combustion of single-particle is first studied and the solution is presented. Each burning/burned particle is considered as a heat source and the amount of heat loss to the walls is assumed as heat sink. Based on the superposition principle, the space-time temperature distribution of particles and the heat loss to the walls are the two factors that influence the temperature of the preheat zone. Heat sources are the burning/burned particles and heat sinks are the heat losses to walls due to the temperature difference between the wall and the gas surrounding the burning particles during the combustion process. The heat released through combustion of particles that their temperatures reach ignition point spreads out into the preheat zone via conduction heat transfer mechanism. The flame front speed is defined as the ratio of the spacing between two adjacent layers to the difference of their ignition times. If the flame quenches in the channel, the channel width will be considered to be equal or less than the quenching distance. The flame will disseminate in the narrow channel if the channel width is more than the quenching distance [23].
In the present model particles are distributed randomly in space. The space between two adjacent layers is estimated from Eq. (20). Because of symmetry, to define the ignition time of all the particles in a layer, a single particle of a layer is taken as the representative of the whole layer. This particle is positioned at the origin of the local coordinate system. The flame spreads out through the layers and ignites successively. By introducing tig,i as the ignition time of layer i, the summation of all the burned and burning particles as heat sources is expressed as:
 (21)
(21)
In this model, the walls play the role of heat sinks (see Fig. 2). The heat transfer to walls by the averaged thermal conductivity of the mixture is considered to be one- dimensional and perpendicular to the wall surface.
Considering the lumped capacitance model, temperature in a layer in the preheat zone is considered uniform. In other words, if temperature of a particle in a layer reachs ignition temperature, which means that all the particles in that layer are ignited at the same time. Walls are assumed to be at the temperature Tw, the lumped capacitance average temperature of layer is T layer and the channel width is D. The rate of heat loss to the walls of the channel is obtained from the work done by BIDABADI [20].

Fig. 2 Schematic of lumped capacitance assumption for layers and one-dimensional heat transfer to walls and the quenching distance obtained for parallel flat plates configuration
 (22)
(22)
The total heat loss of one layer in the preheat zone, from time zero to time t is:
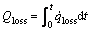 (23)
(23)
Using the lumped capacitance model assumption, the temperature deduction of the layer is expressed as Tsink=Qloss/��cpV. Thus, the temperature reduction becomes:
 (24)
(24)
The temperature of a layer in the preheat zone is the summation of the temperatures of all the heat sinks and sources.
 (25)
(25)

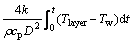 (26)
(26)
In the study, use a backward method to obtain and calculate the integral part of Eq. (26). Since the temperature of the target particle of layer n+1 in the preheat zone reachs the ignition temperature, this layer starts to ignite and burn. Thus, the flame propagation speed is defined as the ratio of the spacing between two adjacent layers, L, divided by the ignition time difference of those layers, tig,i+1-tig,i [25].
4 Results and discussion
The ignition time of each layer and the distance between two adjacent layers is needed to obtain the flame propagating speed. A computer code has been developed to calculate the ignition time and burning velocity of each layer as the logic and algorithm illustrated in Fig. 3. Following the energy release by the ignition system, the temperature of the first layer at the considered location is calculated at the beginning. The igniter��s distance to the nearest layer is assumed to be L and energy release occurs in a plane parallel to layers. When the temperature of the particles in the first layer reaches the ignition temperature, it is recorded as the ignition time of the first layer, and the calculations are continued to find the ignition times of the other layers.
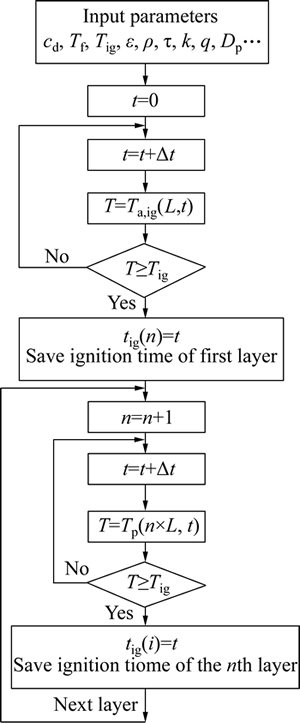
Fig. 3 Flowchart for calculating ignition time of aluminum particles.
Beyond the first layer (n>1), preheating of the layers is influenced by burning of the preceding layers and the ignition system. Thus, when the temperature of a certain layer reaches the ignition temperature, the relevant time is recorded as the ignition time of that layer.
The flame propagation speed is defined as the ratio of the spacing between two adjacent layers, L, divided by the ignition time difference of those layers, tig,i+1-tig,i.
Figure 4 shows variation of flame propagation speed as a function of dust concentration for aluminum particles with 5.4 ��m diameter where particles are randomly distributed in the reaction medium with 11% oxygen concentration by volume at atmospheric pressure and the initial temperature of the reaction zone is considered to be 300 K. With the increase of dust concentration, the value of flame speed tends to rise. The obtained results use the BECKSTEAD��s burning time model with the diameter at the power of 1.8. Three runs of the generated code have been performed and the obtained results shown in Fig. 4 which demonstrate different spatial types of particles distribution in a reaction medium. Therefore, the graphs of flame propagation speed in terms of dust concentration are different from each other.

Fig. 4 Flame propagation speed as function of aluminum dust concentration for random distribution of particles for different values of ��:
The value of flame speed depends on the particular distribution of particles in space for a considered dust concentration. The values of flame speed show fluctuations. The reason of fluctuation is that, in random distribution of fuel particles there are different numbers of particles in each layer and not all particles would fill all existing nodes. In general, flame speed tends to increase with the rise of dust concentration.
Figure 4 also illustrates the fluctuation in the value of flame speed for two different �� values as defined in Eq. (19). It can be seen that by increasing the parameter ��, the amount of flame speed��s fluctuations would be decreased. The reason is that for higher value of ��, more fuel particles would fill the empty nodes; therefore, particles become closer to each other and the scale of fluctuation would be reduced when the flame passes through the dust cloud.
The results are compared with the experimental work of BIDABADI [20], as shown in Fig. 4 and show good agreement with the experiment.
Figure 5 shows the value of flame front speed in terms of dust concentration. However, the percentage of oxygen concentration is considered to be 11% by volume. As seen, the flame speed for a considered dust concentration is lower than that in the case of 16% oxygen concentration. The reason is that in the case of 11% oxygen concentration, the amount of oxidizer required for combustion of fuel particles is insufficient and some particles would not ignite due to the lack of oxidizer; therefore, the value of flame speed decreases remarkably. The parameter �� is assumed to be 0.65 and the BECKSTEAD��s burning time model with the diameter at the power of 1.4 has been employed.
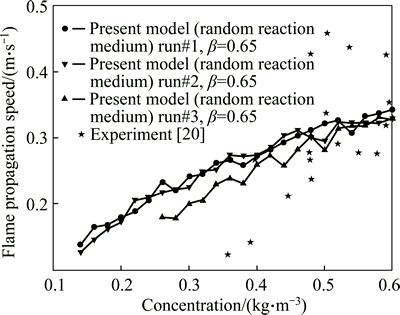
Fig. 5 Flame propagation speed in terms of aluminum dust cloud concentration in a random reaction medium with 11% oxygen for particle diameter of 5.4 ��m at initial temperature of 300 K and atmospheric pressure and ��=0.65
Figure 6 demonstrates the variation of flame propagation speed with equivalence ratio in air for particle diameter of 6.5 ��m. The results from the generated thermal model are in good agreement with the experimental data [27]. The BECKSTEAD��s burning time model with the diameter at the power of 1.8 has been used for this calculation. The random graph in Fig. 6 is the average of the results of 20 runs of the generated code in a random reaction medium.
Figure 7 depicts the change in flame propagation speed under various aluminum dust concentrations using pure carbon dioxide as the oxidizer for two different particle diameters. The thermal diffusivity of carbon dioxide is considered to be a constant value. The results of the thermal model for equivalence ratio of ��=1 and particle diameters of 1 ��m and 3 ��m are compared with the data reported by KING [26]. Figure 7 depicts that the flame speed in pure carbon dioxide is lower than that in oxygen as oxidizer in Figs. 4 and 5 for a considered particle diameter and dust concentration.

Fig. 6 Variation of flame propagation speed with equivalence ratio in air in a random reaction media with for particle diameter of 6.5 ��m under initial temperature of 300 K and under atmospheric pressure and ��=0.65

Fig. 7 Variation of flame propagation speed with equivalence ratio in pure carbon dioxide reaction media at initial temperature of 300 K , atmospheric pressure and ��=0.65
Figure 8 illustrates the quenching distance in terms of aluminum dust concentration in air with ��=0.75. With increase of dust concentration, the amount of energy released per unit volume would be increased; therefore, the temperature of the layers in the preheat zone reaches faster to ignition temperature and the flame speed will increase. With the increase of flame speed, the amount of heat losses to walls reduces and one can conclude that higher values of dust concentration lead to smaller quenching distances. The estimation of the present thermal model is more precise when compared to the theory given by BIDABADI [20] and fits well with the experiment.

Fig. 8 Experimental validation of quenching distance of 5.4 ��m aluminum particles predicted by the presented model in a random reaction medium with ��=0.75
Figure 9 shows the quenching distance in terms of aluminum dust concentration for different particle diameters. Graphs in Fig. 9 are the average of the results of 10 runs of the generated code with ��=0.75. As it can be understood, for a considered dust concentration, for larger particles the value of quenching distance is higher. The reason is that the ratio of the particle��s surface area to its volume A/V for larger particles is less than smaller ones. Therefore the amount of heat released by larger particles lower as the total amount of surface area of larger particles is less and the amount of energy released is lower. It causes that it takes much more time for the next layers in the preheating zone to reach the ignition point to be ignited; therefore, for larger particles the amount of heat losses to walls is higher than smaller particles.
Figure 10 shows the minimum ignition energy (MIE) in comparison with dust concentration for different particle diameters. It can be concluded that for a considered amount of dust concentration, larger particle diameters have higher values of MIE rather than smaller ones. The reason is that larger particles have higher ignition temperatures and they would take much more energy to reach ignition temperature and also the distance between particles would be increased in the 3D space.

Fig. 9 Quenching distance in comparison with aluminum dust concentration for various particle sizes in a random reaction medium with ��=0.75

Fig. 10 Minimum ignition energy in comparison with aluminum dust concentration for various particle sizes
4 Conclusions
In the present study, flame propagation of aluminum dust particles in a quiescent reaction medium with spatially discrete sources is numerically investigated. Fuel particles are assumed to be randomly distributed in space in the reaction medium. An algorithm has been generated in order to study the variation of flame propagating speed as a function of dust concentration and the size of particles for random distribution of particles.
The present thermal model is used to study the influence of random distribution of particles in reaction medium, on the flame speed. Further, the obtained results show that flame speed in aluminum-oxygen suspensions decreases as the particle size increases, owing to the fact that larger particles require much more energy to be ignited as compared to the smaller ones. Considering random distribution of particles, the obtained results are much closer and realistic to the combustion physics of dust flames as compared to the experimental data.
The quenching distance is studied in terms of dust concentrations and particle sizes in a random reaction medium. With an increase in the dust concentration for a considered particle diameter, it leads to a smaller quenching distance. The minimum ignition energy is investigated in terms of dust concentrations. By increasing the dust concentration for a considered particle diameter, the MIE would be decreased as there would be higher number of particles and the distance between particles reduces. The present thermal model is an effective model for estimation of flame front speed in various types of oxidizer at various initial conditions and also for estimation of quenching distance and MIE. It is found that the generated thermal model has a good capability of predicting various flame features such as flame speed and quenching distance with respect to the experimental findings in random reaction medium.
References
[1] ECKHOFF R K. Dust explosion in the process industries [M]. Oxford: Butterworth, 1997.
[2] MOSTAFAVI S A, SALAVATI S, DIZAJI H B, BIDABADI M. Pyrolysis and combustion kinetics of lycopodium particles in thermo-gravimetric analysis [J]. Journal of Central South University, 2015, 22: 3409-3417.
[3] SOLTANINEJAD M, DIZAJI F F, DIZAJI H B, BIDABADI M. Micro-organic dust combustion considering particles thermal resistance [J]. Journal of Central South University, 2015, 22: 2833-2840.
[4] ROCKWELL S R, RANGWALA A S. Modeling of dust air flames [J]. Fire Safety Journal, 2013, 59: 22-29.
[5] BIDABADI M, HAGHIRI A, RAHBARI A. The effect of Lewis and Damkohler numbers on the flame propagation through micro-organic dust particles [J]. International Journal of Thermal Sciences, 2010, 49: 534-542.
[6] PALECKA J, JULIEN P, GOROSHIN S, BERGTHORSON J, FROST D L, HIGGINS A J. Quenching distance of flames in hybrid methane-aluminum mixtures [J]. Proceedings of the Combustion Institute, 2015, 35: 2463-2470.
[7] BELYAEV A, FROLOV Y, KOROTKOV A I. Combustion and ignition of particles of finely dispersed aluminum [J]. Combustion, Explosion & Shock Waves, 1968, 4: 323-329.
[8] FRIEDMAN R, MACEK A. Combustion studies of single aluminum particles [C]// Ninth Symposium (International) on Combustion. VA, USA, 1963: 703-709.
[9] MACEK A. Fundamentals of combustion of single aluminum and beryllium particles [C]// Eleventh Symposium (International) on Combustion. Pittsburgh, PA, USA, 1967: 203-217.
[10] DAVIS A. Solid propellants: The combustion of particles of metal ingredients [J]. Combustion and Flame, 1963, 7: 359-336.
[11] DREIZIN E L, TRUNOY M A. Surface phenomena in aluminum combustion [J]. Combustion and Flame, 1995, 101: 378-382.
[12] BROOKS K P, BECKSTEAD M W. Dynamics of aluminum combustion [J]. Journal of Propulsion and Power, 1995, 11: 769-780.
[13] BECKSTEAD M W. A summary of aluminum combustion [C]// Missile Sciences Conference: Internal Aerodynamics in Solid Rocket Propulsion. NJ, USA, 2002: 5.1-5.46.
[14] SHOSHIN Y L, DREIZIN E L. Particle combustion rates for mechanically alloyed Al-Ti and aluminum powders burning in air [J]. Combustion and Flame, 2006, 145: 714-722.
[15] MARINO T A. Numerical analysis to study the effects of solid fuel particle characteristics on ignition, burning, and radiative emission [D]. The George Washington University, 2007.
[16] MUKASYAN A, HWANG S, SYTCHEV A, ROGACHEV A, MERZHANOV A, VARMA A. Combustion wave microstructure in heterogeneous gasless systems [J]. Combustion Science and Technology, 1996, 115: 335-353.
[17] HWANG S, MUKASYAN A, ROGACHEV A, VARMA A. Combustion wave microstructure in gas-solid reaction systems: Experiments and theory [J]. Combustion Science and Technology, 1997, 123: 165-184.
[18] TANG F D, HIGGINS A J, GOROSHIN S. Effect of discreteness on heterogeneous flames: Propagation limits in regular and random particle arrays [J]. Combustion Theory and Modelling, 2009, 13: 319-341.
[19] VARMA A, MUKASYAN A, HWANG S. Dynamics of self- propagating reactions in heterogeneous media: Experiments and model [J]. Chemical Engineering Science, 2001, 56: 1459-1466.
[20] BIDABADI M. An experimental and analytical study of laminar dust flame propagation [D]. Canada: McGill University, 1995.
[21] SUN J H, DOBASHI R, HIRANO T. Combustion behavior of iron particles suspended in air [J]. Combustion Science and Technology, 1990, 150: 99-114.
[22] HANAI H, KOBAYASHI H, NIIOKA T. A numerical study of pulsating flame propagation in mixtures of gas and particles [J]. Proceedings of the Combustion Institute, 2000, 28: 815-822.
[23] BIDABADI M, ZADSIRJAN S, MOSTAFAVI S A. The discrete heat source approach to dust cloud combustion [J]. Journal of Heat Mass Transfer, 2014, 50: 759-767
[24] MARION M, CHAUVEAU C, GOKALP I. Studies on the ignition and burning of aluminum particles [J]. AIAA J, 1995, 2861: 769-780.
[25] GOROSHIN S, LEE J, SHOSHIN Y. Effect of the discrete nature of heat sources on flame propagation in particulate suspensions [C]// Symposium (International) on Combustion. 1998: 743-749.
[26] KING M. Modeling of single particle aluminum combustion in CO2-N2 atmospheres [C]// Seventeenth Symposium (International) on Combustion. Pittsburgh, USA, 1979, 17: 1317-1328.
[27] YETTER R A, HUANG Y, RISHA G A, YANG V. Effect of particle size on combustion of aluminum particle dust in air [J]. Combustion and Flame, 2009, 156: 5-13.
(Edited by YANG Hua)
Cite this article as: Mehdi Bidabadi, Alireza Khoeini Poorfar. Modeling flame propagation speed and quenching distance of aluminum dust flame with spatially random distribution of particles [J]. Journal of Central South University, 2017, 24(1): 81-89. DOI: 10.1007/s11771-017-3411-2.
Received date: 2015-11-25; Accepted date: 2016-01-26
Corresponding author: Alireza Khoeini Poorfar, Doctoral Candidate; Tel: +98-2177240197; Fax: +98-77240488; E-mail: Alirezapoorfar@iust.ac.ir

